Abstract
Objectives
This study aimed to compare the response rate, progression-free interval (PFI), survival time (ST) and adverse events in cats treated with cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) vs cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP) for intermediate- to large-cell feline lymphoma.
Methods
Medical records of cats treated for intermediate- to large-cell lymphoma between 2015 and 2023 from three referral institutions within the larger Sydney metropolitan area were retrospectively reviewed. Cats were included if they had a confirmed diagnosis of intermediate- to large-cell lymphoma and had received either a CMOP or CHOP protocol as a first-line treatment. Statistical analysis was conducted to determine if PFI or ST differed significantly between treatment groups. A multivariate analysis was also conducted to determine whether differences in prognostic factors between the groups may have biased outcomes.
Results
A total of 123 cats were enrolled, with 41 cats in the CMOP group and 82 cats in the CHOP group. No significant differences were identified between the response rates (66% in both groups), median PFI (CMOP 69 days, CHOP 59 days) and median ST (CMOP 103 days, CHOP 80 days) of cats treated with CMOP instead of CHOP.
Conclusions and relevance
CMOP is a well-tolerated and suitable substitute for CHOP for feline intermediate- to large-cell lymphoma. It is logistically easier to administer as it can be given as an intravenous bolus and carries less risk of catastrophic extravasation injuries.
Introduction
Feline intermediate- to large-cell lymphoma presents a therapeutic challenge in veterinary oncology. Part of the challenge lies in the fact that lymphoma in cats represents a very heterogeneous group of disease. Further, most cats presenting for treatment of lymphoma are symptomatic (ie, substage B). Although long-lasting remissions are documented in some populations of cats, many cats still do not respond favourably to common chemotherapy protocols.
Previous studies researching the benefits of adding doxorubicin into multi-agent protocols for feline lymphoma have yielded mixed results.1 –4 Doxorubicin as a single agent for feline lymphoma is less efficacious5,6 than in dogs and is documented as nephrotoxic in cats.7,8 Considering the prevalence of early-stage chronic kidney disease (CKD) in cats, 9 avoidance of a potential iatrogenic nephrotoxin is appealing as long as this does not result in a reduction in efficacy of treatment.
Mitoxantrone is an anthracenedione with a similar mechanism of action to doxorubicin; inhibiting topoisomerase II, and intercalating with DNA resulting in strand breaks and cross linking. 10 A recent study demonstrated good tolerance and outcomes in cats with substitution of mitoxantrone for doxorubicin 11 in a protocol based around the University of Wisconsin–Madison (UWM-25) protocol. Two recent studies12,13 found that substitution of mitoxantrone in place of doxorubicin in canine lymphoma did not yield any difference in outcomes that reached statistical significance. It has been used as a substitute for doxorubicin in azotemic cats in an adjuvant setting for mammary carcinoma, 14 lower urinary tract transitional cell carcinoma 15 and in feline lymphoma.16,17
The purpose of this study was to compare the response rate, progression-free interval (PFI), survival time (ST) and adverse events when cats are treated with cyclophosphamide, mitoxantrone, vincristine and prednisolone (CMOP) instead of cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP) for intermediate- to large-cell feline lymphoma.
Materials and methods
A retrospective cohort study was conducted of cats treated at three oncology referral centres (Small Animal Specialist Hospital [SASH], University Veterinary Teaching Hospital Sydney [UVTHS] and Animal Referral Hospital Homebush [ARH]) around the Greater Sydney metropolitan area between January 2015 and April 2023. The medical records from these three centres were reviewed for clinical data. The inclusion criteria included cats that had cytologically or histopathologically confirmed intermediate- to large-cell lymphoma and with the intention to treat with either CHOP or CMOP (see Table 1 in the supplementay material). Previous treatment with prednisolone/prednisone was allowed, as was prior surgical intervention to downstage the disease. Modifications allowed included starting therapy with cyclophosphamide or L-asparaginase, completion of protocol in 19 weeks compared with 25 weeks and substitution of vincristine with vinblastine. The exclusion criteria included a diagnosis of small-cell lymphoma, concurrent radiation treatment, prior treatment with chemotherapy and if they were treated with single agent chemotherapy (eg, lomustine, doxorubicin, mitoxantrone) or cyclophophamide, vincristine and prednisolone (COP)-based protocols. Cats were typically given mitoxantrone instead of doxorubicin when there were concerns about renal insufficiency or a need to avoid sedation, due to factors such as heart disease or owner preference.
Information collected from the medical record included age, breed, sex, weight, feline leukaemia virus (FeLV)/feline immunodeficiency virus (FIV) status, complete blood count/biochemistry tests, date of diagnosis, date of first treatment, method of diagnosis/staging, anatomical form of disease, response to treatment, date of progression, rescue therapies utilised, whether completion of the planned protocol was achieved, date of death and cause of death. Baseline creatinine was recorded at the time of diagnosis where available, and subsequent creatinine readings were screened and the highest recorded as the peak creatinine.
The anatomical form of the disease was classified as gastrointestinal (including the stomach, intestines, jejunal, cecal and ileocolic lymph nodes), multicentric (involving peripheral lymph nodes or multifocal visceral disease of unclear origin, such as hepatosplenic disease), nasal and other (including renal, mediastinal, laryngeal, conjunctival and cutaneous/subcutaneous). Cats presenting with disease in two locations at diagnosis (eg, nasal and hepatosplenic, or renal and gastrointestinal) were classified as having multicentric disease.
Cats were determined to have responded to treatment based on a combination of physical examination parameters, clinical/symptomatic improvement and imaging where available. Cats that were noted to have responded either had improvement or resolution of clinical signs, a reduction of palpable disease by more than 30%, or both. A distinction between complete and partial remission was not made, as imaging and cytology to confirm complete remission were not performed in most cases. Weight change (either loss or gain) was assessed 1 month after diagnosis. Cats that did not survive to this time point were excluded from the weight change analysis.
Statistical analysis
Median ST and PFI were estimated using the Kaplan–Meier method, and outcomes were compared between the two groups using the log-rank test. The PFI was calculated from the date of first treatment (either vincristine or cyclophosphamide) to the date of documented progression. ST was calculated from the date of first treatment (either vincristine or cyclophosphamide) to the date of death/euthanasia for any cause. Cats were censored from progression-free analysis if they were still in remission at the time of final follow-up, and from survival analysis if they were still alive at the time of final follow-up. Censorship of cats that were alive or lost to follow-up was carried out within 1 week. Deaths of unknown cause were attributed to lymphoma. A P value <0.05 was considered statistically significant for all comparisons.
Continuous variables (age, weight, creatinine, haematocrit, lymphocyte:monocyte ratio [LMR] at the time of diagnosis) were compared between treatment protocols using the Kruskal–Wallis test, while Fisher’s exact test was used for categorical variables (substage, retroviral status, anatomical form, response to treatment, weight loss >5%).
A bivariate Cox regression analysis was used to identify prognostic factors for PFI (see Table 2 in the supplementary material) and ST (see Table 3 in the supplementay material), and the variables that reached a significance of P <0.1 were selected for inclusion in a multivariate Cox proportional hazards regression model. Two separate Cox proportional hazard regression models were used to quantify the association between prognostic factors that approached significance (P <0.1), and PFI and ST.
A backwards stepwise approach was used for the multivariate logistic regression analysis. The overall significance of the final model was assessed using the likelihood ratio test. Each of the multivariate models was tested to ensure the proportional hazards assumption had not been violated. All statistical analyses were performed using R statistical software version 4.4.1 (R Core Team 2021).
Results
Patient characteristics
A total of 123 cats were identified that fit the inclusion criteria for this study: 91 from SASH (North Ryde, Tuggerah and Prospect), 12 from UVTHS and 20 from ARH (Homebush). The demographic data of all cats enrolled are displayed in Table 1. In terms of breed distribution, in the other category (n = 28) there were five Ragdolls (including crossbreeds), four each of Burmillas, Maine Coone and Siamese cats, two each of Persian, Oriental and British Shorthair cats, and one each of Siberian, Abyssinian, Balinese, Bengal and Australian Mist cats. In terms of the anatomical forms of lymphoma, the ‘other’ category included 11 mediastinal, five renal, two laryngeal, two conjunctival and one cutaneous lymphoma. Notably, nine cats in the multicentric group showed evidence of renal involvement, either cytologically or on ultrasound. Of the 14 cats with renal involvement, nine were treated with CMOP and five with CHOP.
Summary of demographic data of 123 cats with intermediate- to large-cell lymphoma treated with multi-agent chemotherapy

A majority of cats were diagnosed using cytology (n = 86, 70%), while 37 (30%) were diagnosed histopathologically. Of the 86 cats diagnosed by cytology, 13 underwent further testing with either immunocytochemistry or flow cytometry. Immunophenotype was determined in 36/123 (29%) cats, with 26 cases of B-cell lymphoma, seven of T-cell lymphoma and three of T-cell-rich B-cell lymphoma. Only 33/123 (27%) cats underwent FIV/FeLV testing; of those, seven tested positive for FIV and one tested positive for FeLV. Staging was conducted in 114 cats; of these, 54 were staged with abdominal ultrasound alone, nine with thoracic radiographs alone and three with thoracic ultrasound alone. In total, 18 cats had both abdominal ultrasound and thoracic radiographs, and 30 cats were staged with CT scan and/or ultrasound. Nine cats were not staged. Surgery was performed in seven cats before chemotherapy, with five of those involving mass removals (gastrectomy, intestinal resection and anastomosis, splenectomy) and two involving biopsies of masses in the ileocolic junction.
Comparison between treatment groups (Table 2) showed that cats in the CMOP group were significantly older (P = 0.02) and had higher baseline creatinine levels (P = 0.001) than those in the CHOP group.
Comparison of variables between CMOP vs CHOP treatment groups

Data are n or median (interquartile range)
Significant values highlighted in bold and with (*)
CHOP = cyclophosphamide, doxorubicin, vincristine and prednisolone; CMOP = cyclophosphamide, mitoxantrone, vincristine and prednisolone; FeLV = feline lekaemia virus; FIV = feline immunodeficiency virus; LMR = lymphocyte:monocyte ratio
Treatment response, outcome and prognostic factors
For all 123 cats, the median PFI was 60 days (IQR 42–81) and median ST was 80 days (IQR 66–143). The response rates of 27/41 (66%) for the CMOP group and 54/82 (66%) for the CHOP group were not significantly different (P = 1). The median PFI of cats in the CMOP group was not statistically different from that of cats in the CHOP group (median 69 and 59 days, respectively) (Figure 1). Similarly, the median ST for cats in the CMOP group was not significantly different from those in the CHOP group (median 103 and 80 days, respectively) (Figure 2).

Kaplan–Meier curve depicting progression-free interval for cats treated with cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP; orange) or cyclophsophamide, mitroxantrone, vincristine and prednisolone (CMOP; blue)

Kaplan–Meier curve depicting survival time for cats treated with cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP; orange) or cyclophsophamide, mitroxantrone, vincristine and prednisolone (CMOP; blue)
The median starting dose of mitoxantrone was 5 mg/m2 (range 5–6), with 16 cats receiving 5 mg/m2, 14 cats receiving 5.5 mg/m2 and a single cat receiving 6 mg/m2. No dose escalations or reductions were undertaken for any cats that received subsequent doses of mitoxantrone. There was no significant difference (P = 0.821) in the number of anthracycline doses administered between groups, with a median of 1 (mean CMOP 1.683 compared with mean CHOP 1.634) dose being administered in both groups. Of the 41 cats in the CMOP group, eight (19.5%) completed their protocol, compared with 14/82 (17%) cats in the CHOP groups. This was not statistically significant (χ2 = 0.11; P = 0.74).
A multivariate analysis was conducted to identify statistically significant prognostic factors for PFI and ST in this group of cats. Younger cats, nasal form of disease, higher haematocrit, response to treatment and maintenance of weight at weeks 4–6 were positive prognostic factors for PFI (Table 3). Younger cats, higher haematocrit, response to treatment and maintenance of weight at weeks 4–6 were positive prognostic factors for ST (Table 4). Notably, out of these significant prognostic variables, only age was different between the CMOP/CHOP groups.
Regression coefficients and standard errors (SEs) from multivariate Cox proportional hazards regression model identifying risk factors for progression-free interval for all 123 treated cats
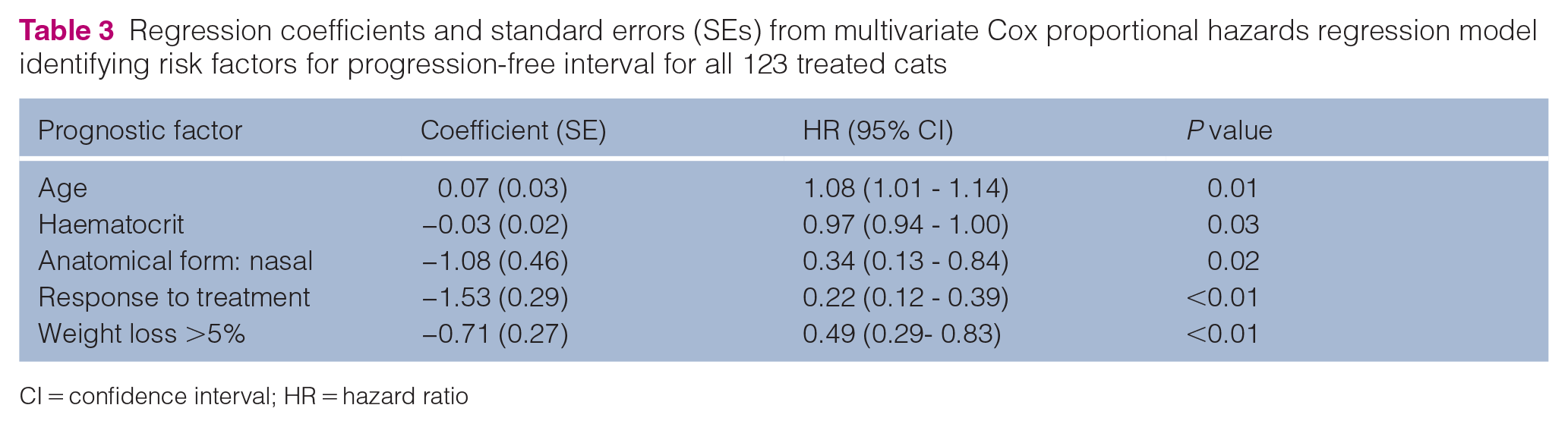
CI = confidence interval; HR = hazard ratio
Regression coefficients and standard errors (SEs) from multivariate Cox proportional hazards regression model identifying risk factors for survival time for all 123 treated cats

CI = confidence interval; HR = hazard ratio
Rescue therapy was attempted in 64 cats, with a majority (n = 40) of these receiving lomustine (1-(2-chloroethy)-3-cyclohelxyl-1-nitrosurea [CCNU]) first. Other rescue protocols that were used included L-asparaginase, doxorubicin or mitoxantrone, chlorambucil, cytarabine, vinblastine and dexamethasone, melphalan, actinomycin D and cytarabine (DMAC). Only two cats that relapsed after completion of their CHOP protocols were re-induced with CHOP. This was successful in one patient. Seven cats received radiation as a form of rescue therapy; in two of these cats, a sustained response was noted leading to substantial improvements in ST compared with PFI (both of these cats belonged to the CHOP group). Attempts at rescue therapy were made in 19/36 (53%) cats and 45/79 (57%) cats with documented progression in the CMOP and CHOP groups, respectively. The rates of rescue were not statistically significant between groups (χ2 = 0.18; P = 0.68).
Toxicity
A total of 85 cats were given either mitoxantrone or doxorubicin. The records of these cats were reviewed for adverse events in the week after administration of either of these drugs. Adverse effects are summarised in Table 5, noting that some of these could be associated with the sedation usually administered before doxorubicin (butorphanol). These events were retrospectively graded using the Veterinary Cooperative Oncology Group’s common terminology criteria for adverse events (VCOG-CTCAE) with the recorded histories. A total of 14/31 (45%) cats in the CMOP group and 33/59 (56%) cats in the CHOP group were noted to have experienced side effects after receiving mitoxantrone/doxorubicin, most of which were mild. A total of 69 doses of mitoxantrone were administered to 31 cats; of these cats, 14 (45%) only ever received a single dose of mitoxantrone. A total of 133 doses of doxorubicin were administered to 59 cats; of these cats, 22 (37%) only ever received a single dose of doxorubicin.
Summary of adverse events after mitoxantrone/doxorubicin dosing (expressed as a percentage of all mitoxantrone/doxorubicin doses administered) graded using the Veterinary Cooperative Oncology Group’s common terminology criteria for adverse events (VCOG-CTCAE)
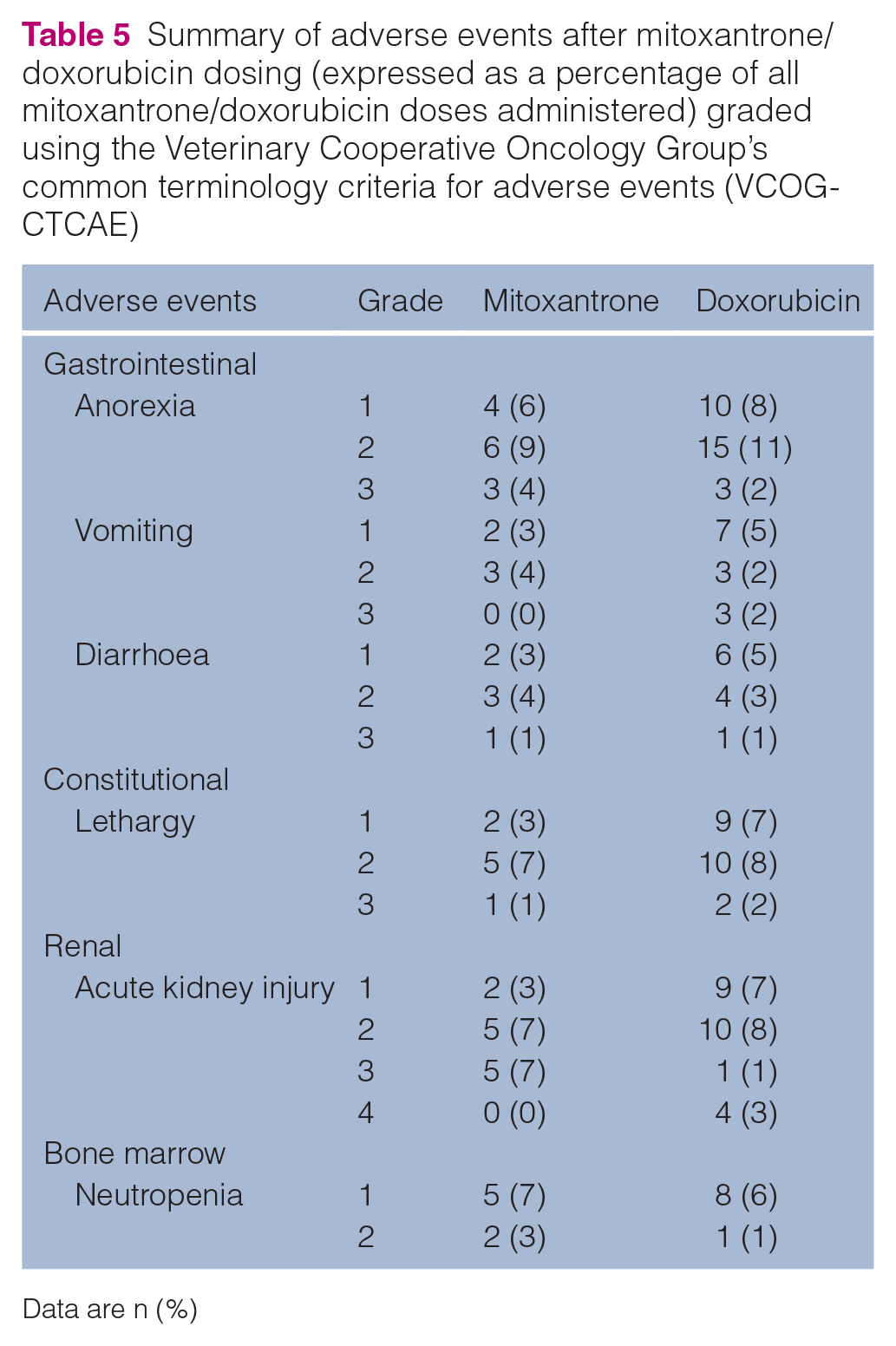
Data are n (%)
Discussion
Mitoxantrone has previously been described as a rescue agent in feline high-grade gastrointestinal lymphoma 17 and as a single agent in feline lymphoma.2,16 Although the previously recorded response rate of 12% (2/17) is poor, it should be noted that a significant number of those cats received doses in the range of 2.5–4 mg/m2, which is notably lower than the doses used in this cohort of cats. The results of this study corroborate the findings in a recent publication suggesting CMOP is a viable alternative to CHOP 11 in terms of maintenance-free protocols. Mitoxantrone used in a multi-agent setting as a first-line treatment for feline lymphoma seems to demonstrate more clinical efficacy than the initial studies indicated.
It is appealing as a substitute for doxorubicin because of the ease of administration; it can be given as an intravenous bolus, as opposed to having to be administered slowly because of the risk of anaphylaxis/hypersensitivity. 18 The consequence of extravasation of mitoxantrone is also typically less dire than that of doxorubicin, given the limited vesicant properties of mitoxantrone.18,19
The inherent limitations associated with the retrospective, multi-institutional nature of this study were the lack of standardisation and randomisation into treatment groups. To account for the lack of randomisation, we attempted to show that there was no uneven distribution of negative prognostic factors in one group compared with the other. The CMOP cats being older and yet having equivalent outcomes to the CHOP cats only supports to the argument for the substitution of mitoxantrone for doxorubicin in feline multi-agent protocols, given the younger age was found to be a positive prognostic factor in this cohort. Between institutions, data collection points (particularly regarding biochemistry/creatinine measurements) were not consistent. In a minority of cases (n = 4), the initial haematology permitting administration of chemotherapy was not available for review.
Staging was not consistently performed because of the associated costs and the lack of consistent prognostic significance of those results.2,3,20 Immunophenotyping was not routinely conducted because of a lack of clear prognostic significance.21,22 However, a recent publication 23 identified T-cell phenotype as a negative prognostic indicator. It is conceivable, though unlikely, that higher-stage disease and/or T-cell phenotypes were allocated to the CHOP group, skewing results in favour of CMOP. Other potential biases include the potential inclusion of Hodgkin’s-like cases between groups; however, with only one cat possibly fitting this criterion, it is unlikely to have significantly affected the results. Two cats in the CHOP group derived substantial clinical benefit from rescue radiation therapy, which could have biased results in its favour, though the small numbers again make significant skew unlikely.
FeLV and FIV status were not established in the majority of cats in this study. Given that previous reports showed FeLV to be a negative prognostic indicator, 2 this could be significant if a higher number of undetected FeLV-positive cases were allocated to the CHOP treatment group. However, given the recorded prevalence rate of 4.6% 24 in Australian cats with lymphoma, it is unlikely that this significantly impacted the results. FIV prevalence in pet cats in Australia is reported to be higher than in other developed countries, in the range of 8–15%,25,26 and up to 20% in cats with lymphoma. 24 Although the distribution of FIV-positive cats into either treatment group could have influenced outcomes, previous studies have not shown prognostic significance of FIV positivity.21,27
Comparison of toxicity data between cats treated with mitoxantrone and those treated with doxorubicin was challenging. A significant number of cats presented with signs, such as malaise/lethargy, hyporexia, vomiting and diarrhoea before treatment. When these signs occurred during treatment, it was hard to elucidate if they were caused by disease or by treatment toxicity. Examination of records in only the week after administration of doxorubicin/mitoxantrone may have missed cases of delayed toxicity. In addition, toxicities that did not require active management or dosage adjustment were inconsistently recorded.
The outcomes observed in this study are consistent with previously reported results for cats with intermediate- to large-cell lymphoma treated using multi-agent chemotherapy protocols.21,28 A recent study describing CMOP treatment reported better outcomes, likely as a result of the exclusion of early protocol failures. 11 Our study explicitly included these early failures and assigned cats to either the CHOP or CMOP group on an intention-to-treat basis to more accurately reflect the median outcome in treatment-naïve cats. Improved outcomes have also been reported in cats with alimentary lymphoma treated with a novel protocol, 23 though this population may have benefitted from an improved prognosis as many had surgical intervention, 29 and started out with higher LMR. 27 Historically and more recently, feline lymphoma populations out of the Netherlands have also reported enviable outcomes,30,31 which may be attributed to better access to veterinary oncological care, availability of drugs such as pegylated asparaginase 32 or a higher proportion of nasal lymphoma forms.31,33
Further avenues of investigation may include an examination of increased dose intensity of alkylating agents in multi-agent protocols, 23 exploring the inclusion of lomustine in such protocols 34 and comparing modes of chemotherapy delivery (intraperitoneal vs intravenous/oral).30,34
Conclusions
CMOP is a viable and well-tolerated alternative to CHOP for treating feline intermediate- to large-cell lymphoma using multi-agent chemotherapy. There was no significant difference in response rate, PFI or ST between cats treated with mitoxantrone vs doxorubicin. Toxicity rates were similar between the two groups. The poor longer-term outcomes in this study – especially when compared with the outcomes achieved in dogs – highlight the need for novel therapeutics to combat this devastating disease.
Supplemental Material
Table 1
CHOP/CMOP protocol broken down into weeks.
Supplemental Material
Table 2
Bivariate cox regression analysis output for progression free interval.
Supplemental Material
Table 3
Bivariate cox regression analysis output for survival time.
Footnotes
Author note
Data from this study were presented as an oral abstract at the 2023 VCS conference in Reno, Nevada, as well as at the 2024 ANZCVS conference in the Gold Coast, Australia.
Supplementary material The following files are available as supplementary material:
Table 1: CHOP/CMOP protocol broken down into weeks.
Table 2: Bivariate cox regression analysis output for progression free interval.
Table 3: Bivariate cox regression analysis output for survival time.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
