Abstract
Introduction
During the COVID-19 crisis, several publications on the treatment of critically ill patients with multiple co-morbidities stand out. The emerging findings point to a dysregulated and “exaggerated” systemic immune response to the viremia that rapidly overwhelms tissue defenses resulting in multiple organ failures and death. 1 During this global crisis, researchers around the world have concentrated on analyzing the “inflammatory soup” common to these patients with the hope of developing specific therapies to reverse the inflammatory cascades set in motion by the virus.2,3 These include sharp increases in plasma interleukin 6 and interleukin 8 along with elevated levels of the chemokines C-C ligands 2, 3, 4, and 5. Because CCL5/RANTES (regulated upon activation, normal T-cell expressed, and secreted) seems to have a more prominent role in creating the “cytokine storm” within the pulmonary system, 4 a few researchers have hypothesized that disrupting the CCL5-CCR5 axis may have therapeutic value.5–7 Leronlimab is a humanized IgG4, kappa monoclonal antibody that blocks the expression of CCR5 with a long safety record treating patients with HIV. 8 Thus, in patients with severe COVID-19, researchers found that Leronlimab also reduced CCL5/RANTES as well plasma IL-6, while normalizing CD4/CD8 ratios.5–7
Coincident with these more recent studies, Lechner et al, have reported significant elevation of CCL5/RANTES in serum as well as from homogenized tissue biopsied from defects in the alveolar bone from tooth bearing regions of the jaw.9,10 Histopathologic features are consistent with fatty degeneration of the residual marrow and osteolysis of the surrounding trabecular bone with dystrophic changes brought on by chronic ischemia, which continue to accumulate and prevent normal bone healing. This condition is now being described as “fatty degenerative osteonecrotic jawbone” (FDOJ). [09, 10] The term aseptic ischemic osteonecrosis of the jaws (AIOJ) is used in ICD-10 to describe the hypoxic condition that persists in the cancellous bone supporting an infected wound in jawbone but essentially has failed to heal.11,12
Questions
The above associations raises the following questions: (1) Is preexisting chronic FDOJ/AIOJ jaw marrow pathology a “hidden” co-morbidity affecting outcomes of some COVID-19 infections as demonstrated in patients with other inflammatory diseases? (2) To what extent does the chronic CCL5/RANTES expression from preexisting ischemic inflammatory areas within the jaws contribute to the development and progression of the acute life-threatening cytokine storm common in COVID-19 infected patients? (3) Has the immunological memory of functional T cells, macrophages, and other cells activated by acute, then persistent inflammation within the jaw’s alveolar bone been pre-sensitized by CCL5/RANTES thus giving the immune system a head start in mounting an exaggerated inflammatory response when confronted with the COVID-19 SARS-2 virus?
5
(4) Do these jaw marrow lesions act as reservoirs of CCL5/RANTES, CCL2 and other inflammatory mediators, thereby contributing to the cytokine storm.
Material and methods
CCL5/RANTES plasma expression in COVID-19 cohorts using CCR5 blocker Leronlimab
As of November 2020, late-stage clinical studies for combating the cytokine storm in severe cases of COVID-19 were in progress and emergency use authorizations granted by the U.S. Federal Drug Administration (FDA). At the time, only Leronlimab targets the CCR5 receptor. They were designed, like Leronlimab, with hopes it would interrupt the intense proliferation of the dysregulated inflammatory phase of the viremia - the so-called “cytokine storm”.3,4 Researchers observed, by blocking the CCR5, the therapeutic monoclonal antibody Leronlimab completely occupied the CCR5 of the macrophages, platelets, endothelial and T-cells thereby contributing to a more rapid reduction of the uncontrolled inflammation and restoration of immunological weakness while reducing the plasma viral load of CCL5/RANTES.5–7 (a) Patterson BK et al, describe CCL5/RANTES expression in patients with mild to moderate symptoms as +/− 5000 pg/mL while the critically ill group (n = 10) averaged +/− 90,000 pg/mL.
5
(b) Agressti N, et al, describe CCL5/RANTES expression in plasma (n = 4) with RANTES peak values diminishing from 2131, 2328, 2122, 2586 pg/mL down to 410, 177, 2122, 1227 pg/mL for each patient after treatment with CCR5 inhibitor Leronlimab while control values were
What is ischemic/inflammatory jaw marrow pathology: FDOJ/AIOJ?
Trophic disorders of bone and particularly the jaw bone are subject to different definitions and classifications,
13
often based on multiple etiologic risk factors.14,15 Chronification of the acute phase of apical periodontitis follows as the already injured alveolar bone and supporting nerve fibers are unable institute repair processes without resolving inflammation, all while still in the hypoxic environment originally created by the acute infection.16–21 The chronic interplay of inflammatory mediators within this alveolar bone pathology is driven by several risk factors which compromise the microcirculation of these tissues: (1) the hypoxic environment brought on by the failure of effective revascularization of the affected zone of bone22–25; (2) the inborn persistency of the adaptive immune system’s mononuclear memory cells to rid the degenerative area of remnants of bacteria or virus, so called pathologic associated molecular patterns (PAMPS) and necrotic cellular debris of bone, fat, nerve and inflammatory cells, so called damage associated molecular patterns (DAMPS).26,27; (3) the presence of CCL5/RANTES which can activate platelets locally triggering the coagulation cascade and thrombosis.28,29 (4) proliferation of eosinophils and mast cells which secrete a variety of chemokines, including CCL2 and CCL5, which impact coagulation and disrupt bone healing.
30
All of these factors keep chronic inflammatory and osteolytic processes active frustrating the body’s innate regenerative capabilities to heal the compromised tissues 30 especially in the presence of heritable thrombophilic risk factors.28–30 This gradual loss of trabecular bone can lead to the development of cavities within the alveolus.31,32 Intravascular coagulation with thrombosis plays a significant role in thwarting the body’s attempt to heal these tissues.33–37 The contents of these cavitations can transform over the years and be filled with what appears clinically as normal adipose tissue but microscopic examination reveals evidence of inflammation and trophic pathology. Consequently, it has been named fatty degeneration of osteonecrosis in jaw, or FDOJ or, aseptic ischemic osteonecrosis of jawbone, AIOJ. 38 These pathologically formed alveolar bone cavitations may take years to form and may remain asymptomatic for a lifetime. Reports range from painful “bone marrow edema” in the early stages of its pathogenesis to large, empty, asymptomatic focal osteoporotic “bone marrow defects in jawbone” (BMDJ) in the late stages, which may or may not be painful depending on the amount of neurodegeneration associated with the nutrient alveolar artery.39–43
Exceedingly high levels of CCL5/RANTES in FDOJ/AIOJ lesions
FDOJ/AIOJ has been shown to be impacted by the overexpression of certain inflammatory mediators, including cytokines and chemokines, which lead to a softening of the bone. [9, 10] Due to the fatty-degenerative softening of the bony substance, the enlarged, coalesced marrow space can be easily curetted.31,44 The sizes of these lesions have been well documented by previous publications over the last years.
12
The histopathology and neurological effects of FDOJ/AIOJ were defined by Bouquot et al.
44
Figure 1(a) shows an FDOJ/AIOJ specimen that exhibits a predominantly fatty transformation of the jawbone. Figure 1(b) shows the corresponding radiograph which shows an apparently unaltered area of the upper left jawbone.
45
Thrombosis indicated ischemia and hypoxia result in adipocytes and fibroblasts degenerating into microscopic bubbles of coalesced, liquefied fat (oil cysts).14,31 Figure 1(b) is a photomicrograph of typical FDOJ/AIOJ lesion: Left panel (1a): FDOJ/AIOJ specimen that exhibits a predominantly fatty transformation of the alveolar bone obtained from tooth site 27, 28 (#15, 16). Ceramic drill with diameter of 0,9 mm demonstrates the extent of the fatty lesions. Right panel (1b): The corresponding 2-D intraoral radiograph shows an apparently normal unaltered area of the upper left posterior jawbone (red circle).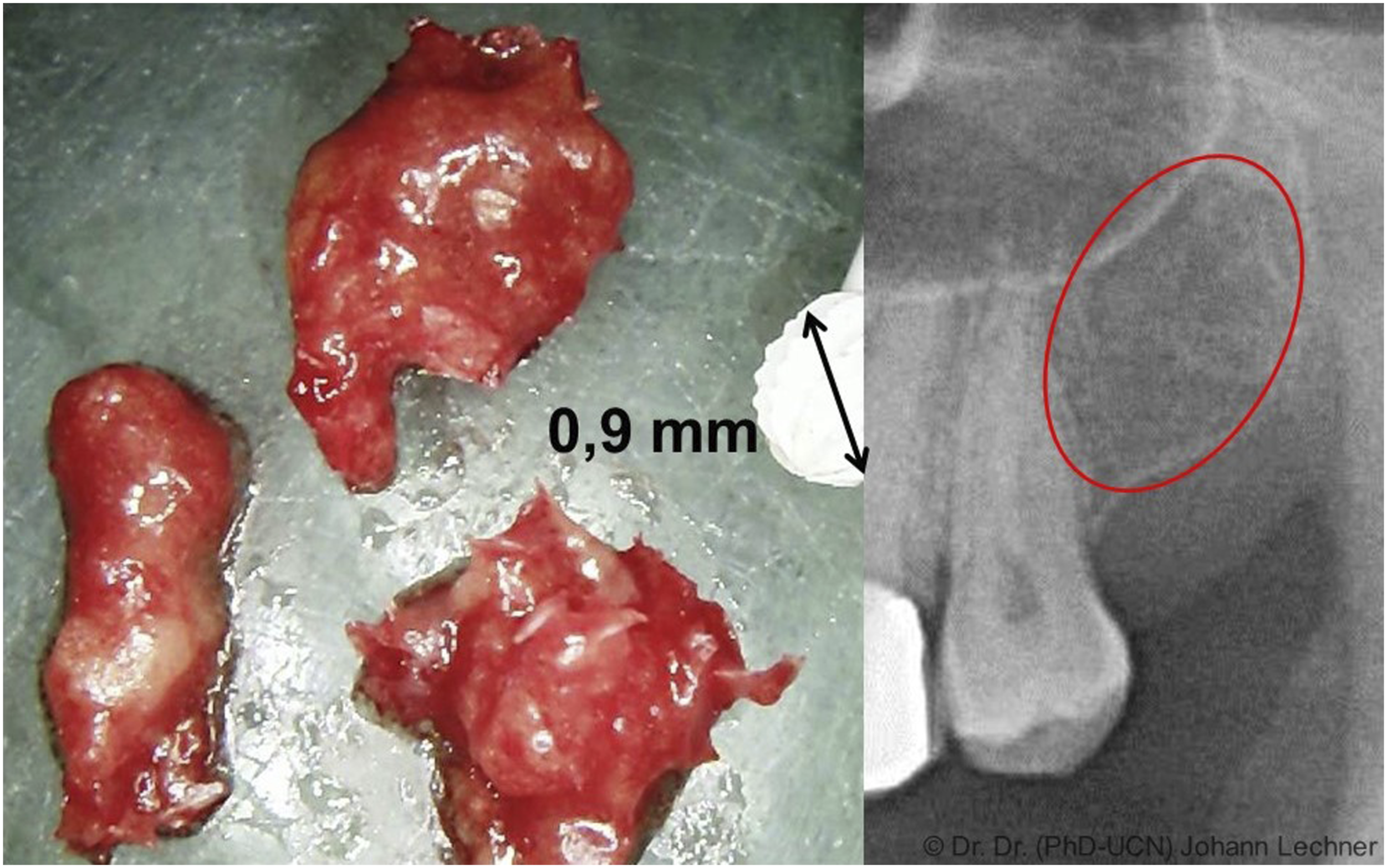
Of great interest, is the discovery of exceedingly high levels of the chemokine CCL5/RANTES (regulated upon activation, normal T-cell expressed, and secreted), fibroblast growth factor 2 (FGF-2), interleukin 1 ra (IL 1 ra) and CCL2 monocyte chemoattractant protein 1 (MCP-1) in homogenized samples of these lesions in apparently healthy and medically compromised controls.38,44 These cytokines are well known and important immune system messengers involved in a great many important physiological functions as well as healing of chronic pathological conditions.46–48 However, the possible systemic effects and the status of cytokine-triggered immune activation in samples of FDOJ/AIOJ have not been widely assessed. 10
Study participants in FDOJ/AIOJ group
The data were collected as part of the normal everyday medical care of the patients and evaluated retrospectively. Written informed consent was obtained from the patients for their anonymized information to be published in this article. The clinical case studies we present here were realized as part of a case-control study and had a retrospective character. The study was in accordance with the Declaration of Helsinki. Ethical approval to report this case series was obtained from the IMD Forensic Accredited Institute (DIN EN 15189/DIN EN 17025) located in Berlin, Germany. All participants provided informed consent to have their medical records reviewed and used in this study.
The aim of the study selection was to visualise the widespread distribution of chronic CCL5/RANTES overexpression from FDOJ/AIOJ areas, as this immunological preload triggers potential immunological overreactions in Covid-19 infections. Selection was random and based on the following criteria a) Reliably confirmed presence of FDOJ/AIOJ lesions in each of the 249 patients; b) Dental surgical evacuation of the greasy, softened FDOJ/AIOJ areas with the suspected CCL5/RANTES accumulations; c) Multiplex analysis of the FDOJ/AIOJ samples in the laboratory for individual documentation of CCL5/RANTES overexpression; d) Subdivision of the patient collective into seven immunologically derailed patient groups in order to use the known susceptibility of this patient group to Covid-19 infections to confirm our working hypothesis. Figure 2 shows the flow diagram of the cohort study. The flow diagram of the cohort study.
Since current Covid-19 research shows that infected individuals with pre-existing immunological conditions show more dramatic courses of virus infection, we chose a chronically burdened comparison group.9,10 All patients were referred by a medical specialist for a dental evaluation to search for any asymptomatic FDOJ/AIOJ chronic inflammatory conditions which may incite the immune system and thereby contribute to the persistence of specific health symptoms. The here demonstrated FDOJ/AIOJ study cohort of 249 patients comprised the following seven disease groups, as diagnosed by a specialist: 1) Autoimmune diseases (including food allergies, chronic rhinitis, and weakened immune systems; n = 33); 2) atypical facial and “idiopathic” trigeminal pain (n = 44); 3) neurodegenerative diseases (including migraine, tinnitus, multiple sclerosis, and amyotrophic lateral sclerosis; n = 29); 4) tumors (breast, prostate, pancreatic, and colon cancers; n = 15); 5) rheumatism (fibromyalgia and Lyme diseases; n = 39); 6) chronic fatigue syndrome (n = 56); and 7) parasympathetic disorders (i.e., disorders related to blood pressure, dizziness, and anxiety; n = 33). The patients’ mean age was 54.05 years (range: 23-75 years). The ratio of women to men was 89:225. All data were collected before routine oral surgery. All patients sought to determine the cause of their respective immune system disorders, which may have taken on the form of a “silent inflammation,” possibly induced by FDOJ/AIOJ. Following the clinically necessary excision of FDOJ/AIOJ, each tissue specimen was divided: half the sample was analyzed to determine the tissue levels of chemokine CCL5/RANTES content by Multiplex analysis.9,10
The data material derived from the FDOJ/AIOJ cohort is divided into ■ CCL5/RANTES expression in serum (in pg/mL) and ■ CCL5/RANTES expression found in homogenized tissue from conspicuously changed FDOJ/AIOJ areas (in pg/mL) (see Figure 3).
Results
Results of CCL5/RANTES expression in serum in systemic diseased patients
In line with our research questions, in 249 FDOJ/AIOJ patients in a group with seven various systemic diseases we found an average serum CCL5/RANTES value of 49.130 pg/mL, with a cut-off value of <30.000 pg/ml in serum in healthy individuals (Figure 3), as previously reported.
13
Green columns show CCL5/RANTES in serum for each disease group. Red column showas mean value of all seven disease groups. Note: The graph shows CCL5/RANTES expression in serum and not in plasma. Please refer to section 4.3 to see the corresponding equitation to CCL5/RANTES in plasma.
Results of CCL5/RANTES expression in FDOJ/AIOJ areas compared to healthy jawbone
To demonstrate the extent and intensity of chronic CCL5/RANTES expression from inflammatory FDOJ/AIOJ cavitations, we collected the bone marrow samples in each of the seven disease groups after jawbone surgery. We measured the specific sample CCL5/RANTES expression through Multiplex analysis. Figure 4 shows the results of the CCL5/RANTES expression in medium values in pg/mL for each disease group. The normal value from healthy jaw bone cancellous bone defined by this research group was 149.9 pg/mL.40,44,47 Medium CCL5/RANTES expression of 3.029 pg/mL from bone marrow defects in FDOJ/AIOJ cohort (Note: red bars). The normal value from healthy alveolar bone cancellous bone defined by this research group was 149.9 pg/mL (Note: green horizontal line). Figures above blue columns (n = 249) show numbers of FDOJ/AIOJ specimen in each disease group.
Comparison of CCL5/RANTES expression in the FDOJ/AIOJ group and COVID-19 patients
(a) In systemic immunologically affected patients of our cohort (see section 4.1) we could demonstrate a 1.5 to approximately 2-fold overexpression of CCL5/RANTES in serum. (b) In FDOJ/AIOJ pre-damaged jawbone marrow, we find a chronic 30-fold overexpression of CCL5/RANTES in the same patient cohort (see section 4.2). (c) Patterson et al.
5
find 500 pg/mL CCL5/RANTES in the plasma of healthy controls and up to 90,000 pg/mL CCL5/RANTES in the plasma of the severely COVID-19 affected group. Thus, up to 180-fold CCL5/RANTES overexpression is present in the plasma of those severely COVID-19 challenged in this study.
5
(d) Agressti N, et al.
6
also find 500 pg/mL CCL5/RANTES in the plasma of the healthy control and up to 2586 pg/mL CCL5/RANTES in the severely COVID-19 affected group. Thus, up to 5-fold CCL5/RANTES overexpression is present in the plasma of those severely COVID-19 challenged in this study.
6
The comparison of the two groups ■ FDOJ/AIOJ group: CCL5/RANTES in serum and in FDOJ/AIOJ samples. ■ COVID-19 group: Patterson and Arrest measure CCL5/RANTES in plasma.
is complicated by the fact that the FDOJ/AIOJ group measures CCL5/RANTES in serum with a norm level at <30 ng/mL, while Patterson and Agressti measure CCL5/RANTES in plasma with a norm level at 500 pg/mL. How do we resolve this issue: Why is CCL5/RANTES expression in serum about 60 fold higher in healthy individuals as in plasma?
As shown in Figure 5, the test kit describes these dimensional differences: A CCL5/RANTES expression in serum of 57.270 pg/ml correlates with CCL5/RANTES expression in plasma with approximately 1000 pg/ml. The coagulation process has been completed in the plasma, and there are no platelets left. Platelets always “store” such chemokines. They are like a sponge. We think it makes more sense to record the serum value in our research. We want to record the “sponge” in the screening because we are not interested in a snapshot of what is currently effective, but rather in the history of the last 1-2 weeks (= platelet lifetime) for the detection of inflammation in FDOJ/AIOJ areas. However, experience has shown that the two values correlate pretty well. The package insert of the test kit describes these dimensional differences out: Serum: 57.270 pg/mL Plasma: approx. 1.000 pg/mL.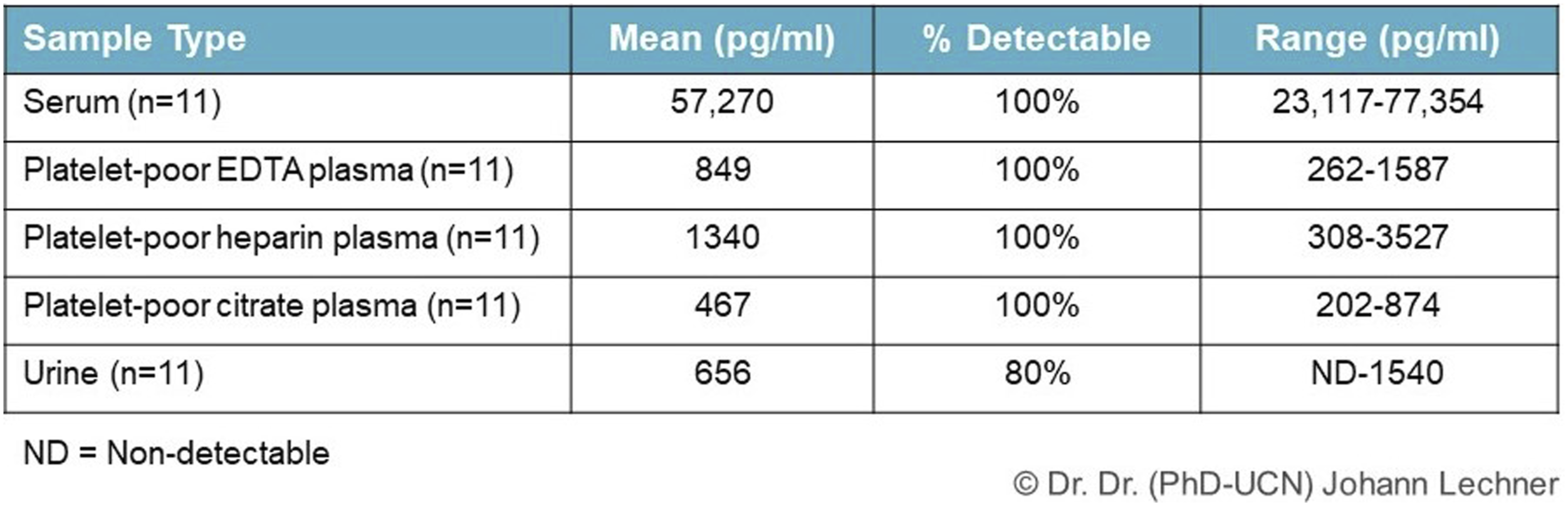
Following the description in the test kit and for proper comparison of healthy CCL5/RANTES expression in the FDOJ/AIOJ group in serum and in the jawbone and in COVID-19 patients in two research papers in plasma, all values were converted to CCL5/RANTES concentration in plasma (= serum values./: 60). Results are shown in Figure 6. Blue columns: Comparison of non Covid-19 CCL5/RANTES expression in the FDOJ/AIOJ group in plasma and in jawbone (Lechner/von Baehr). Red columns: CCL5/RANTES expression converted from serum to plasma according to test kit) and in COVID-19 patients in research paper (Patterson et al.). Green columns: CCL5/RANTES expression in COVID-19 patients in plasma in research paper (Agressti et al.). X-fold columns show the x-fold overexpression of CCL5/RANTES in plasma in FDOJ/AIOJ cohort and in COVID-19 patients.
Discussion
Overview of CCL5/RANTES overexpression
If in each of the seven disease groups mentioned the mean value of 3029 pg/mL of CCL5/RANTES expression in FDOJ/AIOJ areas is up to 25 times the healthy limit of 149,5 pg/mL and at the same time in the sum of these patients the serum level of CCL5/RANTES is significantly increased, we can assume that the chronic CCL5/RANTES signaling pathway from the bone marrow defects is represented by increased serum levels. This conclusion compared with the CCL5/RANTES expressions in severely diseased COVID-19 patients at 49.130 pg/mL 5 shows that the FDOJ/AIOJ collective could also be characterized by a “cytokine storm”, but of course more on a chronic subliminal pathogenesis than on an acute one. Preexisting ischemic and inflamed FDOJ/AIOJ pathology would be of little concern to the patient and the treating physicians while not suffering the acute and subacute manifestations common to contracting the COVID-19-SARS-2 virus.
Comparison of chronic and acute activation of CCL5-CCR5 axis by CCL5/RANTES overexpression
Our presented paper tries to elucidate the possible affection of COVID-19 infections of preexisting ischemic jaw marrow pathology as a “hidden” co-morbidity. Thus, when other investigators find a clear association between elevated serum CCL5/RANTES levels and dramatic courses of COVID-19 disease,5,6 it is reasonable to conclude that chronic CCL5/RANTES induction from FDOJ/AIOJ areas represent a possible co-morbidity of dramatic COVID-19 courses.
In Figure 6, we compare in an overview the fundamental difference of the chronic interference of CCL5/RANTES overexpression in FDOJ/AIOJ areas - as shown in the cohort of 249 patients comprised by seven different immune diseases in section 3.1 – with the critical role of an over-activated CCL5-CCR5 axis in the outcome of COVID-19 infection. Interestingly the final outcome of diminishing CCL5/RANTES signaling pathways results in both aspects in a possible rehabilitation of chronic immune diseases and a possible life-saving effect in severe acute COVID-19 infections. Figure 7 shows the comparison of chronic versus acute activation of CCL5-CCR5 axis as graphical abstract. Graphical Abstract of comparison of chronic and acute activation of CCL5-CCR5 axis by CCL5/RANTES overexpression.
Working hypothesis on the relationship between FDOJ/AIOJ comorbidity and cytokine storm
Many adult tissue samples found to have the highest expressions of CCR5 include the jaw’s lymph drainage to tonsils and lymph nodes, its immune cell interactions within the spleen and hematopoietic tissue of the bone marrow.37,47 Mesenchymal stem cells and progenitor cells within these tissues that receive infected interstitial fluids chronically burdened with DAMPS and PAMPS from recurrent dental, sinus, or oral infections would already have been “primed” to deliver immediate responses after their continued recirculation through the spleen, liver, lungs, bronchi, and bone marrow.26,27 FDOJ/AIOJ lesions represent “end-stages” of unresolved chronic inflammatory processes within diseased alveolar bone. However, when healing mechanisms are persistently violated by macrophage dysfunction only fibroblasts and fat cells can survive the hypoxic environment of these diseased zones of the jawbone (FDOJ). Many of these oxygen starved fat cells will continue to die off, creating further immune system interactions with DAMPS that proceed to the liquefied necrosis and the conversion of the alveolar lesions.9,10
The above data bring the authors to the working hypothesis – and close to the answer of the initial question – that preexisting ischemic jaw marrow pathology might be a hidden co-morbidity affecting outcomes of COVID-19 infections. Our studies of chronic disease patterns and the possible associations of CCL5/RANTES sources of inflammation from the FDOJ/AIOJ areas suggest the hypothesis that the pre-sensitization discussed above may originate from these CCL5/RANTES sources. In Figures 8 and 9 we juxtapose the schematic course of chronic CCL5/RANTES signaling versus acute COVID-19 infection preceded by CCL5/RANTES pre-sensitization. Chronic local CCL5/RANTES expression with accompanying asymptomatic disease. Exceeding the threshold for the induction provokes the start of maladaptation and leads to a specific clinical picture. Chronic CCL5/RANTES expression leads to CCR5 pre-sensitization of the immune system. Acute COVID-19 infection induces the acute CCL5/RANTES signal induced COVID-19 cytokine storm.

A monoclonal CCR5 inhibitor Leronlimab is apparently suitable for attenuating the COVID 19-related currently triggered cytokine storm.4–7 As with any disease, a distinction must be made between the currently triggered infectious event and a chronic pre-sensitizing background. With the assumption that the current COVID-19 pandemic is not the last occurring viral infection wave, the linkage of chronic subthreshold CCL5/RANTES expressions to acute CCL5/RANTES induced cytokine storms discussed above4–7 may be the possibility of preventive mitigation from costly and potentially lethal viral infections.
The limitations of the study lie in the fact that this study represents a hypothesis only. Direct correlations from FDOJ/AIOJ surgery to Covid-19 in a trial were not included in this study. The mentioned immunological correlations require further interdisciplinary clarification.
Why are the CCL5/RANTES sources in jaw marrow extraction sites so little known?
As stated earlier, conventional intraoral dental radiography presents major problems with identifying the location of a FDOJ/AIOJ lesions because they are not easily detected, often considered “invisible” and standard radiographs were often reported as “normal”.40,49–51 So a significant problem is that jawbone with FDOJ/AIOJ bone marrow defects often appears without abnormal findings in x-rays.
52
Undetected FDOJ/AIOJ areas may require diagnostic anesthesia testing,
15
3-D scans, or trans-alveolar ultrasonography (TAU) to rule out such a lesion.40,49–51 Recent research demonstrates that using innovative trans-alveolar ultrasonography pulses, a newly developed TAU device provides reliable bone density data by using ultrasound pulses.50,51 This pulse is generated by an extraoral transmitter and detected and measured by a receiving unit positioned intraorally. The results are displayed on a color screen, which shows different colors depending on the degree of attenuation of the ultrasound pulse (see Figure 10, left and right panel). For the simple detection of FDOJ/AIOJ areas, the reliable, unstressed, and easy-to-use TAU device is science based validated and available. Left panel: TAU device for intraoral use with extraoral ultrasound sender and enoral receiver with 91 piezoelectric sensors. Right panel: Piezoelectric sensors of the TAU device display high and healthy jawbone density in green and diminished bone density in FDOJ/AIOJ areas in red. Figure Indicators: Red circle in radiography marks inflammatory areas of alveolar bone.
Linking neglected CCL5/RANTES overexpression in FDOJ/AIOJ to Covid-19 cytokine storm
Noninvasive, non-radiation producing and sophisticated dental imaging studies are available to rule out these pathological bone cavities within the jaws.51,52 Therefore, the therapeutic conclusion for the practice could be that a systematic pre-screening for FDOJ/AIOJ areas, preferably with the harmless and non-ultrasonic method by TAU devices,51,52 could dampen dramatic peak exposures of the next viral pandemic.
Application of CCR5-blocking antibody Leronlimab often leads to attenuation of the acute COVID-19 cytokine storm via inhibition of the over-activated CCL5-CCR5 axis.4–7 Bone marrow defects in the jaw are a source of chronic CCL5/RANTES overexpression, which may down-regulate the threshold for excessive immune reactions as seen in COVID-19 cytokine storms. Smoldering inflammations in the jawbone may lead to more unfavorable outcomes in the patients contracting the COVID-19 virus. Our data and literature comparisons indicate for the first time that FDOJ/AIOJ may be a risk factor for COVID-19 by promoting local and systemic increases in the proinflammatory cytokine CCL5/RANTES, thus playing an often fatal independent role in triggering a cytokine storm. The above correlations demonstrate an important systemic link between local CCL5/RANTES overexpression in the jawbone and possible immune system derailments by an over-activated CCL5-CCR5 axis. The interruption of the CCL5/RANTES-CCR5 cytokine cascades in dreaded cytokine storms in severe COVID-19 infections may be life-saving.4–6
Conclusion
One way to limit a pandemic is not only to fight the virus but to intercept the cytokine storm and the harmful reaction before it spreads. Future clinical trials should not only be designed to evaluate the efficacy of Leronlimab-mediated inhibition of CCL5/CCR5 axis for COVID-19 in treating the acute phases of the illness. A possible prophylactic focus for future viral-related pandemics may be early surgical clean-up of CCL5/RANTES expression sources in the FDOJ/AIOJ areas. Locating these jaw lesions would not be possible in hospitalized patients suffering from serious life-threatening symptoms common in COVID-19 viremia. However, non-invasive, non-radiation trans-alveolar ultrasonography (TAU) studies could begin on COVID patients who did not require hospitalization, or those recently discharged. Also, COVID-19 patients suffering from “long-haul” COVID symptoms who have not responded to traditional rehabilitation methods may benefit from a more complete dental examination to include trans-alveolar ultrasonography (TAU) to rule out hidden FDOJ/AIOJ lesions which may still be active in spilling CCL5, DAMPS and PAMPS into the general circulation.
The authors propose a new approach to containment of the COVID-19 cytokine storm that considers the antecedent and immunological conditions. Identifying the discussed cavitational jawbone co-morbidities and treating them on a preventive aspect could decrease the morbidity associated with COVID-19 and reduce the cost of treatment. Both the immune and nervous system have been sensitized by the CCL5/RANTES overexpression of the previous yet unknown and undetected FDOJ/AIOJ inflammations.
Footnotes
Acknowledgements
We acknowledge the support of the laboratory team of IMD-Berlin and the authors would like to thank all patients taking part in this study.
Author’s contribution
Conceptualization; JE.B., J.L., and RE.M. Methodology: RE.M., J.L. and JE.B. Formal analysis: F.N., and F.Sch. Investigation: J.L., F.N., and F.Sch. Data curation: F.N., and F.Sch. Writing: J.L., and RE.M. Project administration: RE.M., and J.L. RE.M. and J.L. contributed equally to this work. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. J. Lechner is the holder of a patent used in the new TAU apparatus CaviTAU® as it is introduced to the market.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
