Abstract
During the period from January 2002 to March 2007, infections by melanized fungi were identified with greater frequency in aquarium-maintained leafy seadragons (Phycodurus eques)and weedy seadragons (Phyllopteryx taeniolatus), pivotal species to the educational and environmental concerns of the aquarium industry and conservation groups. The objective of this study was to characterize the pathology and identify fungi associated with phaeohyphomycotic lesions in these species. Samples from 14 weedy and 6 leafy seadragons were received from 2 institutions and included fresh, frozen, and formalin-fixed tissues from necropsy and biopsy specimens. Fresh and frozen tissues were cultured for fungi on Sabouraud dextrose agar only or both Sabouraud dextrose agar and inhibitory mold agar with gentamicin and chloramphenicol at 30°C. Isolates were processed for morphologic identification and molecular sequence analysis of the internal transcribed spacer region and D1/D2 domains of the large subunit ribosomal RNA gene. Lesions were extensive and consisted of parenchymal and vascular necrosis with fungal invasion of gill (11/20), kidney (14/20), and other coelomic viscera with or without cutaneous ulceration (13/20). Exophiala sp. isolates were obtained from 4 weedy and 3 leafy seadragons and were identified to species level in 6 of 7 instances, namely Exophiala angulospora (1) and a novel species of Exophiala (5), based on nucleotide sequence comparisons and phylogenetic analyses. Disseminated phaeohyphomycosis represents an important pathologic condition of both weedy and leafy seadragons for which 2 species of Exophiala,1a novel species, have been isolated.
Keywords
Introduction
The term “phaeohyphomycosis” is collectively used for cutaneous, subcutaneous, and systemic diseases caused by several genera of septate dark-walled fungi, referred to as “dematiaceous,” “phaeoid,” or “melanized.” Melanized fungi exhibit a high degree of molecular diversity with more than 100 species in 60 genera. 40 These agents, classified in various orders of the fungal kingdom, are ubiquitous and primarily recognized as soil saprophytes, plant pathogens, and environmental contaminants. Melanized fungi have been associated with disease in humans, 6,57 mammals, 11,18,28,61 birds, 32,56 amphibians, 12 reptiles, 30 fish, 19,60 and invertebrates. 8,62
Of special interest are members of the order Chaetothyriales, such as Exophiala and Cladophialophora, which are ecologically different and seem to be associated with assimilation of alkylbenzenes, compounds that are also present in vertebrate bodies. 50 These fungi are regularly encountered as causative agents of mycoses of medical and veterinary importance; only Onygenales, the order containing dermatophytes and dimorphic pathogens, has a comparable number of clinically relevant fungi. 16,17
Species of Exophiala represent a source of emerging fungal cutaneous, subcutaneous, and systemic infections, especially in immunocompromised human patients, 9,36,52,53,68 and animals. 10,27,31,39 In teleostean and cartilaginous fishes, Exophiala sp. infection has been reported in Atlantic salmon (Salmo salar L.) 46,54 and cutthroat trout (Oncorhynchus clarkii), 13 caused by Exophiala salmonis, in channel catfish (Ictalurus punctatus) 42 and smooth dogfish (Mustelus canis) 23 due to Exophiala pisciphila, and in captured King George whiting (Sillaginodes punctata) 51 in association with an Exophiala sp. Most recently, a novel species of Exophiala was isolated from Japanese flounder (Paralichthys olivaceus). 33
Seadragons are marine fish of the family Syngnathidae, which include the fused jawed fishes, such as seahorses and pipefish. Seadragons inhabit the shallow, temperate waters along the southern and western Australian coastline and Tasmania and are listed as near threatened on the Red List of Threatened Species by the International Union for the Conservation of Nature and Natural Resources (www.iucn.org). There are 2 main genera with a single species in each genus, namely the weedy seadragon (Phyllopteryx taeniolatus) and the leafy seadragon (Phycodurus eques). Seadragons are significant exhibit species of the aquarium industry and conservation groups because of their importance to research and educational efforts focused on marine coastal habitat conservation and ecosystem sustainability (www.dragonsearch.asn.au). 14,15 Over a period of 5 years from January 2002 to March 2007, slightly more than 400 seahorses, pipefish, and seadragons from several zoos and approximately a dozen commercial aquariums have been submitted to the Connecticut Veterinary Medical Diagnostic Laboratory (CVMDL; University of Connecticut, Storrs, CT) for necropsy. Of these syngnathid submissions, infections with melanized fungi were prevalent in leafy and weedy seadragons from 2 different aquariums. This report describes the characteristics of disseminated phaeohyphomycosis in seadragons caused by Exophiala spp., including infection by a novel (i.e., as yet undescribed) species referred to as “Exophiala sp. nov.”
Material and methods
Animals
Specimens consisted of juvenile and adult, captive-hatched seadragons reared in Australia prior to legal importation into the United States. Gender was undetermined for most animals due to sexual monomorphism when not in breeding condition. Animals were housed in groups of 1–10 animals in both species-specific and mixed species groups. Animals were maintained in filtered, natural, or artificial sea water at temperatures of 14–16°C, pH 8.0–8.2, total ammonia nitrogen <0.07 mg/l, nitrite 0 mg/l, nitrate <30 mg/l, and salinity 29–36 g/l in tanks ranging from 1,000 to 8,000 liters. Life support systems included mechanical and biological filtration with protein skimmers and ultraviolet and ozone disinfection. Diet consisted of live and frozen mysis shrimp (Mysidacea sp.), frozen zooplankton, and live brine shrimp (Artemia salina).
Weedy and leafy seadragons were submitted as part of routine diagnostic investigations to the CVMDL between January 2002 and March 2007. Fish were submitted from 2 different commercial aquariums located in 2 U.S. states and were presented either live or fixed in formalin. Seadragons were euthanized using an approximate dose of 400 mg/l of tricaine methanesulfonate a and were observed for 15–30 min past the last active opercular movement in consideration of the guidelines provided by the American Veterinary Medical Association 3 at the CVMDL or at the submitting institutions, or the animals died naturally. After initial identification of Exophiala sp., some animals were treated with little success using a variety of topical and systemic antifungal agents, including fluconazole, b voriconazole, b itraconazole, c terbenifine, d 37% w/v formaldehyde solution, d methylene blue, d malachite green, e acriflavine, e and Virkon. f Therapy for concurrent bacterial and protozoal infections varied for each case and included at least 1 of the following drugs: ceftazidime, g oxytetracycline, triple sulfa powder, metronidazole, kanamycin sulfate powder, e and chloroquine. h
Necropsy and histopathology
Gross necropsies were performed within 12 hr of death. Representative tissue samples or swabs of lesions were aseptically collected for wet mount preparations and microbial culture. Animals were dissected into multiple tissue samples or had gills and coelomic viscera exposed by removal of the operculums and a ventral midline incision with or without removal of the lateral body wall. Tissue samples or partially opened whole specimens were then fixed by immersion in 10% neutral buffered formalin. Bony tissues were decalcified after fixation using 0.5 M (molar [solution]) ethylenediamine tetra-acetic acid for 24–36 hr prior to trimming.
For preparation of histologic sections, formalin-fixed tissue samples were trimmed to fit plastic cassettes, routinely processed, embedded in paraffin, sectioned at 4 μm, mounted on glass slides, stained with hematoxylin and eosin, and then examined by bright field microscopy. Additional sections were stained with Fontana-Masson (FM), periodic acid–Schiff (modified McManus stain), and Grocott's methenamine silver techniques to highlight histomorphologic and staining characteristics of the fungi. 5,59
Microbial culture and fungus identification
Tissue samples and swabs were submitted for microbial testing, which included aerobic bacterial culture and separate fungal culture. Bacterial cultures from 4 weedy seadragons and 1 leafy seadragon were performed at a commercial veterinary diagnostic laboratory (IDEXX Laboratories, North Grafton, MA). Fungal cultures were performed at the CVMDL, where tissue samples were seared, sliced with a sterile surgical blade, and sampled with a cotton-tipped swab, which was then used to streak plates of Sabouraud dextrose agar i and inhibitory mold agar with gentamicin and chloramphenicol j in duplicate; plates were sealed using parafilm and incubated at 30°C. Swabs submitted for fungal culture were used directly to streak duplicate plates of the 2 types of media, which were then incubated at 30°C. Plates were incubated for up to 4 weeks and examined weekly for growth of mold. In cases in which an olivaceous to black, velvety mold was isolated, 1 plate of the pair was transferred to the Fungus Testing Laboratory (The University of Texas Health Science Center at San Antonio [UTHSCSA], San Antonio, TX) for morphologic and molecular identification.
Isolates were then transferred onto potato flakes agar (PFA) plates prepared in house and incubated at 25°C. 55 Both the macroscopic morphology of the colonies and the diagnostic microscopic features were determined from this medium after approximately 12 days of incubation. Microscopic features were studied using the slide culture technique 26 and modified to contain PFA rather than water agar for both the nutrient and the moisture source. Temperature studies were performed at 30°C, 35°C, and 40°C on PFA slants; nitrate assimilation was assessed using previously described methods. 49 Brown pigment formation was evaluated on Sabouraud dextrose agar.
Molecular fungus identification and phylogenetic analysis
Genomic DNA was extracted from conidia recovered from a 72-hr PFA plate using Prepman Ultra reagent k according to the manufacturer's instructions. Two polymerase chain reaction (PCR) amplifications were performed on each isolate, and molecular analyses were focused on ribosomal genes for phylogenetic inference. The first reaction amplified the internal transcribed spacer (ITS) region located between the 18S and 28S rRNA genes using primers ITS1 (5′ -TCCGTAGGTGAACCTGCGG-3′) and ITS4 (5′-TCCTCCGCTTATTGATATGC-3′). 21,65 The second reaction amplified the D1/D2 region of the 28S rRNA gene using primers NL-1 (5′-GCATATCAATAAGCGGAGGAAAAG-3′) and NL-4 (5′-GGTCCGTGTTTCAAGACGG-3′). 34,48 Both PCR amplifications were performed in 50-μ1 reaction volumes using Triple Master Taq DNA polymerase, d each deoxyribonucleotide triphosphate (dNTP), l and primers (prepared at the UTHSCSA Nuclear Core Facility) at concentrations specified by the manufacturer's instructions. All PCR amplifications were performed in a commercial thermocycler m using a preprogrammed 3-step protocol as the standard program for all reactions. Cycling conditions consisted of an initial denaturation step at 95°Cfor 10 min, followed by 30 cycles, each cycle consisting of denaturation at 95°C for 1 min, annealing at 58°Cfor 1 min, and extension at 72°C for 1 min, with a final extension at 72°C for 10 min. Amplification success was confirmed by agarose gel electrophoresis. The remaining template DNA was then cleaned by polyethylene glycol precipitation. l Sequencing was performed on both strands using the PCR primers as sequencing primers at the UTHSCSA Advanced Nucleic Acids Core Facility. The resultant sequences were compared with available sequences at the National Center for Biotechnology Information using BLASTn (nucleotide database using a nucleotide query, http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) searches of the databases, 1,2 as well as by comparison to the sequence database of black molds at the Centraalbureau voor Schimmelcultures (CBS; http://www.cbs.knaw.nl/databases/). The ITS sequences were aligned using BioNumerics 4.6 software. n A substitution model was calculated using MrAIC (http://www.abc.se/∼nylander), and the tree was built using TREEFINDER algorithm (http://www.treefinder.de) version June 2007 and bootstrapping with 1,000 replicates; values >80 are shown with the branches.
Results
Animals
Twenty aquarium-held weedy (14) and leafy (6) seadragons were evaluated. Animals were in the collection for 2 weeks to 4 years (mean = 32 weeks) prior to onset of clinical signs. Duration of clinical signs ranged from 1 week to 6 months (mean = 8 weeks). Clinical signs included weakness, loss of appetite, lethargy, increased respiratory rate and effort, abnormal buoyancy, listing, piping at the surface of the water, and death. Fungal dermatitis was diagnosed antemortem in 6 cases via cytology or biopsy of lesions, and antemortem fungal culture isolated Exophiala sp. nov. in 2 such cases.
Pathologic findings
Seadragons were submitted to the CVMDL with either antemortem evidence of fungal infection from cytology or biopsy or black lesions suggestive of phaeohyphomycosis. Lesions were identified in skeletal muscle (18/20), skin (16/20), kidney (14/20), gill (11/20), swim bladder (7/20), heart (2/20), liver (3/20), spleen (1/20), muscle coats and serosa of the intestine (2/20), mesentery (1/20), and extradural sinus and spinal cord (6/20); 15 seadragons had lesions in 3 or more of these tissues (Table 1). The most obvious gross lesions were identified in the skin and consisted of 1 to several well-demarcated, occasionally extensive, ulcerations, often with raised black margins, located randomly over the head, trunk, dorsum of the tail, at the base of fins, or in skin around the cloaca (Fig. 1). Microscopically, cutaneous lesions were characterized by ulcerations of the epidermis and extensive mats of fungal hyphae that invaded dermal fibrous connective tissue and extended into the underlying hypodermis, fascia, and skeletal muscle with myonecrosis and mild histiocytic infiltrates.
Multiple, well-demarcated, and occasionally extensive black foci were identified grossly in the kidney, gill, swim bladder, and intestinal wall of seadragons upon internal examination. Microscopically, renal lesions consisted of extensive regions of necrosis involving tubules, hematopoietic interstitium, and sinusoids that were infiltrated throughout by fungal hyphae and corresponded to grossly visible black, friable parenchyma (Fig. 2). Hyphae invaded the overlying epaxial muscle with myonecrosis and mild histiocytic infiltrates. Gill lesions consisted of focally extensive necrosis of filaments, their lamellae, the underlying connective tissue, and blood vessels of the gill arch, with minimal to occasionally moderate loosely organized infiltrates of macrophages (Fig. 3). Lesions in the intestine and swim bladder were limited to the muscle coats and serosa and were characterized by foci of myonecrosis, infiltrated by fungal hyphae, and minimal to moderate, loosely organized infiltrates of macrophages. In the liver, hyphae invaded venules and sinusoids with dissociation and necrosis of hepatocytes and pancreatic acini in 2 instances. Hyphae coursed through venules of the liver and the reticuloendothelial stroma of the spleen and invaded mesenteric blood vessels in 1 specimen. Small numbers of hyphae were present in the extradural sinus of 6 specimens and invaded the spinal cord in 3 of these instances.
Anatomic distribution of phaeohyphomycotic lesions in seadragons in the current study.*
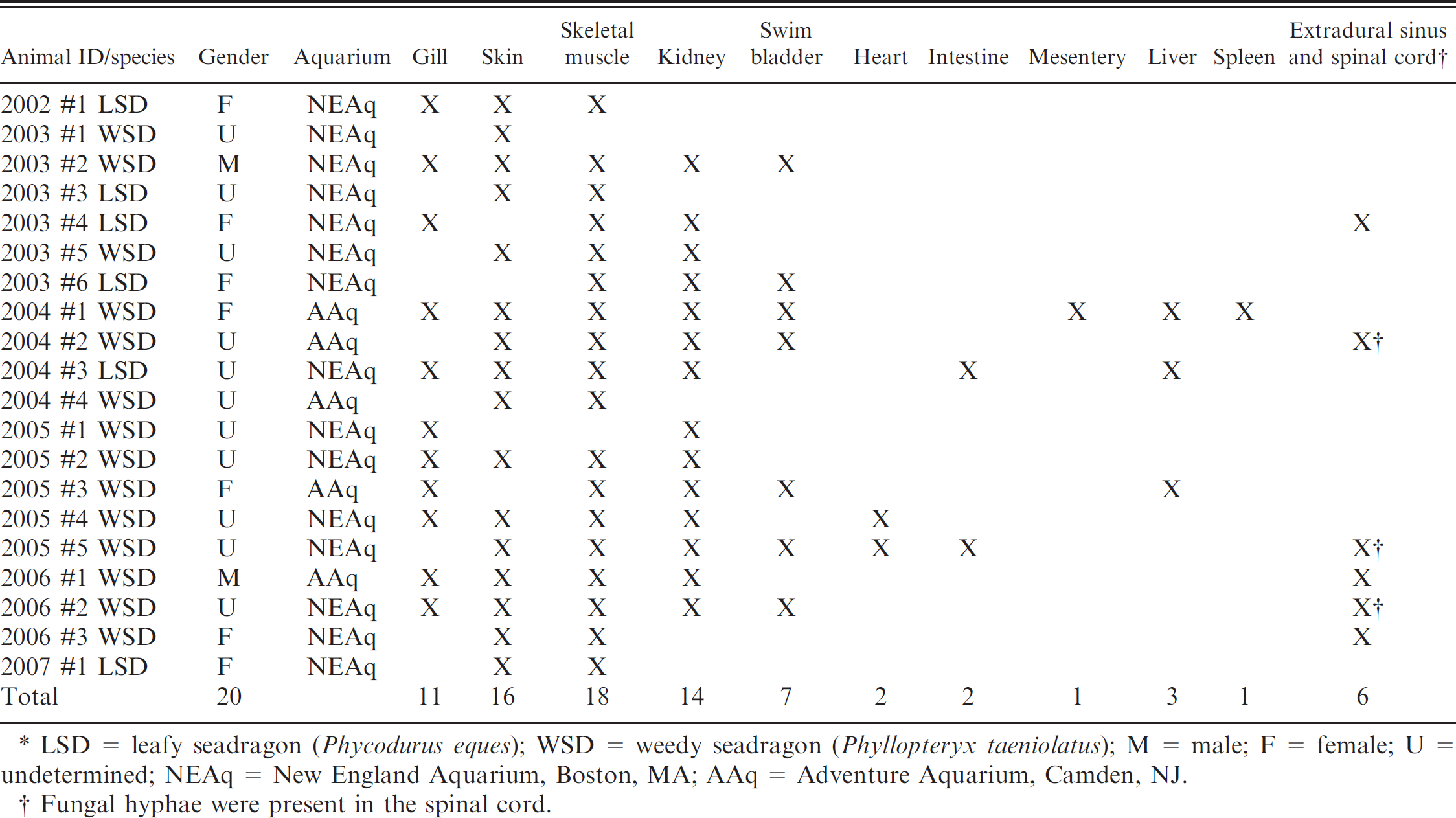
LSD = leafy seadragon (Phycodurus eques); WSD = weedy seadragon (Phyllopteryx taeniolatus); M = male; F = female; U = undetermined; NEAq = New England Aquarium, Boston, MA; AAq = Adventure Aquarium, Camden, NJ.
Fungal hyphae were present in the spinal cord.
Lumina of blood vessels of gill and viscera contained intertwined fungal hyphae together with variable amounts of fibrin, serum protein, and necrotic leukocytes, accompanied by necrosis of vessel walls (Fig. 4). Fungal hyphae were 2–3 μmin width, slender, filamentous, and septate, with right-angle branching and thin parallel walls that stained brown in routine hematoxylin and eosin–stained sections and in sections prepared using the Fontana-Masson technique, consistent with the expected histochemical staining reaction of melanized fungi (Fig. 5). 66,67 Ciliated protozoa, consistent with Uronema sp., and aggregates of Gram-negative, rod- shaped bacteria were identified on cytology of affected gill and skin in 5 cases. Other pathologic findings included enteric coccidiosis in weedy seadragons (2/20), biliary (1/20) and renal myxozoanosis (5/20) in weedy seadragons, and parasitic (3/20) and mycobacterial (1/20) granulomas in visceral organs.
Microbial culture and fungus identification
Fungal cultures were attempted in 4 weedy and 3 leafy seadragons involving 1 or more samples from kidney, skin, liver, and/or spleen; isolates were identified to species level in 6 of 7 instances (Table 2). Isolates grew on both Sabouraud dextrose agar and inhibitory mold agar with gentamicin and chloramphenicol at 30°C over a period of 1–3 weeks. Isolates yielded velvety, olivaceous, or brown-black molds and were referred to the Fungus Testing Laboratory over several months. In most isolates, the colonies were generally restricted, were somewhat moist initially, had a moderate growth rate, were olivaceous to olivaceous-gray with a black reverse, and became velvety at maturity. Both E. angulospora and Exophiala sp. nov. were initially yeast-like but displayed budding cells throughout their growth cycle. These cells soon germinated with the formation of pale, olivaceous torulose hyphae. Conidiation was annellidic, and annelloconidia were either single-celled (2–3 μm × 4–5 μm for Exophiala sp. nov.; Fig. 6) or commonly angular (2–3 μm × 4–6 μm for E. angulospora). Conidia were borne in slimy masses at the tips of flask-shaped to cylindrical annellides and from intercalary conidiogenous loci. All isolates assimilated nitrate, none were able to grow at 35°C, and none produced a brown diffusible pigment on Sabouraud dextrose agar suggestive of E. dermatitidis. Given the very similar microscopic and physiologic profiles of several species of Exophiala, only E. angulospora, which produces distinct angular annelloconidia, could be identified with reasonable certainty based upon morphologic features alone. A variety of Gram-negative bacteria, including Vibrio sp. and Pseudomonas sp., were cultured from lesions. No species of bacteria was consistently isolated from these lesions.
Molecular fungus identification and phylogenetic analysis
The ITS tree was built using TREEFINDER with substitution model GTR+G according to MrAIC calculations. The same program calculated bootstrapping values that gave 100% for all branches. Isolates were found to belong to distinct species of Exophiala. In addition to E. angulospora, a second, hitherto unnamed species was encountered, clearly separate from any other taxon based on rDNA ITS sequences. Because many species of black yeasts differ by mutations rather than by indels or amplicon lengths, greater than 1% ITS sequence diversity exceeds the species level in this group. Figure 7 gives an overview of clades of nearest taxa in the order Chaetothyriales. Sequences dH16401, dH13448, and CBS 119918 represent isolates obtained from 1 leafy seadragon sampled in 2000 by 1 of the 2 aquariums. These sequences were initially identified as E. pisciphila but were subsequently found to be Exophiala sp. nov. when submitted to the CBS for molecular characterization along with sequences from other Exophiala sp. isolates obtained from seadragons and other aquatic animals. Tissue samples for histopathologic evaluation were not received from this 2000 seadragon for inclusion in the current study; nevertheless, inclusion of these sequences together with sequences of Exophiala isolates from other fish contributes to the distinct clade designation and host predilections that characterize Exophiala sp. nov. Nearly all species belonged to the black yeast genus Exophiala having annellidic conidiogenesis, whereas Veronaea botryosa with large sympodial conidiophores was found to be a member of this clade. Most species in the clade have originated from watery environments and have also been isolated from diseased fish and amphibians. In some species, strains from human origins were present and mostly associated with mild cutaneous infections. Efforts are presently under way to provide a formal description of Exophiala sp. nov. in a taxonomic paper, wherein this species will be introduced as a novel taxon (M. J. Harrak, G. S. de Hoog, unpublished data).
Discussion
Environmental fungi are increasingly important sources of infection to humans and animals. The emergence or resurgence of fungi as pathogens, including those previously considered environmental contaminants, have been associated with a wide range of globally relevant medical, societal, and economic factors, such as increasing populations of immunocompromised individuals, 4,22,36,52,53 international travel, changes in land use and agriculture, and even migration of clouds of desert dust in the atmosphere. 25 International commerce of farmed North American bullfrogs (Rana catesbeiana) used for the restaurant trade, for example, has been implicated as a cause of chytridiomycosis in wild amphibians elsewhere around the globe. 41 Ornamental fish and the aquarium industry represent a commercial source of regional and international translocation of innumerable species that can afford environmental fungi the opportunity to infect new hosts.
Exophiala spp. are ubiquitous in soil and aquatic environments and often considered environmental contaminants. Reports of Exophiala spp. infection in domestic and wild animals are few and include subcutaneous 10 and systemic 27 lesions in cats by E. jeanselmei and a subcutaneous mass in the right neck of a dog by E. dermatitidis. 31 In nondomestic animals, E. jeanselmei was isolated from a subcutaneous lesion in a free-ranging eastern box turtle, 30 and an Exophiala sp. with close similarity to E. pisciphila was isolated from systemic lesions in a Galapagos tortoise. 39 Exophiala spp., including Wangiella (Exophiala) dermatitidis, E. jeanselmei, E. oligosperma, and E. spinifera, are more common causes of phaeohyphomycotic lesions in immunocompromised humans and encompass ocular, 47 cutaneous, subcutaneous, 68 and occasional systemic infections, wherein a history of chronic debilitating disease, altered immune status, and/or chemotherapy was reported. 9,36,52,53 Exophiala salmonis, known to be a pathogen of fish, 13,46,54 has been associated with subcutaneous phaeohyphomycosis in a human patient who most likely acquired the infection from a water source. 38 This highlights the zoonotic potential of Exophiala spp. and the potential effect aquatic pathogens may have on human health.
Exophiala spp. have been identified as significant pathogens of cultured fish, such as cutthroat trout, 13 Atlantic salmon, 46 channel catfish, 42 and Japanese flounder, 33 resulting in localized and systemic infections with a notable variety of inflammatory responses. A dematiaceous fungus subsequently named Exophiala salmonis was reported to be the cause of epizootics of cerebral mycetoma in fingerling cutthroat trout. 13 The lesion due to E. salmonis was characterized by granuloma formation with numerous giant cells in the brain and cranial tissues; it developed first in the brain, then extended peripherally to include surrounding cranial structures, such as the eye and gill. Similarly, in Exophiala pisciphila infection associated with high mortality of Atlantic salmon, hyphae invaded cranial structures, including semicircular canals, and the lateral line, accompanied by a granulomatous inflammatory reaction. 35 Contaminated food was suggested as a source of infection by E. salmonis in Atlantic salmon that developed granulomas in the posterior kidney. 54 Exophiala salmonis infection was later described in 3 Atlantic salmon held in a partial reuse system for up to 20 months. 46 Lesions in these salmon were systemic, including the kidney, and differed from earlier reports in that the host inflammatory response was predominantly granulocytic, with the formation of microabscesses. In contrast, lesions in Japanese flounder were limited to the skin. 33
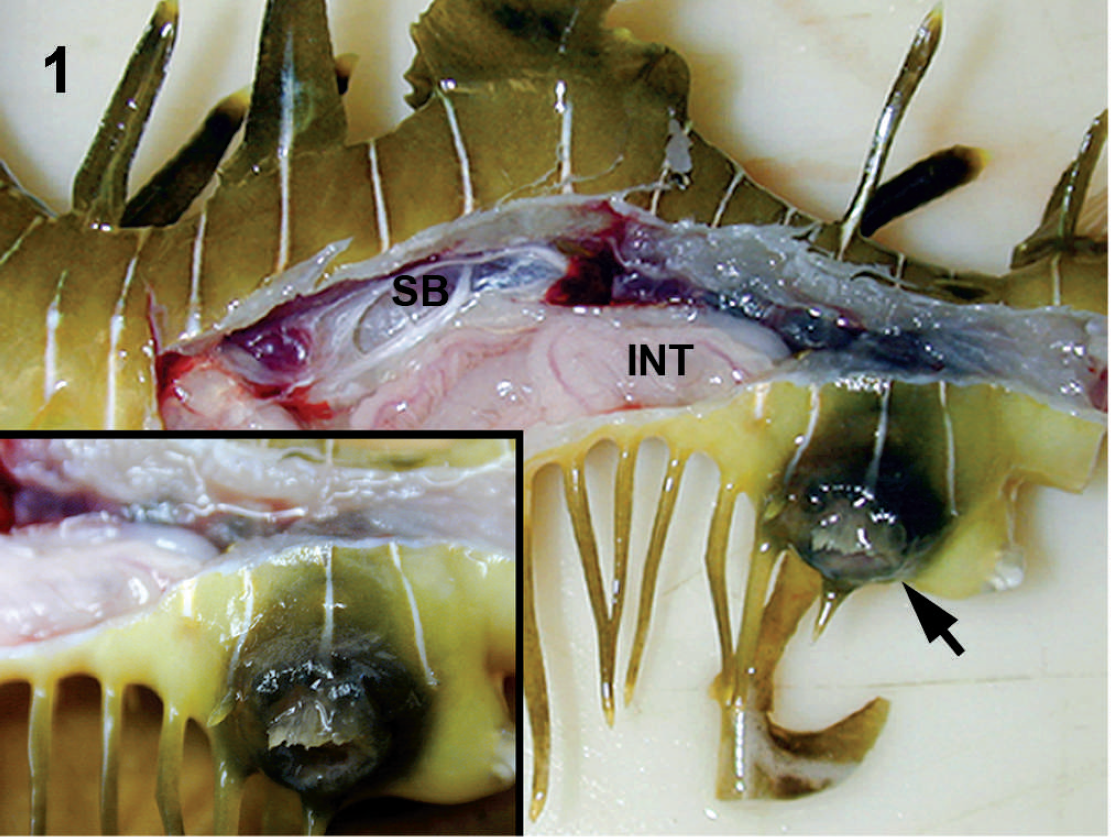
Skin ulcer; leafy seadragon with lateral body wall removed to expose coelomic viscera. An ulcer (arrow) is located in the skin adjacent to the cloaca. Inset: Closer view of the ulcer with raised black margins. SB = swim bladder; INT = intestine.

Transverse section of dorsal trunk; weedy seadragon.
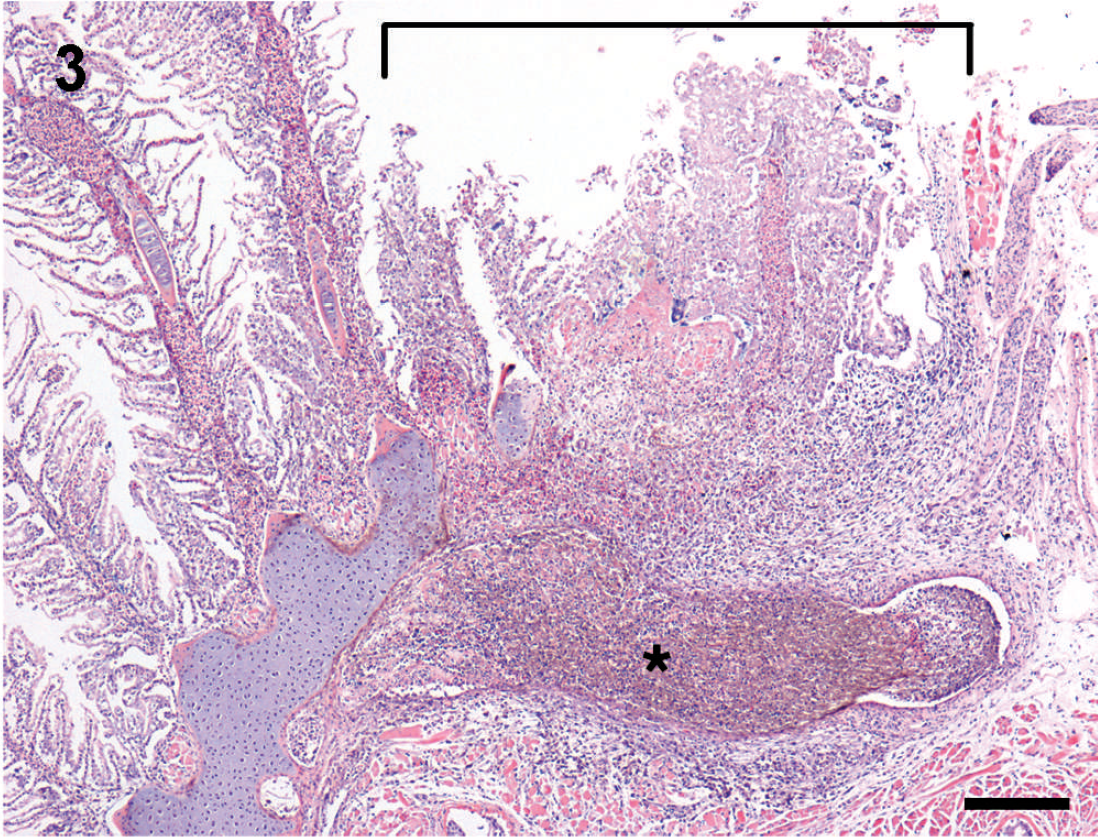
Gill; leafy seadragon. There is focally extensive necrosis of several consecutive filaments and their lamellae (bracket) overlying a region of the arch wherein a mat of densely intertwined brown fungal hyphae (asterisk) resides within the venous sinus. Hematoxylin and eosin. Bar = 200 μm.

Blood vessel, kidney; weedy seadragon. Intertwined hyphae are present in the blood vessel lumen, and there is necrosis of a segment of the wall. Hematoxylin and eosin. Bar = 200 μm.

Fungal hyphae, kidney; weedy seadragon. Hyphae are slender, filamentous, and septate with occasional right-angle branches. Walls of hyphae stain brown, indicative of melanin. Fontana-Masson. Bar = 25 μm.
Exophiala sp. isolated from seadragons in the current study.*
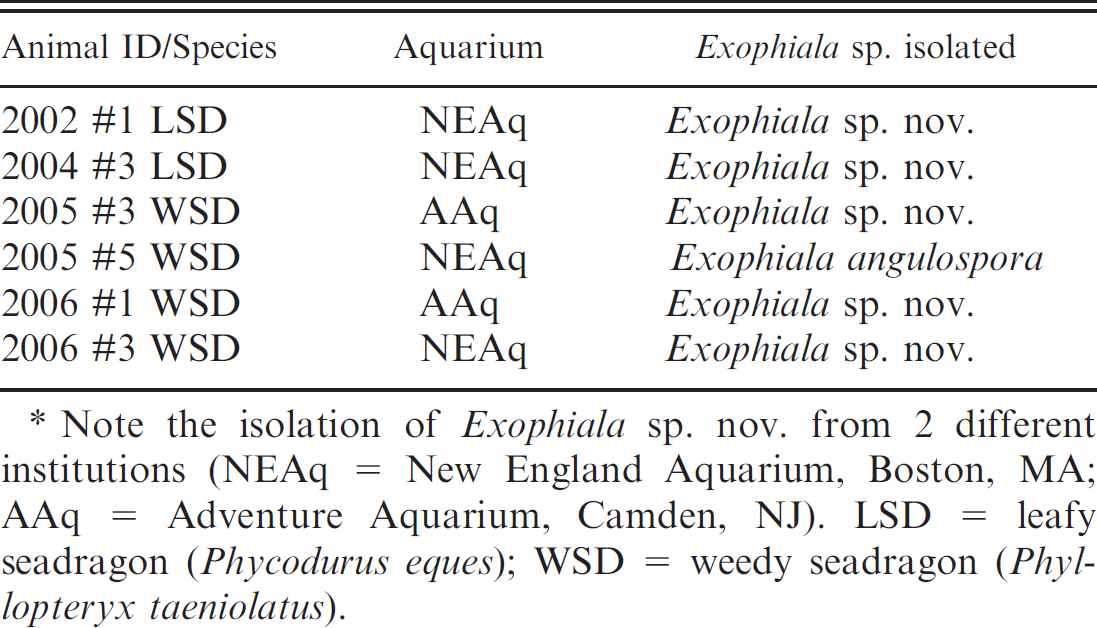
Note the isolation of Exophiala sp. nov. from 2 different institutions (NEAq = New England Aquarium, Boston, MA; AAq = Adventure Aquarium, Camden, NJ). LSD = leafy seadragon (Phycodurus eques); WSD = weedy seadragon (Phyllopteryx taeniolatus).
In the current study, systemic necrotizing lesions and invasion of blood vessels were consistent features of Exophiala spp. infection in seadragons. Necrosis was the predominant pathologic change, and the host inflammatory infiltrates were mild compared with the extent of necrosis and consisted mainly of histiocytes. Granulomas and abscesses were not consistently identified in seadragons, nor was there involvement of the brain, vestibular apparatus, or lateral line. Three of 20 seadragons did, however, have fungal hyphae that invaded the spinal cord, which may have resulted from invasion of the overlying extradural sinus.
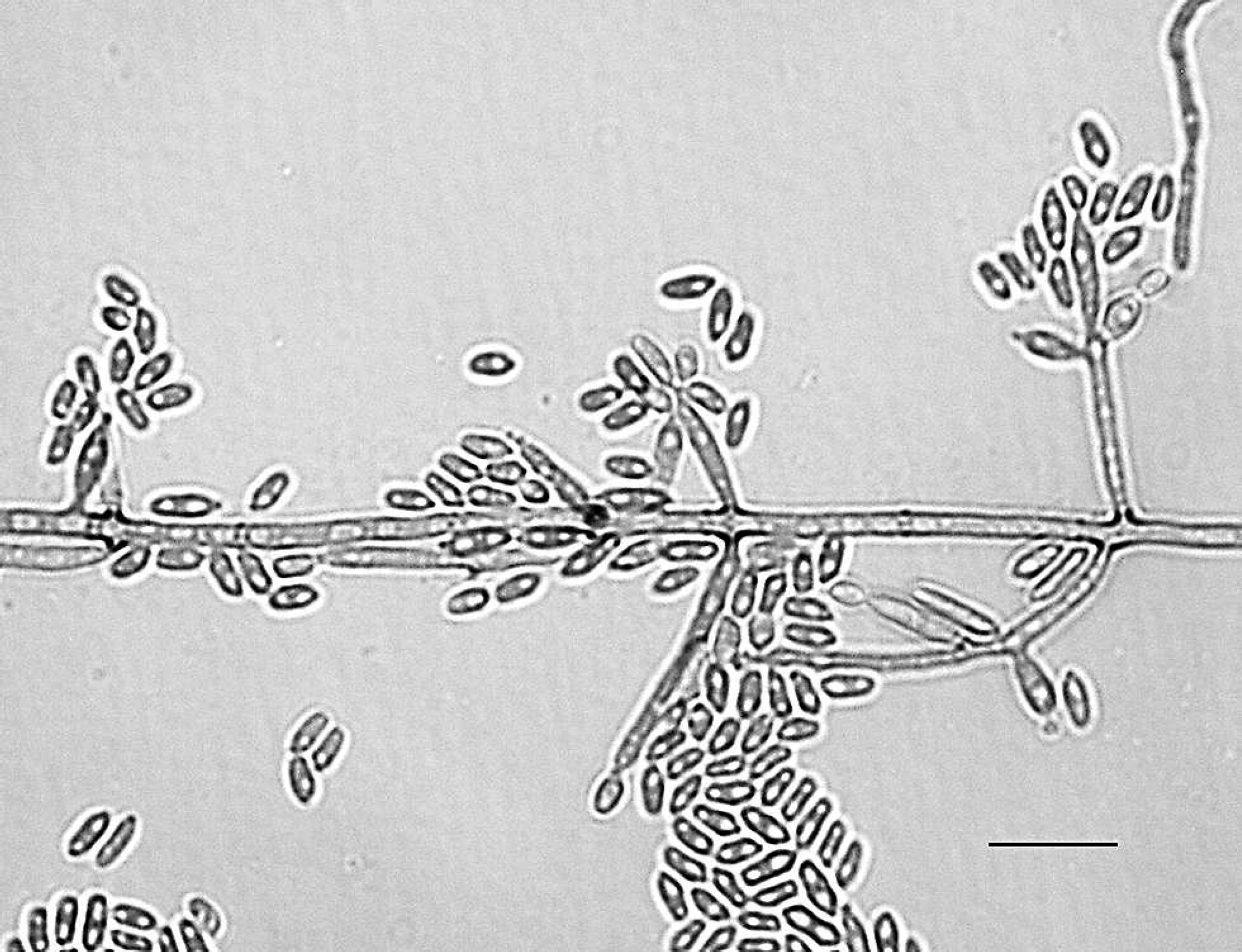
Microscopic colonial morphology of Exophiala sp. nov. showing septate hyphae with multiple annellides and conidiogenous loci bearing single-celled, approximately 2–3 μm × 4–5 μm conidia. Lactophenol cotton blue. Bar = 10 μm.
The portal of entry in these cases is presumed to be gill or skin by traumatic inoculation or secondary inoculation of preexisting lesions, such as abrasions of the tubed snout, with subsequent hematogenous dissemination to visceral organs. Another portal of entry to consider could be direct ingestion of fungi associated with detritus and live or thawed frozen food accumulating in the tank substrate in aquarium settings. In the seadragon cases, however, there were no intestinal mucosal lesions associated with fungal invasion; instead, lesions were limited to the muscle coats and serosa, suggestive of hematogenous or transcoelomic spread.
The absence of granuloma formation or significant host inflammatory reaction to Exophiala spp. infection in these seadragons could be the result of an inadequate or deficient host immunologic response. No studies have been conducted to determine immune function in seadragons, but it could be that stress of captivity may be a contributory factor to reduced immune function. Fish in captive, artificial systems, or intensive rearing operations may be immunosuppressed or otherwise compromised and therefore predisposed to infections by Exophiala spp. and other environmental fungi. 46 Reports of infection with pigmented fungi in tropical marine fish are usually believed to be secondary to immunosuppression resulting from transport, trauma, or confinement-induced stress. 60 There were no mortalities of other species of fish housed in the same tanks as these seadragons, suggesting that this infection is species specific. This hypothesis is supported by the fact that species in the clade of psychotolerant, waterborne melanized fungi (Chaetothyriales) each show somewhat different host predilections (Fig. 7).
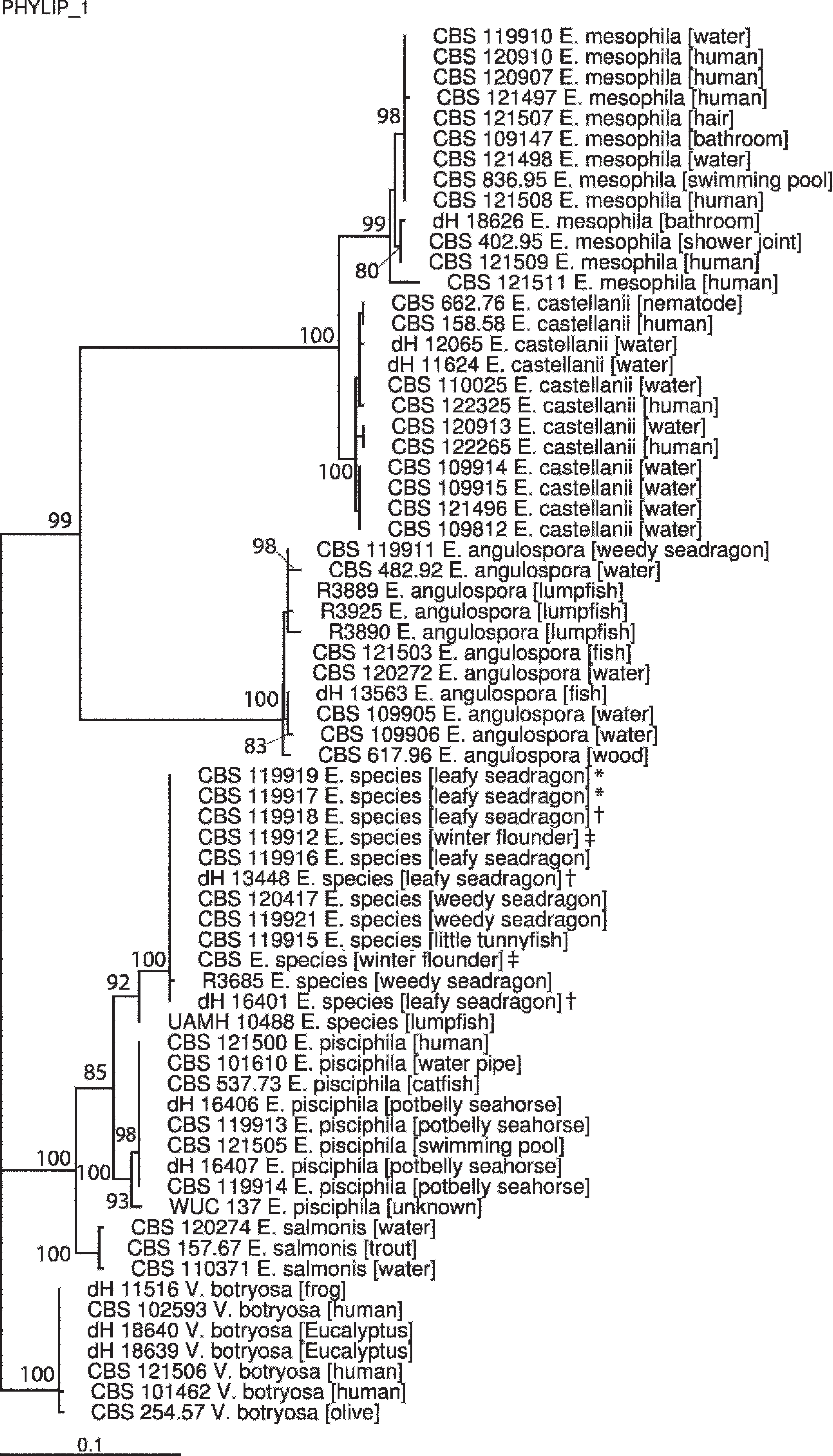
Consensus tree of prevalently waterborne Exophiala species, based on internal transcribed spacer (ITS) ribosomal DNA of 68 strains, constructed with neighbor-joining algorithm under the HKY+G substitution model (according to MrAIC), with 1,000 bootstrap replicates (according to TREEFINDER; values ≥80 are shown with the branches) and edited with Fig Tree version 1.0. Sequences are trimmed at GGCCC to (T/C)AGGGA for comparison. Veronaea botryosa was selected for rooting the tree. ITS sequences from Exophiala sp. nov. isolates form a distinct clade separate from other Exophiala species and supported by very high bootstrap value (100). Symbols following taxa (*, †, ‡) indicate sequences of isolates obtained from the same individual.
Studies on virulence factors in phaeoid fungi, such as Wangiella (Exophiala) dermatitidis, have identified synthesis of melanin within cell walls as the main virulence factor. 20,29,44 Melanin scavenges free radicals and hypochlorite produced by phagocytic cells and/or binds hydrolytic enzymes, and it is also believed to be important in the formation of the fungal appressorium, a structure that aids in entering the host cells. 20,29,63 The ability of these fungi to cause disseminated infection has been associated with the resistance afforded by melanin to oxidative damage by host phagocytic mechanisms. Melanin production in the context of host infection is not limited to phaeoid fungi, however, and melanin has been identified as a virulence factor in certain dimorphic fungi, such as Cryptococcus neoformans, 64 Paracoccidioides brasiliensis, 24 and Histoplasma capsulatum, 45 fungi that are considered emerging pathogens in both domestic animals and humans. Few studies have examined the role of chitin as a virulence factor in melanized fungi. Chitin serves to provide additional strength to fungal cell walls, 37,63 and disruption of chitin synthases has been shown to affect growth of fungi at temperatures of infection. 37 Recently, the assimilation of alkylbenzenes, which occur as environmental pollutants but also in vertebrates as neurotransmitters, has been suggested as a virulence determinant specific to Chaetothyriales. 50
Initial diagnoses in the seadragon cases in the present study were made from gross lesions observed in live and dead animals, cytologic examination of affected tissues, and characteristics of fungi in histologic sections. In instances in which frozen or fresh tissues representing suspected lesions were available, fungal culture of the kidney, skin, liver, and/or spleen consistently yielded velvety, olivaceous, or brown-black molds, which upon further molecular testing were identified as species of Exophiala. The genus is morphologically characterized by the presence of annellated zones producing annelloconidia from nearly undifferentiated conidiogenous cells. However, based on rDNA ITS sequence data, it was found that Veronaea botryosa, with large conidiophores and 2-celled sympodial conidia, is a member of this clade. Given the fact that the entire clade shows an association with watery environments (ranging from ocean water to drinking water) at cold to mild temperatures, it was concluded that ecology is a prime factor in the phylogeny of these species. Species found in somewhat warmer environments, such as bathrooms and swimming pools, are recurrently encountered as agents of mild human disease, infecting external body parts, such as skin of the extremities, and occasionally nasal sinuses (de Hoog, personal communication, 2008).
Such bacteria as Vibrio spp., Pseudomonas spp., and Mycobacterium spp. are commonly found in sea water and likely represent opportunistic invaders or potential members of normal skin flora. 7,43 Although a variety of Gram-negative bacteria were isolated from skin lesions, no species of bacteria was consistently isolated. Cutaneous lesions in the seadragons were often advanced when examined histologically so that a determination as to whether the inciting lesion was bacterial, fungal, or traumatic was not possible. In contrast, visceral lesions and vascular invasion were fungal in nature, and bacteria were not identified.
To the authors' knowledge, this is the first report describing disseminated phaeohyphomycosis with isolation of Exophiala spp. in seadragons. Seadragon habitats, such as algal covered reefs and seagrass meadows, are being adversely affected by human activities, and loss in quality and quantity of habitat has been documented (http://www.dragonsearch.asn.au; www.iucn.org). 14,15,58 Seadragons are difficult to culture and are susceptible to stress of confinement, poor diet, and trauma. Disseminated Exophiala spp. infection, as described in these cases, poses challenges to the management and conservation efforts of these fish.
Acknowledgements
The authors thank Deana Edmunds, Vickie Weidig, and Sharon Gagnon for necropsy support; Ione Jackman and Denise Woodward for histotechnological support; and the animal health and husbandry staff at the aquariums who provided care to these fish. The authors also thank Elizabeth Thompson of the Fungus Testing Laboratory for her work on the morphologic identification of these isolates and acknowledge Drs. Terri Wheeler (IDEXX Laboratories, North Grafton, MA) and Amy Grooters (Louisiana State University, Baton Rouge, LA) for their contributions to the early isolations and characterizations. B. L. Wickes is supported by grant PR054228 from the U.S. Army Medical Research and Material Command, Office of Congressionally Directed Medical Research Programs.
Footnotes
a.
Finquel/MS-222, Argent Chemical Laboratories Inc., Redmond, WA.
b.
Pfizer Inc., New York, NY.
c.
Janssen Pharmaceutica, Beerse, Belgium.
d.
Fisher Scientific Co., Pittsburgh, PA.
e.
National Fish Pharmaceuticals, Tucson, AZ.
f.
Virkon®, Antec International Ltd., Sudbury, Suffolk, UK.
g.
GlaxoSmithKline, Brentford, Middlesex, UK.
h.
Sigma-Aldrich, St. Louis, MO.
i.
Northeast Laboratory Services, Waterville, ME.
j.
Hardy Diagnostics, Santa Maria, CA.
k.
Applied Biosystems, Foster City, CA.
l.
Invitrogen Corp., Carlsbad, CA.
m.
PTC-100, MJ Research, Inc., Waltham, MA.
n.
Applied Maths NV, Sint-Martens-Latem, Belgium.
