Abstract
Clinical relevance:
In contrast to superficial fungal infections, such as dermatophytosis, invasive fungal infections (IFIs) are characterised by penetration of tissues by fungal elements. Disease can spread locally within a region or can disseminate haematogenously or via the lymphatics. The environment is the most common reservoir of infection. Since fungal spores are airborne, indoor cats are also susceptible to IFIs. Some environmental fungi are ubiquitous and present globally, while others are endemic or hyperendemic within specific geographic regions. Zoonotic pathogens include Microsporum canis, Sporothrix schenckii and Sporothrix brasiliensis.
Aim:
In the first of a two-part article series, the approach to the investigation of feline IFIs and oomycoses is reviewed. As well as tips for diagnosis, and information on the ecological niche and distribution of fungal pathogens, the review covers clinical presentation of the most common IFIs, including cryptococcosis, histoplasmosis, blastomycosis, coccidioidomycosis, sporotrichosis, phaeohyphomycosis, aspergillosis and dermatophytic pseudomycetoma, as well as the oomycoses pythiosis, lagenidiosis and paralagenidiosis. In Part 2, the spectrum of activity, mechanisms of action, pharmacokinetic and pharmacodynamic properties and adverse effects of antifungal drugs are reviewed, and the treatment and prognosis for specific IFIs and oomycoses are discussed.
Evidence base:
The review draws on published evidence and the authors' combined expertise in feline medicine, mycology, dermatology, clinical pathology and anatomical pathology.
Keywords
Introduction
The outcome of a fungal infection depends on a complex interplay between the fungal pathogen and the host's immune response. Non-invasive fungal infections are those in which fungal elements are restricted to surface colonisation of the skin, hair, mucosa or airways. By contrast, in invasive fungal infections (IFIs), fungal elements penetrate tissues and can spread locally or by haematogenous and/or lymphatic dissemination. For example, in dermatophytosis, the most common non-invasive mycosis of cats, Microsporum canis infects the stratum corneum of the epidermis, hair shafts and hair follicles. However, occasionally, M canis penetrates the dermis to cause an IFI. 1
Part 1 of this two-part article series describes the general diagnostic approach to cats with suspected IFIs or oomycoses, hereafter termed invasive fungal-like infections (IFLIs). Attention then turns to reviewing clinical features and providing diagnostic tips for the most common IFIs and IFLIs. The fungal pathogen Candida is not reviewed since there have been fewer than 10 cases of invasive candidiasis reported;2-6 by contrast, non-invasive Candida species infections of the lower urinary tract are moderately frequent. 7 Algal infections, such as protothecosis, and other IFLIs, such as microsporidiosis and rhinosporidio-sis, are outside the scope of this review.8-17

Agents of disease
The fungal kingdom is vast and currently includes 19 different phyla. 19 The most common moulds and yeasts that cause IFIs in cats are listed in Table 1. Yeasts are unicellular and reproduce asexually by budding to form a daughter cell, whereas moulds are multi-cellular filaments (hyphae) that reproduce asexually through fragmentation of hyphae or production of asexual spores (conidia). Many yeasts and moulds also reproduce sexually by meiosis. Some yeasts have three vegetative states - yeast, pseudohyphae and hyphae -which can make cytological identification in tissues more difficult. Some fungi (thermally dimorphic ascomycetes, Table 1) exist as moulds in their environmental reservoirs and switch to yeast forms in the higher temperatures of infected hosts. 20
Classification of common invasive fungal pathogens in cats

Previously known as Zygomycetes. †Thermally dimorphic
Oomycetes are the most common fungal-like organisms to infect cats.
Most feline IFIs and IFLIs are not zoonotic. Rather, they are caused by shared environmental pathogens, which lead to disease in humans and animals alike, usually through inhalation or traumatic cutaneous inoculation. Sporotrichosis is a notable, highly zoonotic exception. M canis is also zoonotic in cats with dermatophytic pseudomycetoma and concurrent dermatophytosis.
General diagnostic approach to a possible invasive fungal or fungal-like infection
History and signalment
Knowledge of which fungi are ubiquitous saprophytes, occurring anywhere regardless of geographic location, and which are endemic or hyperendemic in particular regions is helpful when considering the likelihood and ranking of an IFI/IFLI in the differential diagnosis list (Figure 1; see also later discussion of the 'Diagnostic approach for selected invasive fungal and fungal-like infections'). With global warming, the geographic range of many environmental fungi is increasing. 21 Travel history is also important, because some IFIs (eg, cryptococcosis and histoplasmosis) can be latent and subclinical but can reactivate months to years later if the cat is immunosuppressed. 22

The geographic range of many environmental fungi is increasing in association with global warming and will likely continue to expand.
Patient signalment can raise the ranking of IFI/IFLI on the differential diagnosis list. For example, Persian cats and related breeds are predisposed to some IFIs, including aspergillosis and dermatophytic pseudo-mycetoma. 23 Fungal infections can occur in cats of either sex or any age, but some predispositions exist for certain IFIs/IFLIs (see 'Diagnostic approach for selected invasive fungal and fungal-like infections'). Moreover, while IFIs are commonly diagnosed in apparently immunocompetent cats, immuno-suppression from concomitant disease or immunomodulatory drugs, such as ciclo-sporin, 24 is a risk factor for IFIs and also for haematogenous dissemination of an IFI.
Complete blood count, serum biochemistry profile and retrovirus testing
For mould IFIs and IFLIs, an inflammatory leukogram and mild to severe hyperglobuli-naemia are common abnormalities. 25 Hyper-globulinaemia is present in ~65% of cases of sporotrichosis. 26
Mild to moderate non-regenerative anaemia (normocytic, normochromic) is common in cats with cryptococcosis, sporotrichosis and histoplasmosis, but anaemia may be severe in cats with histoplasmosis.26-29 Leukocyte changes are variable among infections caused by thermally dimorphic fungi. Neutrophilic leukocytosis, sometimes with a left shift, is common in both histoplasmosis and sporo-trichosis,26,28 while monocytosis is a common finding in cases of cryptococcosis 27 and coccidioidomycosis. 30 Infiltration of bone marrow in cats with disseminated histoplasmosis may result in neutropenia, thrombocytopenia or pancytopenia. 31
Peripheral eosinophilia is not common overall, but, if present, should raise a flag for possible IFIs/IFLIs of any type. Hypoalbuminaemia is common in disseminated sporotrichosis and histoplasmosis, 26 especially where there is gut involvement. Hypercalcaemia may be present due to monocyte and macrophage activation within pyogranulomas; increased expression of 25-hydroxyvitamin D3 1a-hydroxylase then results in hypervitaminosis D. This has been reported in histoplasmosis, blastomycosis and dermatophytic pseudomycetoma.28,32,33 Other serum biochemical changes in IFIs/IFLIs reflect specific organ involvement.
Results of feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) testing reflect regional prevalence, and, although these retroviruses are not significantly associated with IFIs/IFLIs, testing is recommended. A positive test result is not a contraindication for IFI/IFLI treatment; however, cats with advanced FIV or FeLV infection may require longer courses of treatment.
Fungal-specific tests on urine, serum or other body fluids
Serum antibody or fungal antigen tests, as listed in Table 2, are especially useful in the early stages of a diagnostic investigation since test samples can mostly be procured without the need for general anaesthesia. Their use should be guided by the patient's clinical signs at presentation, as well as the travel history and geographic location. Aspergillosis and crypto-coccosis occur worldwide. While sporadic cases of histoplasmosis and sporotrichosis may also be seen in any geographic region, these, as well as blastomycosis, coccidioidomycosis and pythiosis, usually occur in endemic/hyper-endemic regions (Figure 1). Tests to detect fungal antibodies or antigens in serum or urine are not currently available for dermatophytic pseudomycetoma or phaeohyphomycosis.
Availability of antibody and antigen tests for invasive fungal and fungal-like infections in cats

AGID = agar gel immunodiffusion; BAL = bronchoalveolar lavage; CALAS = cryptococcal antigen latex agglutination system; CF = complement fixation; CSF = cerebrospinal fluid; EIA = enzyme immunoassay; ELISA = enzyme-linked immunosorbent assay; GM = galactomannan; POC LFA = point-of-care lateral flow antigen; RIA = radioimmunoassay; Se = sensitivity; Sp = specificity
Diagnostic imaging
Typical radiographic findings in invasive fungal and fungal-like infections in cats

DIA = disseminated invasive aspergillosis; SOA = sino-orbital aspergillosis
Thoracic radiography is useful when an IFI/ IFLI is suspected because the respiratory tract is the most common portal of entry and may be diseased. A predilection for upper or lower respiratory tract involvement, or both, depends on the infecting pathogen (Table 3). Because of the superimposition of anatomical structures, skull radiography is not sensitive for the detection of upper respiratory tract involvement; if suspected, CT examination of the head is recommended. CT changes in sino-orbital aspergillosis (SOA) may include increased soft tissue attenuation of the nasal cavities and sinuses, as well as turbinate lysis. 51 The degree of nasal cavity involvement ranges from mild to severe. Orbital masses are usually unilateral and ven-tromedial, with heterogeneous and peripheral rim contrast enhancement. Changes in thesinonasal cavity, orbit and paranasal bones are not specific for aspergillosis and may be indistinguishable from neoplasia (eg, nasal carcinoma or lymphoma) or other IFIs/IFLIs such as cryptococcosis, 23 sporotrichosis or pythiosis. 52 If neurological signs are present, MRI of the head or spinal cord (depending on lesion localisation) is the preferred imaging modality. In SOA, orbital masses are hyperintense and heterogeneous on T1- and T2-weighted images, and enhance with con-trast.53-55 If present, central nervous system (CNS) lesions show increased heterogeneous contrast enhancement. In blastomycosis with CNS involvement, there are focal or multifocal intra-axial masses with dural contact; lesions are hypointense on T2-weighted images and diffusion-weighted imaging, and show severe perilesional oedema and strong homogeneous contrast enhancement. 56 In disseminated phaeohyphomycosis with CNS involvement, there may be focal or multifocal fungal abscesses; MRI features of these lesions in humans - which are likely similar in cats -include irregular walls and intracavitary projections caused by fungal hyphae. 57 Fungal granulomas in cases of cryptococcosis are hypointense on T1- and hyperintense on T2-weighted images, often with well-defined rim enhancement after contrast administration.58,59
Advanced imaging is not reliable for definitive diagnosis of fungal granulomas since, for both CT and MRI, features overlap with those of neoplasia.23,51,58,60
DIA = disseminated invasive aspergillosis; SOA = sino-orbital aspergillosis
Cytology
Cytology of fine-needle aspirates from masses, cytocentrifugation (cytospin) preparations of urine, bronchoalveolar (BAL) fluid, cerebrospinal fluid (CSF), brushings from conjunc-tivae or impression smears of ulcerated lesions can provide useful information. Fungal elements, if present, can usually be identified using Romanowsky-type stains, such as Wright, Giemsa and Diff-Quik. Depending on the pathogen, fungal elements may exhibit positive or negative staining (Figure 2). Additional tissue or fluid samples obtained aseptically for cytology can also be submitted for fungal culture to confirm pathogen identity and antifungal susceptibility.

Diff-Quik stained smears showing fungal elements. (a) Positive-staining fungal hyphae in frontal sinus tissue of a cat with invasive fungal rhinosinusitis due to Aspergillus felis infection. (b) Intracellular negative-staining fungal elements in a fine-needle aspirate preparation from a retrobulbar mass of a cat with a phaeohyphomycosis caused by Phoma species
In general, cytology or culture of nasal swabs or washes cannot be used for definitive diagnosis since the nasal cavity can be contaminated with or colonised by fungal spores, which can readily be cultured, giving rise to a false-positive diagnosis of fungal infection. However, if large numbers of fungal elements (hyphae, yeasts) are visualised in the presence of inflammatory cells, there should be a high index of suspicion for fungal infection and a confirmatory test should be performed (eg, fungal antigen test or biopsy).
Surgical biopsies: procurement and transport to the laboratory
Surgical biopsies of involved tissues are often the best samples for definitive diagnosis, especially where less invasive (eg, antigen) tests are not available or where cytology is non-diagnostic. Definitive diagnosis is dependent on taking a representative biopsy of tissue. In many IFIs/IFLIs, fungal elements are only found in the necrotic centre of pathological lesions and are surrounded by a pyogranulomatous inflammatory reaction, which may contain a large proportion of eosinophils. Biopsies that sample only the periphery of a granuloma may yield negative fungal cultures, leading to a misdiagnosis on histology of eosinophilic granuloma complex or non-fungal sterile inflammation. The authors have observed this in cases of SOA and dermatophytic pseudomycetoma.
When performing surgical tissue biopsy from a cat with a possible IFI/IFLI, a good general rule of thumb is either to take one large biopsy and divide it into three portions, or to take three smaller individual representative biopsies. One sample (or portion) is placed in formalin, one is placed in sterile saline in a sterile container for culture and the remaining one is placed in a sterile container and frozen for later molecular diagnostics if fungal culture is negative (Figure 3). Hyphae can be damaged in tissue samples that are homogenised in the laboratory using tissue grinding or beads, as is common practice for bacterial culture, resulting in negative mould cultures. 61 The recommended technique for preparing tissues in the laboratory for fungal culture is to dice them into small (2 mm3) pieces using a sterile scalpel blade. Yeasts are less likely to be damaged by homogenisation than moulds.

Representative tissue biopsies from a lesion (A) are placed in formalin (Bi) for histopathology, in sterile saline in a sterile container (Ci) for culture or in a sterile container (D) for frozen storage at -20°C. After embedding formalin-fixed samples in a paraffin block, a microtome (Bii) is used to cut thin sections for histological examination (Biii[a]). If fungal elements are identified on histology and fungal culture is negative, 6-10 urn thick formalin-fixed sections are cut from the paraffin block using the microtome and a sterile blade, and then placed in a sterile tube (Biii[b]) for DNA extraction and molecular identification. Tissue samples should be diced into small pieces (2 mm3) and cultured on fungal agar (Cii[a]). Molecular identification is most reliable using DNA extracted from fungal culture material (Ciii[a]). DNA can also be extracted from residual fresh tissue (Cii[b]) or from stored frozen tissue (D) for molecular identification

Samples for culture should be submitted to the laboratory at room temperature if they can be delivered within 2 h; otherwise, they should be refrigerated at 4°C until collection. 62
Some fungal elements are hard to visualise on routine haematoxylin and eosin staining of histological preparations. Whenever fungal infection is suspected by the clinician, additional 'fungal' stains should be requested. This includes all cases in which histological reports of biopsied tissues describe a pyogranulomatous or granuloma-tous inflammatory response but where no fungal elements are detected. The two most common special stains used to detect fungal and fungal-like elements are periodic acid-Schiff and Grocott methenamine silver (Figures 4-7).

PYTHIOSIS. Histological sections of a skin biopsy sample from a 6-month-old female domestic shorthair cat with a granulating lesion on the right hindlimb. Pythium insidiosum infection was diagnosed within the dermis and subcutis. (a) On haematoxylin and eosin staining, ghost-like outlines of irregular hyphae (arrows) can be seen embedded in the magenta pink necrotic material. On periodic acid-Schiff (b) and Grocott methenamine silver (c) staining, irregular hyphae with non-parallel walls and tapering ends are identified. Bar = 10 um

PHAEOHYPHOMYCOSIS. Histological sections of a skin biopsy sample from an adult domestic shorthair cat with a large ulcerated nodular skin mass. (a) Haematoxylin and eosin-stained section; within the chronic severe pyogranulomatous dermatitis and panniculitis, hyphal elements (3-4 um in width) are easily identified due to their melanised (brown) cell walls. Septate hyphae stain positively with periodic acid-Schiff (b) and Grocott methenamine silver (c). Bar = 10 um

CRYPTOCOCCOSIS. Histological sections from a mass infiltrating the left external ear canal of a 9-year-old female neutered domestic shorthair cat with Cryptococcus neoformans infection. (a) Haematoxylin and eosin-stained section; sheets of refractile, round-to-oval yeasts, occasionally exhibiting narrow-necked budding (arrow), are surrounded by large unstained capsules. Yeast walls stain magenta with periodic acid-Schiff (b) and black with Grocott methenamine silver (c). Bar = 10 um

DERMATOPHYTIC PSEUDOMYCETOMA. Histological sections from a resected nodule from a 10-month-old female neutered Exotic Shorthair cat. The patient had multiple subdermal nodules on the head and abdomen with pyogranulomatous dermatitis and panniculitis due to Microsporum canis infection. (a) Haematoxylin and eosin-stained section; mats of fungal hyphae with occasional bulbous ends are surrounded by neutrophils and a multinucleated giant cell (asterisk). Fungal hyphae stain magenta pink with periodic acid-Schiff (b) and brown/black with Grocott methenamine silver (c). Bar = 10 um
Adequate tissue should be sampled for submission for histology and for fungal culture. In practice, fungal cultures are often not requested at the time of biopsy, either because an IFI/IFLI is not suspected or because of client financial constraints.
Fungal culture
Optimal conditions for culture
The most reliable way of definitively identifying the cause of an IFI is by fungal culture (Table 4). However, it is underutilised in veterinary medicine. Morphological features of fungal colonies and microscopic examination of cultured fungal elements usually enable identification to genus level. Identification to species level may require one or more PCR assays or matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry (MALDI-TOF MS), using fungal culture material.
Optimal media and incubation temperatures for fungal isolation, and targets for PCR of DNA extracted from fungal culture for molecular identification 63

Non-selective general purpose media are shown in bold
BHIA = brain-heart infusion agar; BSA = bird seed agar; CGB = canavanine glycine bromothymol blue agar; CMA = corn meal agar; Cyt b = cytochrome b gene; CzA = Czapek Dox agar; GPDH = glycerol-3-phosphate dehydrogenase; IMA = inhibitory mould agar; ITS = internal transcribed spacer; MEA = malt extract agar, PDA = potato dextrose agar, SDA = Sabouraud's dextrose agar, TEF = translation elongation factor
The success of fungal culture depends to a large extent on the expertise of the laboratory processing the sample, as well as the condition in which the biopsy is received, and the duration and conditions of transport. Reasons for negative fungal cultures include use of culture media and/or incubation temperatures that are not conducive for growth, as well as overgrowth by commensal or contaminating bacteria or fungi. The use of at least two different incubation temperatures and different types of media has been shown to increase the sensitivity of isolation of fungi from clinical specimens. 64
It is well worth contacting the diagnostic laboratory to ascertain whether it:
✜ Routinely incubates fungal cultures at two different temperatures: most commonly 25° or 28°C, and 37°C; 25
✜ Uses culture media supplemented with antimicrobials (usually gentamicin or chlor-amphenicol) when bacterial contamination of the sample is likely;
✜ Has the capacity to perform molecular testing and/or MALDI-TOF MS;
✜ Submits unusual isolates to a mycology reference laboratory for definitive species identification. Since many of the organisms that cause IFIs/IFLIs in cats are environmental saprophytes capable of infecting humans, mycology reference laboratories are well equipped to identify them.
Most thermally dimorphic species of fungi (eg, Sporothrix species, Histoplasma species) pose an inhalational infection risk to laboratory staff if grown as moulds, and culture of these species is not recommended routinely. If culture is required, laboratory staff should be warned in advance if fungal infection is suspected, as advanced biosafety level (BSL3) conditions to prevent inhalation of spores may be required.
Molecular diagnostics
Many veterinary diagnostic laboratories now offer molecular assays for identification of fungal pathogens. Molecular identification is most reliable when DNA for PCR and sequencing is extracted from fungal cultures of clinical specimens. 65 Fungal DNA can also be extracted directly from formalin-fixed paraffin-embedded tissue (FFPET) or from fresh or frozen clinical specimens (Figures 3 and 8). 66 Panfungal PCR primers are used to target part of the ribosomal RNA (rRNA) gene cluster present in multiple copies in yeasts, moulds and oomycetes (Table 4 and Figure 8). 67
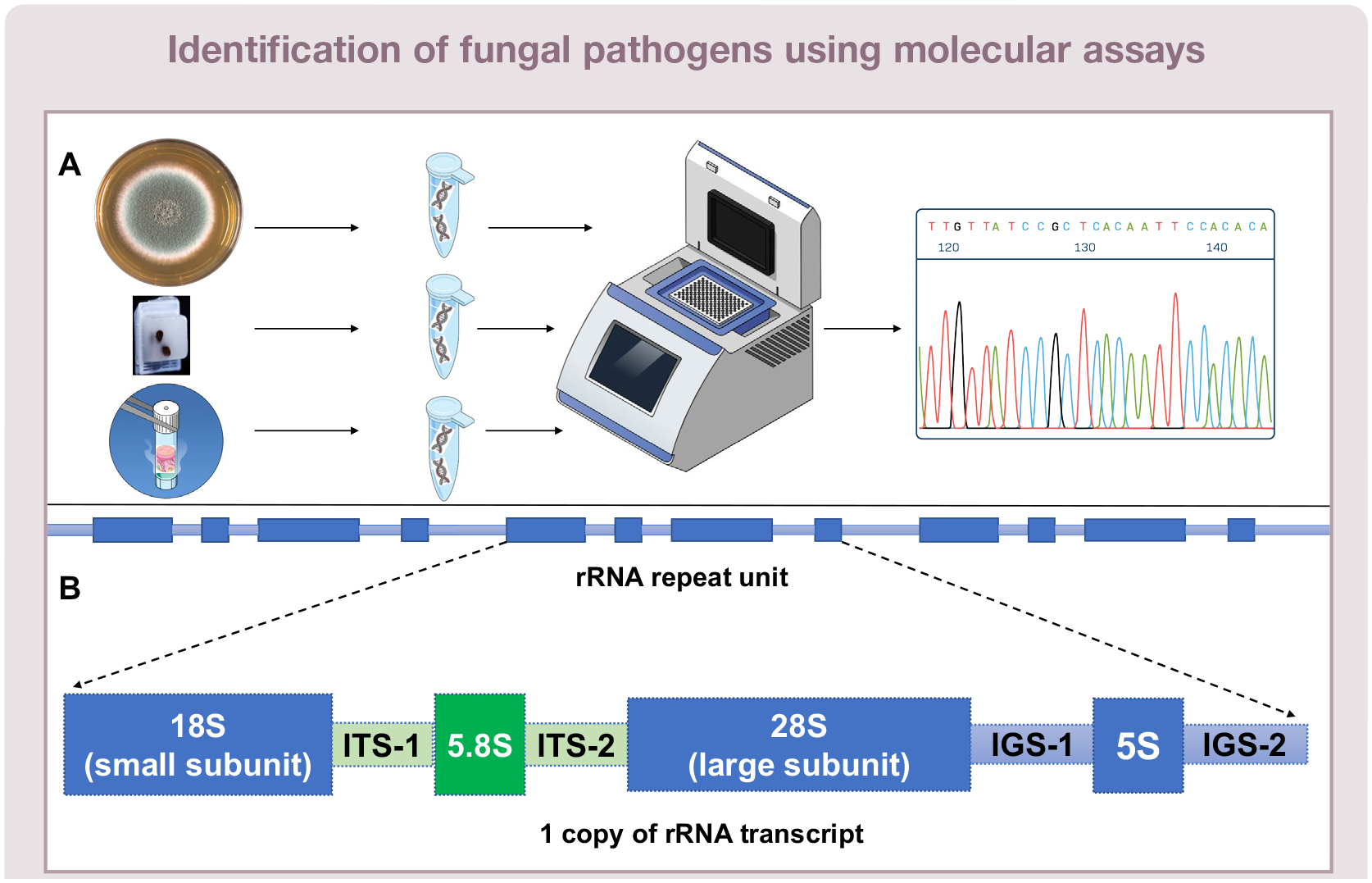
(A) DNA extracted from fungal culture material, formalin-fixed paraffin-embedded tissues or fresh or frozen tissues can be subject to PCR and sequencing to determine fungal genus and species complex or species. (B) The methodology uses panfungal PCR primers to amplify targets within the multicopy ribosomal RNA gene cluster of fungi, which contains hypervariable regions. ITS = internal transcribed spacer; IGS = intergenic spacer
Sequencing the entire ITS1-5.8S-ITS2 region (~700 nucleotides) within the rRNA gene cluster amplified from fungal culture DNA extracts enables identification at least to genus and species complex level (Figure 8).23,65 DNA extracted from FFPET may be degraded or fragmented and DNA quality is also influenced by the duration of formalin contact and number of fungal elements (hyphae, yeasts) present in the FFPET paraffin block, reducing the sensitivity of PCR assays (Figures 3 and 8). However, amplification of the shorter ITS1 (~290 nucleotides) or ITS2 regions (~330 nucleotides) is often possible.25,67 In one study, where fungal elements were identified on histology of lesions from animal tissues, ITS2 PCR of FFPET DNA enabled identification to genus level in 65% of samples; in 96% of these, the identified fungus was consistent with the morphological identification on histological examination. 50
Antifungal susceptibility testing
Broth microdilution methods are the most reliable means of determining in vitro anti-fungal susceptibility. There are two reference methods - one from the Clinical and Laboratory Standards Institute (CLSI) and one from the European Committee on Antimicrobial Susceptibility Testing (EUCAST). Both use a 96-well format, broth media (RPMI 1640) and a minimum inhibitory concentration (MIC) endpoint (ie, complete inhibition of growth determined by visual inspection).
Commercial kits are also available, such as Sensititre YeastOne (TREK Diagnostic Systems), which utilises antifungals in serial two-fold dilutions on a 96-well, dried colori-metric broth microdilution panel. It has shown high interlaboratory reproducibility and moderate to high correlation with the reference methods, dependent on the fungal genus.68,69
Although clinical MIC breakpoints, which correlate in vitro data (including susceptibility testing results and drug pharmacokinetics/ pharmacodynamics) with clinical trial outcomes, are still lacking in veterinary medicine, these are available for many fungal pathogens in humans and are broadly applicable. Also, epidemiological cut-off values (ECVs) of MICs are useful to detect decreased susceptibility or resistance in vitro. ECVs are upper limit values set by determining the distribution of MICs for wild-type isolates. The wild-type population is most commonly defined as those isolates around the modal MIC ± one two-fold dilution, which encompasses >95% of strains.
Diagnostic approach for selected invasive fungal and fungal-like infections
Cryptococcosis
CNS disease - Extension of nasal disease through the cribriform plate or haemato-genously can cause ocular involvement (chorioretinitis, optic neuritis, uveitis), meningoencephalomyelitis and/or fungal CNS granulomas. Presentations include behavioural changes, altered mentation, inappetence, sudden-onset blindness, ataxia, spinal pain and seizures.59,81 Increased intracranial pressure can result in severe acute obtundation.
Systemic and cutaneous disease - Systemic and cutaneous forms of cryptococcosis (Figure 9) signal advanced disease resulting from haema-togenous spread. 75 Cutaneous lesions include non-pruritic, solitary or multiple nodules, abscesses and/or ulcers, with or without peripheral lymphadenopathy. 77 Systemic presentations reflect organ involvement, ranging from skeletal (osteomyelitis, arthritis) to multi-organ disease (gastric and intestinal lesions, hepatitis, pancreatitis, renal abscessation, bladder wall infection, adrenal gland granulomas, generalised lymphadenomegaly).

(a,b) A 6-year-old female neutered domestic shorthair cat in Hong Kong with large fleshy subcutaneous masses infiltrating the external ear canal caused by Cryptococcus neoformans. Images courtesy of Dr Edmund Cheung

Histoplasmosis

Diff-Quik stained smear from the left mandibular lymph node of a cat with cryptococcosis, showing sheets of round yeasts with a non-staining capsule and occasional narrow-necked budding (arrows). An inflammatory response with neutrophils and macrophages is also apparent. x 40 objective

Diff-Quik stained smear from a mesenteric lymph node of a cat with disseminated histoplasmosis, showing numerous intracytoplasmic round yeast forms within macrophages. x 100 objective

Blastomycosis

Coccidioidomycosis


A 5-year-old male neutered domestic longhair cat from Arizona, USA, presented with severe weight loss (50% body weight decline), anorexia, dehydration and progressive hindlimb paralysis. (a) A right lateral thoracic radiograph showed a cranial mediastinal mass (asterisk) and a lytic-proliferative osteomyelitic lesion of the T2 vertebral process (arrow). (b) Lytic and proliferative osteomyelitis of the body and vertebral process of L4, extending along the ventral edges to involve L3 and L5, was causing the paralysis due to compression of the spinal cord at L4. The cat was positive for coccidioidomycosis on serology, with an anti-Coccidioides IgG antibody titre of 1:64. Cytological samples were not available. In areas endemic for Coccidioides species, consistent clinical signs and positive antibody titres are an indication for antifungal therapy. This cat progressively improved after treatment with fluconazole, before being lost to follow-up 7 months after diagnosis
Sporotrichosis

Dermatophytic pseudomycetoma
Occasionally, regional lymph nodes draining primary lesions are involved. 1 Extra-cutaneous disease may also include abdominal, nasal and oral locations.1,33,113,114,126 Large abdominal masses have been diagnosed months to years after ovariohysterectomy, including a uterine stump mass in one cat, 114 and disseminated abdominal dermatophytic pseudomycetoma in another. 113 Clinical signs in affected cats included constipation, tenes-mus and dysuria. Invasive fungal rhinitis with oral cavity involvement, caused by M canis, was reported in an adult cat. 126

Dermatophytic pseudomycetoma in a 6-year-old British Shorthair cat in Hong Kong. The cat, which had a chronic history of dermatophytosis caused by Microsporum canis, developed ulcerated masses on the tail initially, and subsequently on the trunk. Image courtesy of Dr Edmund Cheung

Aspergillosis
Sino-orbital aspergillosis (SOA) - cryptic species (that look alike morphologically) in Aspergillus section Fumigati, especially A felis and A udagawae.
Disseminated invasive aspergillosis (DIA) and invasive pulmonary aspergillosis (IPA) - species in Aspergillus section Nigri and Asper-gillus section Fumigati.
Cats with SOA are usually presented with periorbital swelling; inhaled conidia deposited in the sinonasal cavity germinate into hyphae, which invade the sinonasal mucosa causing granulomatous rhinitis, osteolysis of the orbital bone and spread of infection into the orbit. Occasionally, cats present when infection is still confined to the nasal cavity, but clinical signs at presentation are typically associated with progressive expansion of a ventromedial orbital fungal granuloma, and include unilateral exophthalmos, conjunctival hyperaemia and discharge, nictitating membrane prolapse, central corneal ulceration, an oral mass or ulcer in the ipsilateral pterygo-palatine fossa and mild mandibular lymph-adenomegaly (Figure 14). If nasal signs (eg, sneezing, discharge) are not detected at presentation, there may be a history of these over the previous 6 months. As disease progresses, exophthalmos may become bilateral and other signs may occur, including paranasal soft tissue swelling (Figure 14), which can cause severe facial distortion, and CNS signs, including seizures, nystagmus, circling, facial muscle fasciculation, hyperaesthesia and blindness. Haematogenous or lymphatic dissemination to medial retropharyngeal lymph nodes has been described in association with a ventral cervical mass in two cats.129,130
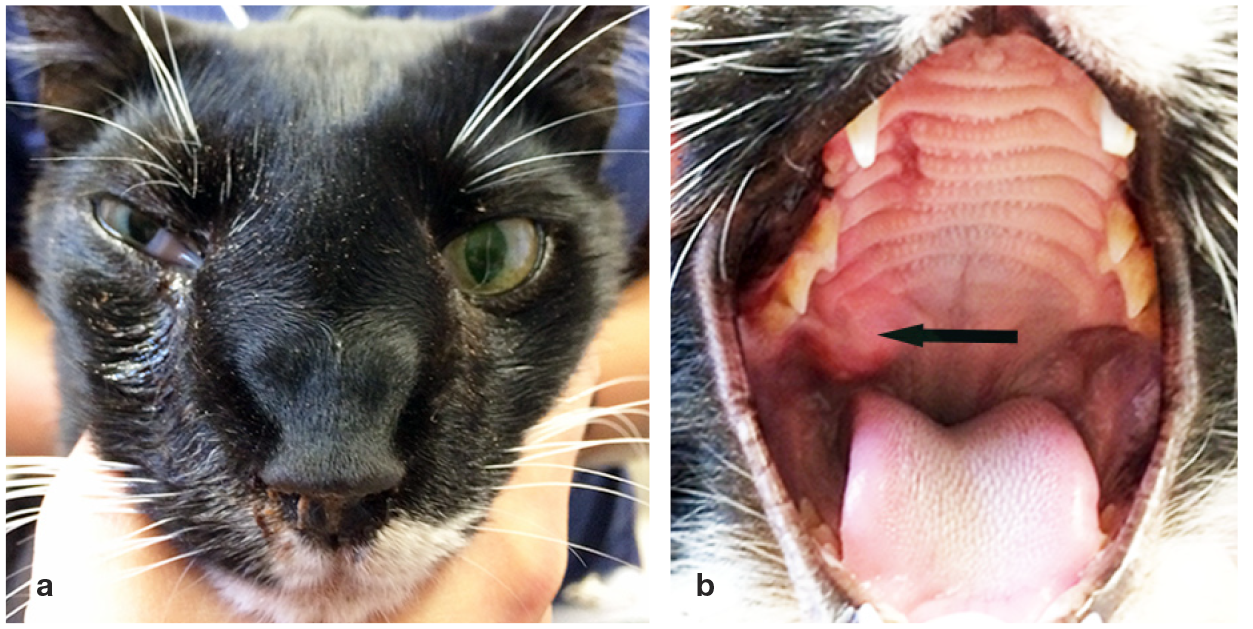
Sino-orbital aspergillosis caused by Aspergillus fells infection in a 2-year-old neutered male domestic shorthair cat in Sydney, Australia. (a) The cat has exophthalmos, third eyelid prolapse and conjunctival discharge in the right eye, as well as right nasal discharge and facial distortion. (b) These signs are associated with a right orbital mass that has invaded the oral cavity, causing a bulge in the right pterygopalatine fossa (arrow)

DIA in immunosuppressed cats has been described as presenting with non-specific signs, including lethargy, anorexia, weight loss and fever. Lung and gastrointestinal involvement is most common. 130
Dematiaceous fungal infections (phaeohyphomycosis)


A 14-year-old female neutered domestic shorthair cat that presented with a subcutaneous mass beneath the right eye, which was identified as a pigmented mould infection. (b) On CT examination of the head, the mass was found to have invaded the right orbit
Mucormycosis and entomophthoromycosis (both previously known as zygomycosis)
Oomycoses (pythiosis, lagenidiosis and paralagenidiosis)
Cutaneous/subcutaneous - Subcutaneous masses are seen, especially in the neck, trunk, inguinal and perianal areas, nasopharynx and / or sino-orbitally. Sometimes these lesions are ulcerated with multiple fistulous tracts. They also occur at the tail base or extremities. Perianal lesions may present as a subcutaneous mass encircling the anus and can extend intrapelvically.
Gastrointestinal pythiosis - Small intestinal masses develop, with mesenteric lymph node involvement. 144 , 149 , 152 , 154 Presenting signs include combinations of anorexia, vomiting, haema-temesis, diarrhoea and weight loss. Palpable abdominal mass(es) may be present and, owing to the insidious course of disease, may be very large (>10 cm diameter) at presentation.

(a) An 8-month-old female domestic shorthair cat, which had been adopted as a stray in Hong Kong, developed firm, progressive swelling of the right hind leg. (b) A CT scan taken by the primary veterinarian showed diffuse subcutaneous tissue infiltrates with increased soft tissue attenuation. (c) The affected tissue was surgically debulked at the time that diagnostic biopsies were collected. Histopathology revealed granulomatous inflammatory tissue containing irregular fungal hyphae. These were identified by PCR as Pythium insidiosum. Images courtesy of Dr Edmund Cheung



Key Points
✜ Invasive fungal and fungal-like infections in cats most commonly present with cutaneous, subcutaneous and/or respiratory signs, but in disseminated disease can have a variety of different clinical presentations, depending on organ involvement.
✜ These infections should be on clinicians' diagnostic radars and can be definitively diagnosed using a combination of different techniques that are readily accessible in clinical practice.
Footnotes
Acknowledgements
The authors would like to thank Dr Edmund Cheung, from Non-Profit Making Veterinary Services, Prince Edward, Hong Kong, for contributing some of the images in this article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This article was funded by a grant (SGP 9380113) from City University of Hong Kong to one of the authors (VRB).
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
