Abstract
Objectives:
Disruption in the natural immune reaction due to SARS-CoV-2 infection can initiate a potent cytokine storm among COVID-19 patients. An elevated level of IL-6 and IL-10 during a hyperinflammatory state plays a vital role in increasing the risk of severity and mortality. In this study, we aimed to evaluate the potential of circulating IL-6 and IL-10 levels as biomarkers for detecting the severity and mortality of COVID-19.
Methods:
This study was conducted according to the Cochrane Handbook and PRISMA guidelines. Authorized databases were searched to extract suitable studies using specific search terms. RevMan 5.4 was applied for performing the meta-analysis. Mean differences in IL-6 and IL-10 levels were calculated among COVID-19 patients via a random-effects model. NOS scoring, publication bias and sensitivity analyses were checked to ensure study quality.
Results:
A total of 147 studies were selected, with 31 909 COVID-19 patients under investigation. In the severity analysis, the mean concentration of IL-6 was significantly higher in the severe COVID-19 cases than in the non-severe cases (MD: 19.98; P < .001; 95% CI: 17.56, 22.40). Similar result was observed for IL-10 mean concentration in severe COVID-19 cases (MD: 1.35; P < .001; 95% CI: 0.90, 1.80). In terms of mortality analysis, circulating IL-6 showed sharp elevation in the deceased patients (MD: 42.11; P < .001; 95% CI: 36.86, 47.36). IL-10 mean concentration was higher in the dead patients than in the survived patients (MD: 4.79; P < .001; 95% CI: 2.83, 6.75). Publication bias was not found except for comparing IL-6 levels with disease severity. Sensitivity analysis also reported no significant deviation from the pooled outcomes.
Conclusions:
Elevated levels of circulating IL-6 and IL-10 signifies worsening of COVID-19. To monitor the progression of SARS-CoV-2 infection, IL-6 and IL-10 should be considered as potential biomarkers for severity and mortality detection in COVID-19.
Systematic review registration:
INPLASY registration number: INPLASY202240046.
Introduction
Novel coronavirus disease (COVID-19) prevalence was first commenced in 2019 by a highly variating virus “SARS-CoV-2” (severe acute respiratory syndrome coronavirus 2) infection. The infection spread rapidly, turning into a worldwide pandemic that caused nearly 5.6 million death in the last 2 years.1-3 A number of diagnostic and treatment approaches have been approved in the fight against COVID-19, although a concrete predictor of disease progression is yet to reveal.4,5 The highly unpredictable nature of this current pandemic has made it difficult to detect the severity of the condition in time. It is crucial to establish a reliable diagnostic marker to follow the pattern of disease development and to halt the process from getting severe, even fatal. Moreover, identifying sensitive and specific biomarkers would create an opportunity to promote stronger preventive and therapeutic strategies.6-8
The key negative prognostic factor of SARS-CoV-2 infection pathophysiology is cytokine storm, a hyperinflammatory process of cytokine releasing that causes acute systemic reactions. This specific immune reactive condition drives the disease state toward acute respiratory distress syndrome (ARDS). Inflammatory cytokines, more specifically interleukins, are found to be the main mediators involved in the cytokine storm development. 9 Although rapid innate immune system reaction following SARS-CoV-2 infection is the first-line defense against COVID-19, excessively active immune reaction generates severe complications. During SARS-CoV-2 infection, irreversibly critical damage occurs in the pulmonary system and lung tissues by higher plasma concentrations of circulating interleukins.9,10 Among the multifunctional proinflammatory cytokines, interleukin-6 (IL-6) and interleukin-10 (IL-10) are suspected to be strongly involved in the COVID-19 related cytokine storm for their potential roles in acute phase immune reactions.11-13
IL-6, an inflammatory mediator with pleiotropic nature, is highly produced during the initial stage of inflammation and rapidly activates multiple acute phases of inflammatory reactants. In COVID-19 patients, IL-6 is produced in response to antigens from several immune cell types, and a number of clinical investigative studies have reported that serum level of circulating IL-6 was critically higher among the COVID-19 patients from severe to the critical stage.11,14-16 Another cross-sectional study stated evidence that serum levels of IL-6 above 24.3 pg/ml might be associated with severe pneumonia in COVID-19 patients. 17 IL-10 exerts powerful anti-inflammatory actions that control severe host immune responses toward antigens by preventing multiple functions of T-cells and neutral killer (NK) cells. Again, dysregulation in IL-10 concentration may influence the immune response and severity of SARS-CoV-2 infected patients. 18 In this context, the serum level of IL-10 also showed significant elevation in severe and critical cases of COVID-19, commensurate with IL-6 serum level. Several studies showed evidence that both IL-6 and IL-10 are positively related to the severity and mortality of COVID-19.8,19-21 According to this evidence, alteration in the normal level of circulating IL-6 and IL-10 can act as potential biomarkers for COVID-19. 13
Although some previous meta-analyses attempted to evaluate the link between circulating IL-6 and IL-10 levels with severity and mortality of COVID-19, they recommended further investigation with a larger sample size to validate their findings. For this reason, we conducted this updated systematic review and meta-analysis with available literature to reveal the correlation between IL-6 and IL-10 elevation with COVID-19 and the effectiveness of testing serum IL-6 and IL-10 levels as clinical biomarkers.
Methods
The recommendations narrated in the Cochrane Handbook 22 and PRISMA (the Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines 23 were followed to conduct this systematic review and meta-analysis. The study is also registered in INPLASY (http://inplasy.com/), and the registration number is INPLASY202240046.
Literature searching strategy
The international scientific authorized databases such as Google Scholar, PubMed, Embase, CNKI, Cochrane Library, and Web of science were used as primary sources to identify and collect the eligible literature. Additional secondary databases were also comprehensively searched to extract more related studies. The specific search terms used for this study were: “COVID-19” OR “SARS-Cov-2”; “interleukin-6 “ OR “IL-6”; and “interleukin-10” OR “IL-10.” The search strategy for each database is enlisted in Supplemental Table 1. All the included literature was selected from December 2019 to December 2021 time period.
Study selection
Two authors individually screened titles and abstracts of the studies from different databases to avoid bias and shortlisted articles with eligibility potentials. The unrelated articles were eliminated from the list after full-text inspection based on the inclusion and exclusion criteria. Any difference of opinions among the authors was resolved via a logical argument with the assistance of the third researcher. The study selection process is outlined in Figure 1.

Study flow chart representing the selection process of eligible studies.
Inclusion and exclusion criteria
Inclusion criteria: (1) clinical studies, case-control investigations or cohort studies; (2) articles representing severity and mortality in COVID-19 patients; (3) articles providing information on IL-6 and IL-10 level among mild-to-severe COVID-19 patients; (4) articles reporting IL-6 and IL-10 level in the COVID-19 survivors and deceased patients.
Critically ill patients, patients with severe dyspnea, critically low oxygen level, patients under mechanical ventilation, or admitted to the intensive care unit (ICU) were considered severe conditions of COVID-19.
Exclusion criteria: (1) meta-analysis, review articles, letters, or comments; (2) articles written in languages other than English or Chinese; (3) incomplete information required in this meta-analysis; (4) unavailability of full texts.
Quality assessment
The Newcastle-Ottawa Scale (NOS) can determine the quality range of studies by rating them from 0 to 10 stars based on some specific features (Available at http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm.). Articles that scored ⩾6 were considered high-quality ones. All the studies included in this meta-analysis were assessed with NOS for quality evaluation by 2 reviewers independently. Studies that scored less than 6 stars were excluded to maintain the quality range for the present analysis.
Data extraction
Data collection from the enlisted articles was conducted by 2 researchers. Basic information like author name, study period, study location, ethnicity, settings, study design, number of COVID-19 patients, age, mild state, severe to a critical state, number of deceased patients, IL-6, and IL-10 concentrations in patients were extracted.
Publication bias assessment
The risk of publication bias was determined by using Review Manager (RevMan 5.4) software for systematic review and meta-analysis. Egger regression test and Begg & Mazumdar test were performed to detect the presence of publication bias. Both the tests were used to verify the significance level of bias among the studies. The presence of asymmetry in the funnel plots also indicates a significant presence of publication bias.
Statistical analysis
The statistical analysis was performed by comparing the concentration level of both IL-6 and IL-10 among the COVID-19 patients according to the disease severity. Patients with mild symptoms or at the recovery stage were considered as the control population and patients with severe or critical conditions were termed as an experimental population. A secondary analysis was also conducted among the survived patients and deceased patients. The control arm showed the IL-6 and IL-10 concentrations among the survived patients, and the experimental arm showed peak concentration (last diagnostic count) levels among the dead patients. The name of the software used to carry out this meta-analysis was RevMan 5.4 from the Cochrane Collaboration, 2020. The unit of concentration measured as pg/ml. We used mean concentration with standard deviation for numerical presentation. The numerical data (mean and SD) was estimated using a validated equation. Estimation of the outcome was pooled as the mean difference with 95% CIs (confidence interval). Two analysis models were used for statistical calculation—the fixed-effects model and the random-effects model. In case of significant heterogeneity (chi-square I2 ⩾ 50 and P < .10), DerSimonian-Laird random-effects model was applied and, in the absence of the heterogeneity fixed-effects model (Mantel-Haenszel) was applied (chi-square I2 = 50 and P > .10). To evaluate the credibility of the results acquired from this study, we performed sensitivity analysis by omitting the studies one by one with the application of RevMan 5.4.
Results
Study selection
We conducted this meta-analysis on overall 31 909 COVID-19 patients from 147 studies.2,4-7,9-18,24-154 Among the recruited patients, 3137 were deceased, and the rest, 28 772 patients, showed mild to severe disease symptoms. The age range of the patients was between 6.25 ± 4.31 and 85.83 ± 7.61 years. All the studies had hospital-based settings; patients under investigation were admitted to the hospitals. No patients under self-quarantine or without hospital admission were included in the study. The recruited studies followed various designs, such as retrospective cohort, prospective cohort, observational cohort, single centered or multicentered cohort, case-control cohort, retrospective observational, prospective observational, prognostic cohort, retrospective longitudinal, non-randomized, cross-sectional observational, and clinical studies. From 147 studies, 107 studies reported IL-6 and IL-10 concentrations in COVID-19 patients and their association with disease severity. On the other hand, 49 studies reported an association of IL-6 and IL-10 serum levels with mortality in COVID-19 patients. Cytokine levels were measured using different biochemical assays—Enzyme-linked immunosorbent assay (ELISA); Electro-chemiluminescent immunoassay (ECLIA); Chemiluminescent immunoassay (CLIA); Online hemodiafiltration (OLHDF), Flow cytometry, Bio plex multiplex immunoassay, Automated immunoassay multiplex array system, and Enzyme-immune assay. The basic information is outlined in Table 1.
Abbreviations: CLIA, chemiluminescent immunoassay; ECLIA, electro-chemiluminescent immunoassay; ELISA, enzyme-linked immunosorbent assay; HB, hospital based; NA, not available; NOS, Newcastle Ottawa Scale; OLHDF, online hemodiafiltration.
Association of IL-6 with the severity and mortality of COVID-19
We assessed 107 studies to verify fluctuation in serum IL-6 concentration among COVID-19 patients in response to disease severity. Comparatively, elderly patients showed severe to critical symptoms than younger patients, according to selected studies. The mean difference in serum IL-6 level was 19.98 higher in the severe patients than in the mild category patients. IL-6 level showed significant elevation in the severe COVID-19 cases (MD: 19.98; P < .001; 95% CI: 17.56, 22.40).
To evaluate the impact of IL-6 level on the mortality of COVID-19 patients, 49 studies of 147 included studies were assessed. The result showed that deceased COVID-19 patients had 42.11 times higher mean concentration than survived patients. IL-6 level was significantly increased in the dead patients (MD: 42.11; P < .001; 95% CI: 36.86, 47.36), and the fluctuation was highly noticeable (Table 2, Figures 2 and 3).
Effect of elevated IL-6 and IL-10 levels on disease severity and mortality in COVID-19 patients.


Forest plots showing IL-6 and IL-10 levels in COVID-19 patients based on disease severity index: (a) IL-6 levels in severe and non-severe COVID-19 cases and (b) IL-10 levels in severe and non-severe COVID-19 cases.

Forest plots showing IL-6 and IL-10 levels in COVID-19 patients based on mortality index: (a) IL-6 levels in dead and survivors COVID-19 cases and (b) IL-10 levels in dead and survivors COVID-19 cases.
Association of IL-10 with the severity and mortality of COVID-19
Fifty-two studies were assessed to identify fluctuation in serum IL-10 concentration according to disease severity among COVID-19 patients. The mean difference in serum IL-10 level was 1.35 between severe and mild COVID-19 patients. IL-10 level showed significantly high concentration in the severe COVID-19 cases (MD: 1.35; P < .001; 95% CI: 0.90, 1.80).
To evaluate the impact of elevated IL-10 level on the mortality of COVID-19 patients, 12 studies of 147 included studies were assessed. The outcome showed that dead COVID-19 patients had an increased IL-10 to a mean concentration of 4.79 than survived patients. IL-10 level was significantly increased in the deceased patients (MD: 4.79; P < .001; 95% CI: 2.83, 6.75), and the fluctuation indicated that IL-10 might be associated with mortality in COVID-19 (Table 2, Figures 2 and 3).
Sensitivity analysis and publication bias
No visual asymmetry was observed during analyzing funnel plots indicating the absence of publication bias (Figure 4). Egger’s regression test showed a significant outcome in IL-6 versus COVID-19 severity model (P = .005). Other analyses did not show any significant publication bias (IL-6 vs COVID-19 mortality: P = .652; IL-10 vs COVID-19 severity: P = .091; IL-10 vs COVID-19 mortality: P = .669). Begg-Mazumdar’s test also showed similar results (IL-6 vs COVID-19 severity: P = .023; IL-6 vs COVID-19 mortality: P = .730; IL-10 vs COVID-19 severity: P = .455; IL-10 vs COVID-19 mortality: P = 1.00). The results are shown in Table 2. Sensitivity analysis was performed by excluding the studies one by one to verify the stability of the final outcome. The final outcome was stable, and none of the studies interfered with the core results (not shown).
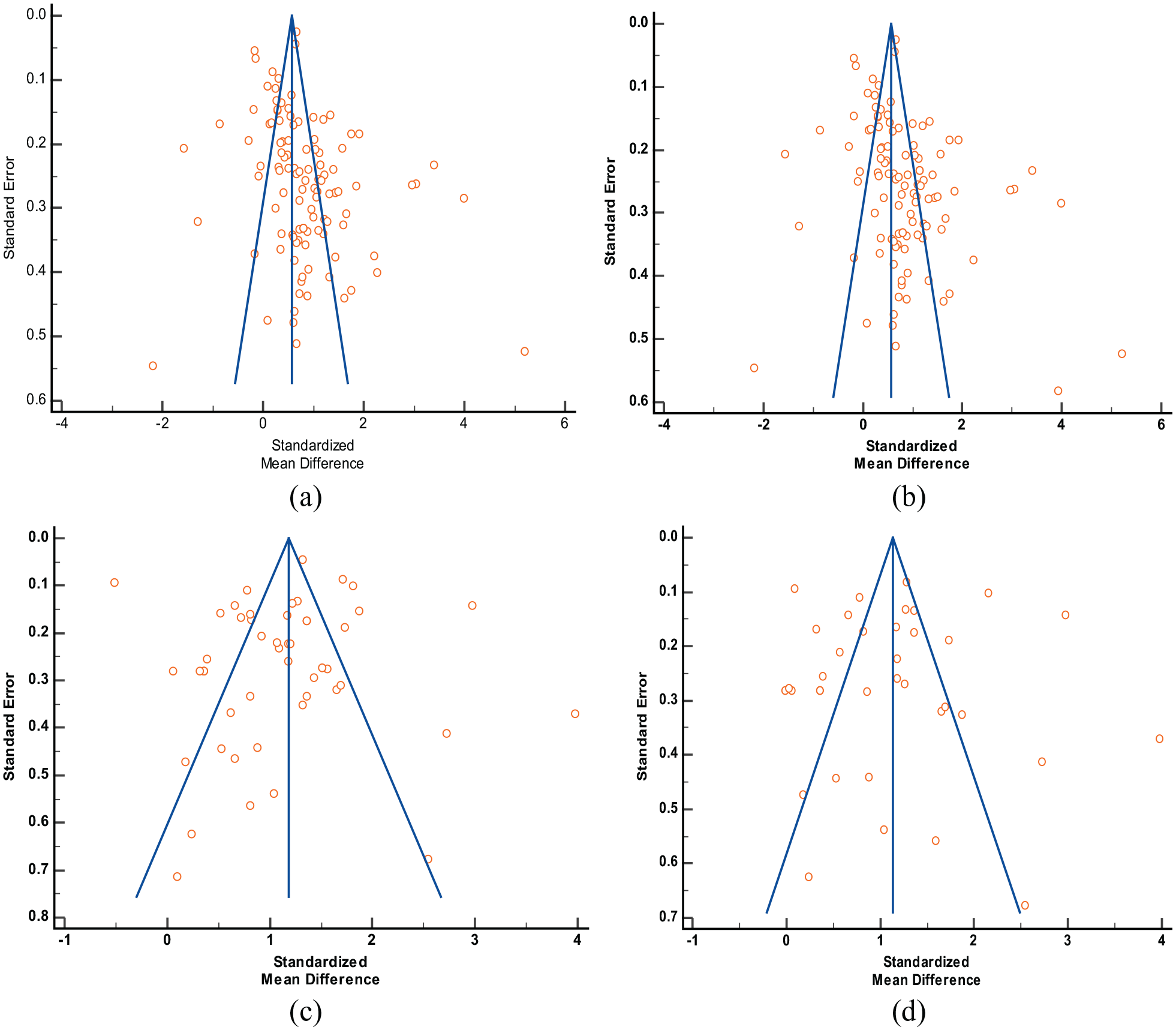
Funnel plots for publication bias analysis in different meta-analysis models: (a) IL-6 and severity of COVID-19, (b) IL-10 and severity of COVID-19, (c) IL-6 and mortality of COVID-19, and (d) IL-10 and mortality of COVID-19.
Discussion
The wave of COVID-19 is still ongoing, with full rhythm failing a number of attempts to control this pandemic situation. Most of the COVID-19 cases remain mild, and patients get their recovery from the fully active natural immune system, but 14% of patients face severe symptoms that lead to ARDS, septic shock and multiple organ failure. 13 The ultimate outcome of severe SARS-CoV-2 infection becomes life-threatening, which is undeniable. Moreover, the rate of survival is minimal in severe to critical cases. The burden of emergency COVID-19 cases is uprising drastically worldwide. SARS-CoV-2 infection has become a threat to human race, and researchers are still struggling to improve this overwhelming situation. Early detection of the severe stage of infection could cease the disease progression toward the critical stage. Checkpoint of severity will also reduce the risk of mortality from COVID-19. 8
In our study, we have accumulated a number of evidence suggesting that cytokine storms developed during SARS-CoV-2 infection intensify the damage rapidly. Elderly patients, children, or patients with a previous disease condition with a weak immunity system mostly show a severe immune reaction. Anti-inflammatory treatments could not instantly reduce the sharp elevation of cytokines in the human body. As a result, the consequences of acute tissue damage and critical lung inflammation become challenging to control. 155 IL-6 and IL-10 show a significant elevation in COVID-19 patients with mild conditions, and the concentration sharply increases manyfold when the condition gets worsens. These 2 biomarkers should be observed as a primary prognostic indicator in COVID-19 patients to understand the disease state.155,156
Efficient immune activity is essential in the fight against any infection, although overproduction and unnecessary activation of active immune cells may cause much more irreversible damage than the actual infection. In COVID-19 cases, cytokine storm in the risk population reduces the lung capacity by flooding lung surfaces with inflammatory cells. The oversensitive immune activity becomes ineffective and fills the air sacks of the lungs with fluid limiting their oxygen uptake ratio, which leads to inevitable deaths.157-159 IL-6 and IL-10 are major pleiotropic interleukins involved in potent inflammatory reactions observed in human body during any infection. Among these 2 cytokines, IL-6 helps to conduct acute phase immune reactions by recruiting immune cells in the infected area. But the excess level of IL-6 is responsible for anaphylactic shock or cytokine storm. This phenomenon will cause additional damage rather than wiping out infectious agents. On the other hand, IL-10 is responsible for maintaining homeostatic balance in the immune system by exerting anti-inflammatory actions. Human immune system can control or inhibit severe inflammation itself when the body starts healing by a homeostatic mechanism. Both IL-6 and IL-10 are closely involved in COVID-19 pathogenesis.160-162 IL-6 is one of the critical inflammatory mediators in patients severely suffering from COVID-19. The level of IL-6 is elevated in these subjects and has been considered an important choice for COVID-19 targeting. Therapeutic agents (eg, sarilumab, tocilizumab) that suppress the IL-6 signaling mechanism have been reported to be effective against COVID-19.163-165
Many studies attempted to find out immune-inflammatory predictors for disease severity in COVID-19. A recent systematic review and meta-analysis that included 19 studies with 3115 participants found that IL-6 and IL-10 level was higher in the severe COVID-19 cases than in the non-severe cases. 166 Another study performed on 24 articles with 6212 participants recommended both IL-6 and IL-10 as potential biomarkers for COVID-19 severity and mortality. 155 Bao et al 167 reported that severe patients had increased levels of IL-6 (1.93-fold) and IL-10 (1.55-fold) serum concentration in a study involving 35 articles (5912 patients). Zawawi et al 168 showed that both the interleukins are associated with the severity of COVID-19 in their recent meta-analysis. In another network meta-analysis with 71 eligible studies involving 8647 patients, a rise in the IL-6 and IL-10 count was observed with worsening of the COVID-19 infection. 169 Other studies with a limited sample size also conducted a similar assessment and reported similar findings.8,19-21 The evidence from these studies was sub-optimal and significant heterogeneity was observed due to the limited sample size. To create valid evidence that sharp elevation in IL-6 and IL-10 levels should be considered as a checkpoint of COVID-19 severity and mortality, we carried out this large-scale updated meta-analysis.
The findings from our present meta-analysis revealed that the mean IL-6 and IL-10 serum level was significantly higher in the COVID-19 patients. Severe category patients faced a sharp increase in the serum level compared to non-severe category patients. A similar result was also observed in the case of mortality. The deceased patients showed abnormally high serum concentrations of IL-6 and IL-10 than the survived patients. Moreover, the concentration of serum interleukins in the dead patients was significantly higher than the severe cases of COVID-19.
The current meta-analysis had some drawbacks that should be mentioned. Most of the included studies were retrospective cohort studies with smaller sample sizes. As the number of studies was huge, some detailed and basic information like—sex, treatment, duration of infection, smoking habit, and body mass index (BMI) could not be added to the meta-analysis. The presence of heterogeneity was another limitation of the study. The heterogeneity may be due to the different ethnic groups, sample size variation, different interventions to treat the symptoms of COVID-19 and variation in the inclusion and exclusion criteria for mild and severe groups selection. In spite of the limitations, our study is methodologically strong. According to our understanding, this is the most comprehensive and updated systematic review and meta-analysis on the association between circulating levels of IL-6 and IL-10 and the severity and mortality of COVID-19.
Conclusion
In summary, this investigative meta-analysis confirmed that sharp elevation in serum IL-6 and IL-10 worsens COVID-19 clinical outcomes. IL-6 and IL-10 are associated with the severity and mortality of COVID-19. The circulating level of both interleukins can act as potential biomarkers for the disease severity and mortality in SARS-CoV-2 infected patients.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719221106600 – Supplemental material for Elevated Levels of Pleiotropic Interleukin-6 (IL-6) and Interleukin-10 (IL-10) are Critically Involved With the Severity and Mortality of COVID-19: An Updated Longitudinal Meta-Analysis and Systematic Review on 147 Studies
Supplemental material, sj-docx-1-bmi-10.1177_11772719221106600 for Elevated Levels of Pleiotropic Interleukin-6 (IL-6) and Interleukin-10 (IL-10) are Critically Involved With the Severity and Mortality of COVID-19: An Updated Longitudinal Meta-Analysis and Systematic Review on 147 Studies by Sarah Jafrin, Md. Abdul Aziz and Mohammad Safiqul Islam in Biomarker Insights
Footnotes
Acknowledgements
This meta-analysis was supported and assisted by the Department of Pharmacy, Noakhali Science and Technology University, Sonapur, Noakhali.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MSI conceptualized this meta-analysis; SJ and MAA wrote the primary draft; MSI carried out the statistical analyses; MSI critically reviewed and revised the manuscript; Before submission, all authors read and approved the final version of the manuscript.
Data Availability Statement
All data generated or analyzed during the present meta-analysis are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

