Abstract
The purpose of this study was to determine the prevalence of pancreatic nodular hyperplasia (NH) and its relation to age in the dog. A total of 101 dogs were enrolled. The pancreas was evaluated by histology and hyperplastic lesions were detected and scored. Age was recorded from the medical records. Correlation of age with inflammation and presence of hyperplastic lesions was evaluated. Of the 101 dogs, 81 (80.2%) had evidence of NH. Twenty-five of the 101 dogs did not have evidence of pancreatic inflammation, necrosis, and/or fibrosis, 17 (68.0%) of which had evidence of NH. Mean ± SD age in dogs with NH was significantly higher compared with dogs without NH (9.5 ± 3.4 years versus 3.4 ± 3.3; p-value < 0.0001). We conclude that NH is a common pathologic lesion in dogs and shows a positive correlation with age regardless of the presence or absence of pancreatic inflammation, necrosis, or fibrosis.
Keywords
Exocrine pancreatic nodular hyperplasia (NH) has been reported in cattle, cats, and dogs. 5, 7 The condition has been considered an incidental finding. 2, 5 Several authors have suggested that the nodules may represent age-related hyperplastic foci rather than either a response to previous injury or an early stage of progressive pancreatic epithelial neoplasia. 5 However, it has been speculated that, in young animals, NH may indicate antecedent inflammation or scarring. 5 Hyperplasia may be manifested as a solitary nodule or, more commonly, as multiple, small, white to tan, well-circumscribed nodules. 5 Microscopic lesions are typically nonencapsulated and minimally or noncompressive of adjacent pancreatic parenchyma. 5 Ductular hyperplasia is an uncommon lesion but may accompany the more common exocrine pancreatic acinar NH. 5 NH can be distinguished from pancreatic adenoma on light microscopy by the multiplicity, the smaller size, the lack of a capsule, and the close resemblance with normal exocrine pancreatic tissue. 5
The goal of this study was to assess numerous pancreatic sections from dogs presented for necropsy to determine the incidence of pancreatic NH, its association with other pancreatic pathology, and any association with age.
A total of 101 pancreata from consecutive dogs presented for routine necropsy examination to the Pathology Service at the Animal Medical Center (AMC) were enrolled into the study. Inclusion criteria consisted of having an antemortem serum sample collected and having the pancreas removed in its entirety within 6 hours of death. Fixation of the pancreas in 10% buffered formalin within the 6-hour period was done in order to eliminate any postmortem degeneration/autolysis of the pancreatic tissue. The right limb of the pancreas was identified with a suture at the time of removal. Pancreata were sectioned every 2 cm, beginning at the tip of the right limb. Each section was routinely processed, stained with hematoxylin and eosin (HE), and assessed with light microscopy by a single pathologist (SJN).
Hyperplastic foci were characterized as nodular to slightly compressive, nonencapsulated masses of variable size (Fig. 1). Histologically, these hyperplastic foci were characterized by aggregates of recognizable exocrine pancreatic epithelial cells or, less commonly, ductular epithelial cells. Hyperplastic lesions were judged as being present or not and were subsequently scored based on size and extent by a single pathologist (SJN). Dogs were given a score of zero (no evidence of NH), one (less than 10% of the section showing NH), two (10–40% of the section showing NH; Figs. 2 and 3), or three (greater than 40% of the section showing NH).
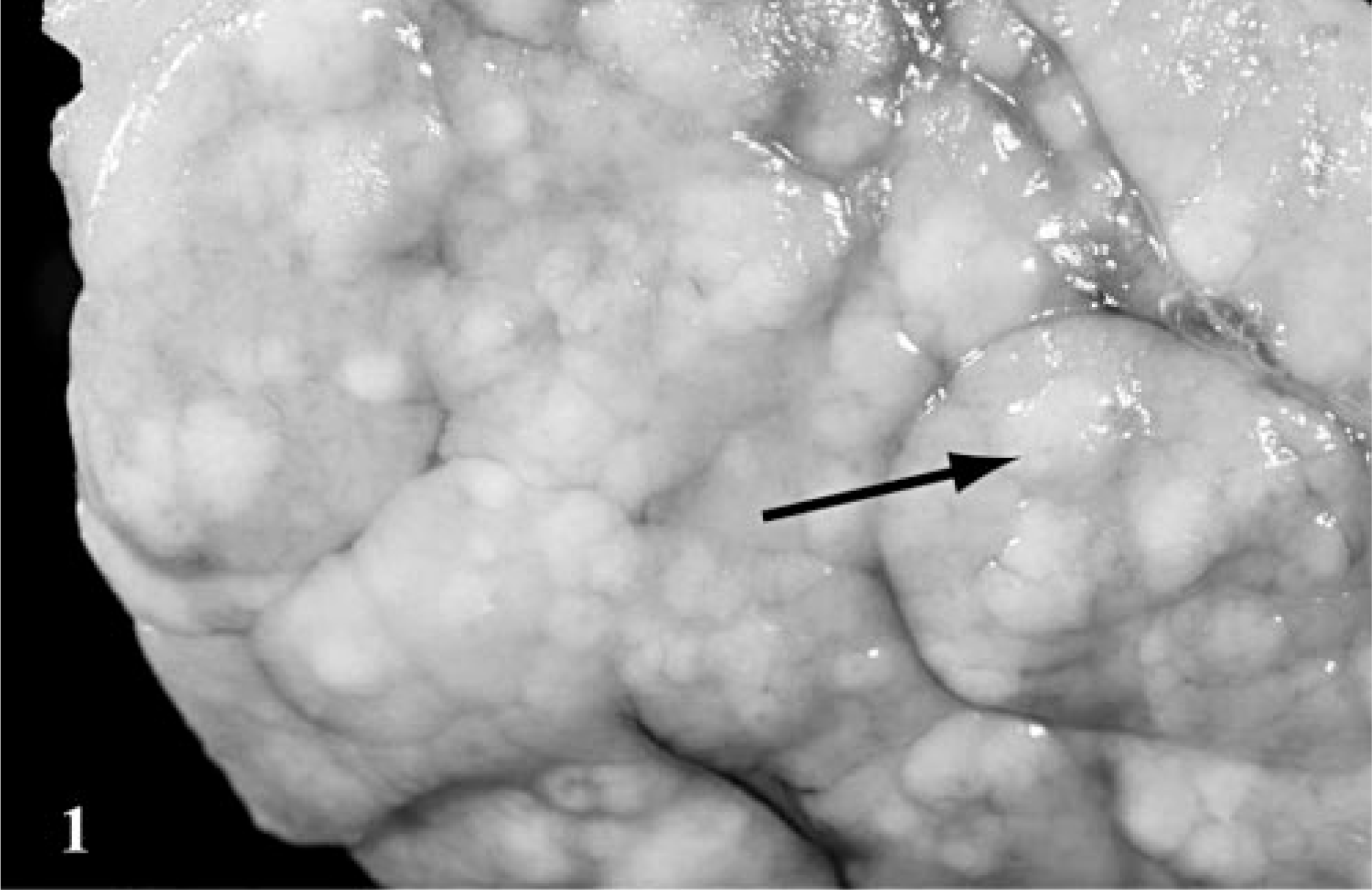
Pancreas; canine, dog 51. Multiple, pale, tan, raised, discrete nodules are present on the pancreatic surface (arrow).
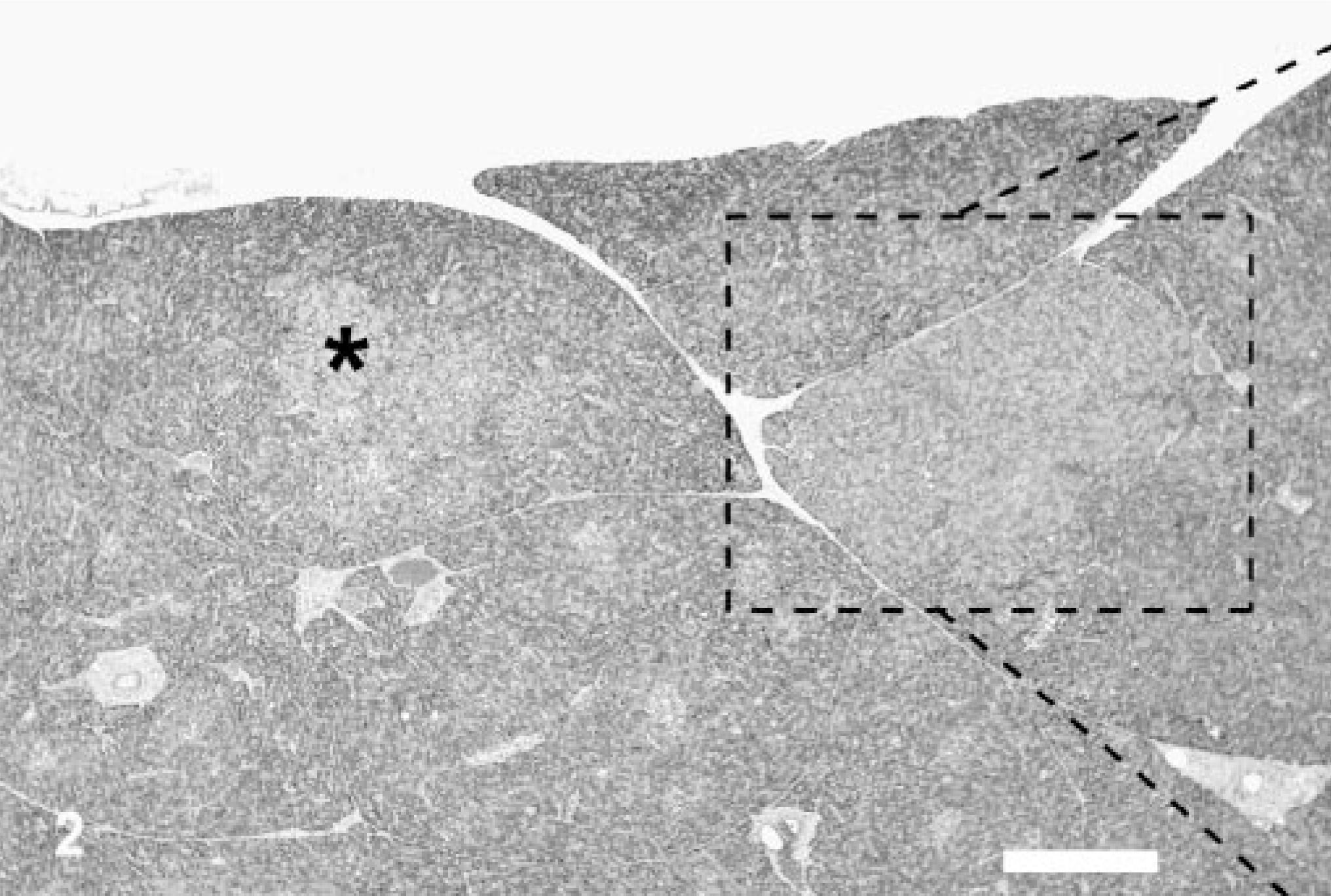
Pancreas; canine, dog 18. Multiple exocrine pancreatic hyperplastic nodules are present (asterisk∗) and comprise 10–40% of this pancreas specimen, thus representing a grade II nodular hyperplasia. Hematoxylin and eosin. Bar = 317 μm.
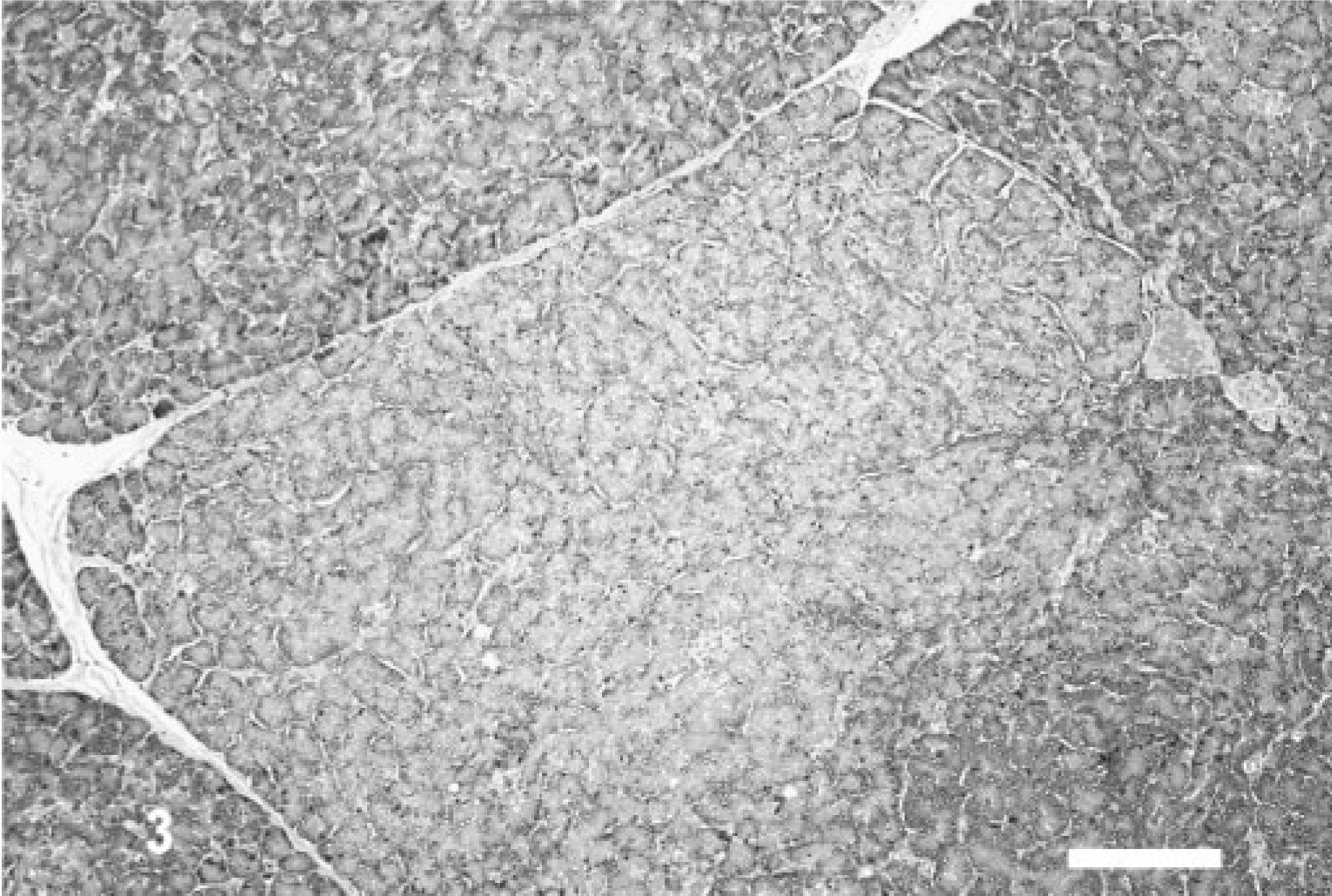
Pancreas; canine, dog 18. Higher magnification of the area within the dashed box from Fig. 2 reveals a well delineated nodule, composed of well-differentiated exocrine pancreatic epithelial cells, most of which contain lesser cytoplasmic zymogen stores than surrounding pancreatic acinar cells. Hematoxylin and eosin. Bar = 135 μm.
A mean NH score was calculated by the sum of the scores of all sections divided by the total number of sections evaluated. Age was recorded from the medical records for all 101 dogs. The mean age of the dogs was compared between those with NH and those that had no evidence of this lesion in any of the sections. Correlation of age and presence of hyperplastic lesions was evaluated by the Spearman r-test in all 101 dogs and also in a subgroup of 30 dogs that did not have any evidence of pancreatic inflammation, necrosis, and/or fibrosis. In addition, correlation of age and mean NH score was evaluated by the Pearson r in both populations. All statistical analyses were performed using a statistical software package (Prism 4.0; GraphPad, San Diego, CA). A P < 0.05 was considered statistically significant.
Hyperplastic foci were characterized as nodular to slightly compressive, nonencapsulated masses of variable size that only occasionally displayed peripheral fibrous connective tissue rimming. Additionally, inflammation, when associated with the nodular lesions, was characterized by the presence of minimal lymphocytes and fewer plasma cells. Histologically, these hyperplastic foci were characterized by tightly packed aggregates of recognizable exocrine pancreatic epithelial cells arranged in acini or, less commonly, as ductular epithelial cells.
Hyperplastic cells resembled the normal exocrine pancreatic epithelial cells, but occasionally, the cellular accumulation of zymogen granules was reduced in the hyperplastic cells compared with those in the surrounding pancreatic tissues. Nuclear alterations were not apparent. Additionally, there were no dogs with pancreatic nodular lesions that demonstrated dysplastic or neoplastic transformation (i.e., adenocarcinoma) in any of the multitude of examined sections.
Of the 101 dogs that underwent necropsy, 81 (80.2%) had evidence of NH. Twenty-five of 101 dogs did not have any evidence of pancreatic inflammation, necrosis, and/or fibrosis, of which 17 (68.0%) had NH. Mean ± SD age of all 101 dogs was 8.3 ± 4.2 years. Mean ± SD age of dogs with NH was significantly higher when compared with those dogs without NH (9.5 ± 3.4 years versus 3.4 ± 3.3; P < 0.0001). There was a positive correlation of age and presence of NH in all 101 dogs (Spearman r = 0.5550; P < 0.0001) and in the 25 dogs without any evidence of pancreatic inflammation, necrosis, and/or fibrosis (Spearman r = 0.6776; P < 0.0002). Furthermore, there was a positive correlation between age and mean NH score in both groups of dogs (Pearson r = 0.6160 and 0.7104; P < 0.0001 and = 0.0001, respectively; Figs. 4 and 5).
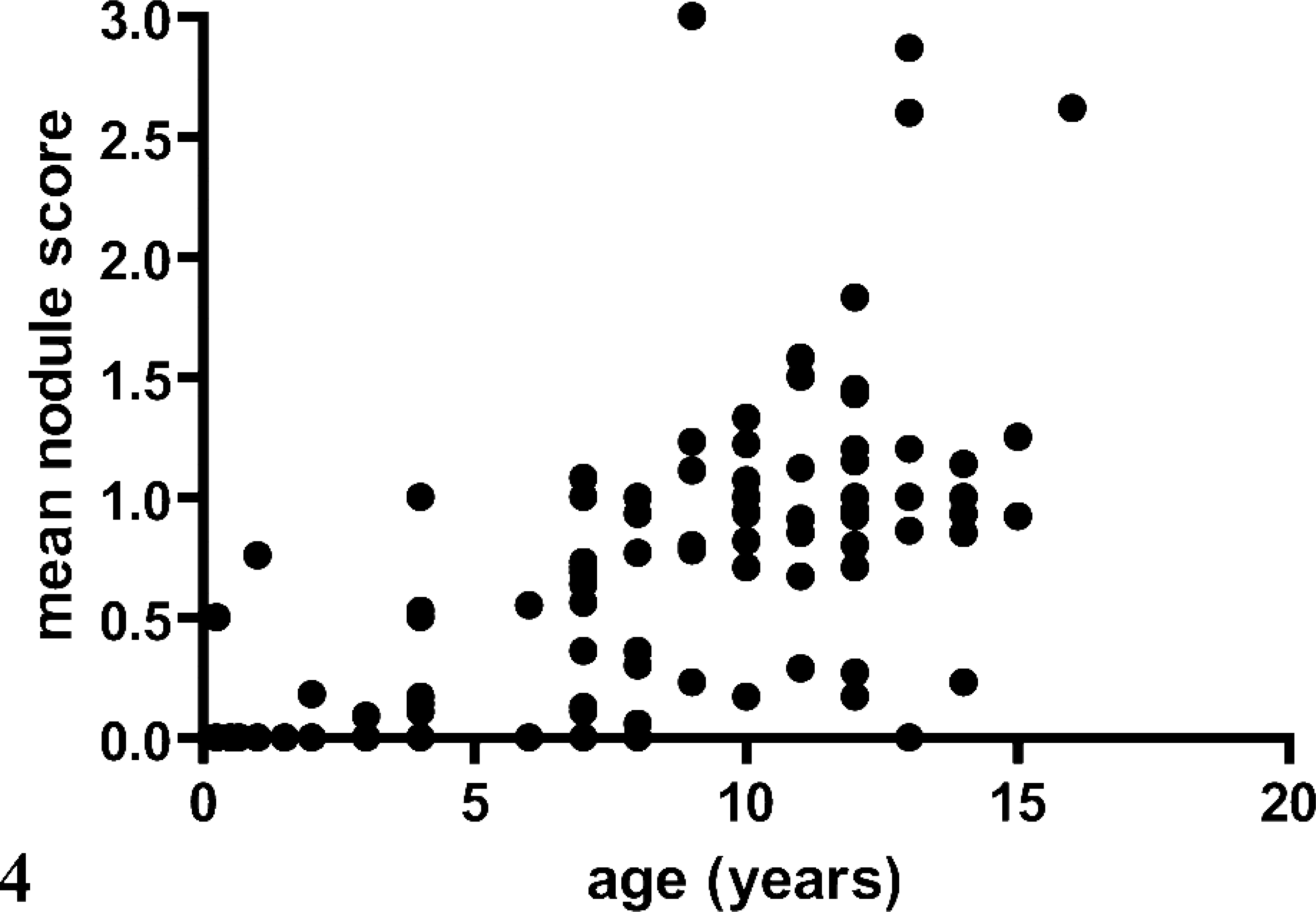
Correlation of age and mean nodular hyperplasia score in 101 dogs that underwent necropsy.
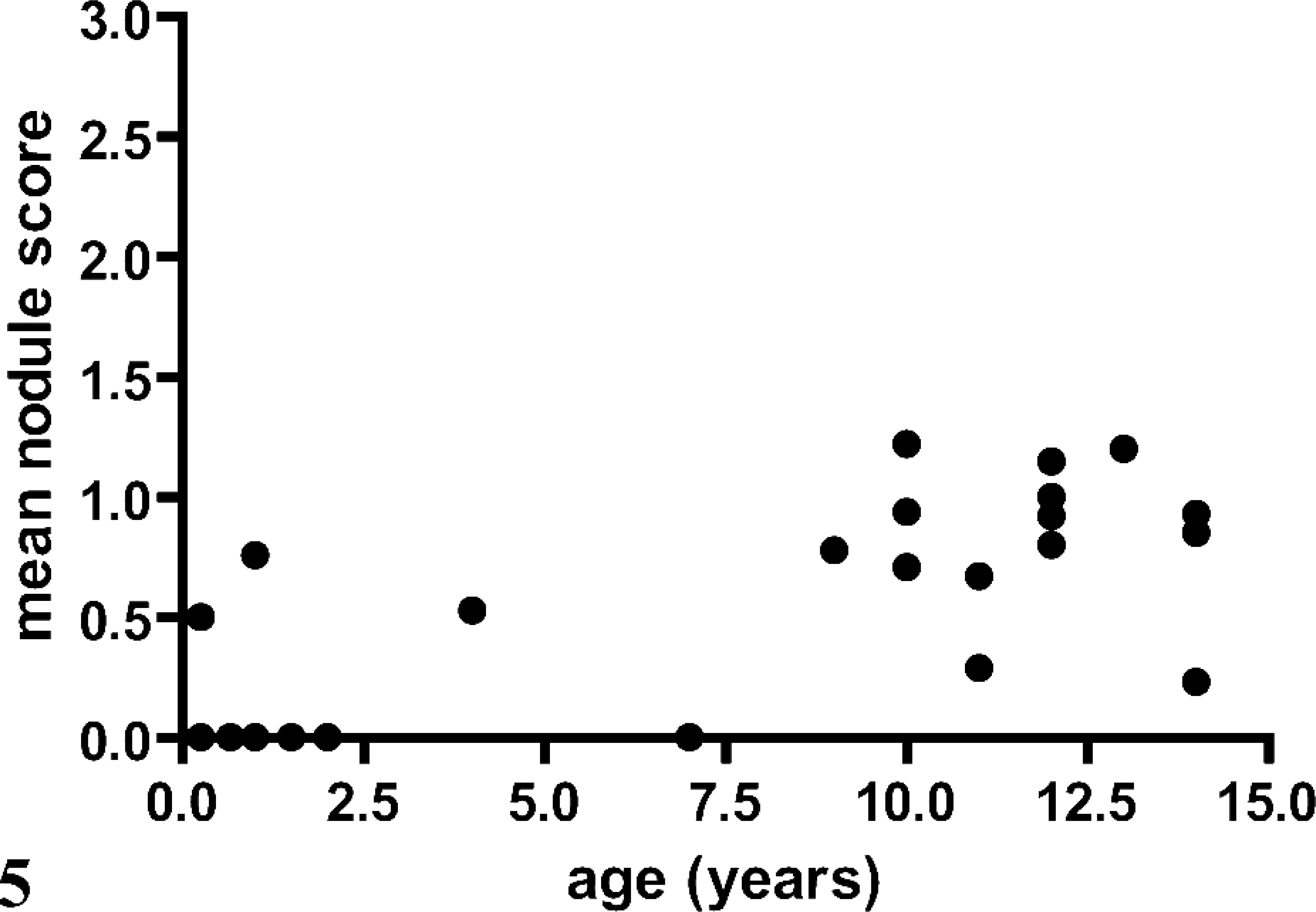
Correlation of age and mean nodular hyperplasia score in 25 dogs that underwent necropsy and did not have any evidence of pancreatic inflammation, necrosis, and/or fibrosis.
In the population of dogs examined here, NH was a very common finding identified during necropsy (81/101 = 80.2%). Also, the prevalence of this lesion increases with age. Similarly, we have previously shown that pancreatic inflammation, necrosis, and/or fibrosis is also a very common histopathologic finding in a similar population of dogs presented for necropsy at the AMC (47/73 = 64.4%). 8
Exposure to traumatic injury or pancreatic inflammation/necrosis would be expected to increase the longer an animal lived. Similarly, if NH were a sequela to pancreatic inflammation or necrosis, a higher occurrence of pancreatic NH could occur with advanced age. However, in approximately 21% (17/81) of our cases, the NH was not associated with recognizable antecedent damage (fibrosis) or concurrent pancreatitis (necrosis or inflammation), despite evaluation of numerous samples. Mean NH shows a positive correlation with age regardless of the presence or absence of pancreatic inflammation, necrosis, and/or fibrosis. Although hyperplasia likely represents a method of tissue repair following pancreatic injury, our findings would suggest that this may not be the only pathogenic mechanism for the development of NH.
None of the pancreatic hyperplastic nodular lesions demonstrated dysplastic or neoplastic transformation (i.e., adenocarcinoma) in any of the multitude of sections examined. Although the size of individual nodules varied, the character of the component cells did not vary from histologically normal exocrine pancreatic epithelial cells other than that, in some nodules, the increased number of compacted epithelial cells was also often smaller and less eosinophilic because of a lesser accumulation of cytoplasmic zymogen granules. The hyperplastic epithelial cells respected the acinar arrangement and nuclei were maintained in a basilar orientation. This would suggest that NH is rarely a preneoplastic lesion in dogs, despite its correlation with increasing age. Additionally, due to the relatively high frequency of pancreatic NH lesions and the relative sparsity of pancreatic adenocarcinoma documented in the canine, these processes are determined to most likely be independent. Although adenocarcinoma is the most common exocrine pancreatic tumor in the dog, the incidence is unreported but perceived as low. The absence of a single case in our sample population of 101 dogs is thus not unexpected.
This correlation of NH and age would also appear to be worth investigating in cats, where the presence of chronic fibrosing pancreatitis is more commonly noted and might result in a more consistent regenerative nodular tissue response in this species compared with the dog. 4
Hepatic NH occurs in the liver of geriatric patients and is interpreted typically as an incidental finding, the specific cause of which is unknown. 1 It is generally thought that hepatic NH is age related in dogs, as initial lesions are often noted by 6–8 years of age and 70–100% of dogs will develop this condition by the time they are 14 years old. 3 The ability of the liver to be repaired by hyperplasia is well documented, and thus, these nodules could represent response to injury, similar to what is seen with hepatic regenerative nodules (hyperplasia of hepatocytes secondary to progressive fibrosis and destruction of hepatic parenchyma, such as with cirrhosis). 2 Similar to our findings in the exocrine pancreas, hepatic NH is also not considered preneoplastic.
Hyperplastic lesions seen in endocrine organs, such as in adrenal or thyroid tissue, are capable of producing clinical syndromes, such as hyperadrenocorticism (Cushing). 6 These hyperplastic changes are not considered a response to antecedent injury but are the result of chronic or persistent stimulation by high levels of pituitary hormones. For example, high levels of adrenocorticotrophic hormone can stimulate hyperplastic responses in the adrenal cortex, which are essentially reversible following removal of the stimulus. It is only following neoplastic transformation of the highly proliferative cells that adrenal adenomas and adenocarcinoma could result. We do not know if or how growth factors control exocrine pancreatic proliferation.
We do not know whether these exocrine pancreatic hyperplastic lesions are functional/secretory and serum testing has not been performed. We hope to be able to correlate the antemortem serum concentrations for various pancreatic markers, specifically canine pancreatic lipase immunoreactivity, trypsin alpha 1 protease inhibitor, canine trypsinlike immunoreactivity, amylase, and lipase to the histopathologic findings in these dogs, in future studies. Canine pancreatic lipase immunoreactivity may be the most sensitive and specific marker for exocrine pancreatic disease, yet its association with proliferative pancreatic lesions is undocumented. 9
We conclude that NH of the exocrine pancreas occurs frequently in the dog and that the presence and severity of lesions correlate with increasing age. Thus, NH is more common in older dogs yet shows a positive correlation with age regardless of the presence or absence of pancreatic inflammation, necrosis, and/or fibrosis.
Footnotes
Acknowledgements
This work was funded by a Morris Animal Foundation grant.
1Present address: Department of Pathology, College of Veterinary Medicine, University of Tennessee, Knoxville, TN.
Presented in part at the American College of Veterinary Internal Medicine Forum, Minneapolis, MN, June, 2004.
