Abstract
Hepatic T-cell lymphosarcoma with involvement of regional lymph nodes and concurrent schistosomiasis were diagnosed in an 11-year-old male neutered mixed-breed dog with a history of chronic weight loss, inappetence, vomiting, and diarrhea. Trematode ova present in the hepatic parenchyma and mesenteric node were surrounded by sheets of neoplastic lymphocytes while those in the intestinal wall were surrounded by large numbers of non-neoplastic lymphocytes. Immunohistochemistry revealed that both the neoplastic and hyperplastic populations were T lymphocytes. The ova were identified by fecal saline sedimentation as Heterobilharzia spp., and fecal ova shedding resolved after praziquantel anthelmintic treatment. The lymphoma progressed despite chemotherapy, and the dog was euthanized after developing neurologic signs and a necropsy was performed. A monomorphic population of neoplastic T cells expanded and replaced normal architecture in the liver and spleen, surrounded nerve roots within the cauda equina, and infiltrated the meninges of the brain. The presence of schistosome ova embedded within neoplastic T-cell infiltrates suggests that, as previously reported in human schistosomiasis, heterobilharziasis may be associated with neoplasia.
Canine lymphosarcoma is one of the most commonly diagnosed neoplasms in dogs, accounting for 5–7% of all canine tumors. 7 Lymphosarcoma may be classified anatomically into multicentric or generalized, alimentary, thymic or mediastinal, cutaneous, and solitary forms, with approximately 80% of dogs having the multicentric form. Approximately 70% of canine lymphosarcomas are of a B-cell phenotype while 10–40% of cases are of T-cell origin. 23 In human beings, the frequency of human T-cell lymphosarcoma in Europe and the United States is estimated to be 12%, while up to 50% of cases in Japan are of the T-cell phenotype. 12 This difference has been partially attributed to the increased incidence of adult T-cell lymphoma associated with Human T-cell leukemia virus type I infection in Japan. Similarly, parasitic infections, including schistosomiasis, have been associated with an increased incidence of neoplasia in human beings.6,17 A causal relationship between schistosomiasis and lymphosarcoma has been proposed in human beings; several cases of splenic lymphosarcoma have been described in patients with advanced hepatosplenic Schistosoma mansoni infection.1,5,10 In addition, a human case of large B-cell lymphosarcoma in the liver with intralesional Schistosoma japonica ova has been documented. 22 The schistosome ova in that report were surrounded by infiltrating neoplastic lymphocytes within the hepatic parenchyma. 22
Schistosomiasis also occurs in dogs where it is caused by Heterobilharzia americana. This trematode parasite utilizes many wildlife species, most often raccoons and dogs, as definitive hosts and aquatic snails as intermediate hosts.2,19 Clinical signs of canine schistosomiasis may include lethargy, weight loss, anorexia, diarrhea, vomiting, polyuria, and polydipsia; reported clinicopathologic abnormalities include hypercalcemia, hyperglobulinemia, anemia, and eosinophilia.8,11,13,18,19,24 In infected dogs, adult schistosoma inhabit and lay eggs in the mesenteric venules. The released ova migrate out of the blood vessel and through the intestinal wall to the intestinal lumen or are spread hematogenously to other tissues, primarily the liver. Intense chronic granulomatous inflammation and fibrosis are usually associated with schistosome ova within tissues.8,11,13,19,24 However, an association between canine schistosomiasis and lymphosarcoma has not been described.
An 11-year-old male neutered mixed-breed dog was presented to The University of Georgia Veterinary Teaching Hospital (Athens, Georgia) with a history of weight loss of 1-month duration, 1 week of inappetence, and 1 known episode of vomiting and diarrhea. The dog predominantly lived indoors but had free access to a fenced yard. Two other dogs that lived in the same environment had no signs of illness. All the dogs in the household had been moved to Georgia from the Texas Gulf Coast region 18 months prior to the onset of clinical signs in the lone sick dog. On physical examination, the dog was quiet but responsive and was slightly thin. Abdominal palpation revealed mild splenomegaly. All peripheral lymph nodes were unremarkable in size and consistency. A complete blood cell count revealed mild thrombocytopenia (228 × 103/µl; reference [ref.] range: 235–694 × 103/µl) and mild basophilia (665 cells/μl; ref. range: 0–140 cells/μl). Serum chemistry analysis showed mild hypoglycemia (62 mg/dl; ref. range: 66–109 mg/dl). Two centrifugal zinc sulfate fecal flotations, wet mount direct smears, and stained fecal smears performed 3 days apart identified no parasites or causative agents to explain the chronic weight loss, vomiting, and diarrhea seen clinically.
Abdominal radiographs and ultrasound revealed mild splenomegaly and a subtle mid-abdominal mass effect. Fine-needle aspirate cytology a of the mid-abdominal mass showed probable reactive lymphocytes with marked eosinophilic inflammation. On laparotomy, mesenteric lymphadenopathy was confirmed, the caudate lobe of the liver was enlarged, and the elongated mid-abdominal mass seen on ultrasound appeared to be a chain of enlarged mesenteric lymph nodes. The small intestine was diffusely congested, with firm tan nodules palpable along the entire length of the mesenteric border. Biopsies of the enlarged liver lobe, jejunum, and mesenteric lymph nodes were obtained.
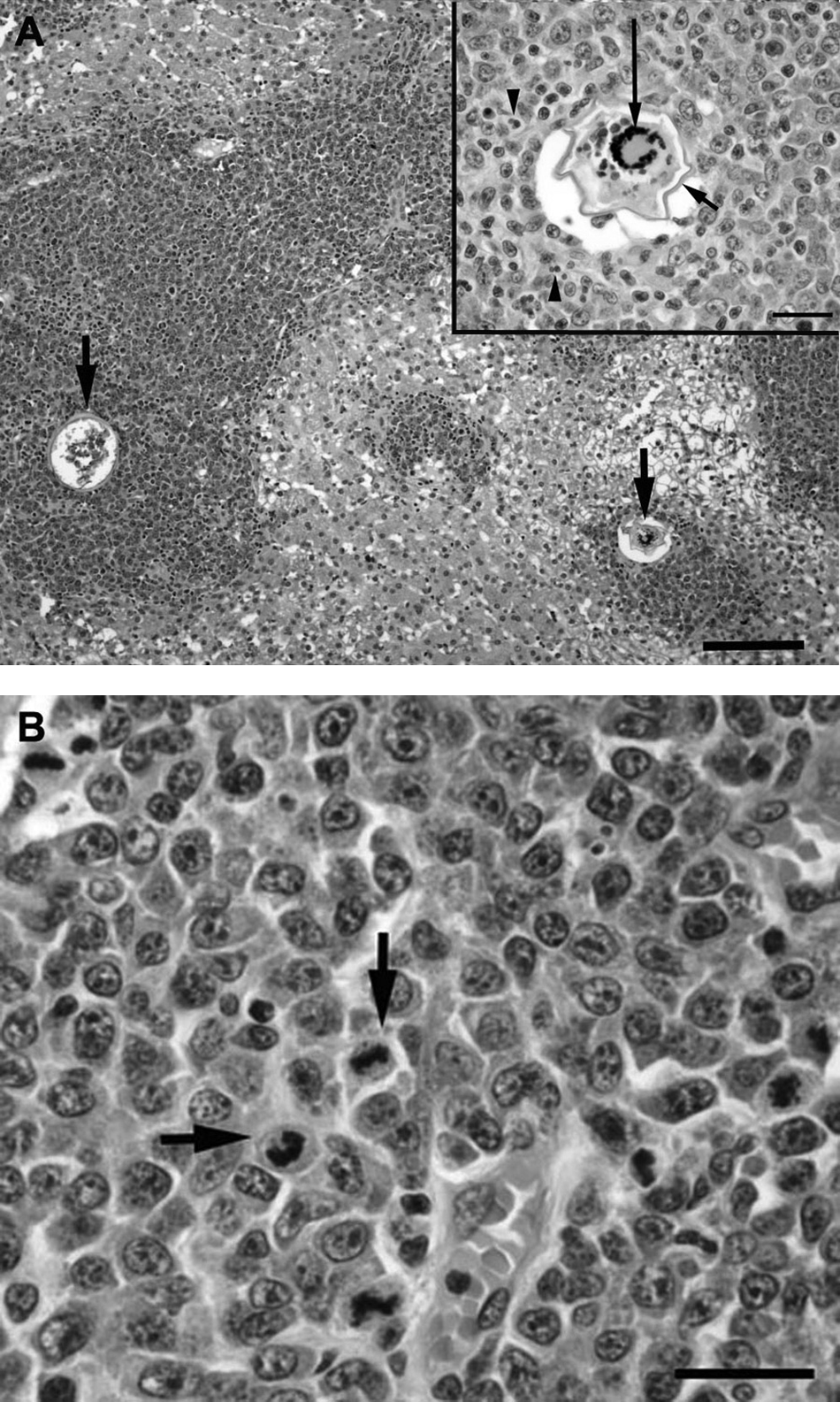
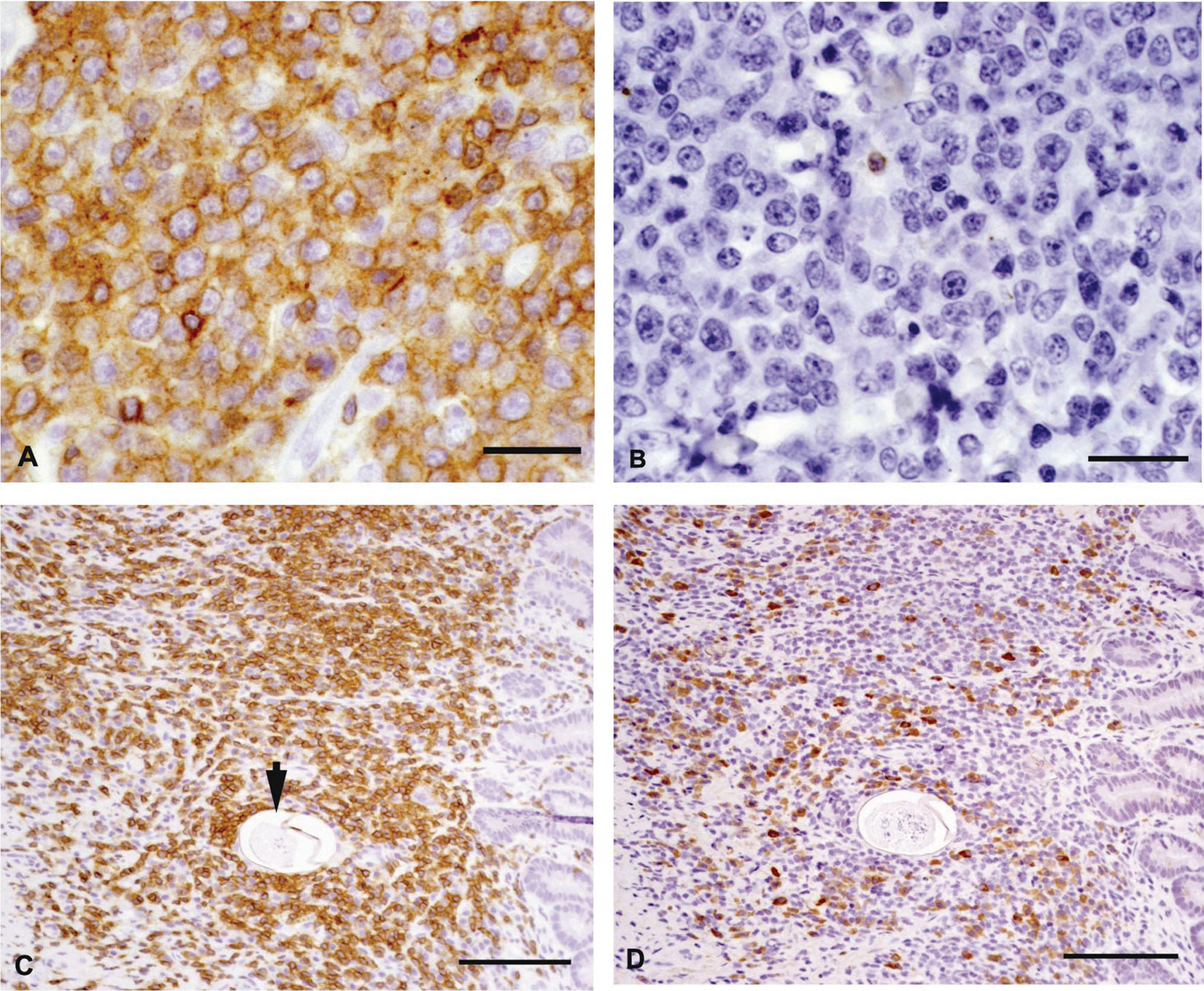
Tissues were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 3 µm, and routinely stained with hematoxylin and eosin. Immunohistochemistry was performed on replicate sections of hepatic and intestinal tissue, with the primary antibodies recognizing cluster of differentiation (CD)3 b and CD79a b antigens. Microscopically, there were extensive centrilobular and portal cellular infiltrates with scattered intralesional ova in the liver (Fig. 1A). The cellular infiltrate was composed of a monomorphic population of large lymphoid cells characterized by large round to oval nuclei, marginated chromatin, single-to-multiple prominent nucleoli, a small amount of cytoplasm, and numerous mitoses (Fig. 1B). Smooth thin-walled trematode ova measuring 60–80 μm in diameter and containing either degenerate mineralized debris or developing miracidia were surrounded by neoplastic lymphoid cells admixed with small numbers of eosinophils, plasma cells, and macrophages (Fig. 1A, inset). Immunohistochemical staining of the neoplastic cell population was positive for the T-lymphocyte marker CD3 and negative for the B-lymphocyte marker CD79a (Fig. 2A, 2B). The jejunum contained multifocal to coalescing aggregates of CD3 immunoreactive cells admixed with fewer CD79a-positive cells, scattered macrophages, and eosinophils within the lamina propria, submucosa, and muscularis (Fig. 2C, 2D). Contained within these areas of transmural jejunal inflammation were parasite ova that were morphologically similar to those demonstrated in the liver (Fig. 2C). The smaller mesenteric lymph node was hypercellular and infiltrated by a mixture of small lymphocytes, eosinophils, and plasma cells while medullary sinuses were expanded by numerous histiocytes. Rare mineralized structures interpreted to be degenerate ova were present within the node. Within the largest mesenteric lymph node previously interpreted to be a mid-abdominal mass, sheets of immature large lymphoid cells were seen. The surrounding nodal tissue was hemorrhagic, presumably from previous fine-needle aspiration. Based on the histopathological and immunohistochemical findings, the diagnoses included T-cell lymphosarcoma of the liver and intra-abdominal lymph node with intralesional trematode ova; severe mural enteritis with intralesional trematode ova; and lymph node reactive hyperplasia with intranodal ova. While the extensive T-cell infiltrate in the jejunum was also suspected to be neoplastic, marked T-cell hyperplasia could not be ruled out.
Mixed-breed dog.
Mixed-breed dog.
As multiple previous routine fecal examinations had been negative despite the presence of trematode ova in tissues, fecal saline (0.9% NaCl) sedimentation was performed, which revealed H. americana ova. Three days following praziquantel c anthelmintic therapy, hepatosplenomegaly had progressed on abdominal palpation. A CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone)-based combination chemotherapy protocol for lymphosarcoma was initiated as described elsewhere.15,26 An initial marked improvement in appetite, stool quality, and normalization of spleen and liver size on palpation was noted; repeat fecal sedimentation 4 weeks into chemotherapy revealed no Heterobilharzia spp. ova. Approximately 7 weeks after initial diagnosis, parapa-resis and left facial nerve paralysis were noted, palpable hepatosplenomegaly had returned, and rapid progression to obtundation and lateral recumbency resulted in humane euthanasia 8 weeks after original diagnosis.
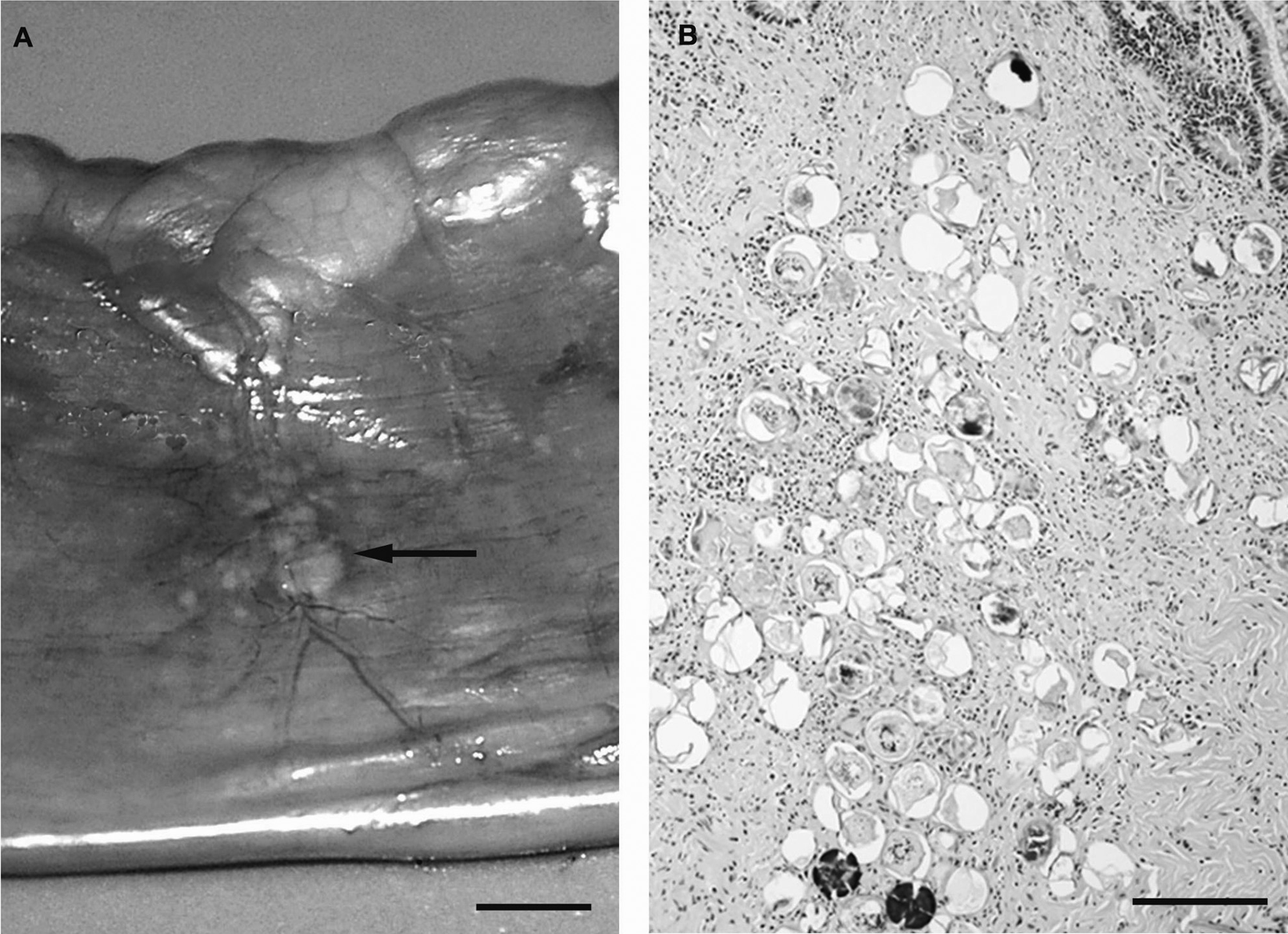
Noteworthy gross necropsy findings included marked splenomegaly, diffuse hepatomegaly, and multiple, 1–2 mm in diameter, raised, white plaques associated with the mesenteric vasculature. The brain and lumbosacral spinal cord were grossly unremarkable. No adult H. americana organisms were detected in mesenteric vessels. Tissue samples harvested at necropsy were processed as described above for the antemortem biopsies. Histopathology revealed sheets of neoplastic lymphoid cells infiltrating centrilobular, portal, and subcapsular liver parenchyma. All examined liver sections showed scattered, mineralized schistosome ova. Normal architecture of the mesenteric lymph node, splenic parenchyma, and bone marrow was effaced by neoplastic lymphoid cells. In addition, neoplastic infiltrates were found in the nerves of the cauda equina and perivascularly within the meninges of the brain. The small intestine contained mural clusters of mineralized and intact schistosome ova corresponding with the raised nodules observed grossly (Fig. 3A, 3B). Fibrous connective tissue, occasional macrophages, and multinucleated giant cells surrounded the egg clusters. Examination of tissue from the site of the previous jejunal biopsy revealed similar degenerate ova and granulomatous inflammation with resolution of the intense transmural inflammation.
Mixed-breed dog.
While schistosomiasis in human beings has been reported to induce neoplastic transformation of tissues chronically infected by the organism, this association has not been previously reported in the dog. The histopathology in the current case report is remarkable, as the location of the Heterobilharzia spp. ova within the sheets of neoplastic T lymphocytes leads to the speculation that the intense T-cell hyperplastic response to the ova may have resulted in malignant transformation of a subset of activated T cells and T-cell lymphosarcoma. The intense T-cell infiltrate in the wall of the jejunum was originally suspected to be neoplastic. However, the infiltrate dissipated following anthelmintic and immunosuppressive chemotherapy, while that in the liver progressed. This response pattern to therapy is consistent with marked hyperplasia in the intestine and neoplasia in the liver.
The anatomic distribution of the T-cell lymphosarcoma in the dog in the current study was unusual. The predominant sites of tumor distribution were the liver, spleen, and regional lymph nodes without involvement of peripheral lymph nodes as typically seen in multicentric lymphosarcoma or intestinal involvement as is present in alimentary forms of the disease. Hepatosplenic T-cell lymphoma (HSTL) has been described as a clinical entity in human beings, and has been reported in dogs.4,14 The disease in both species is characterized by T-cell infiltration of the liver, spleen, and bone marrow without peripheral lymphadenopathy and an aggressive clinical course. The neoplastic T-cells in human beings and in some cases in dogs also express the γδT-cell receptor; immunohistochemical testing for this antigen was not done in the current case. It is also interesting to note that human infection with Human herpesvirus 4 (also known as Epstein-Barr virus) and long-term antigenic stimulation have been identified as possible risk factors for HSTL. 14
In human beings, schistosomiasis has been reported to induce neoplastic transformation of chronically infected tissues. In addition to the association with hepatosplenic lymphosarcoma described above, a strong causal relationship has been demonstrated between Schistosoma haematobium and squamous cell carcinoma of the urinary bladder in human beings.9,17 The urinary bladder lesions begin as granulomas surrounding migrating ova with eventual neoplastic transformation in the overlying urothelium. 6 Excessive oxidative stress and increased DNA damage have been documented in S. haematobium–induced urothelial carcinomas. 25 In one experimental study, S. haematobium total antigen induced sarcomas in subcutaneously inoculated mice, suggesting a specific parasite antigen(s) may be responsible for the neoplastic transformation. 3 The commonly reported distribution of the lymnaeid snail and natural definitive hosts for H. americana makes infection most likely in Gulf Coast states. Based on this distribution, it may be assumed that the dog in the current case report may have contracted schistosomiasis while living in southeastern Texas. This would have theoretically allowed at least 1.5 years of chronic subclinical infection prior to neoplastic transformation of lymphocytes, and subsequent clinical signs of weight loss and enteropathy. However, reports have widened the known range of H. americana to include portions of the southeastern United States. 16 Thus, a less chronic subclinical infection was possible despite the compelling geographic history.
Heterobilharzia americana infection is associated with hypercalcemia in approximately 50% of affected dogs. 8 One case report described increased serum concentrations of parathyroid hormone–related peptide (PTH-rP) with canine schistosomiasis, in the absence of detectable neoplasia; the cause of the increased PTH-rP has not been determined. 13 In addition, PTH-rP mediated hypercalcemia of malignancy is described in 10–40% of canine lymphoma cases.20,26 Specifically, 35% of canine T-cell lymphomas are associated with hypercalcemia. 12 Interestingly, the dog in the present report was not hypercalcemic despite being affected by both diseases simultaneously. Serum PTH-rP concentrations were not evaluated.
There was an initial delay between the anthelmintic treatment and the institution of chemotherapy for lymphoma in the dog in the current study in order to determine if resolution of the parasitic infection would affect the neoplastic disease. In human beings, a causal relationship between chronic Helicobacter pylori infection and gastric lymphoma has been clearly established. 21 Complete remission of low-grade gastric lymphoma after H. pylori infection occurs in approximately 75% of cases, with occasional complete remission of high-grade gastric lymphomas also reported. 21 Unfortunately, while negative fecal sedimentation at follow-up and the lack of detectable adult schistosomes in the mesenteric and portal vasculature at necropsy indicated resolution of the active parasitic infection, this resolution had no apparent effect on the lymphoma, which rapidly progressed despite institution of chemotherapy.
In conclusion, the present case illustrates an association between H. americana and lymphosarcoma in a dog. A similar association with neoplasia has been demonstrated in human schistosomiasis. The present case also demonstrates the need for performing fecal saline sedimentation in dogs with chronic diarrhea, weight loss, and vomiting that are from areas in which canine schistosomiasis has been documented, as schistosome ova will not be observed with routine fecal flotation techniques. Early detection and treatment of schistosomiasis may prevent chronic complications, possibly including tumor development, in affected dogs.
Footnotes
a.
Modified Romanowski, Wescor Inc., Logan, UT.
b.
Dako North America Inc., Carpinteria, CA.
c.
Droncit®, Bayer Healthcare LLC, Animal Health Division, Shawnee Mission, KS.
The author(s) declared that they had no conflicts of interests in their authorship and publication of this contribution.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
