Abstract
Bromodomain and extraterminal domain (BET) proteins are epigenetic readers that play an important role in chromatin remodeling and transcriptional regulation. In this study, we found that BRD4, a BET family member, is significantly upregulated in hepatocellular carcinoma (HCC) tissues compared with adjacent normal tissues. Furthermore, the overexpression of BRD4 in cancer tissues was correlated with poor prognosis in HCC patients. Using shRNA-mediated knockdown of BRD4 or lentivirus-mediated overexpression of BRD4 in HCC cells, we further showed that BRD4 was involved in HCC cell growth and invasion in vitro. Forced expression of BRD4 was sufficient to induce epithelial-mesenchymal transition (EMT) phenotypes in HCC cells. Additionally, BRD4 shRNA significantly inhibited HCC cell proliferation in vivo. Collectively, our study confirmed that BRD4 expression is a valuable predictor of recurrence and survival in patients with HCC. BRD4 can be further used as a potential therapeutic target of HCC.
Keywords
Hepatocellular carcinoma (HCC) is the third leading cause of cancer-related deaths worldwide. 1 This cancer is characterized by resistance to adjuvant chemotherapy and high recurrence after curative liver resection,2,3 therefore, there is an urgent need to obtain a better understanding of the mechanisms relevant to this disease in order to develop new preventive, diagnostic, and therapeutic options. The involvement of epigenetic dysregulation in HCC pathogenesis remains understudied. Thus, investigating the contribution of epigenetic dysregulation to HCC would broaden our understanding of its underlying biology and etiology.
Members of the bromodomain and extra terminal domain (BET) family of proteins (BRD2, BRD3, BRD4, and the testis-specific BRDT) function as important reader molecules that associate with acetylated histones and participate in the assembly of chromatin complexes and transcription activators at specific promoter sites. 4 BRD4 is the best studied member of the BET family. It is a nuclear protein that binds to acetylated histone 3 (H3) and histone 4 (H4) tails, and plays an important role in maintaining chromatin architecture and controls transcription elongation through phosphorylation of RNA polymerase II by CDK9.5–9 BRD4 is also involved in stabilizing nuclear NF-κB and controlling the expression of downstream target genes.10,11 Recent studies have shown that pharmacologic inhibition of BRD4 with BET-specific BD inhibitors effectively inhibits cancer cell proliferation in melanoma, lung cancer, multiple myeloma, Burkitt’s lymphoma and acute myeloid leukemia.12–16
Although BRD4 has been regarded as a potential therapeutic target for several cancers, it has never been investigated in HCC. Thus, in this study, we attempted to elucidate the role and associated molecular mechanism of BRD4 in HCC cell proliferation and invasion. The clinical relevance and prognostic significance of BRD4 in human HCC were also investigated.
Materials and methods
Patients, follow-up, and treatment modalities
A total of 209 pathologically confirmed HCC patients, who received curative resection in the Liver Cancer Institute, Zhongshan Hospital, Fudan University between January 2006 and December 2007, were enrolled in this study. The inclusion criteria and treatments received by patients with standardized follow-up as established by our institute have been described previously. 17 Primary endpoints were time to recurrence (TTR) and overall survival (OS), defined as time intervals between the date of surgery and first report of tumor recurrence or patient death, respectively. Patients without recurrence or death were censored.
Construction of tissue microarrays and immunohistochemistry
Tissue microarray (TMA) was constructed as described in our earlier study. 18 IHC staining for the target genes was carried out on sections of the formalin-fixed samples on the TMA.
Cell culture
The human HCC cell lines Huh-7, HepG2, PLC/PRF/5, SMMC-7721, MHCC97L, and MHCC97H were used. All of the cell lines were maintained in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum and antibiotics in a humidified atmosphere of 95% air and 5% CO2 at 37°C.
Lentivirus production and transduction of target cells
The BRD4 and BRD4 shRNA expression lentivirus were purchased from Shanghai GeneChem Co., and the target shRNA sequences are listed in the supporting information. The lentiviral vector was transfected into cells as described elsewhere. 18
Cell proliferation and transwell invasion assay
Cell proliferation was measured by a CCK-8 Kit (Dojindo Laboratories, Rockville, MD, USA). Cell invasion was measured by a transwell matrigel invasion assay, performed as described in the reference. 19
Western blot analysis
Cell lysates were collected and centrifuged for 15 min at 13,000 rpm at 4°C. The supernatant was transferred to a clean tube, and proteins were quantified using the BCA kit (Pierce, Rockford, IL, USA). Proteins were separated on SDS–PAGE gels and transferred to nitrocellulose membranes. The membranes were blocked with 5% skim milk and incubated overnight at 4°C with primary antibodies. Immune complexes were detected by incubating the nitrocellulose membranes with HRP-conjugated secondary antibody for 2 h at room temperature and subsequent exposure of the membrane to enhanced chemiluminescence reagents.
Tumor growth assay
The tumor growth assay was performed by subcutaneous inoculation of tumor cells into 6-week-old nude mice. Tumor volumes were recorded until mice were sacrificed on the 30th day after the inoculation of tumor cells. The tumor volumes were calculated by using the following formula: (length × width2)/2.
Statistical analysis
Statistical analysis was performed using the SPSS 15.0 software (Chicago, IL, USA). All tests were two-tailed and P <0.05 was considered statistically significant.
Results
High expression of BRD4 correlates with poor prognosis of HCC patients
To explore the association of BRD4 expression in HCC tissues with disease prognosis, TMAs from 209 patients with HCC were examined by immunostaining. As shown in Figure 1A, the BRD4 staining was localized to the nucleus, and the expression of BRD4 was weaker in peritumoral tissues compared with HCC tissues. In HCC tissues, BRD4 expression was strong in 72 (34.4%), moderate in 84 (40.2%), weak in 41 (19.6%), and negative in 12 (5.7%) cases and it was localized to the nucleus.
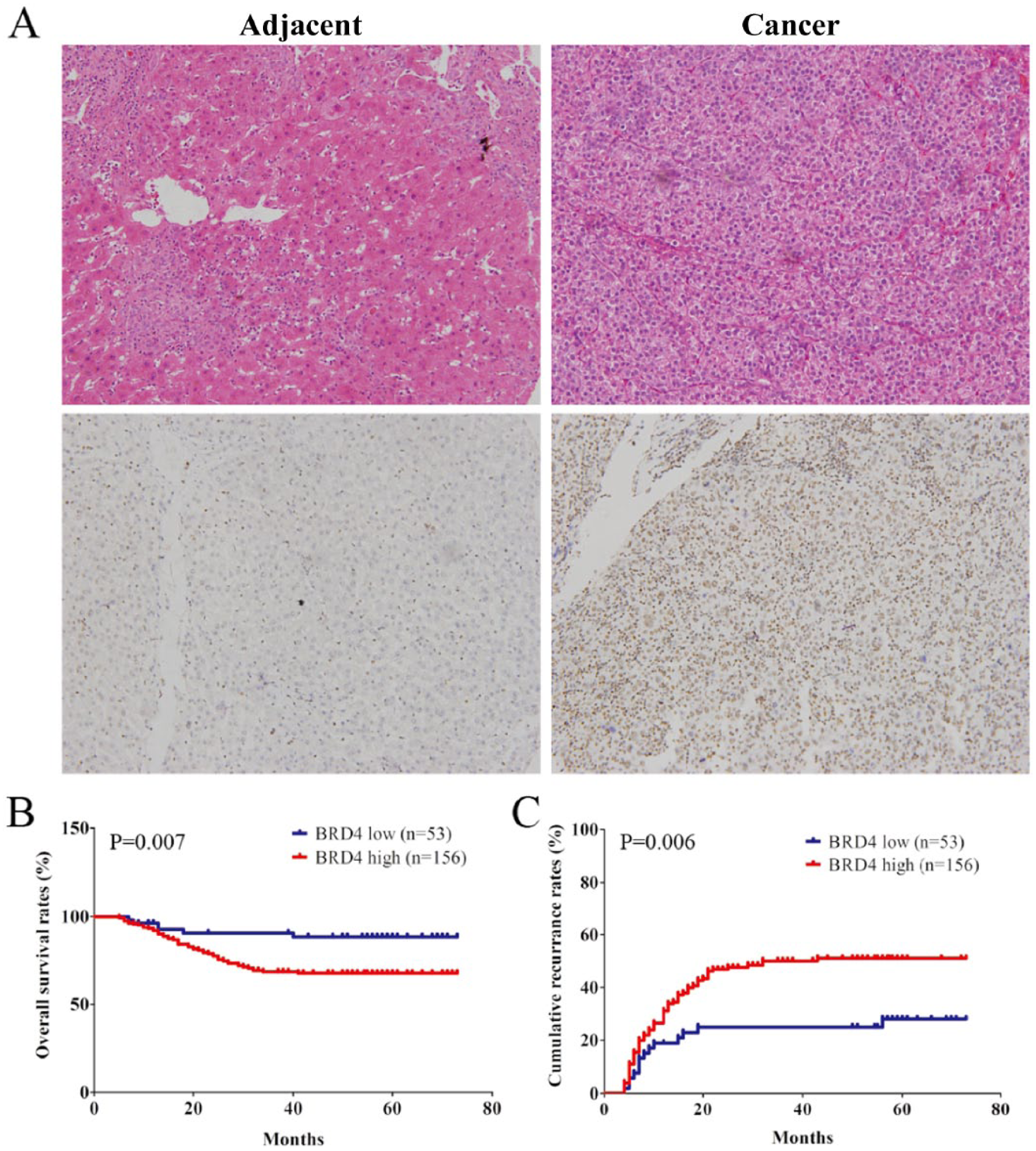
BRD4 is overexpressed in HCC tissues and its expression correlates with clinical outcome of patients with HCC. (A) Immunostaining of BRD4 using 3,3′-diamin.obenzidine (DAB; brown) was shown for adjacent normal liver and HCC tissues. (B, C) the cumulative recurrence and overall survival rates of 209 patients with HCC were compared between BRD4-low or -high groups, using Kaplan–Meier methods (log-rank test).
For further analysis, patients were dichotomized into BRD4-high (moderate and strong; n = 156) or -low (negative and weak; n = 53) groups. Statistically, there was a striking inverse association between BRD4 intensity and overall survival (OS; P = 0.007) and to a less extent recurrence-free survival (RFS; P = 0.006) (Figure 1B and C). The median OS and RFS time for BRD4 high patients were 42.1 and 55.4 months, respectively, compared with 56.6 and 66.3 months for BRD4 low patients, respectively. Multivariate analysis revealed that BRD4 intensity in cancer tissues was an independent prognosticator for both OS and RFS (Tables 1 and 2). Therefore, BRD4 expression is a valuable predictor for recurrence and survival in patients with HCC.
Univariate and multivariate analyses of factors associated with overall survival.

95% CI: 95% confidence interval; AFP: alpha-fetoprotein; BCLC: Barcelona-clinic liver cancer; HBsAg: hepatitis B surface antigen; HR: hazard ratio; NA: not adopted; NS: not significant; OS: overall survival.
Cox proportional hazards regression model.
Univariate and multivariate analyses of factors associated with cumulative recurrence.

95% CI: 95% confidence interval; AFP: alpha-fetoprotein; BCLC: Barcelona-clinic liver cancer; HBsAg: hepatitis B surface antigen; HR: hazard ratio; NA: not adopted; NS: not significant.
Cox proportional hazards regression model.
BRD4 is involved in HCC cell proliferation
To elucidate the role of BRD4 in HCC cell proliferation, BRD4 shRNA was used to reduce BRD4 expression in the Huh 7 and SMMC-7721 cells, which have a high level of BRD4 protein expression (Figure 2A). Our results show that BRD4 shRNA significantly reduced BRD4 expression, as well as the proliferation of Huh 7 and SMMC-7721 cells (Figure 2B and D). To further evaluate whether BRD4 upregulation could promote tumor proliferation, lentivirus-mediated delivery of BRD4 cDNA was used to increase BRD4 expression in the human HCC MHCC97L and PLC/PRF/5 cells, which have lower BRD4 protein expression. The upregulation of BRD4 expression was observed in BRD4 infectants (relative to the mock control). BRD4 upregulation significantly increased the proliferation abilities of MHCC97L and PLC/PRF/5 cells compared with the mock control (Figure 2C and E).
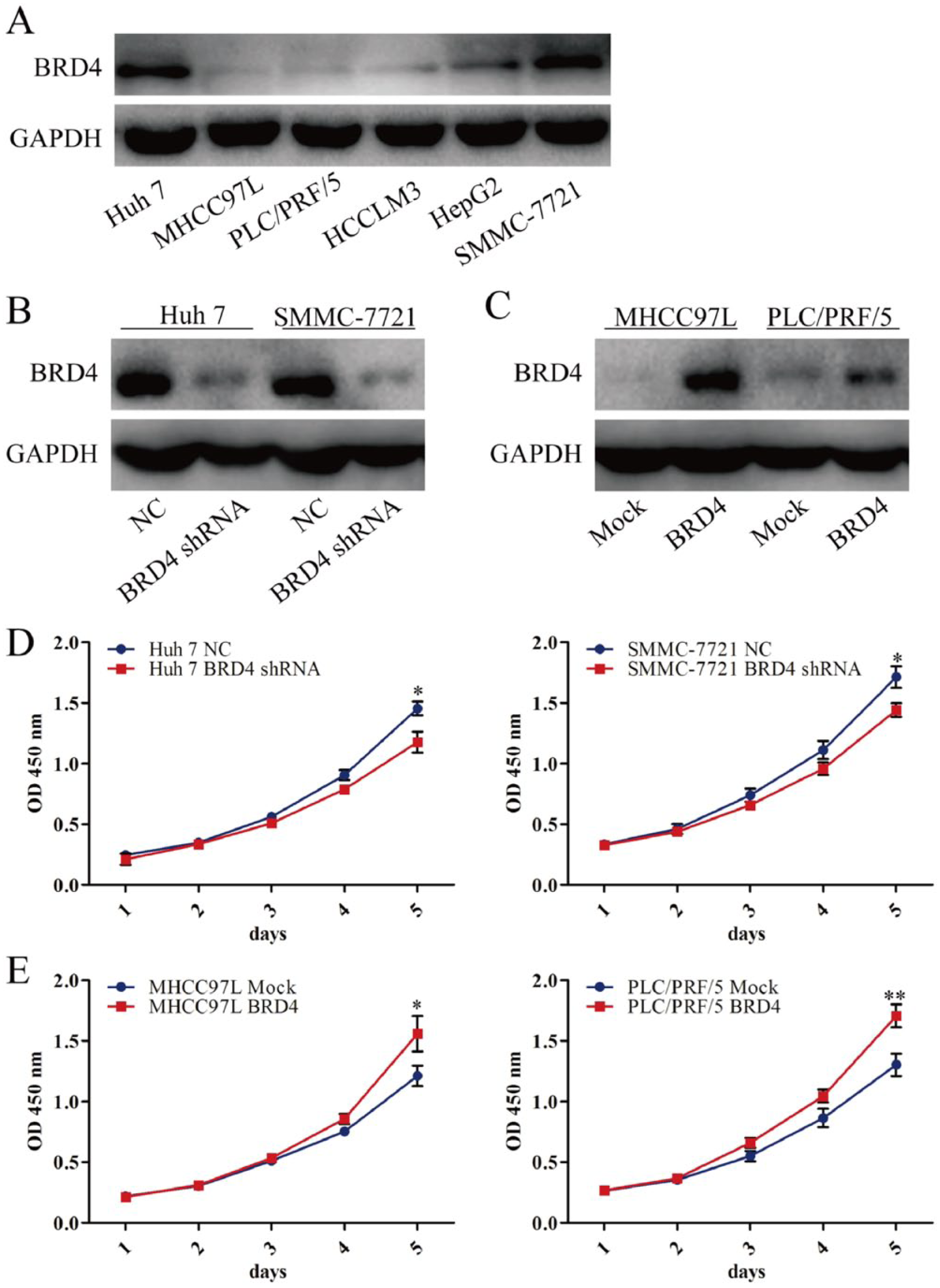
BRD4 promotes HCC cell proliferation. (A) BRD4 expression in several EOC lines was examined by western blotting. (B, C) BRD4 expression in Huh 7, SMMC-7721, MHCC97L, and PLC/PRF/5 transfectants was examined by western blotting. (D, E) The proliferation activity of Huh 7, SMMC-7721, MHCC97L, and PLC/PRF/5 transfectants in vitro was examined by CCK-8 assay. *P <0.05, **P <0.01. Comparisons between two means were evaluated using the LSD method.
BRD4 promotes invasion in vivo and regulates the EMT phenotypes in HCC cells
It has been confirmed in a previous study that BRD4 combined with Twist induces epithelial-mesenchymal transition (EMT) in breast cancer. 20 This finding may indicate that BRD4 could promote HCC cell invasion. Cell invasion was measured by a transwell matrigel invasion assay in this study. The results show that BRD4 shRNA significantly reduced the invasion of Huh 7 and SMMC-7721 cells and that the upregulation of BRD4 expression significantly increased the invasion abilities of MHCC97L and PLC/PRF/5 cells compared with the mock control (Figure 3A and B).
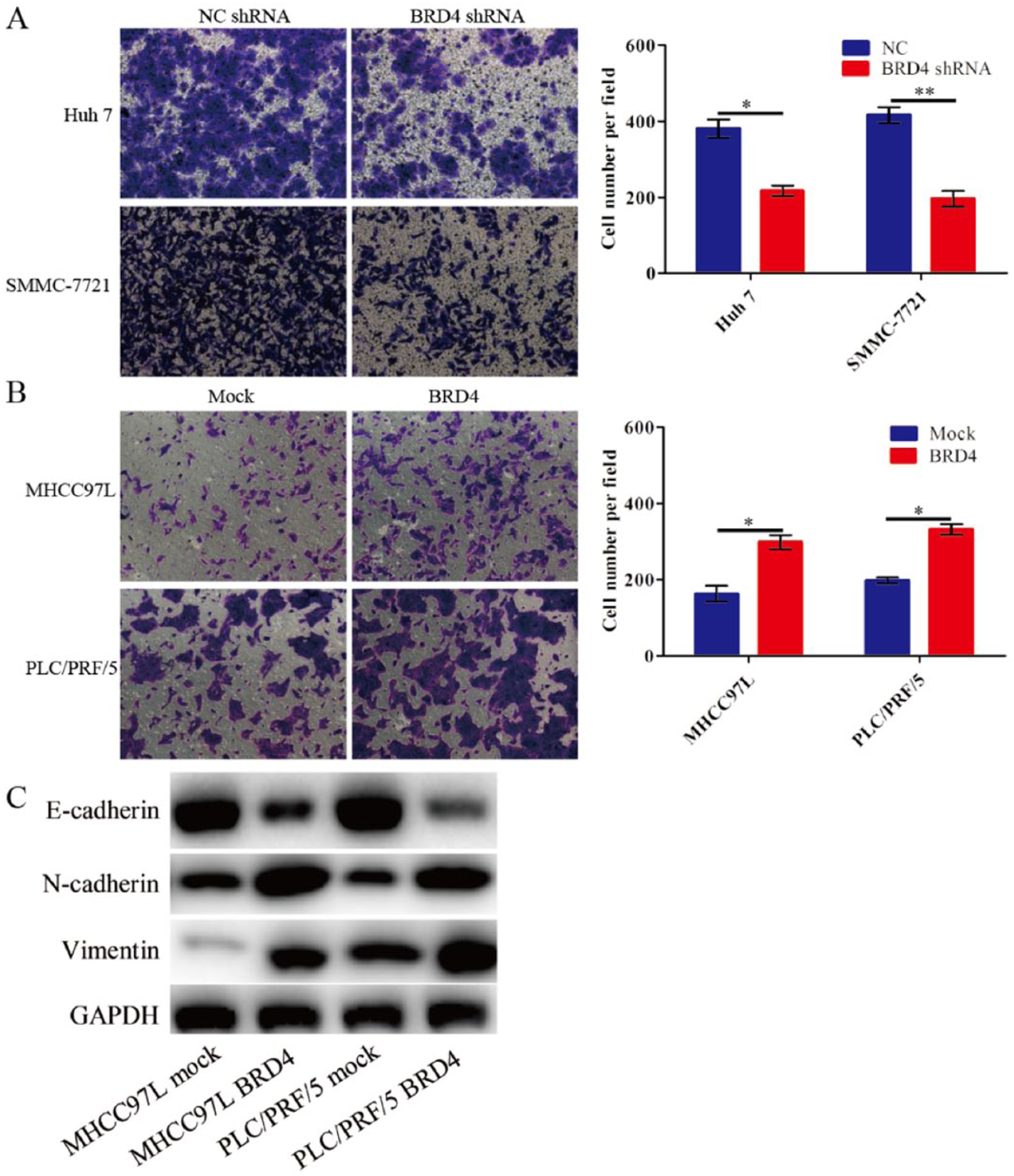
BRD4 promotes invasion and regulates the EMT phenotypes in HCC cells. (A, B) The invasion activity of Huh 7, SMMC-7721, MHCC97L, and PLC/PRF/5 transfectants in vitro was examined by a matrigel transwell assay. *P <0.05, **P <0.01. Comparisons between two means were evaluated using the LSD method. (C) BRD4 regulates the EMT phenotypes in HCC cells.
To further understand the mechanisms involved in the positive regulation of cell migration and invasion by BRD4, we observed the expression changes of EMT markers by using western blot. We showed that BRD4 overexpression decreased the levels of E-cadherin and increased the levels of mesenchymal markers, N-cadherin and vimentin, in BRD4-infected MHCC97L and PLC/PRF/5 cells (Figure 3C).
Knockdown of BRD4 expression by RNAi inhibits growth of HCC cells in vivo
To assess the function of BRD4 expression in HCC cell proliferation in vivo, an in vivo tumor growth assay was performed. The volumes of the tumors were measured for a period of 34 days. At the end of the experiment, the mice were sacrificed. As shown in Figure 4, the tumorigenicity assay results indicate that knockdown of BRD4 expression inhibits cells proliferation in vivo.

BRD4 shRNA inhibits HCC xenografts growth. (A) Huh 7. (B) SMMC-7721. Data are represented as the mean ± SD, n = 6, *P <0.05.
Discussion
The detailed mechanisms of epigenetic modifications in cancer initiation and progression are becoming evident. In HCC, anomalous epigenetic changes have been shown to contribute to tumor initiation and progression.21,22 In this study, we demonstrated that compared with adjacent normal liver tissues, BRD4 was overexpressed in HCC tissues, and a high level of BRD4 expression correlated with poor prognosis in HCC. Similar results have been reported previously. 12
Accumulating evidence from previous studies suggests that EMT plays an important role in cancer invasion. 23 EMT is observed in several carcinoma cells undergoing phenotypic conversion for invasion and is characterized by the loss of epithelial cell markers, such as E-cadherin, and the gain of mesenchymal markers, such as vimentin and N-cadherin. 24 BRD4 has been shown to control the expression of numerous genes involved in the cell cycle and cell growth.25,26 Twist and Snail are two key EMT-activating transcriptional factors. Twist overexpression was correlated with metastasis through the induction of EMT, as well as with cell invasiveness in HCC. 27 Inhibition of the Twist-BRD4 association reduced WNT5A expression and suppressed invasion, cancer stem cell (CSC)-like properties, and tumorigenicity of breast cancer cells. 20 In this study, our results show that the forced expression of BRD4 was accompanied by the upregulation of N-cadherin, viemetin, and reduced E-cadherin in HCC cells. Given our observation that BRD4 is overexpressed in HCC tissues, we hypothesized that the epigenetic and/or transcriptional regulation of certain target genes may support HCC proliferation. This hypothesis was supported further by serial experiments in vitro. Furthermore, we found that BRD4 knockdown in Huh 7 and SMMC-7721 cells reduced cell proliferation and invasion, whereas BRD4 overexpression in MHCC97L and PLC/PRF/5 promoted cells proliferation and invasion. Taken together, these data suggest that BRD4 increased the proliferation and invasion ability of HCC cells. Moreover, we demonstrated that BRD4 overexpression might activate the BRD4/Twist/WNT5A signal to induce HCC cell proliferation and EMT. The above results support the notion that BRD4 plays a vital role in the progression of HCC.
Further experiments are needed to clarify the mechanisms through which BRD4 is involved in HCC proliferation and invasion. Furthermore, we showed that BRD4 overexpression in xenografted HCC cells is effectively inhibited by BRD4 shRNA, suggesting that BRD4 may provide a viable therapeutic target for the treatment of HCC.
In summary, our findings identify BRD4, which functions to promote multiple key processes such as proliferation and invasion, as a predictor for recurrence and survival in patients with HCC. Thus, we propose here that BRD4 is a promising therapeutic target for HCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Key Sci-Tech Project (2012ZX10002011-002) and the National Natural Science Foundation of China (81472840, 81071741, and 81030038); the Shanghai Municipal Natural Science Foundation (14ZR1405800); the PhD Programs Foundation of the Ministry of Education of China (20110072120050); the Open Fund of Key Laboratory of Carcinogenesis and Cancer Invasion, Fudan University, Ministry of Education (KLCCI2014-8); and the China Postdoctoral Science Foundation (2014M561410).
