Abstract
A 9-y-old captive male Pallas’ cat (Otocolobus manul) had a 1-mo history of worsening lameness and was euthanized. The animal was submitted to the North Dakota State University–Veterinary Diagnostic Laboratory for autopsy with differential diagnoses of suspected degenerative joint disease or neoplasia. Autopsy revealed icteric tissues and pinpoint foci in the liver, spleen, and all lung lobes. PCR testing was positive for Francisella tularensis, the causative agent of tularemia. Additional cases of tularemia were later identified in wild eastern cottontail rabbits found dead at the same urban zoo. Tularemia has been reported in captive non-human primates and rodent populations with one case linked to wild lagomorph exposure, which was likely the route of exposure in our Pallas’ cat case. Tularemia is an occupational risk for zoo staff and laboratorians. Pest management and disease surveillance of wild lagomorph populations in zoos are important preventive measures.
Tularemia is a rare zoonotic infection caused by the bacterium Francisella tularensis. F. tularensis is designated as a Tier 1 Select Agent due to its potential as a severe threat to humans and animals and is therefore reportable to the Centers for Disease Control and Prevention (CDC) or the Animal and Plant Health Inspection Service in the United States. Disease caused by F. tularensis has many names, including tularemia, rabbit fever, hare plague, hare disease, and deerfly fever.
The infectious dose of F. tularensis is very low in humans, ~10–50 organisms, and clinical presentation depends on the route of transmission, which makes the diagnosis of tularemia challenging. Arthropods and small mammals, such as rabbits, hares, beavers, and ground squirrels, are potential sources of this disease agent. Sources of exposure include undercooked or raw rabbit or hare meat, arthropod bites, tissue transplants, contaminated water, or occupationally.5–8 Occupational infections have been described in laboratorians, landscapers, and workers who handle animals, such as veterinarians or zookeepers.1,4,11,12
After a month of worsening lameness, decreased range of motion, and swelling in the right forelimb of a 9-y-old captive male Pallas’ cat (Otocolobus manul), staff at the Red River Zoo (Fargo, ND, USA) examined the cat under chemical immobilization and completed diagnostic testing. Radiographs revealed lytic lesions and periosteal reaction of the right distal humerus. Osteoarthritic changes were present in all bones of the elbow joint. Routine bloodwork was unremarkable, except for eosinophilia. Worsening stiffness and weakness were noted. Due to quality-of-life concerns, the Pallas’ cat was euthanized and submitted to the North Dakota State University–Veterinary Diagnostic Laboratory (NDSU-VDL; Fargo, ND, USA) for postmortem examination. Degenerative joint disease and neoplasia were listed on the submission form as possible differential diagnoses.
On gross examination, all mucous membranes and subcutaneous tissues were icteric. On the surface of the liver and spleen and within all lung lobes were dozens of tan pinpoint nodules that extended into the parenchyma on section. Given the size and distribution of the nodules, tularemia or other bacterial infection was favored over neoplasia; lymphoma remained on the differential list given the clinical history of possible neoplasia. Upon microscopic review, the nodules were composed of necrotic debris admixed with degenerate neutrophils, and no bacterial colonies were visible on H&E stain—all classic features of F. tularensis infection. Samples were thus submitted for PCR to confirm the diagnosis of tularemia.
Once tularemia was suspected, sample manipulations occurred only in a biosafety cabinet, and laboratory work was conducted in a biosafety level-3 laboratory. Splenic tissue was swabbed and suspended in 2 mL of PBS. Sample DNA was automatically extracted (KingFisher Duo Prime, MagMAX pathogen RNA/DNA kit; Applied Biosystems, Thermo Fisher) according to the manufacturer’s instructions. Our PCR assay targeted the 23kDa gene. 14 Briefly, each 25-μL reaction consisted of 12.5 µL of master mix (Path-ID qPCR master mix; Thermo Fisher), 1.25 μL (0.5 μM) of each primer, 0.375 μL (0.3 μM) of probe, and 5 μL of template DNA. Cycling conditions consisted of an initial denaturation step of 95°C for 10 min, then 40 cycles of 95°C for 15 s and 60°C for 60 s.
F. tularensis was detected in splenic tissue by PCR assay. Tularemia had not been reported previously at the facility; however, dead ground squirrels and mice had been observed on zoo grounds before the illness of the Pallas’ cat. Pest management procedures within the zoo were reviewed, but no sources of infection were confirmed.
Two years later, a wild eastern cottontail rabbit (Sylvilagus floridanus), was submitted to the laboratory by the zoo. It was 1 of 30 rabbits found dead on zoo premises that appeared to have died suddenly with no obvious trauma and in good body condition. Given the risk of tularemia in lagomorphs, the rabbit carcass was opened in a biosafety cabinet. Samples were processed in biosafety level-3 containment. The spleen tested positive for F. tularensis by PCR assay. A second rabbit was found dead in the camel pond and was submitted for confirmation of F. tularensis infection several days after the first confirmed case; the PCR assay was also positive. A half-eaten rabbit carcass was found in the Pallas’ cat enclosure. Due to previous illness in the other male Pallas’ cat and confirmed tularemia on the premises, the male Pallas’ cat in the enclosure was treated prophylactically with doxycycline (50 mg PO q24h × 18 d). No signs of infection were observed.
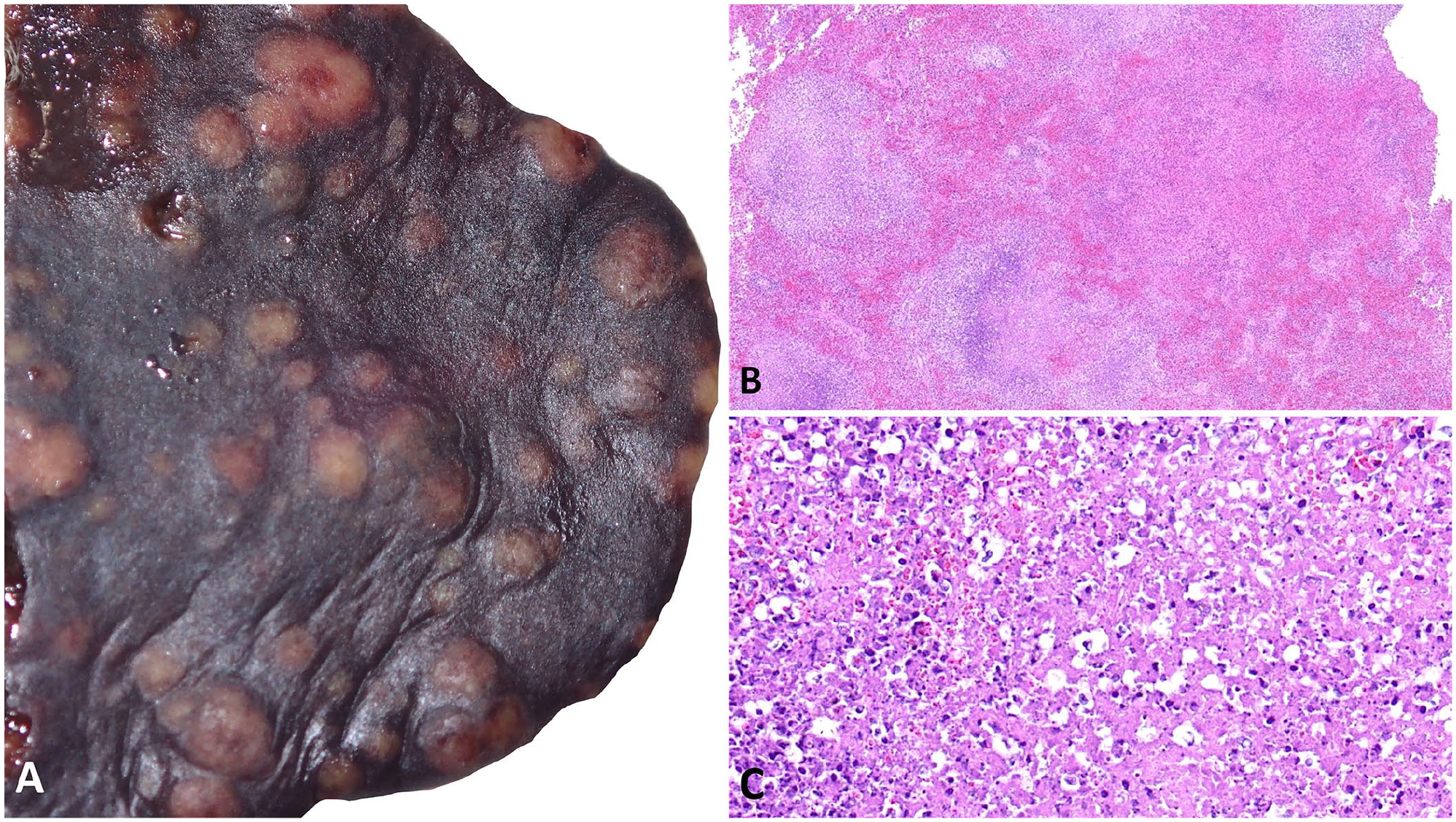
All F. tularensis–positive rabbit tissues were destroyed after confirmation of infection and, therefore, histologic evaluation could not be performed. However, in several unsuspected felid cases of tularemia submitted to the NDSU-VDL in 2020, photographs and microscopic images were obtained before destruction of the case materials; in one adult domestic cat, the spleen contained generalized, up to 1-cm, tan-to-white nodules (Fig. 1A). Similar gross findings in the spleen of the Pallas’ cat corresponded to multifocal-to-coalescing hemorrhage and necrosis (Fig. 1B, 1C). Because CDC requirements are to destroy case materials within 7 d of select agent identification, additional molecular studies were not performed to determine whether infection was caused by F. tularensis subsp. tularensis or subsp. holarctica. F. tularensis subsp. tularensis is the most virulent subspecies and is the type isolated most frequently from human patients in the United States. 3
It is important to consider small mammals, such as lagomorphs, as potential sources of highly infectious zoonotic disease, even in urban environments. Because neoplasia was one of the main differentials listed for the captive Pallas’ cat on the submission form and no rabbit contact had been observed, the carcass was opened on an open-downdraft table for routine autopsy. Once the pathologist suspected tularemia, samples were collected for F. tularensis PCR testing and all other test processes were suspended until testing was complete. The pathologist and autopsy technicians were potentially exposed during autopsy and cleanup; therefore, post-exposure prophylaxis was offered to personnel, and symptom monitoring was initiated. This potential exposure necessitated completion of a CDC Form 3 (https://www.selectagents.gov/forms/index.htm), which facilitates an investigation and process improvements within the laboratory, assisted by the CDC, to ensure that future exposures would be avoided. Fortunately, neither zoo nor laboratory staff reported symptoms after exposure.
Zoos have individual, humane pest management policies to ensure protection of their collection species. 2 These policies are based on institutional needs, therefore policies vary by pest species, but recommendations are to control “insects, rodents, nuisance birds, and certain mammals.” 10 In general, physical barriers, trapping, baiting, biologic control measures, including contraceptives, and pesticides are utilized regularly. Monitoring unusual die-off events of pest animal species may be an appropriate surveillance tactic. For example, F. tularensis was identified in eastern cottontail rabbits found dead on the grounds of a zoological park in Philadelphia, triggering additional monitoring of collection species and zoo staff, with no illnesses reported. 13
Further to our case, physical barriers were implemented at the zoo to prevent wild rabbits from entering exhibits with susceptible collection animals. Live traps were also employed to trap and deflect rabbits from exhibit perimeters. Because a dead rabbit was found floating in the camel pond, the pond was drained and flushed, and the camels were prohibited from accessing it for the remainder of the season. After confirmation of tularemia in the wild cottontail rabbit population, a half-eaten cottontail carcass was found in a Pallas’ cat enclosure, prompting tick prevention application and prophylactic doxycycline administration for several weeks. Physical barriers were employed exclusively for larger animal species.
Transmission of tularemia to veterinarians via animal bite or during autopsy of infected rodents has been documented.12,15 Interestingly, reported tularemia cases in humans increased in the United States in 2011–2019. 3 Most reports of tularemia in captive animal species are in non-human primates6,9,12; however, an outbreak in a colony of captive prairie dogs is also reported. 16 A report linked tularemia in captive orangutans (Pongo pygmaeus pygmaeus) and later discovery of a tularemia-positive rabbit carcass in the orangutan enclosure. 9 Tularemia should be on the differential list whenever there is fever of unknown origin, especially when the animal is icteric.
In our Pallas’ cat case, tularemia was not on the initial differential list. Febrile illness after contact with rabbits is a classic case history for tularemia. Infected animals may have oral or subcutaneous ulcers or enlarged lymph nodes. Pinpoint tan foci in liver or spleen are classic gross lesions for tularemia and, once observed, sample manipulations should be transferred to a biosafety cabinet and rule-out procedures (culture or PCR) should be performed. Laboratories should have occupational health plans and procedures for employees to seek post-exposure care if occupational exposures occur.
Our case emphasizes the need to consider tularemia in susceptible captive species in the presence of wild lagomorphs in zoos and highlights the appropriate use of disease surveillance and prevention measures. Tularemia is an occupational risk factor for zoo staff and veterinary laboratorians.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
