Abstract
Objectives
The aim of this study was to determine the specificity of a rapid point-of-care test for the estimation of feline pancreatic lipase (SNAP fPL) in healthy and sick cats without clinical evidence of pancreatitis. A second objective was to evaluate the agreement between SNAP fPL and serum pancreatic lipase immunoreactivity (fPLI), as measured by Spec fPL.
Methods
A total of 150 cats were prospectively enrolled into this study. Of them, 82 cats were healthy while 68 cats had various diseases but no clinical signs (eg, anorexia, depression, vomiting) raising a suspicion of pancreatitis.
Results
SNAP fPL was normal in 133/150 cats (specificity 89%) without obvious clinical pancreatitis. SNAP fPL was normal in 74/82 healthy cats (specificity 90%) and in 59/68 cats that were sick but without typical signs of pancreatitis (specificity 87%). The agreement between SNAP fPL and Spec fPL was substantial (k = 0.64) in healthy cats and almost perfect (k = 0.93) in sick cats. The overall agreement between SNAP fPL and Spec fPL was almost perfect (k = 0.81).
Conclusions and relevance
The specificity of SNAP fPL in this group of cats was high. There was a substantial and almost perfect agreement between the SNAP fPL and Spec fPL in healthy cats and sick cats without suspected pancreatitis, respectively. In the small percentage of cats with abnormal SNAP fPL and/or Spec fPL results, the possibility of subclinical pancreatitis cannot be excluded.
Introduction
Although pancreatitis is the most common disease of the exocrine pancreas in cats, accurate diagnosis remains challenging in clinical practice. Histopathology is traditionally considered to be the gold standard for the diagnosis of pancreatitis in cats but is associated with several limitations. 1 Most importantly, the collection of a pancreatic biopsy is expensive and invasive, and thus often not in the best interest of the patient. The clinical signs of pancreatitis in cats are ambiguous and variable, the most common being reduced appetite or anorexia and depression, while vomiting, diarrhoea and abdominal pain are less common. 2
The diagnostic approach to cats suspected of having pancreatitis is based on the evaluation of the history, physical examination and routine laboratory findings, as well as pancreatic ultrasonographic findings and the results of sensitive and specific serum tests. The specificity of serum markers for feline pancreatitis has been the subject of several studies but much controversy still exists.3–6
Pancreatic lipase is an enzyme exclusively synthesised and secreted by the pancreatic acinar cells. The measurement of serum feline pancreatic lipase immunoreactivity (fPLI) as measured by the Spec fPL (Idexx Laboratories) has been shown to be highly specific for the diagnosis of pancreatitis in cats, with a specificity in the range of 63–100%.6–10 SNAP fPL, a patient-side test that is based on the Spec fPL, is widely available and is mainly used to exclude pancreatitis. Its reported specificity is in the range of 89–100% according to a recently published study and the manufacturer’s unpublished data. 2
The assessment of the specificity of serum markers for pancreatitis in cats using cats in which even subclinical pancreatitis has been reliably ruled out is quite challenging. For example, one necropsy study showed that mild chronic pancreatitis was common in cats without clinical signs of the disease. However, the clinical importance of this finding is unknown, 1 and necropsy studies may overestimate the prevalence of clinically important pancreatitis. One way to overcome the limitation of using histopathology as the gold standard for excluding feline pancreatitis is to assess the specificity of serum markers in animals that do not have clinical evidence of the disease. This will potentially lead to the enrolment of some cats with mild histopathological evidence of pancreatitis but will exclude animals with clinical disease. We hypothesised that SNAP fPL and Spec fPL are highly specific for the diagnosis of pancreatitis in cats with no clinical evidence of the disease and, based on a previous report, that the agreement between these two methods is high. 2 Therefore, the aim of this study was to evaluate the specificity of SNAP fPL and Spec fPL using healthy cats and sick cats without a clinical suspicion of pancreatitis. A second aim of this study was to evaluate the agreement between SNAP fPL and Spec fPL.
Materials and methods
Ethical approval
The study protocol was reviewed and approved by the Animal Ethics Committee of the Faculty of Veterinary Science, University of Thessaly (13/16-6-15). The handling of these animals was in compliance with the European Communities Council Directive 2010/63/EU and state laws. All owners had to sign an informed owner consent form before enrolment of their cat into the study.
Animals
Cats were prospectively enrolled and divided into two groups. Group 1 consisted of healthy cats and group 2 consisted of cats with various diseases but without clinical evidence of pancreatitis. The inclusion criteria for cats in group 1 were the absence of clinical signs for at least the previous 3 months and no abnormal findings on physical examination. Cats in this group were presented by their owners for vaccination and/or preventive health evaluation. The inclusion criteria for cats in group 2 included the presence of abnormal clinical signs and physical examination findings that could not, however, be attributed to pancreatitis, such as signs associated with respiratory, cardiovascular, cutaneous, haematopoietic or urinary tract diseases. Cats with gastrointestinal signs, such as vomiting, diarrhoea, abdominal pain, depression or anorexia, were excluded from the study. All cats in group 2 had a final diagnosis that was consistent with the clinical presentation.
Sample collection and handling
A thorough physical examination was performed, and blood samples were collected by jugular venepuncture in all cats. All samples were centrifuged at 1000 x g for 10 mins, and serum was separated, transferred into Eppendorf tubes and stored at −80°C until analysis (feline pancreatic lipase is highly stable when stored at −80°C 11 ). Both SNAP fPL and Spec fPL were performed using the same serum sample from each cat.
SNAP fPL
SNAP fPL was performed in all cats included in the study according to the manufacturer’s instructions, 4 and the results were interpreted by the same investigator (PGX) as normal or abnormal. A normal result corresponds to the normal Spec fPL, while an abnormal result can be either in the grey zone or in the range considered consistent with pancreatitis. 12
Spec fPL
Serum Spec fPL was measured at the Gastrointestinal Laboratory at Texas A&M University. 10 The published reference interval for this assay is ⩽4.4 µg/l. Concentrations of 4.5–8.7 µg/l are considered equivocal (‘grey zone’), while concentrations ⩾8.8 µg/l are considered to be consistent with pancreatitis. 12 Therefore, in this study, we used two cutoff values: one for the upper limit of the reference interval (4.4 µg/l) and one as the diagnostic cutoff for pancreatitis (8.8 µg/l).
Statistical analyses
All statistical analyses were performed using GraphPad Prism version 9.2.0 (GraphPad Software). The distribution of data was assessed using the Kolmogorov–Smirnov test. The null hypothesis of the outcome of this study was that the specificity of SNAP fPL and Spec fPL in healthy cats and sick cats without suspicion of pancreatitis is high, and that the agreement between SNAP fPL and Spec fPL is high. The agreement between SNAP fPL and Spec fPL was assessed using the Cohen’s kappa (k) coefficient. Kappa values in the range of 0–0.2 indicate poor agreement, values of 0.21–0.4 indicate fair agreement, values of 0.41–0.6 indicate moderate agreement, values of 0.61–0.8 indicate substantial agreement, and values of 0.81–1 indicate almost perfect agreement. 13 For the comparison of serum Spec fPL concentration and SNAP fPL, the Mann–Whitney test was used.
Results
Animals
A total of 150 cats were included in the study. Of these, 82 (55%) were healthy (group 1), while the remaining 68 (45%) (group 2) were cats with various clinical conditions and diseases (Table 1). Among the cats in group 1, 45 (55%) were male and 37 (45%) were female. Group 2 cats included 31 (46%) males and 36 (53%) females, while for one cat sex was not recorded. Group 1 included 64 (78%) domestic shorthair (DSH) cats, 10 (12%) domestic longhair (DLH) cats, 2 (2%) domestic medium hair (DMH) cats, and 1 (1%) cat each of the Persian, Maine Coon and Siamese breeds, while the breed for 3 (4%) cats was not recorded. Group 2 included 52 (76%) DSH cats, 6 (9%) DLH cats, 5 (7%) Siamese, 2 (3%) DMH cats, and 1 (1%) each of Maine Coon and Birman cross; the breed for 1 (1%) cat was unknown. The median age was 2.0 years (range 0.42–14.0 years) for the cats in group 1 (data available for 75/82 cats) and 4.0 years (range 0.5–18.0 years) for the cats in group 2 (data available for 63/68 cats). The median body weight was 3.5 kg (range 0.6–8.5 kg) in group 1 (data available for 80/82 cats) and 3.8 kg (range 2.2–7.5 kg) in group 2 (data available for 63/68 cats).
Classification of the 68 cats in group 2 according to their suspected disease or condition (some cats presented with more than one disease or condition)

One cat each for the following: anaemia, pulmonary oedema, Horner’s syndrome, tetraplegia, paraneoplastic cachexia, feline upper respiratory disease, periodontal disease, intestinal obstruction due to foreign body
SNAP fPL and Spec fPL
A total of 133/150 cats had a normal SNAP fPL concentration (specificity 89%). SNAP fPL was normal in 74/82 healthy cats (specificity 90%) and 59/68 in sick cats without a suspicion of pancreatitis (specificity 87%). A total of 138/150 cats had normal a serum Spec fPL concentration (⩽4.4 µg/l; specificity 92%), 4/150 (3%) cats had a serum Spec fPL concentration indicative of pancreatitis (⩾8.8 µg/l) and 8/150 (5%) cats had a serum Spec fPL concentration in the equivocal range (4.5–8.7 µg/l) (Figure 1). In group 1 (healthy cats), 78/82 cats had normal serum Spec fPL concentrations (specificity 95%), 1/82 (1%) had a serum Spec fPL concentration indicative of pancreatitis and 3/82 (4%) were in the equivocal range. In group 2 (sick cats), 60/68 cats had normal serum Spec fPL concentrations (specificity 88%), 3/68 (4%) had serum Spec fPL concentrations indicative of pancreatitis and 5/68 (7%) were in the equivocal range (Figure 2).

Comparison between SNAP fPL and Spec fPL results in 150 cats

Scatter plot of serum Spec fPL concentrations in group 1 (healthy cats, n = 82) and group 2 (sick cats, n = 68). The horizontal solid lines represent the medians, the lower dashed line represents the upper limit of the reference interval (4.4 μg/l) and the upper dashed line represents the cutoff value for pancreatitis (8.8 μg/l). Note: the Y axis is split
SNAP fPL was normal in 133/138 (96%) cats with normal serum Spec fPL concentrations and the overall agreement between the two assays was almost perfect (k = 0.81) (Figure 1).
In group 1, SNAP fPL was normal in 74/78 (95%) cats with a normal serum Spec fPL and abnormal in 1/1 (100%) cat with a Spec fPL concentration consistent with pancreatitis. The agreement between the two assays for this group was substantial (k = 0.64). In group 2, SNAP fPL was normal in 59/60 (98%) cats with normal serum Spec fPL concentrations and abnormal in 3/3 (100%) cats with Spec fPL concentrations consistent with pancreatitis. The agreement between the two assays for this group was almost perfect (k = 0.93). The concentrations of Spec fPL were significantly lower in cats with normal SNAP fPL compared with cats with abnormal SNAP fPL (Figure 3). Figures 4 and 5 summarise the results for SNAP fPL and Spec fPL in groups 1 and 2.
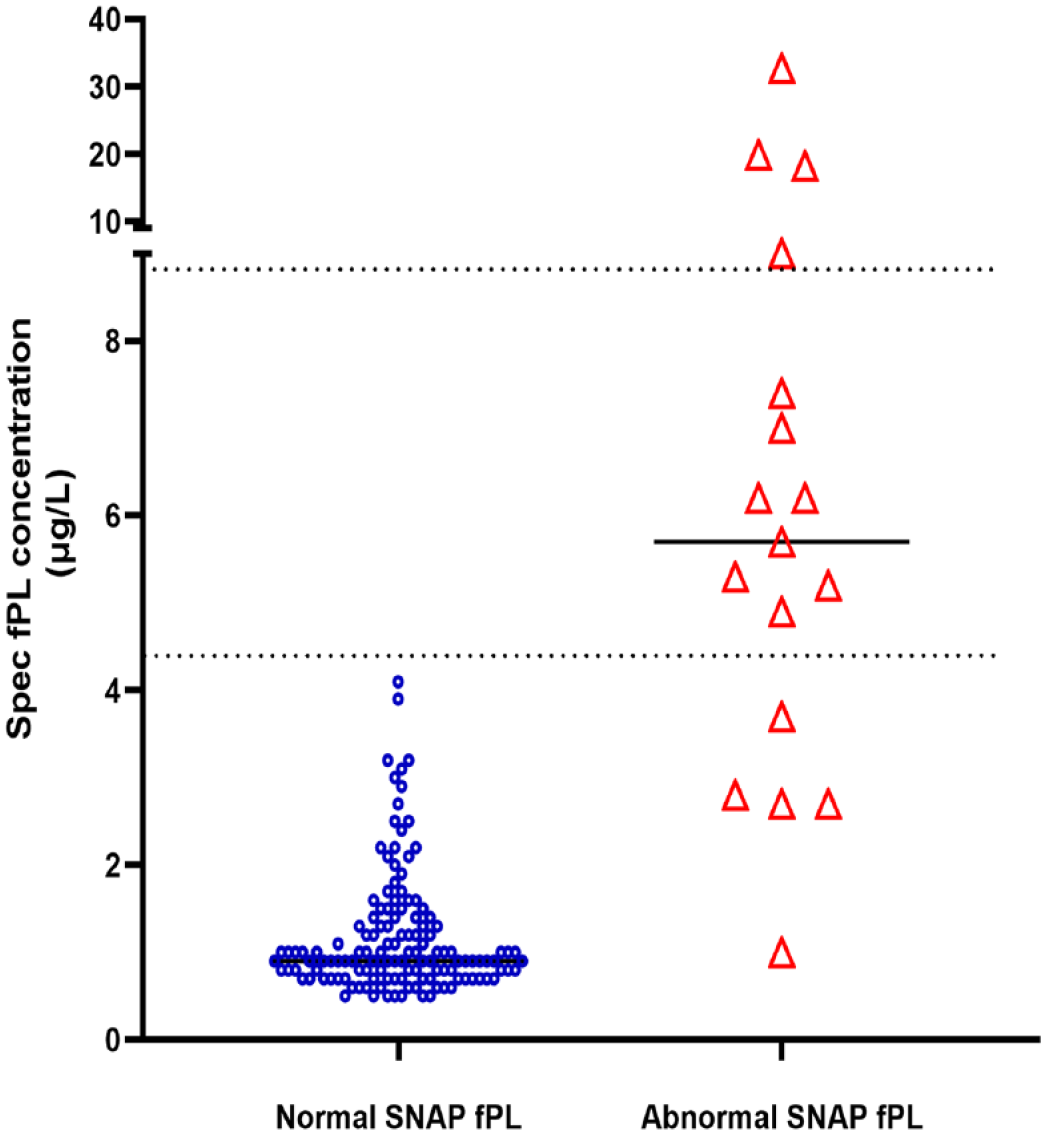
Scatter plot of serum Spec fPL concentrations in cats with normal and abnormal SNAP fPL tests, respectively. The horizontal solid lines represent the medians, the lower dashed line represents the upper limit of the reference interval (4.4 μg/l) and the upper dashed line represents the cutoff for pancreatitis (8.8 μg/l). Note: the Y axis is split

Detailed results of Spec fPL in group 1 (healthy cats) and group 2 (sick cats without suspected pancreatitis)

Detailed results of SNAP fPL in group 1 (healthy cats) and group 2 (sick cats without suspected pancreatitis)
Discussion
Our results indicate that the overall specificity of both SNAP fPL (89%) and Spec fPL (92%) is high, in healthy cats and sick cats with no clinical suspicion of pancreatitis. In addition, the agreement between the two tests is substantial to almost perfect in healthy and sick cats (k = 0.64 and k = 0.93, respectively), with an almost perfect overall agreement of k = 0.81.
SNAP fPL was found to be normal in 133/150 cats, leading to an overall specificity of 89%. The specificity was similar in healthy cats (90%) and in sick cats without a suspicion of pancreatitis (87%). Our results are in agreement with a previous study that found a specificity of 96.5% in cats without a clinical suspicion of pancreatitis. 2
Spec fPL was normal in 138/150 cats, leading to a specificity of 92%. The specificity was slightly higher in healthy cats (95%) compared with sick cats with no suspicion of pancreatitis (88%). Our results are in agreement with previous studies, in which the specificity of Spec fPL was in the range of 82–100%,6,7,10,14 with only one study showing a lower specificity of 63%. 8 Therefore, the specificity of both SNAP fPL and Spec fPL does not seem to differ depending on the presence of clinical signs that might be associated with a suspicion of pancreatitis. This is not unexpected because pancreatic lipase is known to be produced only by pancreatic acinar cells, and diseases of other organs and systems have not been shown to affect its serum concentration.
The small number of cats with abnormal SNAP fPL results and/or increased Spec fPL concentrations is interesting, although not unexpected. One possible explanation is that serum pancreatic lipase was increased due to another condition and not pancreatitis, although, as already detailed above, this is unlikely. 12 Alternatively, these cats might have had mild, subclinical pancreatitis that was not accompanied by clinical signs. Subclinical pancreatitis is relatively common in cats, as shown in necropsy studies, 15 and in this case the positive test results would have been correct and not false positives. In fact, some of the conditions diagnosed in the cats in group 2, such as severe anaemia and intestinal obstruction, could potentially lead to secondary pancreatitis. Unfortunately, this could have been confirmed only by pancreatic histopathology, which was not available for any of the cats.
In our study, SNAP fPL was normal in 74/78 (95%) healthy cats and in 59/60 (98%) sick cats with normal serum Spec fPL concentrations, leading to an almost perfect (k = 0.81) overall agreement between the two tests. This is in accordance with the only other published study that compared these two tests, where an agreement of 89% and a Cohen’s k of 0.65 were reported. 2 In another study (available only as an abstract), 16 where SNAP fPL and Spec fPL were evaluated for the diagnosis of pancreatitis, an agreement in the range of 90–98% was found, but Cohen’s k was not reported. It needs to be pointed out that all the above studies have used the previous cutoffs (reference interval <3.5 µg/l, grey zone 3.5–5.3 µg/l, consistent with pancreatitis ⩾5.4 µg/l) for the interpretation of Spec fPL. 17 In our study, we used the cutoffs based on a recent publication by Wu et al, 12 as these have now replaced the old ones.
In our study, 4/4 (100%) healthy cats and 8/8 (100%) sick cats with increased serum Spec fPL concentrations (ie, in the equivocal range or indicative for pancreatitis) had an abnormal SNAP fPL. Therefore, the agreement for abnormal results was 100%.
There are some studies in dogs evaluating the specificity of SNAP cPL and Spec cPL, and the agreement between these two assays.4,18–25 SNAP cPL and Spec cPL show a high level of agreement according to previous studies, similar to the agreement between SNAP fPL and Spec fPL shown in our study. Based on the studies above, the specificity of SNAP cPL and Spec cPL is in the range of 59–98% and 59–77.8%, respectively, depending on the study and the group of dogs investigated. There are only two studies in dogs evaluating the agreement between SNAP cPL and Spec cPL.20,25 In the first one, 20 the overall agreement between SNAP cPL and Spec cPL was substantial (k = 0.78), similar to the results of our study. In the other study, 25 the authors evaluated the agreement using the interclass correlation coefficient (ICC) and they found that there is an almost perfect agreement between SNAP cPL and Spec cPL (ICC = 0.91).
Care should be taken not to overinterpret our findings. It is important to note that the cats enrolled in our study are not representative of those cats that are typically tested for pancreatitis in the clinical setting. The rationale for the design of our study was to reduce, as much as possible, the possibility of including cats with pancreatitis that would escape the diagnosis in the absence of histopathology. However, studies using different populations of cats (ie, cats with a suspicion of pancreatitis)2,6–8,10,14 found similar results to ours. Therefore, as mentioned above, clinical suspicion of pancreatitis may not affect the specificity of the SNAP fPL, which is reasonable in the face of the studies showing that pancreatic lipase is produced solely by pancreatic acinar cells. 12 Therefore, cats with clinical signs compatible with pancreatitis but without the disease should not differ from cats with no clinical signs of pancreatitis with regard to the amount of pancreatic lipase released.
Our study has two limitations. First, a histopathological examination of the pancreas was not performed in any of the cats enrolled and, for this reason, we cannot exclude with certainty the possibility of subclinical pancreatitis, which could potentially explain the abnormal SNAP fPL and the increased Spec fPL concentrations in some cats. However, a pancreatic biopsy could not be justified in healthy cats or in cats with no suspicion of pancreatitis. Furthermore, pancreatic histopathology is associated with several limitations, including the fact that lesions are often focal and require multiple tissue biopsies and sections of the pancreas to be evaluated. 14 Also, in one study, 45% of healthy cats had histopathological evidence of pancreatitis, a finding that presents challenges in the interpretation of the clinical significance of pancreatic histopathology. A second limitation is that other means of evaluating pancreatic inflammation, especially abdominal ultrasound, were not used. However, ultrasound examination cannot rule out pancreatitis, because the sensitivity of this test has been reported to be as low as 67%. 15
Conclusions
This study shows a high specificity of SNAP fPL and Spec fPL in healthy cats and sick cats with no suspicion of pancreatitis. In addition, there is a high level of agreement between SNAP fPL and Spec fPL. In the small percentage of cats with abnormal SNAP fPL results and increased Spec fPL concentrations, the possibility of subclinical pancreatitis could not be excluded.
Footnotes
Author note
Part of the results of this paper was presented at the 2017 ECVIM Congress.
Conflict of interest
JM Steiner serves as a paid consultant for IDEXX Laboratories and is also the director of the Gastrointestinal Laboratory, which offers diagnostic services (including the Spec fPL) on a fee-for-service basis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedures undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
