Abstract
Objectives
The aim of this study was to determine response rates, median progression-free intervals (PFIs) and median survival times (MSTs) for cats with intermediate–large cell lymphoma treated with a vincristine, cyclophosphamide, mitoxantrone and prednisolone (CMOP) protocol. A secondary objective was to determine the tolerability of mitoxantrone used within this multiagent protocol.
Methods
The medical records of 31 cats treated at a single institution between 2009 and 2022 were reviewed to identify suitable cases. Cats were included in the study if they had a confirmed diagnosis of intermediate–large cell lymphoma, had received a CMOP protocol as first-line treatment and had completed at least one 4-week cycle of this protocol. Modifications allowed in the protocol included the use of l-asparaginase, vinblastine substitution for vincristine, chlorambucil substitution for cyclophosphamide and dexamethasone or methylprednisolone substitution for prednisolone.
Results
The overall response rate was 74% (n = 23), with 45% (n = 14) achieving complete remission (CR), 29% (n = 9) achieving partial remission (PR) and 26% (n = 8) achieving stable disease (SD). The Kaplan–Meier median PFI and MST were 139 days and 206 days, respectively. Responders (CR or PR) had a significantly longer (P <0.001) median PFI and MST compared with non-responders (SD) (176 days vs 62 days, and 251 days vs 61 days, respectively). Cats that achieved CR had a significantly longer median PFI and MST (P <0.001) at 178 days and 1176 days, respectively. The 6-month and 1- and 2-year survival rates in cats with CR were 64%, 57% and 35%, respectively. Treatment with mitoxantrone was generally well tolerated, with no cats experiencing Veterinary Cooperative Oncology Group adverse effects above grade 2.
Conclusions and relevance
The CMOP protocol is an alternative and well-tolerated treatment for cats with intermediate–large cell lymphoma. As demonstrated with previous chemotherapy protocols, cats that respond to treatment, particularly those that achieve CR, are likely to have more durable responses.
Introduction
Lymphoma is the most common neoplasm in cats, for which chemotherapy has been established as the mainstay of treatment.1 –7 Protocols based on cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP) are widely used as a first-line treatment for feline intermediate–large cell lymphoma. Previous response rates, progression-free intervals (PFIs) and median survival times (MSTs) after treatment with a CHOP protocol differ between studies but have been reported to be in the range of 62–72%, 56–156 days and 97–120 days, respectively.2,3,7
Doxorubicin, an anthracycline antibiotic included in a standard CHOP protocol, has known cardiotoxic effects in canine patients, including acute arrhythmias at the time of administration and delayed-onset dilated cardiomyopathy after cumulative doses over time. 8 Mitoxantrone is a synthetic antitumour antibiotic with similar action to doxorubicin, in terms of RNA and DNA intercalation and inhibition of RNA and DNA polymerases and topoisomerase II, but induces less cardiotoxicity in dogs since it does not cause oxidative damage to cells and has reduced potential to form free radicals. 9 The cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) protocol has therefore been proposed as an alternative treatment option to CHOP for dogs where cardiotoxicity is of concern. 10 Comparisons of canine lymphoma patients treated with CMOP have shown comparable response rates, PFIs and MSTs but fewer side effects than those treated with CHOP.11,12 Although cats are less sensitive to the cardiotoxic effects of doxorubicin, the other adverse effects of doxorubicin chemotherapy, including the potential for nephrotoxicity, warrant the assessment of alternative chemotherapeutics.13 –15 For this reason, it may be of benefit to consider a multidrug protocol that replaces doxorubicin with an alternative agent. Mitoxantrone has been shown to be efficacious in other feline neoplasms and is generally well tolerated by cats, with mild gastrointestinal adverse events being the most commonly reported toxicity.16 –19 Therefore, mitoxantrone is often substituted for doxorubicin in clinical practice, particularly in feline patients with pre-existing azotaemia or elderly patients that may be at greater risk for developing azotaemia. However, this substitution as part of a CMOP protocol has not previously been reported in cats. The aim of this study was to assess the efficacy and tolerability of a CMOP protocol in cats diagnosed with intermediate–large cell lymphoma.
Materials and methods
The medical records of cats treated at the University of Glasgow between 2009 and 2022 for intermediate–large cell lymphoma were reviewed. The institution’s database was searched using the keywords ‘cat’, ‘feline’ and ‘lymphoma’, and records were manually screened for suitability. Cats were included in the study if they had a confirmed cytological or histological diagnosis of intermediate–large cell lymphoma, a CMOP protocol (Table 1) was used for first-line treatment and the cat completed at least one cycle (4 weeks) of this protocol. Cats were excluded if they had a diagnosis of small cell lymphoma, if any chemotherapy agents had been administered before the initiation of CMOP (excluding l-asparaginase) or if they did not complete one full cycle (4 weeks) of the CMOP protocol.
Summary of the 25-week CMOP protocol

Prednisolone is started at week 1 at 2 mg/kg POSID and then tapered to 1 mg/kg POSID by week 3. Prednisolone can then be continued at 0.5–1 mg/kg POSID throughout the remaining protocol at the clinician’s discretion
CMOP = cyclophosphamide, mitoxantrone, vincristine and prednisolone
Modifications allowed in the protocol included the use of l-asparaginase, vinblastine substitution for vincristine, chlorambucil substitution for cyclophosphamide and the use of dexamethasone or methylprednisolone acetate instead of prednisolone.
Information obtained from medical records included signalment (age, breed, sex, weight), feline immunodeficiency virus (FIV)/feline leukaemia virus (FeLV) status, haematology, date of first chemotherapy treatment, anatomical location of disease, drugs included in therapy, any rescue treatment used, whether completion of the planned protocol was achieved, the number of mitoxantrone doses given, adverse events after the administration of mitoxantrone and time to endpoints. Where medical records were incomplete, cases were considered suitable for inclusion providing the necessary information regarding time to endpoints was available.
The anatomical location of the lymphoma was classified as alimentary, mediastinal, extra-nodal (including renal, nasal, pulmonary, central nervous system or ocular) or peripheral nodal, depending on where the predominance of disease was located. The lymphoma was classified as multicentric if there were multiple sites and no clear category based on a predominant location.
The response to chemotherapy was classified according to the Veterinary Cooperative Oncology Group (VCOG) response evaluation criteria for peripheral nodal lymphoma in dogs as either a complete response (CR; regression of all measurable disease, clinical signs and/or imaging findings), partial response (PR; improvement >30% but <100%, resolution of measurable disease, clinical signs or imaging findings) or stable disease (SD; no improvement [<30% reduction or <20% increase of measurable disease, clinical signs or imaging findings]). 20 Progressive disease was defined as failure of clinical improvement, >20% increase of measurable disease or increased severity of clinical signs. Where disease was primarily internal, or if chemotherapy was initiated when microscopic disease was present, repeat imaging to assess disease response was performed after 1–2 cycles of chemotherapy, at the discretion of the primary clinician. Where imaging was not performed (due to owner finances, patient temperament or other mitigating factors), the response was judged using improvement or resolution of clinical signs. The response rate was calculated as a percentage of cats showing a response out of all those receiving the treatment.
Adverse chemotherapy events that were recorded at each visit were graded according to VCOG common terminology criteria for adverse events. 21 Dose adjustments and treatment delays were made at the clinician’s and owner’s discretion, as was the selection of any relapse protocols.
The Kaplan–Meier product limit method was utilised to estimate PFIs and MSTs. PFI was defined as the time from the date of the first chemotherapy treatment to the first reported date of progression, via measurable disease, clinical signs and/or imaging findings. Overall survival time was defined as the time between the date of the first chemotherapy treatment to the date of death from any cause. Cats were censored from survival analysis if they were still alive at the end of the study period or if they were lost to follow-up. Log-rank testing was utilised to compare outcome between response groups (CR vs PR vs SD). Statistical analyses were performed using commercial software (SPSS Statistics; IBM Corp) and a P value <0.05 was considered significant.
Results
Patient characteristics
A total of 91 cats were identified using the search criteria and 31 cats met the inclusion criteria for the study. There were 22 (70.9%) castrated male cats, one (3.2%) entire male cat and eight (25.8%) spayed female cats. The median age and body weights at the time of diagnosis were 11.5 years (range 1.5–18) and 4.49 kg (range 2.4–6.4), respectively. Breeds included domestic shorthair (n = 24), Siamese (n = 3), Oriental (n = 2), Maine Coon (n = 1) and British Shorthair (n = 1). Retroviral status assessment was available for 15 cats, with 4/15 cats positive for FIV and 0/15 cats positive for FeLV on either PCR or ELISA. Anatomical locations included alimentary (n = 13), mediastinal (n = 4) and extra-nodal (n=14), which included renal (n = 4), renal and intestinal (n = 3), nasal (n = 2), pulmonary (n = 2), renal and nasal (n = 1), renal and CNS (n = 1) and renal and ocular (n = 1). All cats started chemotherapy with macroscopic disease present, with the exception of one cat with alimentary lymphoma that started chemotherapy with only microscopic disease. Nine cats received l-asparaginase as part of their protocol, 2/31 cats received vinblastine (one cat showed incomplete remission at week 19 and was given a total of one dose; the other cat was given the substitution due to gastrointestinal adverse events after vincristine and received a total of seven doses of vinblastine) and 1/31 cats received chlorambucil from the start of the protocol due to a previous urinary tract infection (total of two doses given).
Treatment response and outcomes
In total, 8 (26%) cats completed the 16 treatments included in the 25-week CMOP chemotherapy protocol, including four doses of mitoxantrone. For those that did not complete the protocol, the median number of treatments was 8 (range 4–15). Reasons for failure to complete the protocol were progressive disease (n = 14), financial reasons (n = 3), owner withdrawal due to adverse events (n = 2), a shortened protocol of eight treatments due to clinician preference (n = 2), a change of protocol as only PR (n = 1) and death from an unrelated cause (n = 1).
A total of 74 doses of mitoxantrone were administered to the 31 cats; 10 cats received one dose (one full cycle), seven cats received two doses (two full cycles), six cats received three doses (three full cycles) and eight cats received four doses (complete protocol). The actual dose administered was available for 54 of the doses. The median dose administered was 4.97 mg/m2 (range 4.6–6.5).
In total, 19 (61%) cats received a rescue protocol following evidence of incomplete response to treatment or progressive disease. Rescue protocols were not standardised and were chosen based on owner and clinician preference. A total of 15 cats received a single rescue protocol, one cat received two rescue protocols and three cats received three rescue protocols. Rescue protocols included the ALP (l-asparaginase, lomustine and prednisolone) protocol (n = 5), the DMAC (dexamethasone, methotrexate, actinomycin D, cytarabine) protocol (n = 1), a combination of l-asparaginase, lomustine and cytarabine (n = 1), and single-agent chemotherapy with l-asparaginase (n = 4), lomustine (n = 4), prednisolone (n = 5), mitoxantrone (n = 2), doxorubicin (n = 2) or chlorambucil (n = 1). Radiotherapy was used as a rescue treatment in one cat with nasal lymphoma.
The overall response rate to CMOP was 74% (n = 23), with 45% (n = 14) of cats achieving CR, 29% (n = 9) achieving PR and 26% (n = 8) achieving either SD or no response. The Kaplan–Meier median overall PFI was 139 days (Figure 1) and the MST was 206 days (Figure 2). Of the 31 cats included in calculating the MSTs, two cats died of unrelated causes and seven cats were censored from survival analysis (two cats were lost to follow-up and five cats were alive at the end of the study period). Approximately one-third of the cats included in this study received a dose of l-asparaginase; however, this inclusion did not result in a significant difference in PFI (120 days vs 140 days; P = 0.831) or MST (176 days vs 206 days; P = 0.659).

Kaplan–Meier curve of progression-free interval (PFI) for cats with intermediate–large cell lymphoma, treated with a cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) chemotherapy protocol (n = 31). Median PFI was 139 days
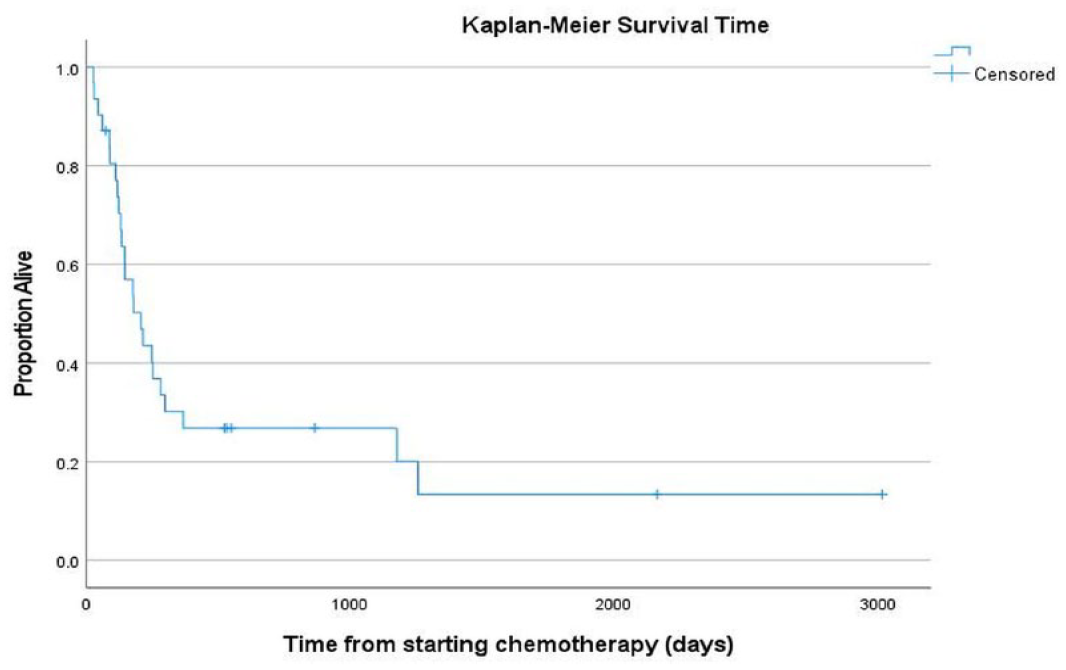
Kaplan–Meier curves of overall survival time for cats with intermediate–large cell lymphoma, treated with a cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) chemotherapy protocol (n = 31). Median survival time was 206 days
Cats that achieved CR had a significantly longer PFI (178 days vs 100 days; P <0.001) and MST (1176 days vs 113 days; P = 0.003) compared with those that achieved only PR or SD. There was a significant difference in PFI and MST based on response to treatment (CR vs PR vs SD) with a calculated PFI of 178 days vs 111 days vs 62 days (P <0.001) and MST of 1176 days vs 215 days vs 61 days (P <0.001), respectively (Figure 3). The 6-month and 1- and 2-year survival rates in cats with CR were 64%, 57% and 45%, respectively.

Kaplan–Meier curve of progression-free interval (PFI) for cats with intermediate–large cell lymphoma, treated with a cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) chemotherapy protocol that achieved complete remission (n = 14; solid green line), partial remission (n = 9; dotted red line) or stable disease (n = 8, dashed blue line). Median PFI was significantly longer (P <0.001) in cats achieving complete remission
Adverse events
The medical records of 30 cats were available for review of the adverse events after mitoxantrone treatment. A total of 13/30 (43%) cats experienced side effects after mitoxantrone, the most common of which was gastrointestinal (Table 2). All the side effects were grade 1 or 2; there were no grade 3, 4 or 5 side effects in the study population.
Frequency of toxicity in 30 cats after administration of mitoxantrone as part of the CMOP protocol, graded according to VCOG criteria 21
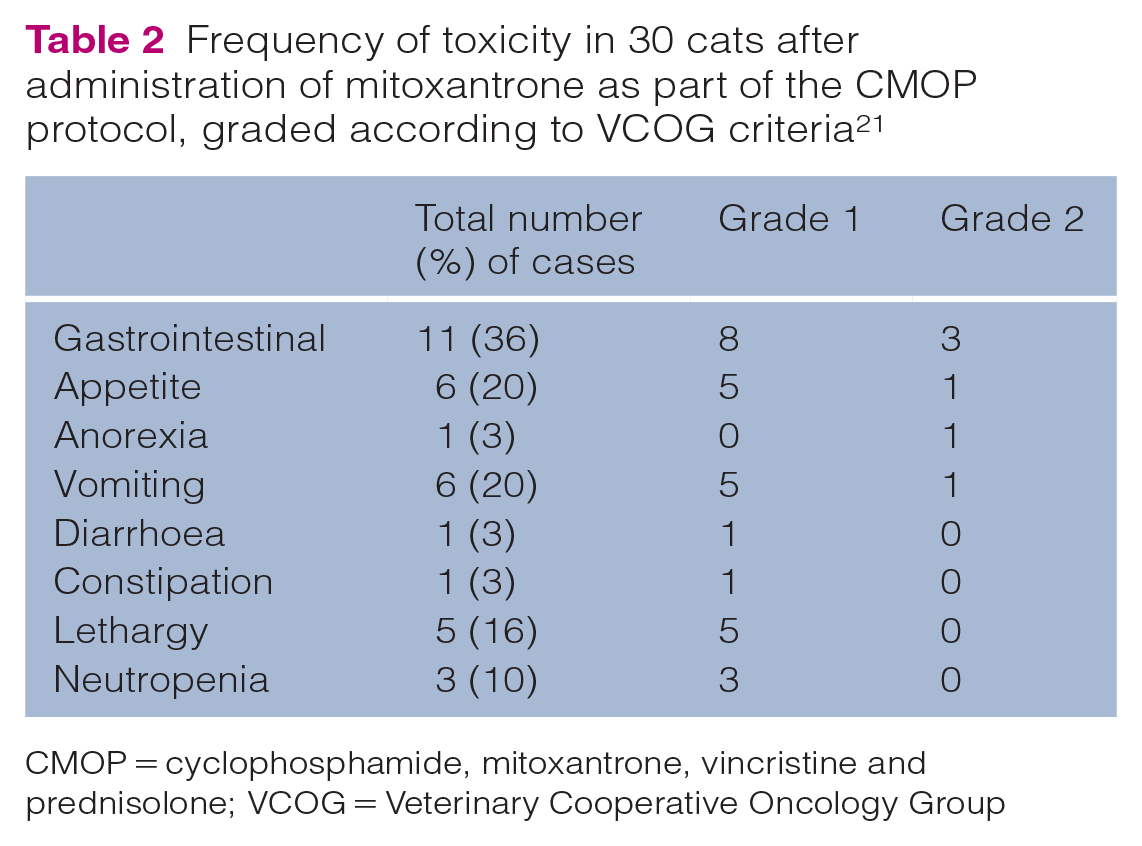
CMOP = cyclophosphamide, mitoxantrone, vincristine and prednisolone; VCOG = Veterinary Cooperative Oncology Group
Discussion
The CHOP protocol was initially designed for use in humans, and modifications are widely used to treat canine lymphoma.22 –24 COP-based protocols (vincristine, cyclophosphamide and prednisolone) are also widely used in canine and feline lymphoma, and provide good response rates and remission times. 25 By excluding doxorubicin, the COP protocol provides an alternative treatment option for patients in which the side effects of doxorubicin could be detrimental (including cardiotoxicity in dogs and nephrotoxicity in cats).8,13 –15 However, in canine lymphoma, the addition of doxorubicin to a multiagent protocol has been shown to yield more favourable outcomes, hence the substitution of doxorubicin for an alternative drug in a CHOP-based protocol is perhaps more desirable than changing to a COP-based protocol.25,26 The substitution of doxorubicin for mitoxantrone in a multiagent protocol (CMOP) in dogs has been evaluated in two different studies that did not show any negative impact on survival times in these patients compared with CHOP, and would suggest that CMOP is an acceptable alternative chemotherapy protocol for canine lymphoma patients that cannot tolerate doxorubicin.11,12 However, less is known about the clinical implications of this substitution in cats, in which the use of doxorubicin can be limited by its nephrotoxic effects, particularly in the older population predisposed to cancer. While multiagent chemotherapy is the mainstay of treatment for feline lymphoma, there is currently no consensus on the optimum chemotherapy protocol as currently no prospective evidence exists to definitively conclude a superior protocol as, unlike in their canine counterparts, the benefits of the addition of doxorubicin to a multiagent chemotherapy protocol has not been well established in cats. Cats receiving multiagent protocols that included doxorubicin showed improved remission durations compared with cats that received COP in some studies, although this was only on univariate analysis in one study and doxorubicin was used only in the maintenance phase in another study.5,27 In other studies, the addition of doxorubicin showed no beneficial effect compared with a COP protocol.7,28 Consequently, in cats in which nephrotoxicity is of concern, clinicians may opt for a COP protocol rather than seeking an alternative chemotherapeutic agent to doxorubicin, and thus there is less information available on multiagent protocols with an anthracycline substitution. To the authors’ knowledge, there are no published studies that assess the CMOP protocol in cats; therefore, the purpose of this study was to evaluate the use of a CMOP protocol for the treatment of feline intermediate–large cell lymphoma. Our results suggest that 74% of cats with intermediate–large cell lymphoma responded after treatment with a CMOP protocol, with an overall PFI and MST of 139 days and 206 days, respectively. These values are not dissimilar to those in published studies using maintenance-free CHOP protocols (see Appendix 1 in the supplementary material), suggesting that CMOP is a viable alternative.2,3,7
As has been demonstrated with other chemotherapy protocols, the results of this study suggest that cats that achieve CR have a more durable response to treatment with significantly longer PFIs and MSTs.2 –4,7 The MST for cats achieving CR was 1176 days, which is greater than the MST previously reported with the CHOP protocol in other studies (see Appendix 1 in the supplementary material).2,3,7 However, this MST is based on only 14 cats in this study achieving CR, and direct comparisons to other studies cannot be made as many factors may have contributed to this difference, including the substitution of mitoxantrone, different institutions and the retrospective nature of these studies. In addition, the inclusion of, but lack of standardisation of, rescue protocols that were used in this cohort and in previous published studies is a further reason why direct comparisons of MSTs cannot be made. Only 25% of the study population completed the entire 25-week protocol and rescue treatments were not standardised. A large proportion (61%) of cats went on to receive some form of rescue therapy, which may have influenced the MST; therefore, the PFI could be considered a more reliable endpoint when comparing CMOP with alternative published protocols for the treatment of feline lymphoma.
The definitions of the anatomical locations in cats are not standardised and can overlap between categories. In this study, 41% of the cats included had alimentary lymphoma. Alimentary lymphoma has been reported as the most common form across several different studies, published in the post-FeLV era, and alimentary lymphoma has been associated with a shorter survival time compared with mediastinal or renal lymphoma in cats.2,3,29,30 Therefore, the non-uniformity of the study population could have influenced the survival time.
Vincristine, cyclophosphamide and prednisolone used within multiagent chemotherapy protocols have already been established as well tolerated in cats.2,4,6 Therefore, our study did not examine the adverse events after the administration of these drugs and instead focused on the adverse events after mitoxantrone. In previous publications, the most common toxicities after the use of mitoxantrone in cats have included vomiting, anorexia, diarrhoea, lethargy and myelosuppression.17,31 This is similar to the recorded toxicities in this study. Our retrospective study population also contained a large proportion of cats with alimentary lymphoma, hence it could have been difficult to distinguish treatment-associated toxicity from clinical signs associated with disease. All the adverse events in our study population after the administration of mitoxantrone were mild to moderate (grade 1 or 2), with no severe events documented (grade 3 or higher).
A study that reviewed the use of mitoxantrone in cats with mammary carcinoma documented that in addition to the gastrointestinal and myelosuppressive side effects, azotaemia was one of the more common adverse events. 16 Azotaemia was not reported in an early study of 87 cats receiving mitoxantrone, and azotaemia is not an unexpected finding in any study population of elderly cats. 17 As biochemistry was not part of routine monitoring at the authors’ institution, it was performed only infrequently and at the discretion of the overseeing clinician, thus an assessment of azotaemia could not be made within our population. Likewise, urinalysis was not performed routinely in the cohort and therefore information regarding urine concentrating ability was also not available for assessment. This is a limitation of the retrospective study design, and further prospective studies evaluating the impact of mitoxantrone on the development of azotaemia (similar to those performed for the use of doxorubicin) would be useful to investigate the potential for nephrotoxicity in cats receiving mitoxantrone within a multiagent protocol for lymphoid neoplasia.
Other limitations of the study include those inherent to its retrospective nature, lack of standardisation of rescue protocols and potential bias in case selection (cats had to have completed at least one cycle of CMOP, thereby excluding any cats that died within the first cycle or switched protocols due to poor response/progressive disease).
The lack of a control group is also a limitation of this study. Only six cats at the authors’ institution received CHOP during the same period. Further, sufficiently powered, randomised prospective trials would be recommended to directly compare the efficacy of mitoxantrone with that of doxorubicin as part of a multiagent chemotherapy protocol in the treatment of feline lymphoid neoplasia.
Conclusions
The CMOP protocol produces acceptable response rates, PFIs and MSTs and therefore is an alternative treatment choice for cats with intermediate–large cell lymphoma. It is well tolerated and, in line with previous literature, complete responders are likely to have more durable responses and significantly longer MSTs.
Supplemental Material
Appendix 1:
Response rates, PFI and MST of cats treated with maintenance-free CHOP compared to CMOP.
Footnotes
Author note
This paper was presented as an abstract at the BSAVA Congress 2023.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
TThe work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or nonexperimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
Supplementary material
The following file is available as supplementary material: Appendix 1: Response rates, PFI and MST of cats treated with maintenance-free CHOP compared to CMOP.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
