Abstract
Clinical, laboratory and tissue findings from 37 horses with lymphoma were investigated. Horses ranged in age from 0.3 to 20.5 years (median 5.0 years) and included 18 females and 19 males. Weight loss (n = 25) and ventral edema (n = 21) were the most common historical and physical abnormalities. The most common laboratory abnormalities were hyperfibrinogenemia (n = 26), hypoalbuminemia (n = 19), anemia (n = 19), leukemia (n = 14), hyperglobulinemia (n = 13), and thrombocytopenia (n = 13). Thirty-four tumors involved multiple lymphoid tissues and abdominal or thoracic organs, and 3 tumors were restricted to cutaneous and subcutaneous sites. Histopathologically, all tumors diffusely effaced normal lymph node architecture. Tumor cell morphology was heterogeneous in 17 tumors, and 8 tumors had marked histiocytic and multinucleated giant cell infiltrates. Extensive necrosis or focal fibrosis was present in 22 and 4 lymphomas, respectively. Staining of tumor sections with antibodies against CD3 and CD79α molecules resulted in classification of T-cell (n = 26) or B-cell (n = 7) origin. Four tumors could not be classified. Most T-cell tumors comprised small to medium CD3+ lymphocytes, whereas 5 of 7 B-cell tumors were infiltrated by numerous small T lymphocytes and classified as T-cell-rich B-cell lymphoma. Neither estrogen nor progesterone receptor expression was consistently identified by immunochemical assessment of tumor tissues. Fresh tumor cells from 6 horses bound antibodies reactive with equine CD4, CD5, CD8, CD21, or major histocompatibility class II molecules, confirming T-cell (n = 5) or B-cell origin (n = 1). These findings suggest that T-cell lymphoma is more common than B-cell lymphoma in horses and that inflammation, possibly from tumor cytokine production, is frequent.
Equine lymphoma is a heterogeneous disease entity with much variation between animals in presenting signs, clinical course, laboratory data, and pathologic findings. 6, 8, 26, 28, 31, 32, 43 Hemolymphatic tumors in horses appear to be relatively uncommon, but lymphoma is among the most frequent. 37 Involvement of retroviral or corynebacterial infections in malignant transformation has been speculated but remains unproven because transmission experiments failed to induce tumors. 7, 38, 42 Lymphoma has been reported in horses ranging from fetal to advanced age, and limited surveys have identified neither gender nor breed predispositions. 13, 31 Typically, tumors are multicentric and involve lymph nodes and thoracic and abdominal organs. 26, 31, 43 Solitary tumors at extranodal sites have been reported to occur in the tongue, bladder, leg, and ovary but appear to be much less common than multicentric forms. 12, 24, 34, 40 Subcutaneous forms of lymphoma that regress and recur in response to steroid hormone stimulation have been described. 15, 16 In these cases, a small proportion of tumor cells expressed nuclear progesterone receptors, which was implicated to account for cell proliferation in response to hormone binding. 16 Characterization of tissues predominantly from abattoir submissions suggested that most horse lymphomas were of B-cell origin and that presence of 40–80% small T lymphocytes was common. 18 Tumors with such T-lymphocyte infiltrates composed approximately half of all B-cell lymphomas and were classified as T-cell–rich B-cell lymphoma (TCRBCL) because the T lymphocytes were considered to be benign and the B lymphocytes malignant. 18
Paraneoplastic conditions, such as immune-mediated hemolytic anemia, thrombocytopenia, mucosal ulceration, pruritus, and alopecia, have been reported in horses with lymphoma, but their frequency among affected horses is undetermined. 1, 9, 22, 25, 33 Furthermore, some horses with lymphoma were reported to have immunoglobulin deficiencies. 11, 30 Most horses with multicentric lymphoma are euthanized because of poor condition weeks to months after diagnosis, but the natural history of subcutaneous lymphoma could involve months to years with regression and redevelopment of masses. One horse with an apparent solitary mediastinal tumor was successfully treated with chemotherapy for 10 months. 36 Thus, lymphoma in the horse appears to be a clinically and pathologically heterogeneous neoplasm with features distinct from lymphoma in other species.
A modern and widely accepted classification scheme of lymphoma in humans is based on morphologic, immunophenotypic, genetic, and clinical features of the neoplasm. 14 This flexible format allowed variable weighting of a wide range of tumor characteristics, emphasized the consistent presence of unique characteristics, and resulted in a highly reproducible classification scheme that incorporated the natural history of the tumor. 14, 39 Classification systems for lymphoma of animals, such as adaptations of the Working Formulation or the Kiel classification, most commonly relied on morphologic features of the tumor and, in some instances, incorporated assignment of T- or B-cell origin or proliferative indices. 21, 41, 44 Limited ability to distinguish behaviorally distinct neoplastic lymphocyte populations, lack of recognition of molecular changes associated with specific types of lymphoma, and incomplete outcome assessments have limited establishment of prognostically useful, reproducible, and widely accepted classification schemes for lymphoma in animals. However, progress in comparative genomics and analysis of animal genomes might allow eventual development of such comprehensive schemes in the future. 23, 29 In this study, comprehensive assessment of 37 horses with lymphoma is reported.
Materials and Methods
Cases
Thirty-seven horses with lymphoma and complete history, physical examination, histopathologic assessment, and follow-up information were identified at the Veterinary Teaching Hospital or the Animal Health Laboratory of the University of Guelph between 1990 and 2005 for inclusion in this study. Formalin-fixed tissues for histopathologic evaluation were available for each tumor. For 19 horses, neoplastic cytopathology specimens were reviewed, and for 6 horses, flow cytometric assessment of malignant effusions was performed. The diagnosis of lymphoma was based on histopathologic assessment of biopsy or necropsy samples.
Laboratory assessment
Blood samples from horses were assessed in an automated hematology analyzer (Cell Dyn 3000, Abbott, or Advia 120, Bayer, both Mississauga, ON, Canada) and by manual differential counts. Pathologists reviewed blood and bone marrow smears. Serum clinical chemistry analysis was performed on automated analyzers (Dacos, Coulter or Hitachi 911, both Mississauga, ON). Fibrinogen was measured as the refractometric difference after heat precipitation of plasma proteins. Serum protein electrophoresis was performed on cellulose acetate films stained with amido black.
Cytopathology and histopathology
Cytopathologic tumor samples were obtained by tissue aspiration or imprinting, and effusion fluid samples were prepared by cytocentrifugation and direct smear. All slides were stained with Wright's stain and routinely reviewed. Tissue samples were fixed in 10% formalin, embedded in paraffin, and stained with hematoxylin and eosin (HE). Sections with histiocytic infiltrates or multinucleation were further assessed after Ziehl–Neelsen and Periodic Acid Schiff (PAS) staining. Assessments of tumor tissue architecture (diffuse or follicular), nuclear shape, presence of necrosis, fibrosis or inflammation, and estimates of cell size and mitotic index were performed on HE-stained sections as previously described. 18 Lymphocytes with nuclei <1.5 red blood cell (RBC) diameters were classified as small, 1.5–2 RBC diameters as medium, and large lymphocytes had nuclei >2 RBC diameters. Tumor cell size was defined according to the predominant (>70%) cell population; if small and large cells were present in roughly equal proportions, the lymphoma was classified as a mixed-cell tumor. The mitotic figures were counted in 10 fields at 40× magnification, averaged, and an index of “low” was assigned for 0–2, “intermediate” for 3–5, and “high” for >5 mitotic figures per field.
Immunochemistry
Immunohistochemistry was performed on 4-μm sections mounted on silane-treated glass slides. Control tissues were normal equine lymph node, spleen, small intestine, and diestral and estral endometrium. Rabbit polyclonal antibody against CD3 (DakoCytomation, Mississauga, ON) and monoclonal antibodies against CD79α (clone HM57, DakoCytomation), BLA36 (clone A27-42, BioGenex, San Ramon, CA), and estrogen and progesterone receptors (ER and PR; clones 1D5 and 1A6, DakoCytomation) were applied to separate tissue sections. These antibodies are directed against human antigens but labeled appropriate cells in equine tissue sections. Omission of the primary antibody was used as a negative reagent control in all procedures with monoclonal antibodies; for CD3, nonimmune rabbit serum with protein concentration equivalent to the anti-CD3 antiserum was substituted for the primary antibody. Prestaining treatment involved 3 sequential xylene washes to deparaffinize the sections, followed by serial graded ethanol washes for rehydration. Sections were treated with 3% hydrogen peroxide for 10 minutes to block endogenous tissue peroxidases. Heat-induced epitope retrieval was accomplished with a commercial steamer and tissue sections incubated in target retrieval solution (pH 6.0; DakoCytomation) at 95°C for 20–45 minutes. After cooling to room temperature over 10 to 20 minutes, sections were treated with universal blocker (DakoCytomation). Target retrieval was not performed for BLA36 staining. Tissue sections were incubated for 30 minutes at room temperature with primary antibody. Both streptavidin–biotin (LASB+, DakoCytomation), and non–streptavidin–biotin horseradish peroxidase–labeled polymer (EnVision HRP, DakoCytomation) secondary antibody systems were used in CD3 and CD79 assays, with identical results in control tissues. After primary antibody incubation in streptavidin–biotin procedures, tissues were incubated with link antibody for 10 minutes, followed by a 10-minute incubation with streptavidin–peroxidase and 5-minute incubation with diaminobenzidine. In the non–streptavidin–biotin method, sections were incubated with polymer for 30 minutes followed by 10-minute incubation with Nova red chromogen (Vector Laboratories, Burlington, ON, Canada). Slides were then counterstained with Harris hematoxylin and mounted with a nonaqueous permanent mounting medium.
In 25 horses, multiple tissues with lymphoma were assessed by immunohistochemistry. Three pathologists, unaware of the case history and clinical data, independently assessed immunochemical results for each horse. Immunophenotypic assignment was based on an estimate of the percentage of positively staining cells for T- and B-cell markers in each tumor and the morphology and distribution of stained cells. The proportion of nonstaining cells in tumors was noted. Tissues stained for ER and PR expression were examined in a similar manner.
Neoplastic effusions from 6 tumors were assessed with fluorescent antibodies reactive with equine CD4, CD5, CD8, CD14, CD21-like, and major histocompatibility class (MHC) II antigens (all from Serotec, Raleigh, NC) in a FACScan cytometer (BD Biosciences, Oakville, ON, Canada).
Results
Cases
Complete data were available for 37 horses with lymphoma. These included 19 male and 18 female animals ranging in age from 0.3 to 20.5 years. Eighteen horses were ≤5 years old (Fig. 1). The most common historical abnormalities were weight loss (n = 25), edema (n = 21), and respiratory disease (n = 10). Physical exam identified edema in 25 horses, abnormalities of the respiratory system in 12 horses, and enlarged peripheral lymph nodes in 9 horses.
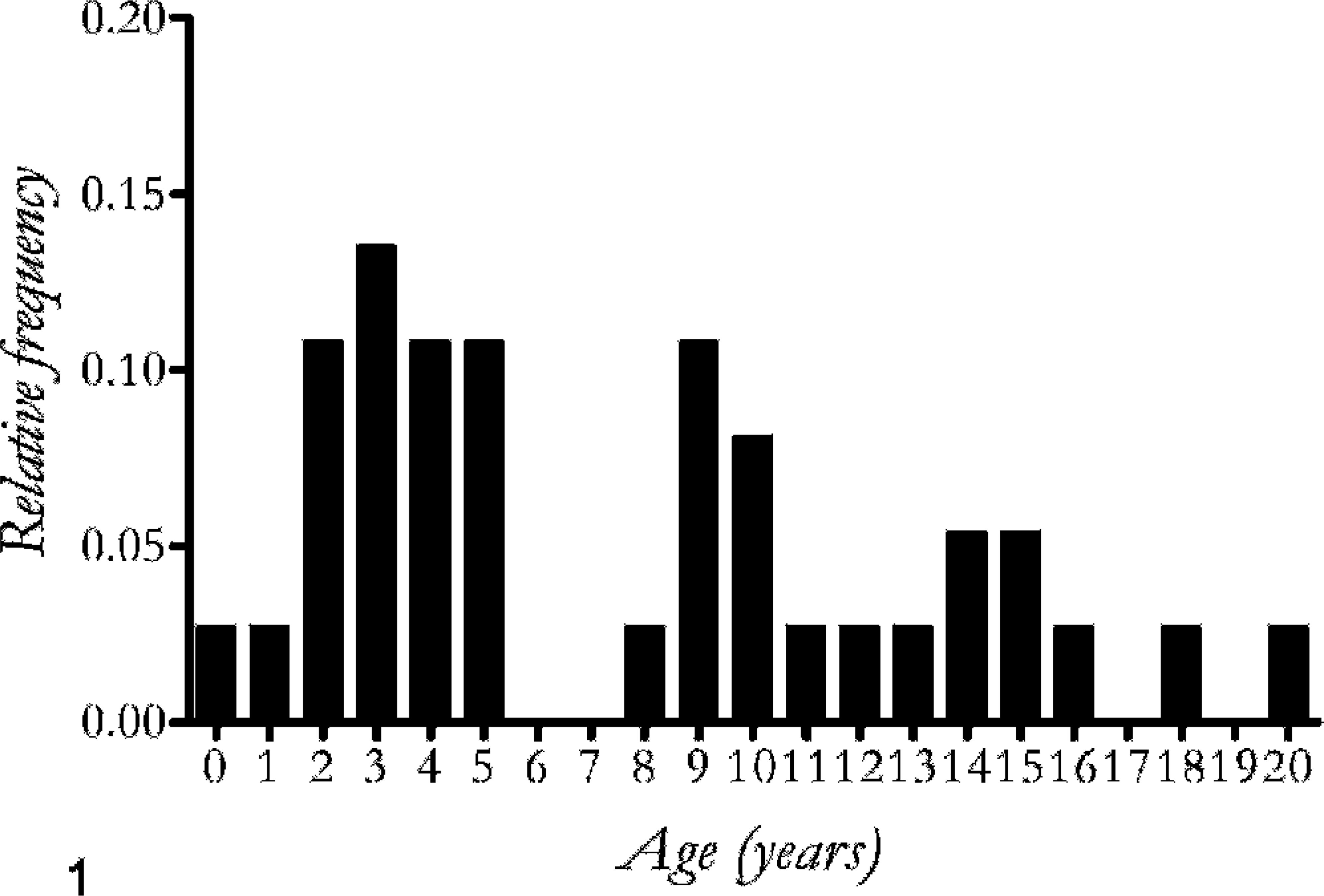
The age frequency of 37 horses diagnosed with lymphoma ranged from 0.3 to 20.5 years; median 5.1 years, mean 7.5 years.
Laboratory assessment
Hematologic and clinical chemistry data were available from 34 horses. Cytopenia was identified in 23 animals and consisted of anemia (n = 19), thrombocytopenia (n = 13), neutropenia (n = 5), or a combination of conditions (Table 1). The anemia was frequently severe (hematocrit 0.28–0.44 [liter/liter], n = 14), and slide agglutination was present in 11 samples. Leukemia was diagnosed from 14 blood samples (Fig. 2). Six horses had mild lymphocytosis ranging from 1.3 to 4.7 × 109 lymphocytes/L, and 8 horses had normal lymphocyte concentrations but cells with abnormal morphology. Two horses had erythrocytosis (hematocrit 0.60 and 0.82 [liter/liter]. The most common clinical chemistry changes were increased fibrinogen (n = 26), decreased albumin (n = 19), and increased globulin (n = 13). Serum electrophoresis of 5 hyperglobulinemic serum samples (85–147 g/liter, reference interval 58–75 g/liter) identified polyclonal (n = 3) and monoclonal (n = 2) protein patterns.
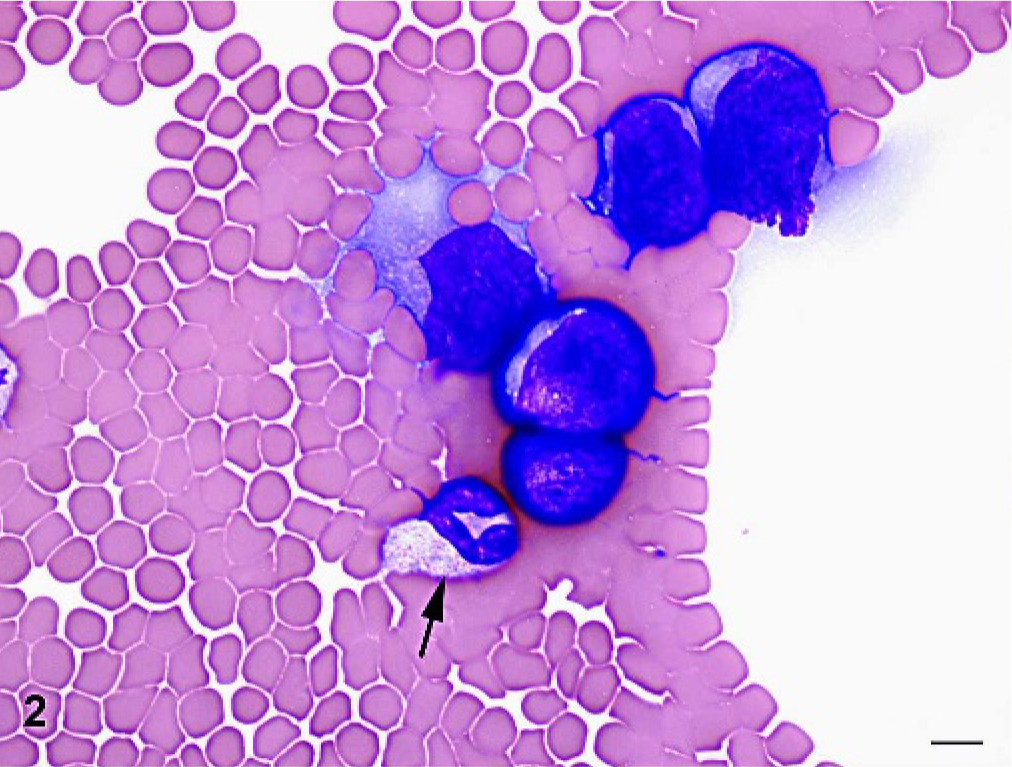
Venous blood; horse with T-cell lymphoma. Mild lymphocytosis with leukemic lymphocytes characterized by large size and basophilic cytoplasm. A single neutrophil is present (arrow). Wright stain. Bar = 10 μm.
Laboratory and tumor characteristics in horses with lymphoma.
Assessed in 24 horses with T-cell, 6 horses with B-cell, and all horses with unclassified lymphoma.
Features of the presumed neoplastic cell population.
Cytopathology and histopathology
Cytopathology of tumor specimens or effusion fluid established a diagnosis in 9 of 17 specimens. Pleural effusion fluid was evaluated in 8 of the 9 cases in which a diagnosis was established by cytopathology (Fig. 3). Bone marrow aspirates and biopsies were evaluated in 5 of 12 horses with leukemia, and identified tumor myelophthisis in 4 horses. Antemortem histopathologic assessment of a tumor biopsy established a diagnosis of lymphoma in 24 horses, although in 2 horses, biopsies from enlarged mesenteric lymph nodes was initially interpreted as “granulomatous inflammation.” Four animals were euthanized without biopsy because of identification of abdominal masses by rectal palpation or imaging.
Pleural fluid smear; horse with T-cell lymphoma. Marked anisocytosis among tumor lymphocytes, apoptotic cells (arrowhead), mitotic cell (arrow). Wright stain. Bar = 10 μm.
The majority (n = 30) of lymphomas were multicentric, involving thoracic and abdominal lymphoid tissues, liver, spleen, intestine, or other abdominal organs (Table 1). Four tumors were restricted to the abdominal cavity, and 3 tumors involved subcutis, muscle, peripheral lymph nodes, or a combination of tissues. In most lymphomas with thoracic involvement, the predominant clinical sign of respiratory disease was referable to a large thoracic mass (range 15–36 cm diameter) associated with a large volume of effusion fluid.
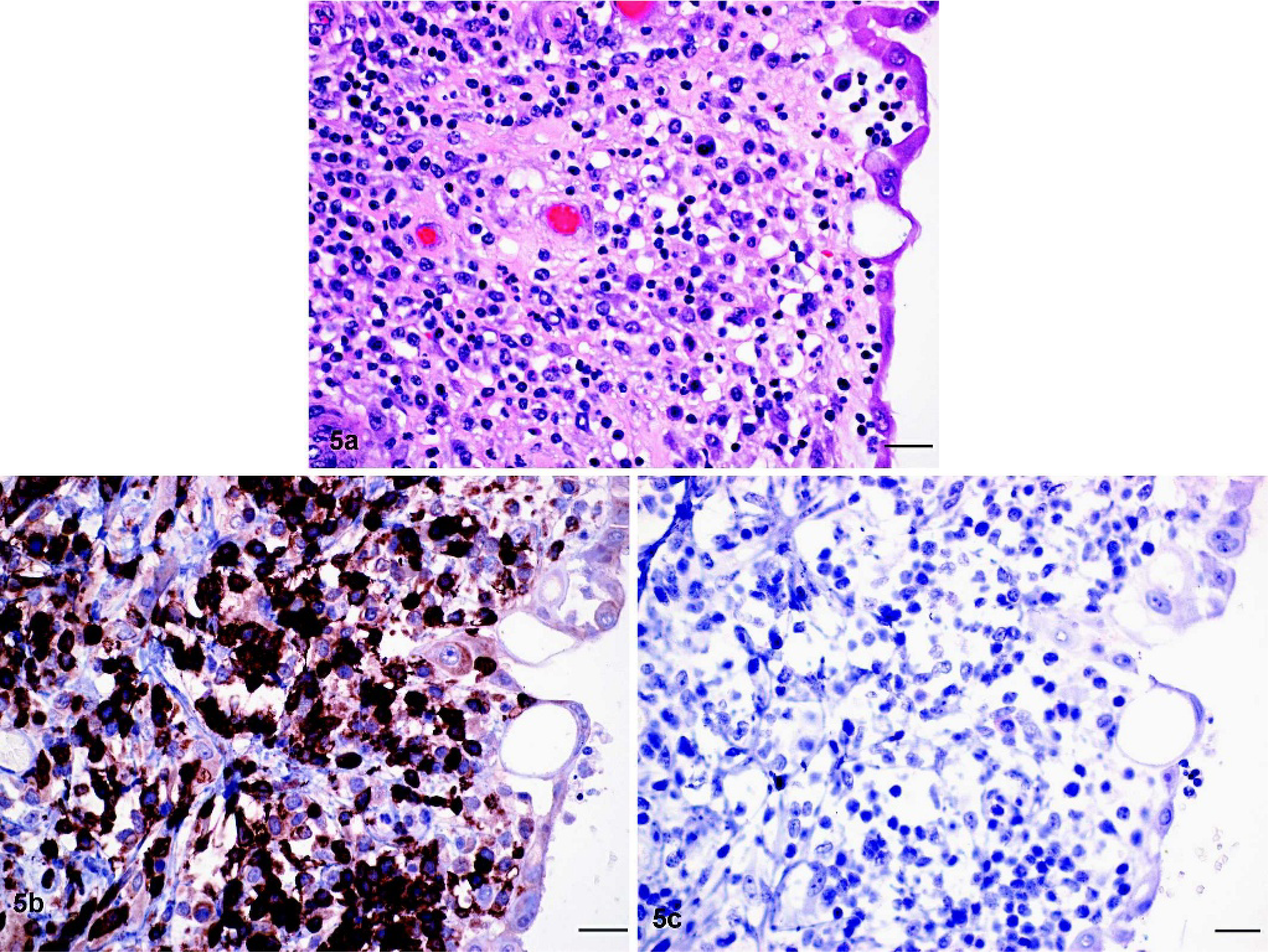
Tumor architecture was diffuse in each case, although remnant follicular structures were present in 8 of 24 tumors affecting lymph nodes. Most tumors (n = 22) had areas of necrosis, apoptosis, or both, and in some cases (n = 8), these were associated with numerous histiocytes as well as multinucleate giant cells. The number of nuclei in the latter cells ranged from 10 to 20, and nuclei were arranged peripherally and circumferentially within the giant cells (Fig. 4). Organisms were not identified in these tumors after Ziehl–Neelsen and PAS staining. Fibrosis was less common (4 tumors) and varied from diffuse finely fibrillar eosinophilic extracellular stroma to wide bundles of mature collagen. Plasma cells were present singly or in large clusters throughout 10 tumors (Table 1) but were morphologically homogeneous and lacked mitotic activity. Twenty tumors consisted of a relatively uniform population of medium to large lymphocytes, arranged closely together with few interspersed inflammatory cells or stromal tissue. These lymphocytes had little cytoplasm, and nuclear shape varied from round to oval to convoluted. Seventeen tumors consisted of a pleomorphic population of round cells that ranged from small lymphocytes with dense chromatin and sparse amounts of cytoplasm to large cells with round or angular nuclei, vesicular chromatin, and abundant pale cytoplasm (Fig. 5a). Cells in many of these tumors were arranged loosely. All tumors involving subcutaneous tissue, muscle, and superficial lymph nodes (n = 3) had extensive infiltrates of large histiocytic cells but did not involve the epithelium.
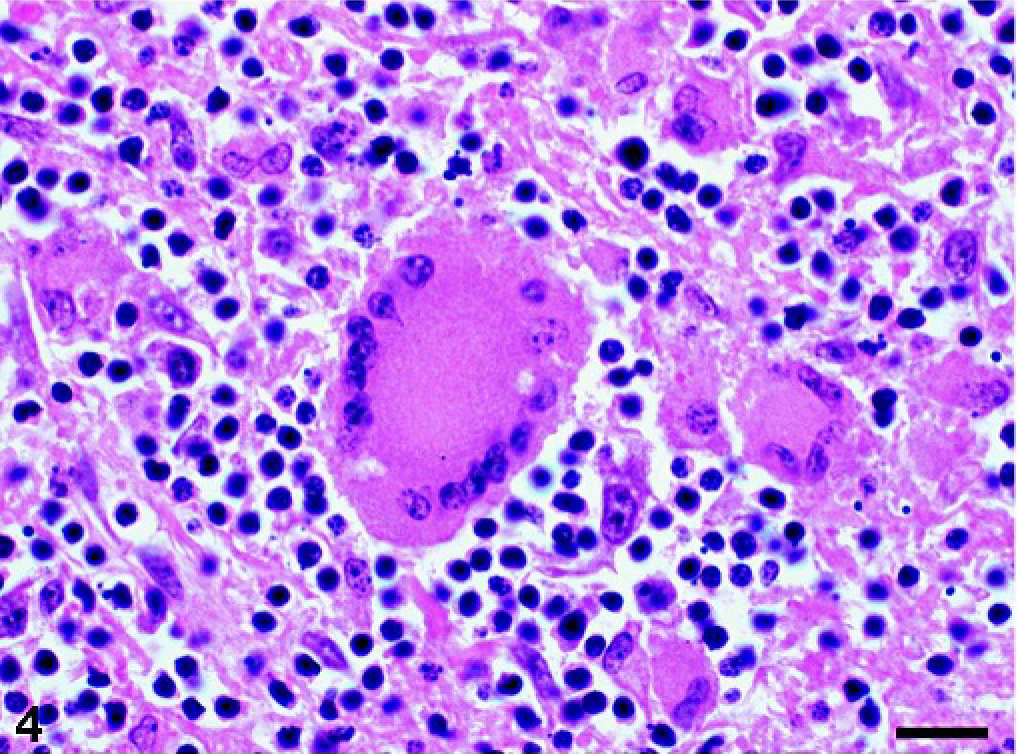
Neoplastic mesenteric lymph node; horse with T-cell lymphoma. Multinucleated giant cell surrounded by histiocytes and lymphocytes. HE stain. Bar = 20 μm.
Thoracic mass; horse with T-cell lymphoma.
Immunochemistry
Control tissue sections stained with CD3, CD79α, PR, and ER antibodies stained the appropriate cells or nuclei in lymph node and uterine tissues. Staining with the BLA36 antibody was inconsistent in control and neoplastic tissues. Positive cells were much fewer than those stained with CD79α antibody and were scattered, single, large cells with ill-defined cell borders and occasional nuclei stained. Therefore, this antibody was not used for lymphoma classification.
Twenty-six tumors were assigned T-cell origin because of expression of CD3 on the predominant cell population (Table 1). These tumors consisted of small to medium-sized lymphocytes with a moderate amount of cytoplasm. In 19 of these tumors, lymphocytes were tightly packed, whereas in the remainder, lymphocytes were loosely arranged with variable amounts of extracellular matrix. The mitotic rate ranged from low to intermediate, and tumor necrosis was common. More than 80% of cells stained positively for CD3 in 20 T-cell tumors, and either none or only cells in remnant follicular structures stained with the CD79α antibody (Fig. 5b, c). In 6 tumors, the proportion of CD3-staining lymphocytes was lower (50–80%), but these were still classified as T-cell tumors because tumor invasion or metastasis into extranodal tissues (liver, mesenteric fat, kidney, heart) was exclusively due to CD3-expressing lymphocytes; CD79α+ cells were of small size, occurred in clusters, and composed fewer than 20% of cells in the mass. Nine T-cell tumors involved the intestine in addition to other abdominal and thoracic tissues (Fig. 6).
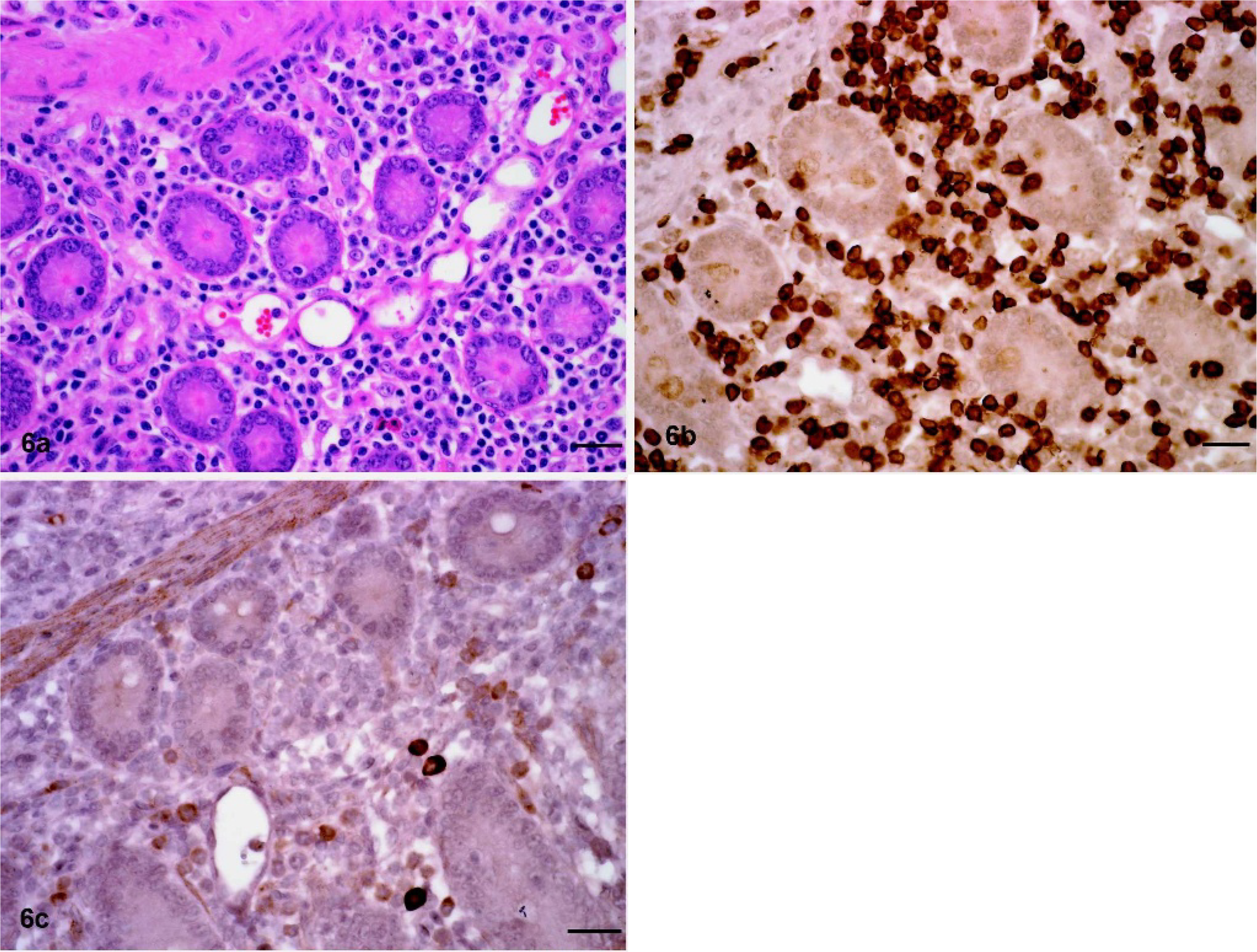
Intestinal mass; horse with T-cell lymphoma.
Seven tumors were classified as B-cell origin because of either diffuse expression of CD79α (n = 2) or expression of the antigen by individual, very large, morphologically atypical and mitotically active cells (n = 5). B-cell tumors were either composed of homogeneously large cells with a high mitotic rate or of a pleomorphic population in which small lymphocytes outnumbered large lymphocytes (Fig. 7a). In the latter cases (n = 5), the large cells stained for CD79α and the small lymphocytes for CD3, and the tumors were classified as TCRBCL (Fig. 7b, c). This category included 2 tumors that affected predominantly skin and underlying tissue with involvement of local lymph nodes, 2 tumors that involved the heart and diaphragm in addition to thoracic and abdominal lymphoid tissue, and 1 tumor that involved multiple abdominal organs and skeletal muscle. The homogeneous B-cell tumors (n = 2) involved multiple abdominal and thoracic organs, including the small intestine (n = 1).
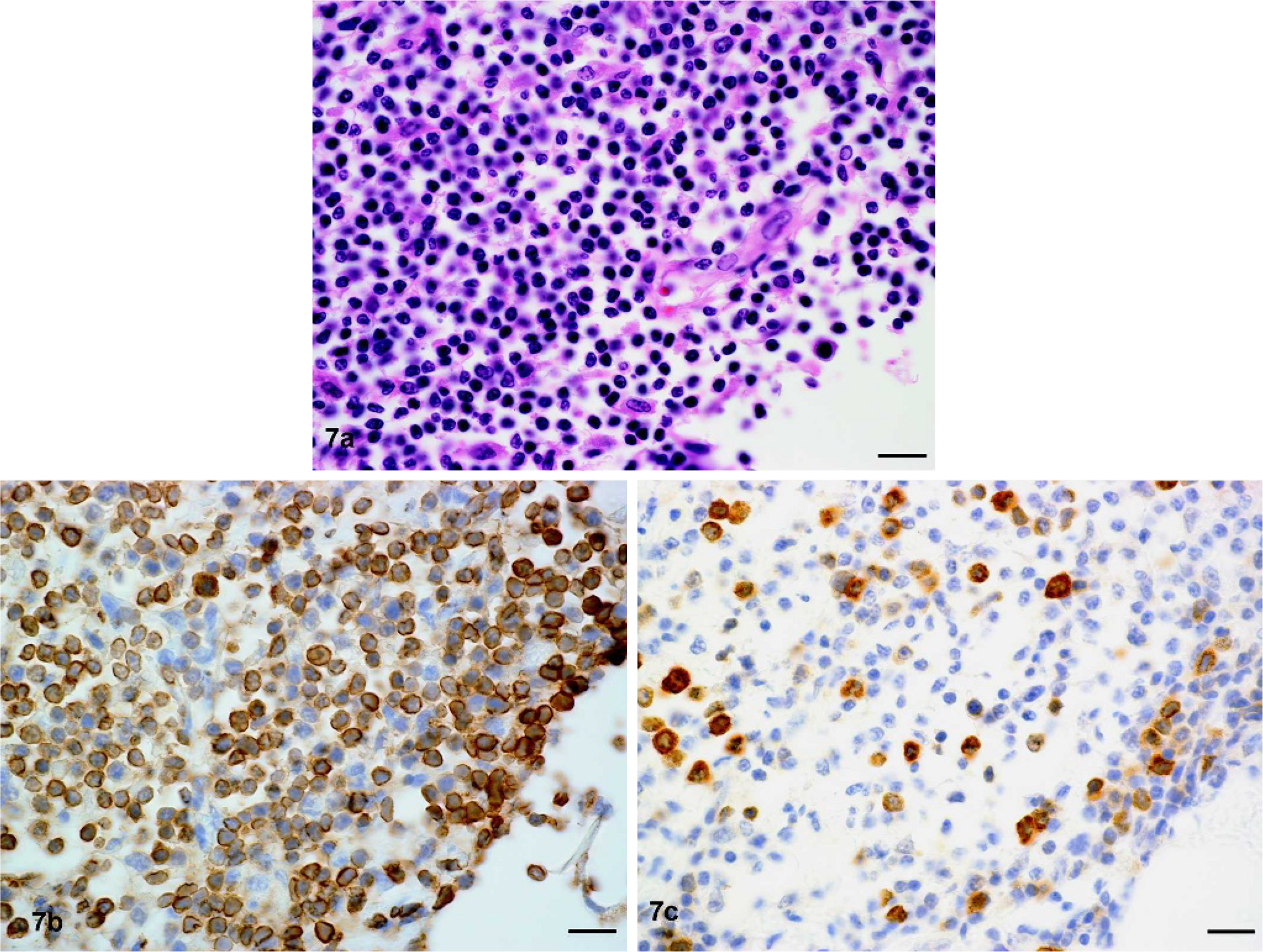
Mesenteric mass; horse with T-cell–rich B-cell lymphoma.
Four lymphomas could not be classified as of either T- or B-cell origin because less than 2% of cells expressed CD3 or CD79α (n = 3) or because both antigens were expressed concurrently by most tumor lymphocytes (n = 1). Tumors that did not stain immunochemically were pleomorphic with extensive areas of necrosis. Lymphoma in one of these horses was associated with hyperglobulinemia and a monoclonal serum electrophoretic pattern.
Neither ER nor PR receptors were immunochemically identified in tumor tissue aside from rare, single, positively stained nuclei (<0.1% of cells) in 2 B-cell tumors. Flow cytometric assessment of 6 samples of fresh neoplastic effusion fluid with a panel of antibodies identified surface expression of CD5 and MHC II on 4 T-cell lymphomas; CD4, CD5, and MHC II antigens on 1 T-cell lymphoma; and CD21 and MHC II antigens on 1 B-cell lymphoma.
Seven horses with a diagnosis of lymphoma were treated with glucocorticoids and euthanized 1–3 months (n = 5) or 7–8 months (n = 2) after diagnosis because of clinical deterioration.
Discussion
Equine lymphoma has been recognized as a lymphoproliferative disease with unique features for decades. 6, 27, 28, 32 Relatively infrequent enlargement of peripheral lymph nodes; typical multicentric distribution; and extensive extranodal organ involvement, heterogeneous morphologic features, and associated hematologic abnormalities have previously been recognized in several surveys. 6, 27, 28, 31, 32 Morphologic evaluation and classification into T- or B-cell origin were reported for 31 tumors, derived largely from slaughterhouse specimens. 18 In the current study, clinical, laboratory, and morphologic findings of a large series of equine lymphoma tumors were evaluated.
The ages of affected horses ranged from neonatal to old, and gender preference was not identified. Lymphoma is a neoplasm affecting all ages in animals and humans, with relatively less differentiated lymphocyte phenotypes predominant among pediatric patients. 3 Similarly, a report of lymphoma in an aborted equine fetus and in 2 horses <1 year old in this study suggest that potentially congenital genetic changes could contribute to tumor development. 13 Juvenile horses in this study were too few to assess the natural history in this age group, but each foal was in poor condition, with tumor involvement of multiple organs and leukemia. More than half of horses in this study were 5 years old or younger, suggesting that lymphoma is relatively common in younger horses.
Laboratory findings in horses with lymphoma confirmed a high frequency of abnormalities that were previously suggested from individual case reports. 1, 24, 25, 33, 43 Cytopenia affecting 1 or several hematopoietic cell lineages occurred in 23 of 34 horses. Anemia was most common and frequently was associated with agglutination of RBC and hyperglobulinemia. The cause for anemia might be premature destruction of RBC coated with antibody, inadequate RBC production because of myelophthisis, or a combination of both mechanisms. Agglutinating anemia occurred in horses with T- and B-cell lymphoma and might therefore result from paraneoplastic polyclonal rather than neoplastic monoclonal antibody production. Anemia was not restricted to horses with intestinal involvement by lymphoma. Thrombocytopenia and neutropenia occurred less frequently but were associated with bone marrow infiltrates in each case. Leukemia was diagnosed from blood smear review in 14 horses, and in each case, it was associated with extensive bone marrow involvement at necropsy. Neutrophilia in 7 horses might have resulted from inflammation secondary to tumor necrosis, and erythrocytosis in 2 horses, not associated with tumor involvement of the lung or blood hypoxia, might have been due to tumor or paraneoplastic erythropoietin production.
The most common clinical chemistry abnormalities were hyperfibrinogenemia, hypoalbuminemia, and hyperglobulinemia. Fibrinogen is an acute phase reactant induced in hepatocytes in response to cytokine stimulation, in particular interleukin (IL)-6, whereas albumin is a negative acute phase reactant. 10 Furthermore, IL-6 is an essential cytokine for induction of immunoglobulin synthesis by plasma cells. 20 Although immunoglobulin isotypes were not quantified in serum samples from horses with lymphoma in this study, polyclonal increases in proteins migrating in the beta and gamma region on electrophoresis likely involved acute phase as well as immune globulins. Therefore, it is conceivable that tumor cytokine production rather than a microbial infection could result in some of the clinicopathologic changes in the horses in this study. Subclinical bacterial infection, in particular mycobacterial infection, could induce such inflammatory changes, but was unlikely because neither abscesses, granulomas, nor microscopic organisms were identified. Dependent edema was a common clinical finding and occurred in horses with decreased, normal, and increased serum protein concentrations. The cause was likely impaired lymphatic drainage because of widespread tumor involvement of lymph nodes rather than reduced oncotic pressure.
Histopathologically, tumors were heterogeneous and it was difficult to assign individual tumors to the distinct categories that were previously suggested for equine lymphoma. 26, 28, 31 For this study, multiple tissues with neoplastic involvement were morphologically and immunohistochemically evaluated for each tumor. Heterogeneity was evident in cellular density and in the presence of necrosis, fibrosis, or inflammation between different tissues of the same animal. Tumors with similar cell morphology (medium size, densely packed, no histiocytes) composed approximately 50% of all tumors and could be analogous to the previously described category of lymphoblastic lymphoma. 31 However, neither “follicular” nor “plasmacytic” nor “lymphoblastic leukemia” types of lymphoma were recognized as distinct entities in this study. Follicular arrangement with homogeneous expansion of germinal center cells or uniform plasmacytoid tumor cell appearance was not observed, although infiltrates of cells of typical plasma cell appearance were common among tumor lymphocytes. Involvement of bone marrow was not associated with any distinct morphologic cell type. Histiocytic lymphoma, composed of regions of tightly packed lymphocytes interspersed by large areas of histiocytes and multinucleated giant cells, made up a distinct category. All tumors of this type were categorized as T-cell lymphomas and had a multicentric distribution. Core needle biopsies of enlarged lymph nodes resulted in several initial diagnoses of granulomatous inflammation, which could be attributed to a relative lack of lymphoid infiltrates in small-size biopsies. Multinucleate giant cells are derived by fusion from monocytic precursors and classified either as Langhans or foreign body giant cells. 2 Morphologically, Langhans cells have <20 nuclei arranged peripherally in a circular fashion, are surrounded by lymphocytes in tissue, and arise by fusion of monocytes in response to indigestible microbial components, interferon-γ stimulation, or both. 2 On the other hand, foreign body giant cells typically have >20 nuclei arranged irregularly throughout the cytoplasm, lack lymphocyte association, and can be experimentally derived from monocyte precursors in the presence of IL-4 and granulocyte-macrophage colony stimulating factor (GM-CSF) or IL-3. 2 The giant cells present in lymphoma tissue from horses consistently had the appearance described for Langhans cells and were frequently in the vicinity of lymphocytes. Thus, it is possible that these multinucleate cells arose in response to interferon-γ production by neoplastic T lymphocytes.
Immunohistochemical staining for markers typically expressed by T or B cells allowed confident assignment of cell origin in 28 tumors. In the remainder of tumors, the proportion of stained lymphocytes was either not uniform or relatively low. Five of these tumors were classified as TCRBCL because of cellular atypia and mitotic activity among CD79+ cells. The proportion of T cells in TCRBCL of humans can range from 10 to 90%, and assignment of B-cell origin was only possible after intricate molecular manipulation to identify clonal receptor gene rearrangement in single microdissected cells. 35 TCRBCL is an uncommon diffuse B-cell lymphoma variant in humans and appears rare in animals other than horses. In a previous study of equine lymphoma, 11 of 31 tumors were classified as TCRBCL, but only 5 of 37 tumors in this study met similar classification criteria. 18 The reasons for this difference are unclear, but the ages of many horses in the previous study were either unknown or advanced relative to horses in this study, and possibly TCRBCL is more common in older horses, as it is in older humans. Although the proportion of cells with proliferation-associated nuclear antigens was estimated in the previous study, concurrent cellular immunophenotype was not determined, and the index did not appear to discriminate between different morphologic lymphoma variants. 18 Therefore, we did not consider such markers to be helpful for classification of equine lymphoma.
Most tumors classified as T-cell origin were associated with large mediastinal masses, and many occurred in animals <5 years of age. T-cell origin of mediastinal lymphoma is recognized across species and, in cats, is frequently associated with retroviral infection. Two lymphomas classified unequivocally as diffuse B-cell tumors were associated with >90% CD79α cells. Phenotyping of effusion fluid with an expanded range of antibodies reactive with surface molecules on equine lymphocytes was consistent with immunohistochemical findings in each tumor and might have merit for future more detailed analyses.
Lack of CD3 or CD79α expression in 3 tumors might have been because of a lack of reactivity of the tissue from prolonged formalin exposure or because of a lack of expression of these molecules by tumor cells. CD79α is part of the B-cell antigen receptor complex and is expressed in all but late differentiation stages of B-cell development. 4 Natural killer lymphocyte populations lack CD3 and CD79α expression. Therefore, lymphomas might arise in horses that cannot be classified with antibodies against these molecules.
BLA36 is a monoclonal antibody derived by immunization of mice with human Hodgkin's lymphoma cells and has been described to interact with a B-cell epitope on equine lymphocytes. 5, 19 However, staining of nonlymphoid cells and inconsistent reactivity with B cells have been observed in human tissues. 17 In this study, with the use of normal equine lymphoid tissues, the antibody labeled cells that resembled macrophages rather than lymphocytes. Therefore, the specificity of the BLA36 antibody for equine lymphocytes appears inconsistent.
Clinical observations have indicated that cutaneous lymphoma in the horse could persist over years without apparent progression or might progress in response to steroid hormone changes. 6, 12, 15 Therefore, we sought to systematically assess equine lymphoma tissues for expression of estrogen or progesterone receptors. However, hormone receptors were rarely identified in neoplastic tissues, which included 2 tumors with cutaneous involvement. Previous reports described such receptors on 1 to 10% of tumor cells, but there was no apparent correlation to the hormone produced by an ovarian tumor. 15, 16 Thus, the link between hormonal states and neoplastic lymphocyte proliferation or regression remains incompletely understood.
A report of a horse with lymphoma treated by chemotherapy indicated a disease-free interval of 10 months, and in this study, glucocorticoid treatment of 7 horses resulted in survival times of 1–8 months. 36 Because many horses eventually diagnosed with lymphoma had weight loss and respiratory impairment for several months and were subject to multiple diagnostic procedures before establishment of a definitive diagnosis, the implication is that some cases of lymphoma in the horse may progress relatively slowly.
Hematologic and clinical chemistry changes were common in horses with lymphoma. Histopathologic features of lymphoma were diverse and frequently included marked histiocytic inflammation. Immunophenotypic assignment was not readily achieved in all tumors on the basis of expression of typical T- and B-cell markers. Assessment of cytokine production by tumor cells, identification of unique genetic changes, and more detailed immunophenotypic assessment might be helpful to better understand and prognosticate this tumor.
Footnotes
Acknowledgements
We thank M. Hazlett for critical review of the manuscript and B. Jefferson for assistance with immunohistochemical methods.
