Abstract
Clinical relevance:
Invasive fungal infections (IFIs) and oomycoses (hereafter termed invasive fungal-like infections [IFLIs]) are characterised by penetration of tissues by fungal elements. The environment is the most common reservoir of infection. IFIs and IFLIs can be frustrating to treat because long treatment times are usually required and, even after attaining clinical cure, there may be a risk of relapse. Owner compliance with medication administration and recheck examinations can also decline over time. In addition, some antifungal drugs are expensive, have variable interpatient pharmacokinetic properties, can only be administered parenterally and/or have common adverse effects (AEs). Despite these limitations, treatment can be very rewarding, especially when an otherwise progressive and fatal disease is cured.
Aim:
In the second of a two-part article series, the spectrum of activity, mechanisms of action, pharmacokinetic and pharmacodynamic properties, and AEs of antifungal drugs are reviewed, and the treatment and prognosis of specific IFIs/IFLIs - dermatophytic pseudomycetoma, cryptococcosis, sino-orbital aspergillosis, coccidioidomycosis, histoplasmosis, sporotrichosis, phaeohyphomycosis, mucormycosis and oomycosis - are discussed. Part 1 reviewed the diagnostic approach to IFIs and IFLIs.
Evidence base:
Information on antifungal drugs is drawn from pharmacokinetic studies in cats. Where such studies have not been performed, data from ‘preclinical’ animals (non-human studies) and human studies are reviewed. The review also draws on the wider published evidence and the authors’ combined expertise in feline medicine, mycology, dermatology, clinical pathology and anatomical pathology.
Abbreviations for antifungal drugs:
AMB (amphotericin B); FC (flucytosine); FCZ (fluconazole); ISA (isavuconazole); ITZ (itraconazole); KCZ (ketoconazole); PCZ (posaconazole); TRB (terbinafine); VCZ (voriconazole).
Keywords
General pretreatment considerations
For invasive fungal infections (IFIs) and oomycoses (hereafter termed invasive fungal-like infections [IFLIs]) in cats, antifungal susceptibility testing should be performed when possible, as outlined in Part 1. 1 Targeted antifungal therapy based on the susceptibility profile of the pathogen isolated is an important factor in determining treatment success.

Penetration of antifungal drugs at the site of infection to achieve minimum inhibitory concentrations (MICs) against the target pathogen is essential for efficacy. In general, only free drug is considered biologically active; thus, drug concentrations in interstitial fluids (eg, cerebrospinal fluid [CSF]) are important determinants of antifungal efficacy. 2 Small polar compounds with low plasma binding (eg, fluconazole [FCZ], isavuconazole [ISA], flucytosine [FC]) have volumes of distribution similar to total body water, penetrate well into aqueous sites (eg, ocular fluid, CSF) and have body fluid:plasma concentration ratios of ~1. In contrast, highly lipophilic drugs (eg, itraconazole [ITZ], posaconazole [PCZ]) that are also highly protein-bound have much higher volumes of distribution, penetrate best into tissues with high lipid content and have tissue:plasma concentration ratios of >1, but may not penetrate aqueous sites. 2

For azoles, FC and echinocandins, the ratio between the area under the plasma concentration-time curve (AUC) and the MIC (AUC:MIC) for the fungal pathogen best correlates with antifungal efficacy. For amphotericin B (AMB), which is a concentration-dependent antifungal with a post-antifungal effect, the ratio between peak concentration (Cmax) and the MIC (Cmax:MIC) correlates best with efficacy. 3

Antifungal drugs used in the treatment of invasive fungal and fungal-like infections
Amphotericin B
AMB is a polyene macrolide antibiotic.
Antifungals used in the treatment of invasive fungal infections in cats
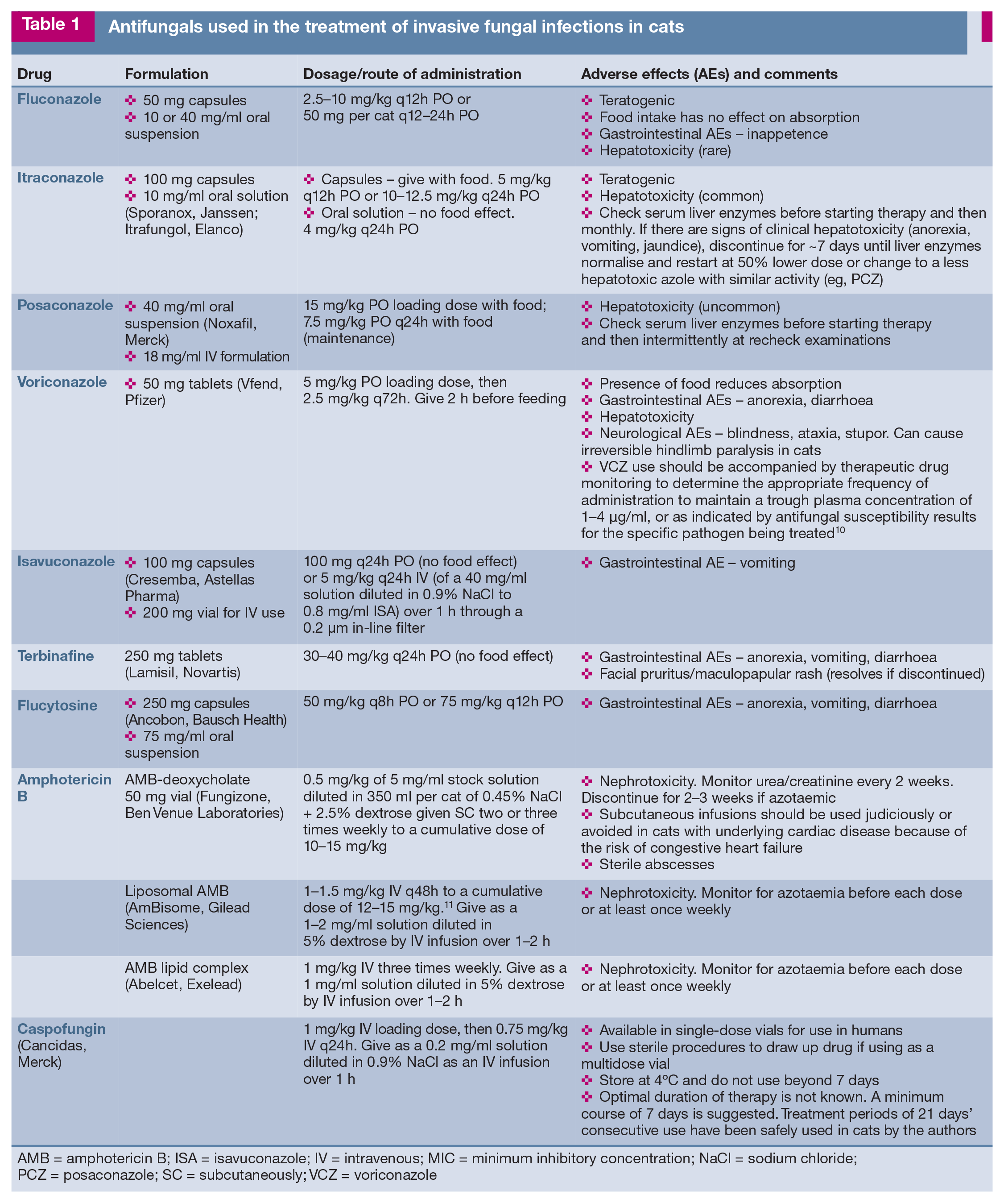
AMB = amphotericin B; ISA = isavuconazole; IV = intravenous; MIC = minimum inhibitory concentration; NaCl = sodium chloride; PCZ = posaconazole; SC = subcutaneously; VCZ = voriconazole
Flucytosine
Flucytosine (FC), also known as 5-flucytosine, is a pyrimidine analogue.


Synthesis of ergosterol – an essential fungal cell membrane component – commences with the conversion of squalene to lanosterol by the enzyme squalene epoxidase (1). Lanosterol is converted to ergosterol by binding with the cytochrome P450 (CYP450)-dependent enzyme 14α-demethylase (2). Antifungal drugs exert their effects by interfering with these biosynthetic processes:
Azole antifungals
Azole antifungals inhibit 14a-demethylase, an enzyme belonging to the cytochrome P450 family (Figure l), which is also known as CYP51 in moulds and ERG11 in yeasts. It is encoded by CYP51A/CYP51B and ERG11 for moulds and yeasts, respectively. 3 Azoles bind to the haem group of 14a-demethylase to prevent conversion of lanosterol to ergosterol, an essential fungal cell membrane component. Ergosterol depletion and the accumulation of methylated sterol precursors results in inhibition of fungal cell growth and/or fungal cell death.
Fluconazole
- yeasts (Cryptococcus, Candida and Malas se zia species); and
- thermally dimorphic fungi (Histoplasma, Blastomyces, Coccidioides and Sporothrix species).
FCZ has no or limited activity against moulds (including Aspergillus species), dema-tiaceous fungi and Zygomycetes. 17
Pharmacokinetic parameters (mean ± SD) and properties of triazole antifungals in cats after oral administration

After a mean dose of 17.5 mg/kg q24h 24
51% in dogs, 58% in humans 25
AUC0-∞ = area under curve from time 0 to infinity; Cmax = peak concentration; Css = concentration at steady state; Tmax = time to peak concentration;
β T1/2 = elimination rate half-life; NA = not available
Itraconazole
ITZ is the most common antifungal drug used in feline practice and has replaced KCZ for the treatment of IFIs because of its higher potency, broader spectrum of activity and improved safety profile.
- yeasts (Cryptococcus, Candida and Malassezia species);
- thermally dimorphic fungi (Histoplasma, Coccidioides and Sporothrix species);
- moulds (dermatophytes and some Aspergillus species); and
- some dematiaceous fungi.
ITZ has no or limited activity against cryptic Aspergillus section Fumigati species that cause sino-orbital aspergillosis (SOA) in cats, Fusarium species and Zygomycetes (eg, Mucor and Rhizopus species). 27
Owing to high protein binding, ITZ concentrations in CSF, ocular fluid and urine are negligible, but in the presence of inflammation ITZ can penetrate the blood-brain barrier. ITZ is distributed extensively in tissues throughout the body where active drug concentrations accumulate and persist far longer than in plasma, especially in skin and claws. 30 ITZ is metabolised in the liver. Excretion is primarily in bile; <1% of active drug is excreted in urine.
Posaconazole
Although absorption of PCZ is not affected by gastric acid, administration with food increases bioavailability of the oral adult suspension (up to four-fold in humans). PCZ is highly protein-bound and is widely distributed into tissues, especially the liver, kidneys, lungs and myocardium.3,33 Similar to ITZ, concentrations in CSF are low or negligible, but PCZ can penetrate the blood-brain barrier in the presence of inflammation. In humans, ~65% is excreted unchanged in faeces, while ~15% is metabolised in the liver by uridine diphosphate glucuronidation, with inactive metabolites being eliminated in faeces and urine. 34 Only trace amounts of PCZ are excreted unchanged in urine. 35 Cats are deficient in some uridine diphosphate glucuronosyltransferases compared with humans and dogs, 36 which may contribute to the relatively longer half-life of PCZ in cats (Table 2). PCZ inhibits cytochrome P450 enzymes to a lesser extent than ITZ. 37
Voriconazole
Similar to FCZ, VCZ has very good penetration into tissues, including CSF, ocular fluid and lungs. 40 In contrast to FCZ, elimination of VCZ is predominantly by metabolism rather than by renal clearance of unchanged drug. Saturation of metabolic clearance results in non-linear dose-dependent pharmacokinetic parameters, such that a greater than proportional increase in exposure is seen with increasing VCZ dose. Metabolites, which are mostly inactive, are excreted in urine (~80% in dogs and humans) and to a lesser extent in faeces (~20%).
In humans, the major metabolic pathways include fluoropyrimidine N-oxidation by hepatic cytochrome P450 enzymes, followed by glucuronide conjugation, as well as fluoropyrimidine hydroxylation and methyl hydroxylation. 25 Multiple oral doses of VCZ in rats and dogs result in lower systemic drug concentrations due to autoinduction of VCZ metabolism, with liver cytochrome P450 enzymes leading to increased drug clear-ance. 25 By contrast, repeated oral doses of VCZ in cats (12.5 mg q48h after a loading dose of 25 mg) resulted in increasing plasma drug concentrations over time; even by day 14, when dosing was stopped, steady state had not been reached. 10
Isavuconazole
Similar to VCZ, ISA has high MICs ( activity) against clinical isolates of cryptic Aspergillus species that cause SOA in cats. 45
In contrast to VCZ, ISA has linear dose-dependent pharmacokinetics, and improved safety and tolerability.
Terbinafine
A synergistic effect has been reported in vitro for TRB combined with ITZ or FCZ against isolates of dermatophytes, Tricho -phyton species, Aspergillus species, dematia-ceous fungi, C neoformans/C gattii species complexes and Pythium species.48,55,56 Synergism of VCZ and TRB combination therapy was shown in vivo for Aspergillus species in an invertebrate model of infection. 57 A randomised controlled clinical trial found that combination therapy with ITZ and TRB was superior to use of either drug alone for the treatment of dermatophytosis in humans. 58 Patients treated with TRB plus VCZ for Lomentospora (formerly Scedo sporium) prolifi-cans had better survival rates than those treated with other antifungal regimens. 59
Echinocandins
Representative drugs in this class include caspofungin acetate, micafungin, anidulafun-gin and rezafungin. Caspofungin is the only echinocandin for which pharmacokinetic data are available in cats. 60

Severe facial excoriations in a cat being treated with terbinafine (TRB) and posaconazole for sino-orbital aspergillosis. The severe pruritic ulcerative lesions on the face, which developed after 6 months of therapy, resolved after withdrawal of TRB
Echinocandins show no activity against fungal genera that have p-(1,6)-D-glucan as the most abundant cell wall component, including Cryptococcus, Mucorales and Tricho -sporon species.61,63 Activity is limited against Histo plasma, Blastomyces and Coccidioides species.
Caspofungin
Management of invasive fungal and fungal-like infections in cats
Dermatophyte pseudomycetoma
Cryptococcosis
If LCAT titres remain persistently elevated or fail to decline after 6 weeks of FCZ administration, switching to an alternative azole is indicated. ITZ is the next best choice, with a median treatment duration of 9 months. 79 Isolates exhibiting high MICs for FCZ or ITZ may be susceptible to other azoles, such as KCZ, 80 VCZ or PCZ.
Surgical excision or debridement of large cryptococcal granulomas may be considered for some patients, if deemed safe, since antifun-gal drugs penetrate poorly into hypoperfused tissues. However, there is currently no evidence that this approach decreases treatment times or is superior to medical therapy alone.
First-line therapy for CNS (including ocular) or disseminated disease is AMB combined with FC, or with FCZ if FC is unavailable.79,81 Critical patients with increased intracranial pressure may benefit from adjunctive judicious glucocorticoid (eg, dexamethasone) administration for the first 48-72 h of treat-ment. 82 Clinical improvement is expected within 7-10 days, and the AMB/FC combination is typically continued for 4-8 weeks. Treatment is then de-escalated to oral FCZ (or ITZ), which is administered until LCAT titres decrease to zero; prolonged treatment (≥2 years) may be needed to achieve this. Periodic follow-up monitoring of LCAT titres (eg, every 3-6 months) should be performed to detect recrudescence, which can occur in up to 30% of cases. 79
In some cases of cryptococcosis (any form), LCAT titres may not decline to zero despite switching azoles (eg, from FCZ to ITZ). In such refractory cases, combination therapy with an azole and TRB (Table 1), or TRB monotherapy, may be effective. 83 If LCAT titres decline four- to five-fold and then remain static despite these approaches, treatment response may be deemed adequate if accompanied by clinical remission. 83 Serum LCAT titres should be monitored and treatment re-instituted if they increase.
Sino-orbital aspergillosis
The reliably low MICs of caspofungin against infecting species make it attractive for adjunctive initial treatment, although this requires hospitalisation for daily IV infusions for 7-21 days. Placement of a central venous line enables daily infusions to be given for up to 3 weeks.11,38 Pharmacokinetic modeling data support the use of caspofungin in SOA, with effective target drug concentrations likely achieved when used as recommended in Table 1. 60 The combination of caspofungin and PCZ has also been used to treat invasive aspergillosis in humans caused by the same agents as feline SOA (eg, Aspergillus udagawae). 88 Some feline cases have shown improvement with the addition of orbital exenteration or debridement of orbital granu-lomas to the treatment regimen;42,89 overall, however, surgery has not been shown to improve outcomes compared with antifungal therapy alone. 11
In cats treated successfully, oral antifungal therapy was administered for 4-8 months in some cases, 90 and up to several years in others based on evidence of residual disease on serial head CT scans.11,38 In the light of the treatment duration, the authors strongly recommend therapeutic drug monitoring to enable individualised dose adjustments if PCZ levels are not optimal. In addition, monitoring for hepatotoxicity is recommended.
Blastomycosis
Coccidioidomycosis
Histoplasmosis
Median treatment durations of 6 months are typical, but longer treatment is often necessary to effect a cure, especially for disseminated disease.100-102 Low anti-inflammatory doses of short-acting glucocorticoids have been advocated by some as additional therapy for cats with severely compromised airways. 95 In the same study, the use of glucocorticoids was also a negative prognostic indicator, as was the use of supplemental oxygen, which may have reflected the selection bias for severe disease. 95
Sporotrichosis
AEs have occurred in 50% of cats receiving the second-line therapy of ITZ plus potassium iodide, including anorexia, lethargy, vomiting, diarrhoea and hepatotoxicity.103,106 The anti-fungal mechanism of action of potassium iodide - which is used for first-line therapy of cutaneous sporotrichosis in humans 107 - is incompletely understood.
Although clinical data are lacking, based on antifungal susceptibility testing and clinical efficacy in other hosts, the authors suggest the following alternative options for second-line therapy of S brasiliensis infection:
- TRB monotherapy. TRB was shown to be non-inferior to ITZ in a randomised controlled trial of human cutaneous sporotrichosis. 108 In addition, TRB MICs are low for feline isolates of S brasiliensis. 109
- In cases of severe or refractory disease, ITZ or PCZ could be combined with TRB, or PCZ could be given as monotherapy, since low MICs of this drug suggest susceptibility. 110 In vitro, ITZ combined with TRB is synergistic against Sporothrix species, 111 while PCZ/ITZ combinations have not been evaluated.
- AMB could also be added to the treatment regimen (Table 1). In an uncontrolled intervention study, feline cases of cutaneous sporo-trichosis with one to three non-resolving lesions were cured after treatment with oral ITZ combined with once-weekly AMB-deoxycholate administered intralesionally. 112 Complications included pain on injection and sterile abscess formation.
In contrast to S brasiliensis, isolates of S schenckii from Malaysia and Thailand have much higher MICs for azoles, TRB and AMB, and may be refractory to treatment.110,113 The combination of ITZ and TRB is synergistic, resulting in low MICs; hence it is recommended by the authors for first-line therapy. 111

Phaeohyphomycosis
Given that >100 species of pigmented fungi cause phaeohyphomycosis in humans and animals, antifungal susceptibility testing is recommended to guide therapy. If this is not possible, molecular identification of the infecting agent can usually be achieved by panfun-gal PCR of formalin-fixed biopsy tissues (see Part 1 1 ). This can help inform the best empirical therapy since antifungal susceptibility profiles vary between different fungal genera and species. Of 126 clinical and environmental isolates of Exophiala species, PCZ (0.063 μg/ml) and ITZ (0.125 μg/ml) had much lower MIC90 values than VCZ (1 μg/ml) and ISA (2 μg/ml), suggesting better suitability of ITZ or PCZ for first-line therapy. 115 In one case series, recurrence of cutaneous lesions caused by Alternaria species was common after surgical excision and ITZ therapy for 2-4 months. 116
Mucormycosis
Pythiosis
- PCZ, TRB, azithromycin and doxycycline combination therapy; or
- PCZ, minocycline and clarithromycin combination therapy.

In dogs, an agricultural fungicide, mefen-oxam, used to control oomycete infections in plants through inhibition of RNA polymeras-es, has had some success as part of the treatment protocol for gastrointestinal and cutaneous pythiosis; it was variously used after surgical resection in combination with ITZ and TRB, or with minocycline.127-129 Mefenoxam has potent in vitro activity against Pythium and Lagenidium species, but pharma-cokinetic studies have not been performed in animals; nor has it been used in cats. 130
Adjuvant immunotherapy using commercially formulated P insidiosum antigens administered as a subcutaneous injection has been successful in treating pythiosis in horses and humans. Its efficacy in dogs appears to be poor. 131 One cat with extensive ulcerated cutaneous pythiosis showed dramatic shrinkage of the lesion after immunotherapy, but the cat was euthanased because of severe pleural effusion of unknown cause. 132 Immuno-therapy has been used as adjunctive therapy in four successfully treated cats with pythiosis in Hong Kong, and was well tolerated (authors’ unpublished observations); however, its contribution to treatment success in these cases is unknown.
Key Points
✜ Targeted antifungal therapy based on the susceptibility profile of the isolated pathogen is an important factor in determining treatment success.
✜ Therapeutic drug monitoring to measure plasma concentrations of azoles can be used to investigate lack of treatment response due to underdosing or antifungal resistance, and to optimise drug dosage in cases of drug-drug interactions or toxicity.
✜ MICs for azithromycin, clarithromycin, doxycycline and minocycline are generally low in vitro for Pythium and Lagenidium species, and these antimicrobial drugs show promise for use in treatment regimens for oomycoses.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
