Abstract
Interleukin-10 (IL-10) is a key anti-inflammatory cytokine, and it is associated with the pathogenesis of inflammatory bowel disease (IBD). However, the relationship between serum IL-10 level and IBD remains controversial. In this study, a meta-analysis was performed using STATA 12.0 software. Articles were gathered by searching PubMed, Web of Science, Embase, and the Chinese Biomedical Database. Relevant studies were examined to identify their eligibility. Finally, eight studies met the inclusion criteria; these studies consisted of 211 patients diagnosed with ulcerative colitis (UC), 134 patients diagnosed with Crohn’s disease (
Keywords
Introduction
Inflammatory diseases (IBD), containing ulcerative colitis (UC) and Crohn’s disease (CD), are chronic inflammatory intestinal diseases. The chronically relapsing characteristics of IBD irreversibly damage the structure and functions of the intestine. IBD is a global disease, prevalent in Europe (UC, 505 per 100,000 persons; CD, 322 per 100,000 persons) and North America (UC, 249 per 100,000 persons; CD, 319 per 100,000 persons). The incidence of IBD in Asia continues to increase. 1
The pathogenesis of IBD is not completely elucidated. Nonetheless, interactions among genetic susceptibility, environmental triggers, and dysregulated immune response are believed to contribute to the initiation and progression of IBD. Cytokines are key modulators in the intestinal immune system. An imbalance of pro- and anti-inflammatory cytokines facilitates the disorder of the intestinal immune system. Interleukin-10 (IL-10) is an anti-inflammatory cytokine secreted by CD4+ T helper 2 (Th2) lymphocytes. IL-10 inhibits the secretion of various pro-inflammatory cytokines, such as IL-1, IL-6, tumor necrosis factor (TNF)-α, and IL-12. IL-10 also regulates innate and adaptive immune responses. Hence, IL-10 is a critical mediator in maintaining intestinal immune homeostasis, but its contribution to IBD is not fully understood. IL-10 knockout mice are well established as genetic models for enterocolitis. 2 Loss-of-function mutations in the IL-10 receptor can occur in children with severe and early-onset enterocolitis. 3 IL-10 plays an important role in the pathogenesis of IBD.
Serum IL-10 level is vital in immune regulation. The level of IL-10 in the blood was measured to explore its association with IBD. However, characterization of serum IL-10 level in IBD patients provided inconsistent results. Therefore, in this study, the serum IL-10 level of IBD patients was evaluated to elucidate the relationship between IL-10 and IBD.
Materials and methods
Data sources
Studies that investigated cytokines in IBD (including UC and CD) were systematically searched in PubMed, Web of Science, Embase, and the Chinese Biomedical Database in July 2016. The primary search strategy was “(‘interleukin 10’ OR ‘interleukin-10’ OR ‘interleukin10’ OR ‘IL10’ OR ‘IL 10’ OR ‘IL-10’) AND (‘inflammatory bowel disease’ OR IBD OR ‘ulcerative colitis’ OR UC OR ‘Crohn’s disease’ OR CD).”
Study selection and quality assessment
Articles were included if all the following criteria were met: (1) the study design is a case–control study, (2) all patients meet the diagnostic criteria of UC or CD, and (3) the study should offer quantitative results about the serum IL-10 level. Potential duplicates or overlapping data were excluded. Exclusion criteria were as follows: (1) studies that assessed the IL-10 level of messenger RNA (mRNA) and data from cell supernatant or intestinal mucosa; (2) patients diagnosed with autoimmune diseases (e.g. rheumatoid arthritis); (3) reviews, letters, comments, editorials, animal studies, case reports, and meeting abstracts that cannot provide sufficient data.
Two reviewers independently evaluated these studies. Abstracts were scanned first, and the full texts of potential eligible studies were reviewed subsequently. Full texts in English or Chinese were evaluated for eligibility. If any discrepancy was observed, a third investigator will join in the discussion until a consensus was reached. The Newcastle–Ottawa Scale (NOS) criteria were adopted to assess the methodological quality of included studies. An NOS score of ⩾7 indicates good quality.
Data extraction
The related information were obtained from the included studies: (1) first author’s name, (2) year of publication, (3) origin of the study population, (4) ethnicity, (5) method, (6) IL-10 levels of serum, (7) sample size, and (8) mean age.
Statistical analysis
STATA software (version 12.0; STATA Corporation, USA) was used in the meta-analysis. IL-10 levels were extracted as mean difference ± SD or median (interquartile range or IQR), and data were converted into mean difference ± SD type. Het-erogeneity was assessed by chi-square Q test and I2 statistics. Given
Results
Study selection and characteristics
A total of 11,073 potentially relevant studies were initially identified in the systematic search. However, 11,064 studies were eliminated for various reasons. Finally, eight studies4–11 were eligible for the meta-analysis. The selection process is illustrated in Figure 1. A total of 211 UC patients, 134 CD patients, and 131 controls were included in the meta-analysis. Most of the controls were healthy volunteers, and others were patients diagnosed with dysbacteriosis or diarrhea. All the studies were conducted on Caucasian populations. The most common technology applied to quantify IL-10 concentration is enzyme-linked immunosorbent assay (ELISA). Bio-Plex technology and Luminex assay were also used. Five and three studies were conducted on adults (>17 years old)4,5,7,9,11 and children (<17 years old)6,8,10 with UC, respectively. The characteristics of the studies are shown in Table 1.

Flow chart of study selection in the meta-analysis.
Characteristics of studies included in the meta-analysis.
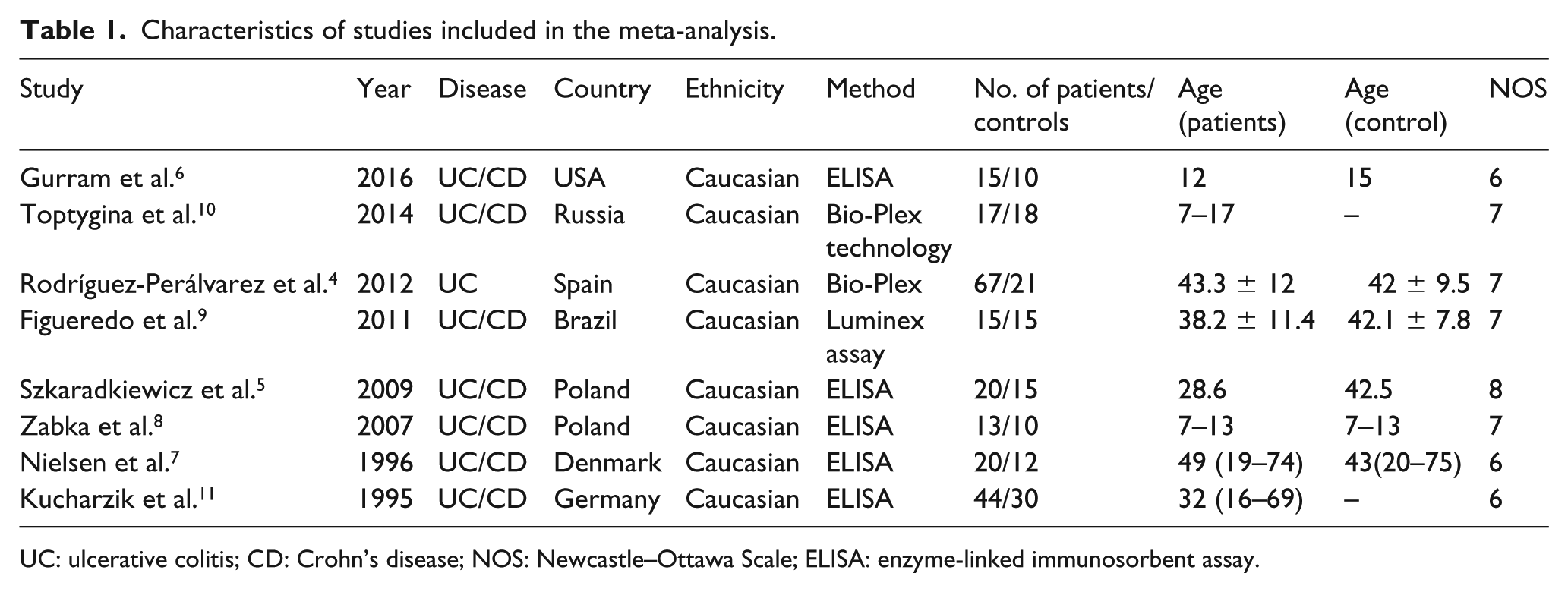
UC: ulcerative colitis; CD: Crohn’s disease; NOS: Newcastle–Ottawa Scale; ELISA: enzyme-linked immunosorbent assay.
Meta-analysis
IL-10 level and UC
Heterogeneity between studies was quantified by chi-square Q test and I2 statistics. The results showed that significant heterogeneity exists among the studies (χ2 = 27.66, I2 = 74.7%,
A moderate increase in the IL-10 level was observed in UC patients unlike that in the controls (pooled SMD = 0.55, 95% CI: 0.08 to 1.03,
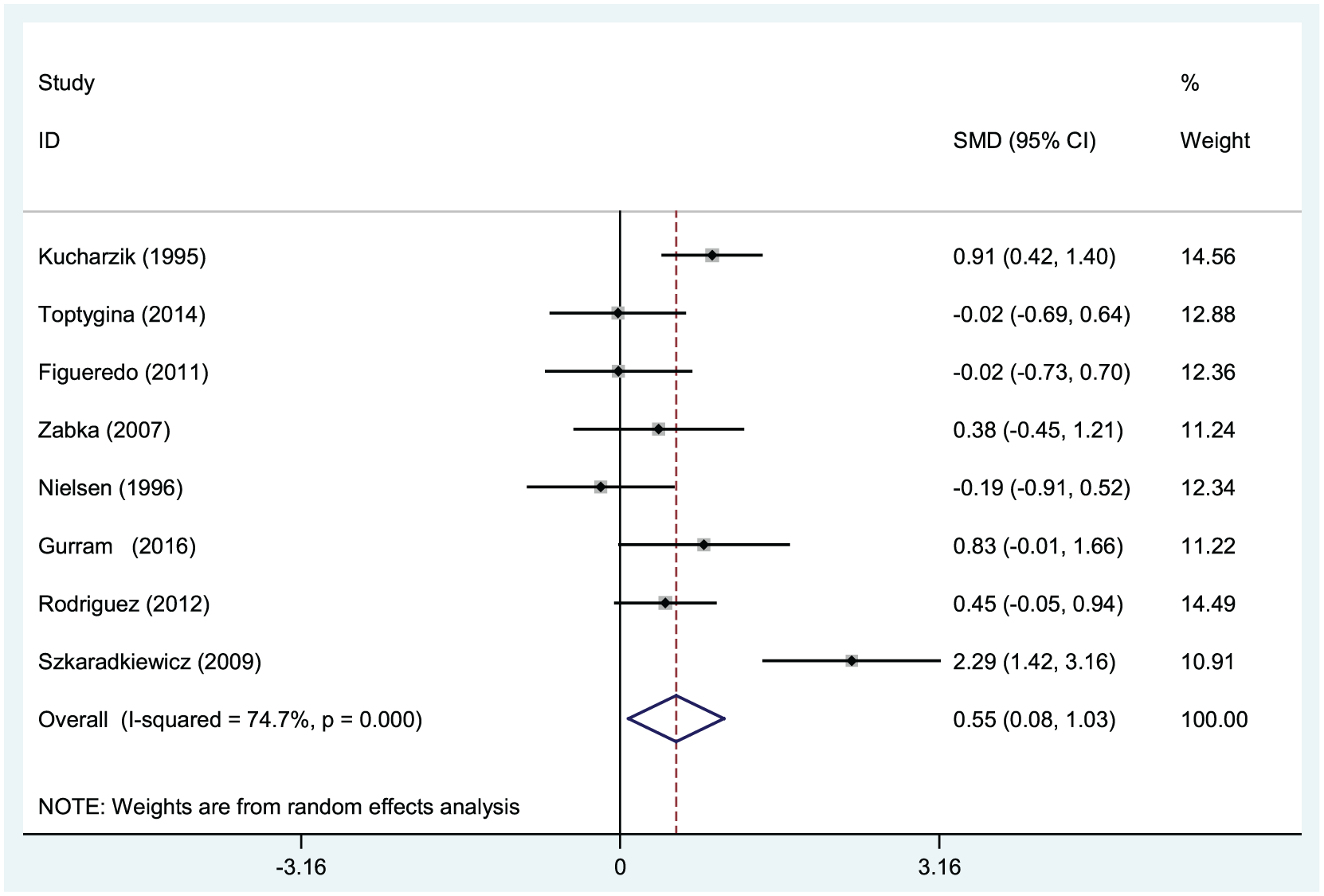
Forest plots of the correlation between serum interleukin-10 levels and ulcerative colitis.
The funnel plot was asymmetrical (Figure 3), and the more sensitive Begg’s test (

Begger’s funnel plot of publication biases.
UC and CD
The comparison of IL-10 levels between patients with UC and CD were conducted in Caucasian studies. A significant heterogeneity was detected among these six studies5–7,9–11 (χ2 = 19.65, I2 = 74.6.0%,

Forest plots of the correlation of serum interleukin-10 levels between ulcerative colitis and Crohn’s disease.
Discussion
IBD is characterized by a typical relapsing–remitting course. An imbalance of pro- and anti-inflammatory cytokine release contributes to the pathogenesis of IBD. IBD patients often experience intermittent remission and reactivation during the course of the disease, suggesting an imbalance between pro- and anti-inflammatory cytokines. IL-10 acts as a key anti-inflammatory cytokine in the human immune response. IL-10 inhibits immune responses and reduces the damage caused by inflammation. Mice deficient in IL-10 or IL-10 receptors can develop severe enterocolitis. 2 IL-10 supplementation was applied in CD animal models and humans in recent years. Recombinant human IL-10 (rhuIL-10) has been produced and used clinically. IL-10 treatment decreased the CD activity which was measured by the CD activity index. 12 Thus, exploration of the characteristics of serum IL-10 levels in IBD patients may be significant to improve IBD treatment.
In the meta-analysis, a moderate increase in IL-10 level was observed in UC patients unlike the control group. A genetic research which includes 159 pediatric inflammatory IBD patients also suggests that IL-10 gene and IL-10/STAT3 signaling pathway contribute to the risk of pediatric IBD. 13 The comparison of serum IL-10 levels between patients with UC and CD indicates that no statistical significance was observed in our study. Nearly a third of patients were diagnosed with IBD during childhood or adolescence. However, our subgroup analysis suggested that the IL-10 level was not associated with both adult (>17 years old) and PUC (<17 years) patients. Hence, differences of serum IL-10 levels and its potential mechanisms in patients at different ages remain to be studied. In terms of test method, the association between serum IL-17 level and UC did not differ as determined by ELISA. ELISA proves to be a stable and sensitive technology.
This study was the first to conduct a quantitative analysis to assess the relationship between the IL-10 serum level and IBD and explore the difference between age and test technology. However, this study presents some limitations. Selection biases and unidentified confounders are unavoidable. The studies included were restricted to be published in English and Chinese, and studies in other languages were eliminated. Significant heterogeneity was detected among the studies. A small sample size will affect the results. And the use of different test methods and the limited sensitivity of the method can cause heterogeneity.
The meta-analysis further validated that the serum IL-10 level increased in UC patients unlike in the control group, and IL-10 contributes to the pathogenesis and progression of UC patients. Therefore, serum IL-10 level may be a noninvasive biomarker for UC patients. However, the current evidence is insufficient to identify the relationship between serum IL-10 level and CD patients. High-quality and multi-center studies must be performed to validate the results.
Footnotes
Acknowledgements
The authors would like to thank Department of Gastro-enterology, People’s Hospital of Guangxi Zhuang Auton-omous Region, Nanning.
Author contributions
All authors have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by Construction of Guangxi Clinical Medical Research Center for Digestive Diseases (Project Number: AD17129027).
