Abstract
Objectives
The primary goal of this study was to characterize the clinical presentation of feline cutaneous lymphoma. The secondary aims included determining if treatment or initial response to treatment affected the overall survival of patients, and understanding if disease characteristics such as immunophenotype, cell size or the presence of epitheliotropism influenced response to treatment.
Methods
Veterinary medical oncologists at four academic veterinary teaching hospitals submitted cases of feline patients with cutaneous lymphoma diagnosed by histopathology or cytology. Signalment, feline leukemia virus (FeLV)/feline immunodeficiency virus (FIV) status, physical examination findings, clinical signs, diagnostic tests, therapy, response and outcome, and necropsy findings, when available, were recorded.
Results
Forty-one patients were identified and described. The majority of patients were domestic shorthair cats (n = 29). The median age at diagnosis was 12.3 years. Males were over-represented in the population (n = 30). In the majority of patients (n = 33), the FIV/FeLV status was unknown. Twenty patients were fully staged. Thirty-four patients were treated with a variety of modalities, including surgery, radiation, single-agent or combination chemotherapy, or prednisolone only. In multiple patients, surgery or radiation was combined with a systemic therapy. Of 34 patients treated with some form of therapy, 20 responded (achieving either a partial response or complete remission).
Conclusions and relevance
Clinical signs and physical examination findings varied among patients. Response to therapy appeared to be associated with survival (P = 0.0025); however, this population was highly censored. Immunophenotype, cell size and the presence of epitheliotropism did not influence treatment response. Results were limited by small numbers of patients, heterogeneous disease manifestations and treatment protocols. Further studies are necessary to evaluate the effect of specific treatment modalities and disease subtype on prognosis.
Introduction
Feline cutaneous lymphoma is a rare disease, accounting for approximately 0.2–3% of lymphoma diagnoses in feline patients. 1 It primarily affects cats between the age of 10 and 14 years, with no confirmed sex or breed predilections.2–6
Cutaneous lymphoma is more thoroughly described in dogs and is primarily stratified as epitheliotropic or non-epitheliotropic disease. 7 Cutaneous epitheliotropic lymphoma is a histopathologic diagnosis, which is characterized by neoplastic cells exhibiting tropism for the epidermis with secondary extension into the dermis. Epitheliotropic disease can be further classified as mycosis fungoides or pagetoid reticulosis on histopathology, and previous literature suggests that histopathologic subtype may carry prognostic significance in canine patients. 8 In contrast, non-epitheliotropic lymphoma affects the middle and deep portion of the dermis or subcutis, traditionally lacking involvement of the epidermis. Most documented cases in cats are reported to be similar to the traditional mycoses fungoides subset of epitheliotropic tumors; however, a single case report found that in cats with non-epitheliotropic lymphoma, 5/6 cases were T cell in origin. 9 Relatively little is known about the significance of epitheliotropism, cell size or immunophenotype in terms of prognosis for feline patients.
Because of the similar presentation to other diseases, feline cutaneous lymphoma can present a diagnostic challenge. Solitary or multifocal alopecia, erythema, crusting, nodules, ulcers, plaques and/or scaling are often present (Figure 1), and these skin changes can mimic numerous non-neoplastic conditions, including eosinophilic granuloma complex and eosinophilic plaques.1,3–5,10,11 Previous reports suggest that there may be a predilection for development of cutaneous lymphoma lesions on the face, eyelids, mucocutaneous junctions, elbows, tarsus and trunk (Figures 2 and 3). 3 Historically, disease affecting the head and face was thought to exhibit an indolent disease course.
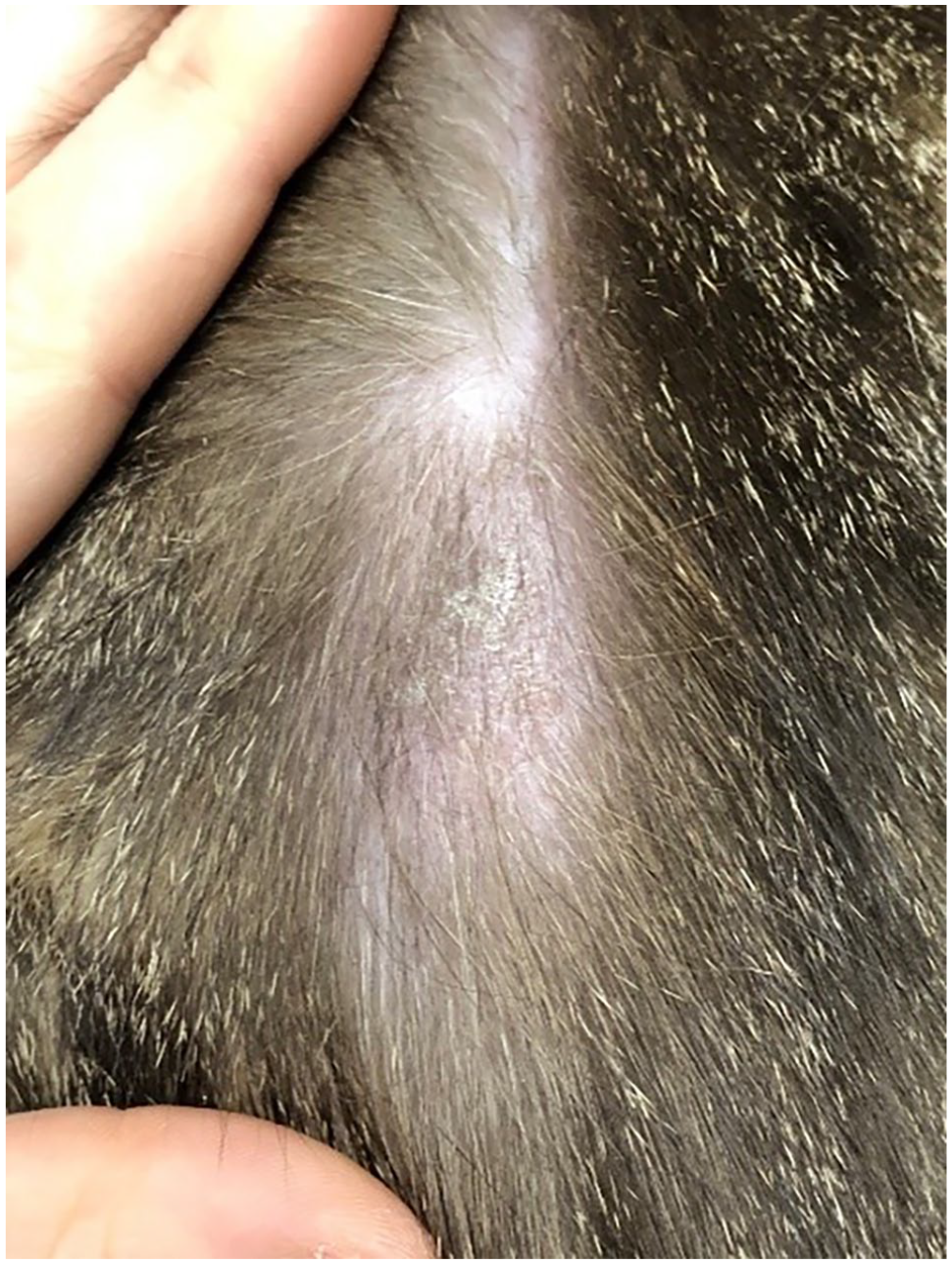
Alopecia and scaling. Image courtesy of University of Minnesota

Perioral feline cutaneous lymphoma. Image courtesy of University of Minnesota
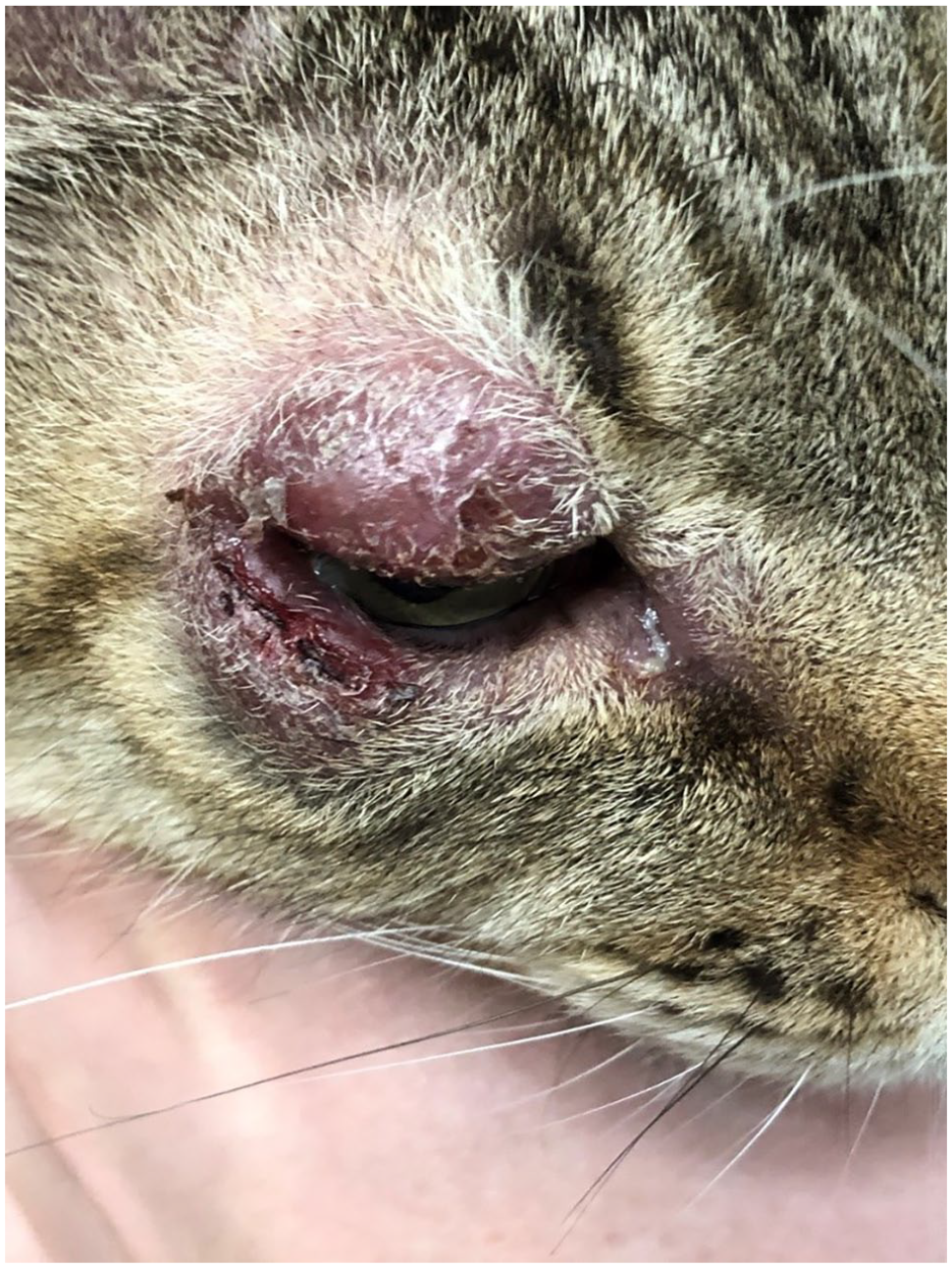
Periocular feline cutaneous lymphoma. Image courtesy of University of Minnesota
The varied presentation of cutaneous lymphoma likely contributes to delays in appropriate diagnosis and under-diagnosis, which may inhibit timely appropriate treatment. Gold standard treatment for cutaneous lymphoma in veterinary patients has yet to be defined. In dogs, lomustine (CCNU) is often successful in mitigating disease for a time; however, the amount of research regarding its use in feline populations is limited. 11 Overall, the prognosis for feline cutaneous lymphoma remains poor, with a reported median survival time of 10.25 months (range 2.5 months to 4 years). 12
The primary purpose of this study was to perform a multi-institution retrospective analysis to further describe the presentation of feline cutaneous lymphoma. Given the relative rarity of this disease, any form of lymphoma affecting the skin was included. The secondary goals included determining if the initial response to treatment affected the overall survival of patients and analyzing whether disease characteristics such as immunophenotype, cell size or the presence of epitheliotropism influenced response to treatment. A better understanding of this relatively rare disease process will aid practitioners in the earlier identification and treatment of cutaneous lymphoma in cats, and also provide information on disease variables that may affect expected response to treatment.
Materials and methods
Case selection
This was a multi-institutional retrospective study of cats diagnosed with cutaneous manifestations of lymphoma. Participating institutions included Colorado State University, University of Minnesota, Oregon State University and the University of Wisconsin-Madison. The medical records of client-owned cats were searched for cases of cutaneous lymphoma from January 2008 to January 2018. Patients were included if they had a cytologic or histopathologic diagnosis of cutaneous lymphoma. As the primary goal of this study was to better describe the presentation of cutaneous lymphoma, any subtype of cutaneous lymphoma was included for analysis. Because epitheliotropism is a characteristic traditionally noted on histopathology, patients were classified as having non-epitheliotropic lymphoma if epitheliotropism was not specified on the histopathology report or they were specifically called non-epitheliotropic on histopathology (K Hughes, 2020, personal communication). Immunophenotype was determined via immunohistochemistry using CD3 for T-cell and CD20 or CD79a for B-cell staining, or PCR for antigen receptor rearrangement (PARR). Patients diagnosed via cytology only (n = 10/41) were included if cytology was consistent with large-cell lymphoma or if PARR was performed to support diagnosis; these patients were included in the non-epitheliotropic group. Two patients with a diagnosis of round-cell malignancy and one patient with a diagnosis of probable lymphoma were included for evaluation as the most likely diagnosis was lymphoma based on pathologist assessment and the patients were treated as having lymphoma. Patients were excluded if there was no cytologic or histologic diagnosis of cutaneous lymphoma. Patient selection is outlined in Figure 4.

Patient selection
If patients were considered eligible, the following information was collected: breed, age at diagnosis, neuter status, feline leukemia virus/feline immunodeficiency virus (FeLV/FIV) status, weight and body condition score, abnormal physical examination findings on initial presentation, presenting clinical signs, duration of clinical signs prior to diagnosis, previous/concurrent therapy, diagnostics performed, method of diagnosis, time between diagnosis and initial treatment, therapy protocol, length of treatment, best response achieved, progression-free survival, rescue therapy and time to death/euthanasia. For patients with gross disease, response was determined using the Veterinary Cooperative Oncology Group’s Response Evaluation Criteria in Solid Tumors (RECIST) criteria. 13 If pertinent information was missing from the hospital record, the primary veterinarian or owner was contacted to clarify points of interest. Owing to a lack of follow-up information, 14/41 patients were censored. Patients were stratified by treatments, to illustrate various therapeutic options for feline cutaneous lymphoma. Owing to the heterogeneity in disease characteristics and treatment the population size was deemed inadequate to examine statistical differences in survival between treatment groups.
Statistical analysis
Overall survival time was calculated as date of diagnosis to date of death from any cause and was used to analyze association between factors. Fourteen patients were lost to follow-up and censored at the time of last evaluation. The impact of treatment on survival was determined via log-rank analysis. The impact of treatment response on survival was evaluated via the Kaplan–Meier methodology and analysis was compared using the stratified log-rank test. Treatment response was defined as patients who had a complete excision with surgery, or if a complete remission (CR) or partial response (PR) was achieved when treated in the gross disease setting. The effect of cell size, cell type (B vs T cell), and presence of epitheliotropism upon treatment response was determined with Fisher’s exact test. Owing to the small number of patients and the heterogeneous nature of many of the characteristics, multivariable analysis and analysis of all factors was deemed inappropriate.
Results
Collectively, 47 patients were identified by the initial search of historical records at four institutions. In total, the records of 41 patients were evaluated; the remaining six patients were excluded owing to a lack of complete medical records. The population comprised 30 male castrated and 11 female spayed patients. No patients were intact. Domestic shorthairs were most common; additional breeds included domestic mediumhair, domestic longhair, American Shorthair, Russian Blue, Ragdoll, Maine Coon, Siamese mix and Dexon Rex. One patient was FeLV positive (unknown FIV status), and two patients were FIV positive/FeLV negative. Five patients were FeLV/FIV negative. The FeLV/FIV status was unknown for 33 patients. The median age at diagnosis was 12.3 years (range 3.8–19.8). Comprehensive signalment information is provided in Table 1.
Signalment data

Data are reported as n (%)
DSH = domestic shorthair; DMH = domestic mediumhair; DLH = domestic longhair; MC = male castrated; FS = female spayed
Case presentation
Patients presented with a wide variety of clinical signs, including hyporexia, pruritus, inappropriate elimination, excessive grooming, lameness, lethargy, weight loss, vomiting, polyuria/polydipsia, hiding, ataxia, head tilt, diarrhea and vocalizing. Additionally, physical examination findings were variable and included skin changes, characterized by nodules/masses, crusting/scabbing, ulceration, pustules, thickened skin, plaques, miliary dermatitis, alopecia, bruising, edema, non-healing wounds and erythema. Twenty-six patients had multifocal skin lesions described. The distribution of skin changes was highly variable, and it was common for individual patients to have a multifocal disease distribution. Common regions reported included limbs (n = 15), caudal trunk (n = 15), cranial trunk (n = 12), head (n = 9), tarsus (n = 6) and neck (n = 4). In addition, 10 patients presented with varying types of lesions. The size of nodules, masses, crusting, scabbing, ulceration and pustules was variable, ranging from 0.5 cm to 6 cm. Non-skin changes included lymphadenopathy, heart murmurs, sarcopenia, ocular discharge, anisocoria, uveitis, head tilt and nasal discharge. In total, 20 patients were fully staged with evaluation of skin lesions, lymph node assessment, thoracic radiographs and abdominal ultrasound. Of the fully staged patients, one had evidence of lymphoma at additional sites confirmed by cytology (mandibular lymph node and abdominal mass). Three had changes concerning for lymphoma at distant sites characterized by splenic and hepatic changes on abdominal ultrasound that were not sampled. A variety of concomitant diseases were present, including hyperthyroidism, chronic kidney disease, inflammatory bowel disease, hypertrophic cardiomyopathy, cholangiohepatitis, eosinophilic granuloma, pancreatitis, mast cell tumor, diabetes mellitus and cerebellar hypoplasia. A summary of these findings and their frequencies is available in Table 2.
Clinical signs, physical examination findings and concomitant diseases
Treated patients
Of 41 patients evaluated, 34 patients were treated. In 11 patients, local therapy was performed, including either surgery or radiation. In two patients, surgery was combined with a systemic therapy, including CCNU (n = 1) or chlorambucil and prednisolone (n = 1). Radiation therapy was combined with adjuvant CCNU in two patients and CHOP chemotherapy in one. In patients that received solely systemic therapy, therapeutic protocols varied, including CCNU, doxorubicin, cyclophosphamide, CHOP, modified CHOP, cyclophosphamide/vincristine and prednisolone only (see Table 1 in the supplementary material). In total, 20 patients received prednisolone in conjunction with systemic chemotherapy. Of the 34 patients treated with some form of therapy, 20 responded, with five patients rendered disease-free after surgery (denoted as a treatment response for statistical purposes), seven patients achieving a PR and eight patients achieving a CR when treated in the gross disease setting. Of the seven patients not treated, the median survival time was 14 days. Fourteen patients were lost to follow-up, and these patients were censored at the date of last follow-up. On statistical analysis it was found that treatment (vs no treatment; P = 0.0025) and treatment response (P = 0.0025) both significantly affected overall survival.
Lymphoma characteristics such as immunophenotype (B cell vs T cell), cell size, presence of epitheliotropism and their influence upon treatment response were evaluated via Fisher’s exact test. Of the 34 treated patients, three had B cell immunophenotype, 10 had T cell, two had a mixed population and 21 were unknown. Two of these patients were diagnosed via PARR, with the remainder being determined with immunohistochemistry; these patients are depicted in the Table 2 in the supplementary material. No significant difference was seen between immunophenotype and response to treatment (P = 0.0684). Of the 34 treated patients, 17 were classified as large cell, eight as small cell and in nine patients the cell size was unknown. No significant difference was observed between cell size and response to treatment (P = 0.0621). Cell size was determined by histopathology in 20 patients and cytology in five patients. Of the 34 patients treated patients, five were classified as epitheliotropic and 29 were classified as non-epitheliotropic. No significant difference was observed with epitheliotropism and response to treatment (P = 0.0608).
Discussion
The primary goal of this study was to better describe the manifestations of cutaneous lymphoma in the feline population. Physical examination findings and clinical signs of patients that were lost to follow-up were included to better characterize this disease presentation. In addition, patients were evaluated to determine if the overall survival was affected by treatment and treatment response, or disease characteristics such as immunophenotype, cell size or the presence of epitheliotropism.
This collection of patients confirmed the previously reported diverse array of clinical signs and physical examination findings in patients with this disease process. The most prevalent clinical signs within the cohort were hyporexia, pruritus, inappropriate elimination, excessive grooming and lameness. As this was a population of older cats, it is possible that some of these clinical signs were due to concurrent diseases and not directly related to cutaneous lymphoma, which highlights the need for a thorough history and examination in geriatric feline patients. Pruritus has not been consistently documented in previous reports of cats with cutaneous lymphoma; however, while still variable, it was one of the most common clinical signs in this population of cats. 12
The most common physical examination findings were skin changes, most commonly characterized by nodules, erythema, crusting, scabbing, alopecia and discrete masses (Figures 1–3 and 5–7). Mucocutaneous involvement was not reported in this feline population as has been described in previous literature. 10 Furthermore, lesions affecting the head and face are previously described as being consistent with a more indolent disease process, which was not displayed in this population. 2 Of 41 patients evaluated, nine had lesions on the head and face, and the median survival of those patients was 127 days. This group of cats exhibited a relatively rapid disease course, and further studies examining the prognostic significance of lesion location is warranted.

Patient 33: swelling and alopecia of the dorsal nasal planum. Diagnosed as T-cell lymphocytic lymphoma on histopathology
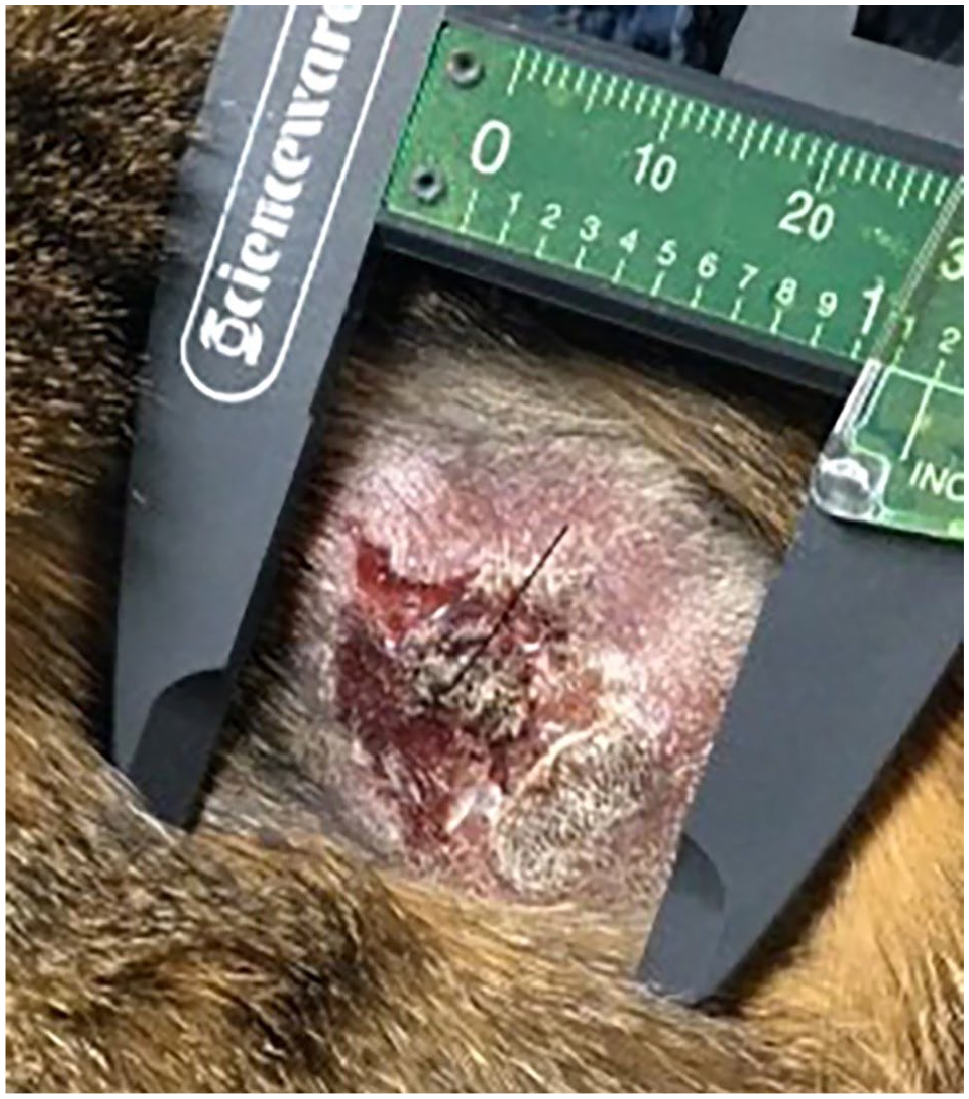
Patient 32 following its first radiation treatment. Ulcerated cutaneous mass in region of left shoulder. Diagnosed as cutaneous T-cell lymphoma on histopathology

Patient 17: ulcerated subcutaneous mass in left axillary region. Diagnosed as large cell lymphoma on cytology
It is worth highlighting the variability of clinical signs and physical examination findings for the presentation of this disease process. As with the canine population, feline cutaneous lymphoma can mimic many common skin diseases. The collection of clinical signs in our population is variable and non-specific. Because of the ambiguity of presentation, many patients in the cohort were treated with therapies prior to diagnosis, including antibiotics, steroids, antifungals, shampoos, opioids and anthelmintics. Delayed recognition or diagnosis of a neoplastic process appears common and defers the appropriate treatment of these patients. Therefore, practitioners should be mindful of cutaneous lymphoma as a differential diagnosis in feline patients who present with a dermatologic disease, especially in those where the condition does not respond or worsens with empirical therapy.
Patients were evaluated to determine if the overall survival was affected by treatment or whether disease characteristics such as immunophenotype, cell size or presence of epitheliotropism affected response to treatment. Given the rarity of the disease, any form of cutaneous lymphoma (epitheliotropic, non-epitheliotropic and patients with extracutaneous involvement) was considered. The majority of patients had a clinical diagnosis of primary cutaneous lymphoma with one patient having confirmation of systemic disease and three others having suspicion of systemic disease. However, a limitation is that only 20/41 patients had full staging. In more advanced disease it can be challenging to determine the original site of involvement. It is also likely that the varying forms of cutaneous lymphoma carry different prognoses; therefore, it is difficult to make conclusions regarding survival times given this heterogeneous population. Further investigation into disease classification and response to treatment of homogeneous forms of cutaneous lymphoma is needed.
Of the 41 patients evaluated, 34 were treated with some form of therapy. Of these, 20 responded, with five patients having all disease surgically removed. In the gross disease setting, seven patients achieved a PR and eight patients achieved a CR. Of the seven patients that were not treated, all developed progressive disease and had a median survival time of 14 days. While statistically this suggests that treatment is beneficial (P = 0.0025), the number of patients that were not treated was very small. This resulted in a serious imbalance between treated and untreated patients within the population (34 treated patients vs seven untreated patients), making it difficult to draw definitive conclusions about the benefit of treatment. Notably, no untreated patients had an improvement in their disease. Importantly, statistics were performed with regard to overall survival rather than progression-free survival (PFS). While PFS is viewed as a more reliable measure of disease progression and outcome, owing to the retrospective nature of the study, this was not possible to discern in all patients. Overall survival time, while potentially biased in veterinary medicine by euthanasia, rescue therapy and other disease processes, is a more definitive number to determine retrospectively. However, there can still be difficulty in determining overall survival as patients had variable duration of clinical signs prior to diagnosis and the time between first onset of signs and diagnosis was not always known. Furthermore, in veterinary medicine the decision to treat neoplasia also introduces bias as those owners may be more likely to delay the decision for euthanasia than owners who do not elect to pursue therapy. Therefore, caution should be used to apply these results to larger populations of cats.
Currently, a gold standard treatment of feline cutaneous lymphoma has yet to be defined. Reported treatments include surgical excision of solitary nodules, radiation therapy of localized disease, retinoid therapy, linoleic acid, glucocorticoids and a milieu of chemotherapy protocols, including CCNU, CHOP, l-asparaginase and VELCAP.1–5,8,10–12 This patient group represents a heterogeneous collection of all forms of cutaneous lymphoma and was likewise treated with a variety of therapies, including surgery, radiation therapy, systemic chemotherapy and multimodal therapy. This highlights the lack of established standard of care therapy for this heterogeneous disease. Owing to the small patient population and heterogeneous treatments, patients were not stratified by their specific therapies for statistical analysis.
Subjectively, the treatments that were associated with the longest patient survival in our study were surgery, single-agent cyclophosphamide, single-agent doxorubicin and surgery in combination with chlorambucil and prednisolone. These results are likely misleading, however, as these are based on small sample sizes. The patient that received adjuvant chlorambucil and prednisolone had a PFS of 547 days. This is a considerably longer PFS time than the previously reported median survival time (10.25 months). 14 This same patient went on to receive rescue therapy of cyclophosphamide and then CCNU, and was ultimately lost to follow-up 927 days after the initial diagnosis. For this single patient it is impossible to determine how either the surgical intervention or adjuvant therapies contributed to the prolonged survival. Furthermore, this patient had a histopathologic diagnosis of non-epitheliotropic, low-grade T-cell lymphoma and it is possible that this patient’s disease represented a more indolent form of lymphoma. Likewise, the patient who received cyclophosphamide (250 mg/m2 every 2 weeks) had subsequent rescue therapy with CCNU (40 mg/m2 every 3 weeks), which may have contributed to prolonged survival. It is also likely that the lone patient with a prolonged survival following single-agent doxorubicin (median survival time 604 days) is a product of small sample size, rather than superiority of this chemotherapy protocol over patients who received a multidrug protocol such as CHOP. This should be interpreted with caution, and further studies evaluating the efficacy of single-agent doxorubicin in feline patients with feline cutaneous lymphoma are worth consideration. Furthermore, it is likely that patients with disease amenable to surgery would have a better prognosis, as their disease is likely more localized. Further studies are necessary to assess whether surgical intervention significantly prolongs the survival time for patients diagnosed with feline cutaneous lymphoma. Table 1 in the supplementary material is provided to display individual patient’s outcomes with regard to specific treatment protocols and rescue therapies; however, additional studies are warranted to further evaluate the survival outcome of patients with certain treatment protocols in specific forms of cutaneous lymphoma.
In patients where the immunophenotype was known, the majority were T cell. This is consistent with previous literature, which suggests that both canine and human epitheliotropic lymphoma are both primarily T cell in origin, with 80% of canine cases being CD8+ and 90% of human cases developing from CD4+ T cells. 8 In the three patients with B-cell lymphoma, one was specifically classified as non-epitheliotropic, while epitheliotropism was not noted in the remaining two patients. This also correlates with previous literature that non-epitheliotropic lymphoma can carry a B- or T-cell immunophenotype. 1 For the purposes of our study, patients without epitheliotropism explicitly noted on the histopathology report were classified as having non-epitheliotropic disease, as is it standard practice to describe epitheliotropism on histopathology (K Hughes, 2020, personal communication). In addition, 10/41 patients were diagnosed via cytology only and placed in the non-epitheliotropic group, which is a major limitation and potential source of bias. Therefore, further work evaluating the impact of epitheliotropism upon disease response is warranted.
Immunophenotype, cell size and/or presence of epitheliotropism were not found to have a significant influence upon response to treatment. Owing to the heterogeneous disease and treatment population, these results should also be interpreted with caution. It is possible that these factors would influence treatment response and survival if they could be evaluated in a larger group of patients with more similar disease characteristics or with a homogenous treatment regimen. Several patients did not have the immunophenotype determined, representing another limitation.
Besides those already mentioned, this study had other limitations. First, while this is the largest known collection of cases of feline cutaneous lymphoma, the patient population size is still relatively small. This corresponds to the rarity of the disease; however, it made statistical analysis and the determination of prognostic factors difficult. In addition, it precluded us from accurately examining how specific treatment protocols, including surgery, radiation and chemotherapy, affected survival in these patients. Additional work evaluating the efficacy of various chemotherapeutic protocols on this disease is warranted.
Owing to the rarity of the disease, patients were recruited across multiple institutions, which may have resulted in inconsistent and heterogeneous diagnostic and therapeutic protocols, and further contributed to difficulty with the evaluation of the patient group. Furthermore, all contributing institutions were academic veterinary hospitals, which typically represent tertiary referral hospitals. This suggests that there is likely a population of cats with cutaneous lymphoma in which disease is either not accurately diagnosed or in which owners did not elect therapy that are not represented here. Extrapolation of our findings to a larger population of cats should be performed with caution.
Other limitations include the lack of histopathologic diagnosis in all patients and the fact that necropsies were performed only in a limited number of patients. A small number of patients were included without definitive diagnosis of lymphoma but with disease that was considered most likely to be cutaneous lymphoma. While these cases appeared to represent cutaneous lymphoma as based on pathologist assessment, alternative neoplastic diagnoses, such as histiocytic diseases, or benign processes, such as lymphocytic dermatitis or cutaneous lymphocytosis, cannot be completely ruled out. Finally, there are inherent limitations of any retrospective study. The variable reporting of physical examination findings, clinical history, clinical signs at diagnosis and interpretation of progressive disease all represent additional limitations.
Conclusions
Feline cutaneous lymphoma is a rare disease in feline patients that can manifest in a diverse group of clinical signs and skin abnormalities. Nodules, masses and erythema were the most common clinical presentations in this population. Given its ability to mimic other more benign skin diseases, it is possible that proper diagnosis and treatment is often delayed, which may contribute to the poor prognosis in these patients. This study suggests feline patients respond to treatment, but further study evaluating the effect of specific treatment modalities on prognosis is warranted. Immunophenotype, cell size and the presence of epitheliotropism was not found to be associated with response to treatment; however, this should be evaluated in a larger population of cats.
Supplemental Material
Supplementary Table 1
Individual patient treatment.
Supplemental Material
Supplementary Table 2
Treated patients with known immunophenotype
Footnotes
Acknowledgements
The authors acknowledge the following clinicians for their contributions to the study: Cecilia Robat DVM, DACVIM (Oncology), and Jack O’Day DVM. Special thanks to the Robert and Eva Knight Residency in Oncology Endowment for supporting Dr Siewert’s medical oncology training program.
Supplementary material
The following files are available online:
Supplementary Table 1: Individual patient treatment.
Supplementary Table 2: Treated patients with known immunophenotype.
Author note
The preliminary findings of this work were presented at a poster session during the Virtual VCS conference in October 2020.
Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent for their use in the publication (verbal or written) was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
