Abstract
Objectives
The objectives of this study were to describe the clinical features of cats diagnosed with tularemia, and to compare the clinical features of cats with and without tularemia and identify characteristics increasing the likelihood of a tularemia diagnosis.
Methods
Diagnostic laboratory data and medical records were retrospectively reviewed of cats tested for tularemia between 2000 and 2021. Clinical features including signalment, presenting complaint(s), physical examination findings and clinicopathologic data were described for cats with tularemia diagnosed by culture and/or PCR. Clinical features were also compared between PCR+ vs PCR− cats and logistic regression was used to identify features that predicted PCR+ status.
Results
A total of 18 cats were diagnosed with tularemia. The affected cats had a mean age of 5.8 ± 4.9 years, with a slight male predominance. Most were domestic shorthair cats and had outdoor access. Common presenting complaints included lethargy and anorexia, while physical examination findings frequently revealed fever, peripheral lymphadenomegaly and oral ulcers. Laboratory findings varied, with notable abnormalities including neutrophilic inflammation observed on cytology and widespread acute systemic pathologic changes in necropsy results. Leukopenia and neutropenia were more common in PCR+ vs PCR− cats, and these factors were 7.4 times (confidence interval [CI] 1.2–62.3) and 10.0 times (CI 1.4–83.2) more likely to occur in PCR+ vs PCR− cats, respectively. There were no significant differences between the groups in other parameters including anemia, thrombocytopenia, band neutrophilia and hyperbilirubinemia.
Conclusions and relevance
This study provides a more comprehensive description of tularemia in cats than has been available to date. In particular, leukopenia and neutropenia are significantly associated with tularemia-positive cats. These clinical features should be considered and raise suspicion for tularemia in cats. These findings may be valuable for the timely diagnosis, treatment and prevention of tularemia outbreaks in cats and humans.
Keywords
Introduction
Tularemia, a zoonotic disease caused by Francisella tularensis, affects various vertebrates such as humans, rabbits, mice and domestic mammals.1 –3 Transmission occurs through several routes, including vectors (ticks and other biting insects), direct contact with the blood or tissues of infected mammals, ingestion of contaminated food or water, or inhalation of the pathogen. 1 Cats are notably susceptible to tularemia. In particular, outdoor, young adult cats stand a higher risk of encountering F tularensis because of their frequent exposure to ticks and small mammal reservoirs.
Overall, tularemia is a rare disease in North America. However, several hotspots exist for human cases, mostly in central and north-central regions of the USA, including Arkansas, Missouri, Kansas, South Dakota and Oklahoma.1,4,5 Massachusetts, particularly Martha’s Vineyard, 6 is another significant hotspot, with documented outbreaks also occurring on Long Island, New York. 2 Tularemia infections in cats have been described in case reports from Oklahoma and New Mexico.4,5,7 Tularemia in animals mirrors the distribution seen in humans, with cases reported in Kansas, 8 Montana, 9 Missouri, 10 Massachusetts, 11 central Virginia 12 and eastern Oregon. 13 Relevant to this paper, tularemia also occurs in Illinois in both people and cats. Between 2011 and 2022, 63 human cases from Illinois were reported and Illinois had the eighth highest incidence of human tularemia in the country in 2022. 14 Outside the USA, tularemia is prevalent across most of Europe and has frequently exhibited signs of local emergence and re-emergence in both humans and wildlife. 15
The disease’s severity in cats varies, with clinical findings ranging from fever, anorexia, lethargy and vomiting to marked depression, oral/lingual ulceration, regional or generalized lymphadenomegaly and hepatosplenomegaly.1,3,5 The most commonly reported laboratory findings include pancytopenia, toxic neutrophils, high band neutrophil counts, thrombocytopenia and hyperbilirubinemia.1,5 Tularemia should be suspected in febrile cats with a history of contact with wild animals or ticks. The high susceptibility of cats also poses a public health concern, given that cats can transmit this disease to people, usually through biting. Cats’ susceptibility makes them a sentinel species in tularemia-endemic areas.6,16
The diagnosis of tularemia in cats and humans starts with identification of consistent clinical findings and a history of exposure. A definitive diagnosis in both species can be made through organism isolation in culture or by demonstrating a four-fold rise in acute and convalescent titers. However, because of its highly infectious nature, the culture of F tularensis requires special precautions, and antibodies may not develop for up to 3 weeks after infection; thus, neither of these tests are appropriate for acute detection. In the clinical setting, where rapid detection is essential to appropriate management, PCR is the most common method of diagnosing tularemia. A single positive titer (⩾1:160 in cats) with consistent clinical signs also suggests active infection. 17
In humans, the preferred treatments for tularemia are streptomycin or gentamicin. However, dosage formulation and toxicity preclude their use in some patients. Doxycycline and ciprofloxacin are the recommended alternatives in these cases and are also used for post-exposure prophylaxis. A recent meta-analysis classified aminoglycosides, fluoroquinolones, tetracyclines and chloramphenicol as highly effective drug classes against tularemia, while penicillins, cephalosporins, macrolides and sulfonamides were considered minimally effective or ineffective.17,18 In cats, treatment recommendations for tularemia are limited by lack of evidence. However, gentamicin is considered the drug of choice, with doxycycline and fluoroquinolones as effective alternatives, for a duration of 2–4 weeks. 17
Most of the known clinical manifestations of feline tularemia are drawn from scattered case reports and small case series, with the majority of cases diagnosed post mortem. Such an approach does not provide a comprehensive or definitive description of the clinical picture of this disease and highlights the need for more in-depth research on tularemia in cats. Based on this rationale, this study aimed to provide insights into the clinical characteristics of feline tularemia and identify significant clinical signs that should raise suspicion for this disease in individual patients. The objectives were two-fold: (1) to describe the clinical findings of all cats diagnosed with tularemia in a large, retrospective cohort; and (2) to compare the clinical features of cats diagnosed with and without tularemia based on PCR and identify characteristics that increase the likelihood of tularemia diagnosis. We hypothesized that the descriptive findings in our study would be similar to those in previous, smaller case reports of tularemia in cats. We also hypothesized that cats with a positive tularemia PCR test would exhibit hyperbilirubinemia, lymphadenopathy and leukopenia more frequently than those with a negative PCR test. Enhanced knowledge of feline tularemia will facilitate faster diagnosis and treatment, benefiting cats and minimizing human transmission risks.
Materials and methods
Study population and data retrieval
A retrospective study of cats tested for and/or diagnosed with tularemia was conducted. The Laboratory Information Management System of the University of Illinois Veterinary Diagnostic Laboratory was searched for accessions with (1) F tularensis-positive in bacterial culture or (2) F tularensis PCR test performed between 2000 and 2021. Duplicate accessions or non-cat cases were excluded to generate a cat-only list. The medical data, including presenting complaint(s), physical examination findings, clinicopathologic data, and cytology and histopathology results, were further investigated in each case. These data were collected from the electronic medical records system of the University of Illinois Veterinary Teaching Hospital or by contacting the veterinary hospital that submitted the F tularensis culture or PCR by email or phone and requesting the medical records for 6 months before and after the diagnosis. If no medical record was available, the case was excluded from the study.
Diagnostic testing for Francisella tularensis
Before 2015, culture was the exclusive diagnostic test employed in the University of Illinois Veterinary Diagnostic Laboratory for tularemia. F tularensis culture-positive samples were handled in line with Laboratory Response Network procedures, including biosafety and destruction of select agents. F tularensis was identified based on the results of Gram stain, biochemical reactions or matrix-assisted laser desorption ionization time-of-flight mass spectrometry (Bruker).
Since 2015, PCR has been used as a diagnostic tool for tularemia. For PCR detection, DNA from tissue and EDTA blood was extracted using the Maxwell 16 Tissue and Blood DNA Purification Kits (Promega), with tissues (approximately 100 mg) homogenized before extraction using a mini-beadbeater (Biospec Products). Primer sets used included those for Francisella species (F 5’–CCT TTT TGA GTT TCG CTC C –3’, R 5’–TAC CAG TTG GAA ACG ACT GT –3’), North American strains of F tularensis (F 5’–CAA GGT TAA TAG CCT TGG GGG A –3’, R 5’–GCC TTG TCA GCG GCA GTC TTA –3’) and European strains of F tularensis (F 5’–CAA GGT TAA TAG CCT TGG GGA–3’, R 5’–CCT TGT CAG CGG CAG TCT CA–3’). 19 PCR products were amplified from the samples using a PCR mix containing 1X Qiagen Multiplex Master Mix (Qiagen), 0.4 μM each of forward and reverse primers and 1 μl of DNA template to a final reaction volume of 20 ml. Thermal cycling conditions were as follows: 95°C for 15 mins; followed by 35 cycles of 94°C for 30 s, 65°C for 90 s and 72°C for 90 s; and a final extension step of 72°C for 10 mins. The quality of the DNA template and PCR inhibition were monitored by amplification of the mitochondrial 16S gene. PCR products were electrophoresed on a 2% agarose gel and genus- and species-specific amplicons identified as 1100 base pair (bp) and 690 bp bands, respectively.
Statistical analysis
Continuous data are presented as both mean ± SD and median (range). To characterize the clinical presentation of tularemia in cats (objective 1), features of all cats diagnosed with tularemia by culture, PCR or both were described. These included age, breed, sex, neuter status, presenting complaint(s), physical examination findings, bloodwork parameters, cytology and histopathology. Treatment measures and clinical outcomes were also reported, when available.
To identify the differences in clinical presentation between cats with and without tularemia (objective 2), the features of cats tested for tularemia with a positive (PCR+) vs negative (PCR−) result were compared. The Kolmogorov–Smirnov test was used to assess normality of continuous data, including age, temperature and clinicopathologic variables. For data that were normally distributed, the t-test was employed. For data that were not normally distributed, the Mann–Whitney U-test was used. Categorical data including sex, indoor/outdoor status and the presence of anorexia, fever, oral ulceration and lymphadenomegaly were compared using Fisher’s exact test or the χ2 test, as appropriate. In addition, several clinicopathologic variables, selected a priori based on previous reports of feline tularemia, were analyzed as binary data using Fisher’s exact test (the presence of anemia, thrombocytopenia, leukopenia, neutropenia, high band neutrophil count and hyperbilirubinemia).
Univariate logistic regression analysis was performed to identify variables that predicted PCR status. Those variables with a P value <0.20 were included in a multivariate, backwards stepwise, logistic regression analysis. All statistical analyses were performed using commercially available software (SAS OnDemand for Academics, SAS Institute; Prism 10, GraphPad Software). Statistical significance was set at P <0.05.
Results
Clinical features of cats diagnosed with tularemia
A total of 18 cats were diagnosed with tularemia in this retrospective cohort study, comprising eight cats diagnosed by culture alone, nine cats diagnosed by PCR alone and one cat diagnosed by culture and PCR. The mean age of the affected cats was 5.8 ± 4.9 years (median age 5 years; range 0.2–15 years). The sex distribution was 10 (56%) males, 7 (39%) females and one (6%) cat of unknown sex. Of the study cats, 15 (83%) were neutered, one (6%) was intact and two (11%) had an unknown neuter status. Represented breeds included 16 (89%) domestic shorthair cats, one (6%) domestic mediumhair cat and one (6%) domestic longhair cat. The housing status was known for 12 (67%) cats, all of whom had some access to the outdoors.
The presenting complaints were known for 11 cats. The most common were lethargy (9/11, 81%) followed by anorexia (8/11, 72%). Physical examination findings were available for 13 cats and the most common abnormalities were fever (11/13, 86%) followed by peripheral lymphadenomegaly (8/13, 62%) and oral ulcers (8/13, 62%). The mean body temperature of tularemia-positive cats was 104 ± 3°F (40 ± 1.68°C), with a median temperature of 104.9°F/40.5°C (range 96–106.9°F, 35.6–41.6°C). Table 1 provides summary statistics of bloodwork parameters for cats diagnosed with tularemia.
Summary statistics for complete blood count and serum biochemistry parameters in cats diagnosed with tularemia

ALP = alkaline phosphatase; ALT = alanine transaminase; BUN = blood urea nitrogen; HCT = hematocrit; PLT = platelets; TBILI = total bilirubin; WBC = white blood cells
Abdominal ultrasound results were available for one case, revealing generalized splenomegaly, bilateral renomegaly, mesenteric lymphadenomegaly and scant peritoneal effusion (Figures 1 and 2). Abdominal radiographs were available for two cases: one showed segmental small intestinal distension and a small right kidney; and the other was unremarkable. Thoracic radiographs were available for six cats. One showed cardiomegaly, pulmonary nodules and a bronchial lung pattern; another showed an interstitial lung pattern; and a third displayed both bronchial and interstitial lung patterns. The remaining three cats had unremarkable thoracic radiographs.

Abdominal ultrasound scan of a tularemia-positive cat. Splenomegaly with scant peritoneal effusion was noted

Abdominal ultrasound scan of a tularemia-positive cat. An enlarged ileocolic junction lymph node was noted
Cytology results were available for four cats. All had undergone mandibular lymph node cytology and one cat also had cytology of the liver and spleen. All cytology results showed neutrophilic inflammation.
Necropsy results were available for six cats, all of whom exhibited widespread acute and systemic pathologic changes. Necrotizing inflammation was a common finding across multiple organs. Notably, all six cats exhibited necrotizing lymphadenitis and splenitis. Pulmonary involvement was significant, with 5/6 cats showing embolic pneumonia. Gastroenteritis varied across the cases, from acute necrotizing to pyogranulomatous inflammation, affecting all six cats to varying degrees.
The spleens in all six cases demonstrated varying degrees of necrotizing splenitis, from miliary to coalescing necrosis. Similarly, the mesenteric lymph nodes in all cats showed severe necrotizing lymphadenitis, often accompanied by granulomatous changes. Liver pathology, predominantly multifocal necrotizing hepatitis, was observed in five cats. Consistently, all six cats had some form of enteritis, often necrotizing and granulomatous. Renal pathology was more varied: three cats exhibited moderate lipidosis; four had mild lymphoplasmacytic interstitial nephritis; and two presented with severe membranoproliferative glomerulonephritis alongside tubular mineralization.
Of the 18 cats diagnosed with tularemia, two cats survived for extended periods after diagnosis, living for 2 and 4 years, respectively. One of these cats was treated with pradofloxacin, while the other received enrofloxacin. Three cats improved while hospitalized and were discharged but were then lost to follow-up. Of these three cats, two received pradofloxacin and one was treated with doxycycline. Eight cats were euthanized; four cats received treatment before euthanasia, while the other four did not receive any treatment. Of the four treated cats, antimicrobial regimens included ampicillin/sulbactam + clindamycin, gentamicin + enrofloxacin, amoxicillin–clavulanate and doxycycline. One cat in the study died despite treatment with enrofloxacin and doxycycline. No data were available for the remaining four cats regarding treatment or outcome.
Comparison of clinical features of PCR+ vs PCR− cats
A total of 63 cats were tested for tularemia using PCR, with 10 cats being PCR+ and 53 being PCR−. Nine of the PCR+ cats were diagnosed ante mortem on blood PCR; the 10th PCR+ cat had a negative blood PCR but had a positive PCR on splenic tissue taken at necropsy. Of these cats, only one had culture performed; this was a blood culture and was positive for F tularensis. Of the PCR− cats, ante-mortem PCR was performed in 38 cats: 35 on blood; one on urine; one on a lymph node aspirate; and one on a lymph node biopsy. Tularemia PCR was performed post mortem for the remaining 15 cats: five on liver alone; four on spleen and liver; three on kidney; one on spleen alone; one on lymph node; and one on tissue that was not identified. Of the PCR− cats, 18 also had bacterial cultures performed of at least one site and none had results positive for F tularensis. A final diagnosis was available for 15 PCR− cats. These included feline infectious peritonitis (n = 3), pneumonia (n = 2), cholangitis (n = 2), bacterial sepsis (n = 2) and one of each of bartonellosis, feline immunodeficiency virus infection, acute gastroenteritis, Mycoplasma haemominutum infection, nasal nematodiasis and endocarditis/myocarditis.
Demographic, historical and physical examination data are presented in Table 2; there were no significant differences between PCR+ and PCR− cats for these parameters. Continuous clinicopathologic variables are presented in Table 3; there were no significant differences between PCR+ and PCR− cats. However, when treated as binary parameters, leukopenia (4/6 vs 7/33, P = 0.042) and neutropenia (3/6 vs 3/33, P = 0.036) were significantly more common in PCR+ cats compared with PCR– cats (Figure 3). There were no significant differences between PCR+ and PCR− cats in the frequency of anemia (3/6 vs 16/34, P = 0.999), thrombocytopenia (5/6 vs 26/33, P = 0.999), band neutrophilia (2/4 vs 12/26, P = 0.999) or hyperbilirubinemia (5/6 vs 23/31, P = 0.999).
Demographic, historical and physical examination data for cats positive and negative for tularemia on PCR
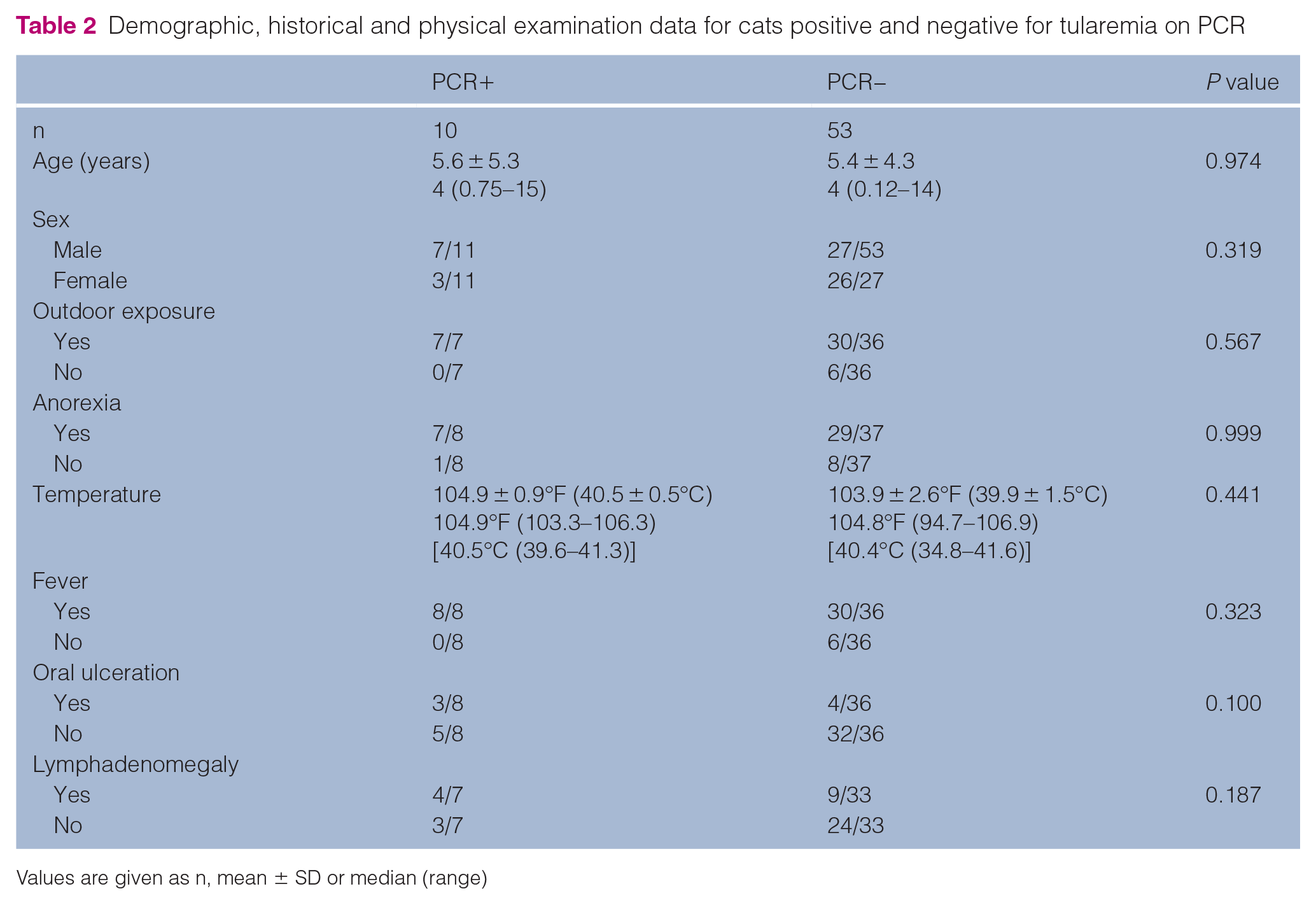
Values are given as n, mean ± SD or median (range)
Clinicopathologic data for cats positive and negative for tularemia on PCR. There were no significant differences between groups

Values are given as n, mean ± SD or median (range)
ALP = alkaline phosphatase; ALT = alanine transaminase; BUN = blood urea nitrogen; HCT = hematocrit; PLT = platelets; TBILI = total bilirubin; WBC = white blood cells

Comparative analysis of clinical findings in PCR+ vs PCR− cats. (a) Leukopenia and (b) neutropenia were significantly more common in PCR+ cats compared with PCR− cats
The results of the univariate analyses are presented in Table 4. Of the assessed parameters, cats with leukopenia (P = 0.038) and neutropenia (P = 0.024) were more likely to be tularemia PCR+. These variables, as well as oral ulceration (P = 0.082), lymphadenomegaly (P = 0.139), anemia (P = 0.133) and BUN (P = 0.190), were included in the multivariate logistic regression analysis. However, none of these variables reached statistical significance in the final model.
Univariate logistic regression analyses of historical, physical examination and clinicopathologic variables predicting PCR status in cats tested for tularemia

P values of parameters included in the multivariate model are in bold
ALP = alkaline phosphatase; ALT = alanine transaminase; BUN = blood urea nitrogen; CI = confidence interval; HCT = hematocrit; PLT = platelets; TBILI = total bilirubin; WBC = white blood cells
Discussion
Feline tularemia is a rapidly progressive, often fatal, zoonotic disease. Thus, prompt identification is essential to reaching a better prognosis and avoiding human exposure. There are no pathognomonic signs of this disease; therefore, initial recognition is reliant on integration of clinical findings. To this end, the current study aimed to identify clinical features of tularemia in cats that should increase the index of suspicion for this disease for clinicians. In our cohort of 18 cats diagnosed with tularemia, key clinical features included fever, oral ulceration and lymphadenomegaly. In addition, all cats for whom housing information was available had access to the outdoors. Cats with PCR-diagnosed tularemia were significantly more likely to have leukopenia and neutropenia than PCR− cases.
These features of feline tularemia are similar to those documented in previous case reports and are consistent with our current understanding of disease pathogenesis.4,5,8,9 For example, cats with tularemia in this and other studies generally had a history of outdoor activity. This accounts for F tularensis exposure, which often occurs through contact with infected animals such as rodents and rabbits, drinking contaminated water, aerosol exposure from infected animals or bites from infected insects or ticks. Outdoor hunting cats are especially at risk. 16 Oral ulceration is commonly described in case reports and occurred in more than half of the cats with tularemia in our study for which physical examination findings were available.4,5,11 In contrast, only 2% of human tularemia cases develop oral ulceration 20 and this may represent a species difference in route of exposure. Unlike cats, people are usually exposed through arthropod vectors or skin contact with tissues from infected animals, and often develop skin ulcers at the site of inoculation. 1 The clinical findings of pyrexia, leukopenia and neutropenia in our cats likely result from the septic nature of F tularensis infection, and the toxic and band neutrophils reported in other cases complement these findings.4,5,11 There was also a high incidence of lymphadenomegaly here and in other reports, consistent with preferential infection of lymphoreticular organs. This is further supported by suppurative inflammation and necrosis of the lymph nodes, liver and spleen in cytologic and histologic samples.4,10,11
Our study demonstrated that leukopenia and neutropenia were significantly more common in cats with tularemia (PCR+) compared with cats without the disease (PCR−). However, we did not find significant differences in other common clinical findings of tularemia, including oral ulceration, lymphadenomegaly, band neutrophilia, thrombocytopenia and hyperbilirubinemia. In addition, the final multivariate regression failed to identify any clinical variables that predicted PCR status. It is possible that our study was underpowered to detect such differences. However, it is also possible that no differences were found because the control group was composed of cats who were tested for tularemia by PCR but had a negative test. The fact that the PCR test was performed in these cats suggests that the clinical or pathologic findings identified by the primary clinician were consistent with the disease and the test was ordered for a definitive diagnosis. Thus, common clinical findings that were not statistically significant are likely still important in raising the suspicion for feline tularemia. Still, leukopenia and neutropenia may allow for further discrimination between positive and negative cases.
The significance of this study lies in its potential to improve the identification of tularemia in domestic cats, particularly considering that this is generally a rapidly progressive disease and many cats with tularemia are only diagnosed at necropsy. Having this information will enable clinicians to achieve quicker identification and initiate prompt treatment. Of the 10 cats with tularemia who received antimicrobial therapy, five survived to discharge. Although the prognosis is guarded, pursuing treatment is reasonable and earlier identification may improve outcomes for affected cats. In addition, earlier recognition would allow for faster institution of isolation protocols, reducing the risk of transmission to owners and veterinary personnel.
Our study is not without its limitations. The retrospective nature and a relatively small cohort size are important limitations, although they represent a step forward compared with previous case reports. Because tularemia is a rare disease, we had to use various methods to collect clinical information, which resulted in incomplete diagnostic data for some patients. In particular, imaging, cytology and histopathology were not performed or available for many patients. The selection of cases based on availability of PCR and culture results was driven by the submitting clinicians’ suspicion of tularemia. This introduces a potential selection bias in the analysis, as it may not represent the broader population of cats who might have been exposed to or infected with F tularensis. Another limitation of our study was the inability to subspeciate the samples used. This is because the PCR assay employed was designed as a diagnostic test and does not have the capability to differentiate between subspecies of F tularensis. Finally, different diagnostic methods were used (culture vs PCR) when compiling the tularemia-positive cohort because of a change in Veterinary Diagnostic Laboratory safety policy. Future investigations should aim to address these limitations, perhaps through prospective studies or broader geographical sampling. Continued exploration into the clinical aspects of tularemia could give further insight into the diagnosis and management of this disease.
Conclusions
This study enhances our understanding of feline tularemia, emphasizing the importance of early recognition of key clinical signs such as leukopenia and neutropenia for timely diagnosis and treatment. These findings are crucial for clinicians in suspecting tularemia, especially given the disease’s rapid progression and potential for fatal outcomes. Despite limitations such as its retrospective nature and small cohort size, the study offers valuable insights for future research aimed at improving tularemia diagnosis and management, ultimately reducing the risk to both feline patients and humans.
Footnotes
Acknowledgements
The authors thank Ms Dardi Miller for assistance with data collection and Dr Arnon Gal for assistance with statistical analysis.
Author note
Since 2017, informed consent has been obtained upon hospital admission for all cats seen at the University of Illinois Veterinary Teaching Hospital. Before this, informed consent for research was not included in the admission paperwork. Verbal informed consent was obtained by most other veterinary hospitals at the time records were released. All data from this study were collected from medical records systems; no archival samples were used.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication and therefore additional informed consent for publication was not required.
