Abstract
Objectives
The aim of this study was to determine whether ultrasonographic pancreatic echogenicity, especially hypoechogenicity, has an impact on the prognosis of cats with suspected pancreatitis. We hypothesised that cats with a hypoechoic pancreas have a longer duration of hospitalisation, higher treatment costs and a higher mortality rate.
Methods
This was a retrospective study, which included cats with clinical signs of pancreatitis, a SNAP fPL test result above the reference interval and ultrasonographic abnormalities consistent with pancreatitis. Medical records and follow-up information were retrieved from the local electronic database. Cases were assigned to one of three groups based on pancreatic echogenicity: hypoechoic, hyperechoic or mixed echogenicity. Statistical analysis aimed to assess differences in outcome, ultrasonographic abnormalities, historical features, physical examination findings, laboratory results, concurrent diseases or treatment costs.
Results
Thirty-six (64%) cats with a hypoechoic, seven (13%) with a hyperechoic and 13 (23%) cats with a mixed echoic pancreas were included. Cats with a hypoechoic pancreas had a significantly lower median body weight (P = 0.010) and lower median body condition score (P = 0.004) compared with the other cats. Furthermore, they were presented as being lethargic significantly more often (P = 0.014), were more likely to have a homogeneously enlarged pancreas (P <0.001) and were less likely to have concurrent abnormalities or diseases, such as ultrasonographic enteric abnormalities (P = 0.020), triaditis (P = 0.013) and diabetes mellitus (P = 0.041). However, there was no difference in treatment costs (P = 0.961), duration of hospitalisation (P = 0.898), survival of hospitalisation (P = 0.419) or survival time (P = 0.256) between groups.
Conclusions and relevance
In this study, echogenicity was of no prognostic value in cats with suspected pancreatitis. Cats with a hypoechoic pancreas were not associated with a poorer outcome than cats with a hyperechoic or mixed echoic pancreas.
Introduction
Histologically, feline pancreatitis can be divided into acute and chronic disease.1,2 In chronic pancreatitis, fibrosis, acinar atrophy and mild-to-moderate lymphocyte infiltration are the predominant features, whereas acute pancreatitis is characterised by necrosis of acinar cells, neutrophilic inflammation associated with interstitial oedema and peripancreatic fat necrosis.1,2 Irrespective of its classification, history and physical examination findings are often non-specific, and severity of the disease may vary from asymptomatic to severe.2,3
The prognosis is considered to be dependent on the severity, as well as duration of the disease, amount of pancreatic necrosis, the presence of systemic complications (eg, disseminated intravascular coagulation, pulmonary thromboembolism and multiorgan failure) and concurrent disorders.1,4–6 In human medicine, patients with pancreatic necrosis have been shown to have a higher mortality rate than patients without necrosis, and, in dogs, pancreatic necrosis is associated with longer hospitalisation and more relapses.7,8
In ultrasonography, the normal feline pancreatic echotexture is homogeneous and the echogenicity is nearly isoechoic to the surrounding mesenteric fat, isoechoic or slightly hyperechoic to the liver and hypoechoic to the spleen. 9 The pancreatic duct is visible in the centre of the pancreatic parenchyma as an anechoic, tubular structure with a surrounding thin hyperechoic wall. 9 The thickness of the pancreas ranges from 0.5 to 0.9 cm in the left lobe and the pancreatic body, and from 0.3 to 0.6 cm in the right lobe. 10
The most common ultrasonographic findings in feline pancreatitis are an increased echogenicity of the mesenteric fat bordering the pancreas, peritoneal effusion, increase in pancreatic thickness, irregular pancreatic margins, hypoechoic or hyperechoic appearance, mixed echogenicity and pancreatic masses/cysts.11–13 The peripancreatic hyperechogenicity is consistent with peripancreatic steatitis and saponification of fat. 14 Decreased echogenicity in acute pancreatitis is thought to be a result of oedema, inflammatory infiltrate and necrosis. In contrast, increased echogenicity suggests fibrotic changes.15,16
The primary objective of this study was to determine whether ultrasonographic pancreatic echogenicity can be used as a prognostic marker of disease severity. As a hypoechoic pancreas indicates necrosis, at least to some degree, our hypothesis was that cats with a hypoechoic pancreas would have a poorer outcome (longer duration of hospitalisation, higher treatment costs and a higher mortality rate).
Materials and methods
The database of the University of Veterinary Medicine Vienna was searched for cats suspected of having pancreatitis based on abdominal ultrasound findings between July 2011 and May 2017, with pancreatic echogenicity described as either hypoechoic, hyperechoic or mixed. The ultrasonographic diagnosis of suspected pancreatitis was made based on the presence of any of the following: hyperechoic peripancreatic fat, peritoneal effusion, increase in pancreatic thickness, irregular pancreatic margins and change in echogenicity.
Cases with a SNAP fPL test (IDEXX) above the reference interval (>3.5 μg/l) were included. Each patient’s medical records were reviewed and cases with at least two historical features or physical examination findings consistent with pancreatitis were included. Historical features compatible with pancreatitis were determined as lethargy, vomiting, anorexia, diarrhoea and/or weight loss. Physical examination findings consistent with pancreatitis were hypothermia, hyperthermia, dehydration, icterus, pale mucous membranes and/or a painful abdomen.
As many cats with pancreatitis have concomitant diseases such as hepatic lipidosis, cholangitis, inflammatory bowel disease (IBD), triaditis and diabetes mellitus,3,17 and hyperthyroidism and chronic kidney disease are common in older cats, 18 cats with uncomplicated diabetes mellitus, mildly elevated creatinine concentrations (International Renal Interest Society [IRIS] stage ⩽2), as well as hyperthyroidism, IBD, suspected and confirmed hepatobiliary disease, or suspected triaditis were included. Cats without hospitalisation, asymptomatic patients, patients with previous surgery, a history of trauma, neoplasia or concurrent severe systemic diseases (eg, feline infectious peritonitis, diabetic ketoacidosis and IRIS stage ⩾3 renal failure) were excluded.
All ultrasound examinations were performed in right lateral recumbency by a single radiologist with over 25 years of experience in the field of ultrasonography. An 8–5 MHz curved array transducer and/or a 12–5 MHz linear array transducer (HDI 5000 and iU22; Philips) were used.
Included cats were assigned to one of three groups. Group A included all cats with an ultrasonographic hypoechoic pancreas compared with the surrounding fatty tissue, the liver and the spleen. In the case of hyperechoic peripancreatic fat, only the liver and spleen were used for echogenicity comparison. Group B included all cats with a hyperechoic pancreas compared with the surrounding fat, liver and spleen. Cats with mixed echogenicity were assigned to group C. Mixed echogenicity was defined as an echo intensity (brightness) ranging from hypoechoic to hyperechoic. Cases were included if the pancreas had patchy hypoechoic and hyperechoic areas throughout the parenchyma (in this example, the echotexture was termed ‘heterogeneous’); they were also included if a portion of the pancreas had a different echogenicity than the rest of the pancreas and when the pancreas had a normal echogenicity but other signs consistent with pancreatitis (Figures 1 and 2).

Ultrasound images of a (a) hypoechoic, (b) hyperechoic and (c) mixed echoic pancreas. Pancreases are between the measuring calipers in all images. (a) The hypoechoic pancreas being measured between the calipers (14 mm) has a homogeneous parenchyma and irregular margins; note the increased echogenicity of the mesenteric fat bordering the pancreas. In (b), the left lobe of the pancreas is diffusely hyperechoic compared with the surrounding fat; the pancreatic duct is visible as an anechoic tubular structure running through the pancreatic parenchyma. The left pancreatic lobe in image (c) has a mixed echogenicity; it is partially isoechoic to the mesenteric fat bordering the pancreas and has multifocal areas of increased echogenicity, resulting in a heterogeneous parenchymal texture
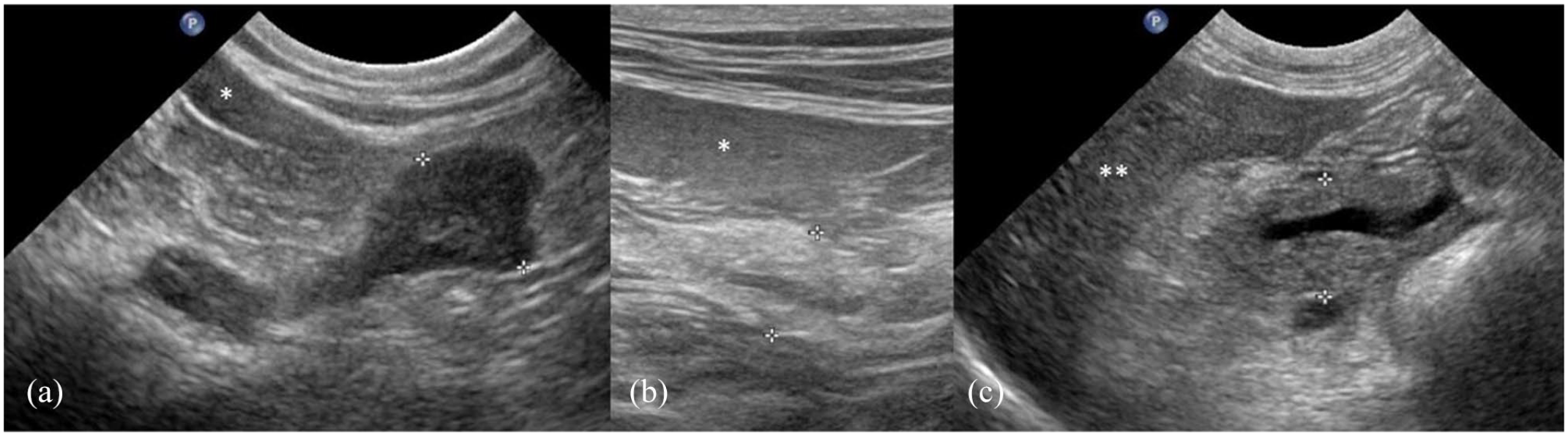
Ultrasound images of the pancreas (between the measuring calipers) of a (a) group A, (b) group B and (c) group C cat obtained with a (a,c) micro-convex and (b) linear array probe, respectively. Comparison of echogenicity to parenchyma of the liver and spleen: (a) the pancreas is hypoechoic to the spleen (*) with hyperechoic fat surrounding the pancreas; (b) hyperechoic pancreas in comparison to the spleen (*); and (c) the pancreas is isoechoic to slightly hyperechoic to the liver (**), is enlarged (13.5 mm thickness), has irregular margination and hyperechoic peripancreatic fat is evident
Parameters used to characterise disease severity and outcome were duration of hospitalisation, survival of hospitalisation, survival time, time from admittance to voluntary intake of food, recurrence, time to recurrence and treatment costs. Recurrence was diagnosed if the cats re-presented with clinical signs consistent with pancreatitis and a SNAP fPL test result above the reference interval within 3 weeks and up to 4 years after hospital discharge. Time to recurrence was measured from the day of discharge to the day of re-presentation at the clinic. Owners were contacted by telephone to obtain follow-up information, if it was not documented in the local electronic database.
Sex, breed, age, body condition score (BCS), weight and duration of clinical signs prior to presentation were recorded. Patient assessment also included historical features (lethargy, vomiting, anorexia, diarrhoea, weight loss); results of the physical examination (hypothermia, hyperthermia, dehydration, icterus, pale mucous membranes, painful abdomen); serum biochemistry and electrolytes when available (lipase, alanine transaminase [ALT], gamma-glutamyl transpeptidase, glutamate dehydrogenase, alkaline phosphatase, total bilirubin, glucose, calcium and potassium); haematocrit, erythrocytes and leukocytes; concurrent diseases (diabetes mellitus, hyperthyroidism, IBD, suspected or confirmed hepatobiliary disease and triaditis); and ultrasonographic findings (enlarged pancreas, heterogeneous parenchymal texture, hyperechoic peripancreatic fat, peritoneal effusion, pancreatic nodules/cysts, enlarged lymph nodes, hepatic abnormalities, dilated common bile duct, gallbladder abnormalities and enteric abnormalities). Enlargement of the pancreas was defined as measuring >1 cm in diameter at the pancreatic body and/or left lobe. 10 Lymph nodes were measured according to previously published literature. 19 Ultrasonographic hepatic abnormalities were defined as changes in echogenicity or echotexture or increase in size. The common bile duct was defined as dilated if >4 mm in diameter. 20 Gallbladder abnormalities were noted as wall thickness >1 mm and/or the presence of sludge. 21 An increase in intestinal mucosal layer echogenicity and thickening of the muscularis propria (when the width of the muscular layer was greater than half the thickness of the submucosal layer) were summarised as ultrasonographic enteric abnormalities. 22
A BCS from 1 (emaciated) to 5 (obese) was applied. 23 Hyperthermia and hypothermia were defined as a body temperature >39.3°C and <38.0°C, respectively. 24 A diagnosis of diabetes mellitus was made when blood glucose concentrations exceeded the renal threshold (230–290 mg/dl) and fructosamine concentrations were above the upper limit of the reference interval (400 mol/l).25–27 A thyroxine concentration >48 nmol/l was considered diagnostic of hyperthyroidism. 28 Histological diagnosis of IBD was determined via full-thickness biopsy. Suspected hepatobiliary disease was defined as the presence of ultrasonographic abnormalities of the liver and/or the gallbladder in combination with elevated bilirubin and/or ALT levels. Hepatobiliary disease was confirmed by cytological or bacteriological examination of specimens obtained by ultrasound-guided fine-needle aspiration (FNA) of the liver and/or cholecystocentesis. Triaditis was suspected when cats had serum biochemistry and ultrasonographic changes consistent with inflammation of the hepatobiliary system, pancreas and small intestine.
Statistical analysis
IBM SPSS Statistics 24 was used for the analyses. Metric data (eg, age, weight, survival time and duration of hospitalisation) were tested for normality using the Kolmogorov–Smirnov test. Comparison between groups was performed using Kruskal–Wallis and Dunn’s post-hoc test. Categorical variables (eg, historical features, physical examination, ultrasonographic abnormalities and concurrent disease) were compared using contingency tables and Fisher’s exact test. Survival data were examined by Kaplan–Meier survival analysis and the log-rank test. Associations between variables were tested in a pairwise fashion using the Spearman’s rank order correlation coefficient. Results were considered statistically significant if P ⩽0.05.
Results
Study population
Fifty-six cats met the inclusion criteria. Thirty-six (64%) cats were assigned to group A, seven (13%) to group B, and 13 (23%) to group C.
There were 29 (52%) neutered males, one (2%) intact male, 23 (41%) spayed females and three (5%) intact females in the study. Differences in sex distribution identified between groups were not significant (P = 0.165). Breeds included domestic shorthair (n = 46), domestic longhair (n = 2), Maine Coon (n = 2), Burmese (n = 1), Birman (n = 1), Maine Coon mixed breed (n = 1), Siamese (n = 1), Persian (n = 1) and Norwegian Forest Cat (n = 1).
Median (range) age, BCS, weight and duration of clinical signs are provided in Table 1. The median body weight of cats in group A was significantly lower compared with those in group B (P = 0.018) and group C (P = 0.045). Median BCS was significantly lower in cats in group A than in cats in group B (P = 0.006), with no difference between cats in group A and group C (P = 0.091).
Age, body condition score (BCS), weight and duration of clinical signs

Data are median (range)
Group A = hypoechoic pancreas; group B = hyperechoic pancreas; group C = mixed echogenicity
There were no significant differences in clinical pathology results or therapy between groups (see Table 1 in the supplementary material).
Historical features and physical examination
Owing to some missing data, groups B and C were combined for comparison of history and physical examination findings, which are summarised in Table 2. Results of the individual groups can be found in Table 2 in the supplementary material. Lethargy was the only finding that differed significantly between groups (P = 0.014).
History and physical examination findings

Data are n (%)
Group A = hypoechoic pancreas; group B = hyperechoic pancreas; group C = mixed echogenicity
Ultrasonographic findings
Ultrasonographic abnormalities are given in Table 3. The most consistent finding was enlargement of the pancreas, identified in 84% of all cases. Cats in group A were significantly more likely to have an enlarged pancreas (P <0.001) with homogeneous parenchyma (P = 0.012) than the other two groups. Cats in group C were associated with ultrasonographic enteric abnormalities (P = 0.020).
Ultrasonography findings

Data are n (%)
Group A = hypoechoic pancreas; group B = hyperechoic pancreas; group C = mixed echogenicity
Concurrent diseases
The frequency of concurrent diseases is summarised in Table 4. Ultrasonographic abnormalities of the small intestine (P = 0.020) and suspected triaditis (P = 0.013) occurred more frequently in cats in group C than in cats in the other two groups. There was no difference in the frequency of ultrasonographic hepatobiliary abnormalities, or suspected or confirmed hepatobiliary disease. In the eight cases in which hepatobiliary disease was confirmed, FNA cytology revealed cholangitis, cholecystitis and hepatic lipidosis. Bacteria were cultured from the gallbladder in one cat and from the liver in another. When groups B and C were considered together, significantly more cats in those two groups had concurrent diabetes mellitus compared with cats in group A (P = 0.041). Only four cats in the entire study population had no additional ultrasonographic abnormalities other than those suggestive of pancreatitis and were free of suspected concurrent disease.
Occurrence of concurrent diseases

Data are n (%)
Group A = hypoechoic pancreas; group B = hyperechoic pancreas; group C = mixed echogenicity; US enteric abnormalities = ultrasonographic abnormalities of the small intestine; confirmed IBD = histological diagnosis of inflammatory bowel disease (IBD) determined via full-thickness biopsy; US hepatobiliary abnormalities = ultrasonographic abnormalities of the liver and/or the gallbladder; suspected hepatobiliary disease = ultrasonographic abnormalities of the liver and/or the gallbladder in combination with elevated bilirubin and/or alanine transaminase (ALT) levels; confirmed hepatobiliary disease = cytological or bacteriological confirmation of hepatobiliary disease by ultrasound-guided hepatic fine-needle aspiration and/or cholecystocentesis; suspected triaditis = ultrasonographic abnormalities of the hepatobiliary system, small intestine and the pancreas as well as elevated bilirubin and/or ALT levels
Outcome
Duration of hospitalisation, survival, survival time after discharge, time from admittance to voluntary intake of food, recurrence and the time to recurrence did not differ between groups (Table 5).
Outcome parameters

Survival of hospitalisation and recurrence data are n (%); all other data are median (range)
P values are given for comparison of outcome variables between groups
Group A = hypoechoic pancreas; group B = hyperechoic pancreas; group C = mixed echogenicity
Thirty of 36 cats in group A, 5/7 cats in group B and 12/13 cats in group C survived hospitalisation. No significant difference was detected for survival times between groups (Figure 3). There was no correlation between survival times and duration of hospitalisation (P = 0.636), or between survival times and time from admittance to voluntary intake of food (P = 0.216).

Kaplan–Meier survival curves. Vertical marks represent censored cats. The x-axis shows survival time in days since discharge and the y-axis the proportion surviving.
As cats in group C had significantly more frequent ultrasonographic enteric abnormalities and suspected triaditis, and diabetes mellitus was detected more frequently in groups B and C, these parameters were compared to survival of hospitalisation, independently of group. There was no significant difference in mortality for the presence of ultrasonographic enteric abnormalities (P = 0.288), triaditis (P = 0.676) and diabetes mellitus (P = 0.671).
Discussion
Acute and chronic feline pancreatitis can vary from mild to severe and show overlapping ultrasonographic abnormalities.1,2,10,29,30 Based on the assumption that a hypoechoic pancreas represents, at least to some extent, necrosis, we hypothesised that this specific pancreatic appearance is associated with a poorer outcome and can be used as a prognostic marker. The results of this study provide evidence against the proposed hypothesis. There was no difference in outcome between cats with either hypoechoic, hyperechoic or mixed echoic parenchyma.
Necrosis is present in 96% of dogs with fatal acute pancreatitis. 31 In an experimental study on five Beagle dogs, necrosis was demonstrated by Maier 32 to be hypoechoic, where cases with 75–100% necrosis of the pancreas on microscopy were hypoechoic on ultrasound. Conversely, ultrasonography of the pancreas was normal in most cases of minimal necrosis (⩽25%). 32
As necrosis cannot be distinguished from oedema in B-mode ultrasonography, 33 we speculate that the homogeneously enlarged hypoechoic pancreas on ultrasound in this study could have, in fact, represented oedema. The typical ultrasonographic sign for pancreatic oedema in dogs is the so-called ‘tiger-stripe’ pattern, which represents poorly circumscribed hypoechoic foci to well-defined linear anechoic fissures and subcapsular anechoic spaces. 34 In human medicine, an enlarged diffusely hypoechoic pancreas is consistent with oedematous pancreatitis on endoscopic ultrasound, whereas in necrotising pancreatitis, the gland is also enlarged but tends to show a heterogeneous parenchymal texture with hypoechoic circumscribed areas. 35 In our study, a hypoechoic pancreas was associated with enlargement and homogeneous parenchyma in most cases, and thus would also fit the description of oedematous pancreatitis in human medicine. 35
In people, the gold standard method to distinguish necrosis from oedema is CT with intravenous contrast administration; in oedematous pancreatitis the gland enhances homogeneously, whereas necrosed areas do not enhance. 36 This has clinical relevance as necrosis is associated with a higher mortality rate. 7 In dogs, heterogeneous contrast enhancement of the pancreas in CT is consistent with necrotic areas and associated with a longer duration of hospitalisation, more relapses and a higher frequency of portal vein thrombosis.8,37 Limited availability and the need of anaesthesia, but most of all the very low sensitivity for diagnosing feline pancreatitis, inhibits the use of CT in feline practice.38,39 A previous study with MRI showed promising results in the detection of feline pancreatitis, but there have not been any studies correlating the results with necrosis or patient outcome. 40 Using contrast-enhanced Doppler ultrasonography, Rademacher et al 41 identified a difference in vascularity and blood volume in healthy cats and cats with pancreatic disorders. However, to our knowledge, there is no literature correlating contrast-enhanced ultrasonography (CEUS) results with feline patient outcome. In people and dogs, CEUS can distinguish necrosis from interstitial pancreatitis.42–44 Studies in cats are strongly encouraged.
In the current study, body weight and BCS were lower in cats with a hypoechoic pancreas than either hyperechoic or mixed echoic pancreas. Weight loss is a common complaint in cats with pancreatitis; thus, it could be concluded that these cats had a longer duration of disease. The results of this study contradict this explanation. It is possible that cats with a hypoechoic pancreas had a lower initial weight. Conversely, cats with a higher BCS could have an increase in echogenicity of the pancreas owing to fatty infiltration. In people, such an increase in pancreatic echogenicity is described as a result of an increase in age and body weight.45–47 The terminology fatty pancreas, pancreatic lipomatosis or steatosis is used, and has also been described in a single case report in a dog.48–50 A hyperechoic pancreas also correlated with dogs having hyperadrenocorticism. 51 In clinically healthy cats, there was no increase in echogenicity with age; 52 however, no studies have investigated the relationship of body fat and echogenicity of the feline pancreas.
Although feline pancreatitis is associated with many different concurrent diseases,1,3,5,13,14,29,53 their impact on patient outcome is uncertain. Similar to previous studies, more than half of the cats in this study had confirmed concurrent disorders and 93% had at least suspected concurrent disease.13,53 This is comparable to a study in which 92% of cats were found to have concomitant disorders. 3 In this study, 54% of the cats had ultrasonographic hepatic abnormalities, 39% had gallbladder abnormalities and 30% had a dilated common bile duct. Group differences were not observed. As a normal liver size, echogenicity and normal biliary system on ultrasound does not exclude feline cholangitis, these results should be interpreted with caution. 54 Interestingly, cats with a hypoechoic pancreas were statistically less likely to have concurrent diseases, such as ultrasonographic enteric abnormalities, triaditis and diabetes mellitus. Although it is possible that this improved the outcome in cats with this specific pattern, cats with diabetes mellitus or the suspicion of triaditis did not have a poorer outcome in this study.
Of note, cats with a hypoechoic pancreas tended to be more lethargic than cats with hyperechoic or mixed echoic pancreas. An association with pain seems possible. Lethargy, as well as decreased appetite or decreased grooming, can be a sign of pain. 55 In dogs, a painful abdomen is a common finding on physical examination. 31 In people, acute and chronic pancreatitis are also characterised by persistent and severe pain. 56 This is caused by fibrosis, ischaemia, recurrent acute inflammation with necrosis of the pancreas or increased pancreatic pressure, which also has been demonstrated in an experimental study with cats.57–61
This study had several limitations. The first was the small sample size, particularly in groups B and C. This was mainly due to the retrospective nature of the study design and the strict exclusion criteria. The small number of cases weakened the statistical analyses and conducting a power analysis would have been ideal. A further limitation of this retrospective study was that it was not possible to determine the actual number of cats with concurrent diseases because histopathology is required for diagnosis. For this reason, we included the parameters with suspected concomitant disorders, which is probably closer to the actual number. However, it was not possible to determine the severity of these concomitant diseases. Another limitation was the lack of histopathology, which, again, was based on the retrospective study design. FNA or biopsy of the pancreas were only performed in four cats, and only three cats underwent necropsy. Histopathology of incisional pancreatic biopsies is currently considered the gold standard for ante-mortem diagnosis of feline pancreatitis.2,3,62 However, because of the frequently multifocal nature with lesions scattered throughout the pancreas, a single biopsy may be insufficient to exclude pancreatitis.2,62–64 Based on necropsy examinations, chronic pancreatitis is also evident in clinically sound cats and, therefore, the clinical significance of histopathologically mild pancreatitis is debatable. 38 In addition, cats with severe pancreatitis are often not suitable candidates for anaesthesia.62,64
Although a SNAP fPL test was performed in all patients, Spec fPL or 1,2-o-dilauryl-rac-glycero-3-glutaric acid ± (6′-methylresorufin) ester (DGGR) lipase measurements to further support a diagnosis of pancreatitis were performed in only a small number of these cats. Of note, the primary aim of this study was not to diagnose feline pancreatitis, but to investigate whether pancreatic echogenicity, especially hypoechogenicity, has an impact on the prognosis in cats with ‘suspected’ pancreatitis.
Conclusions
Echogenicity was of no prognostic value in cats with suspected pancreatitis in this study. Cats with an ultrasonographically hypoechoic pancreas were not associated with a poorer outcome than cats with a hyperechoic or mixed echoic pancreas. Studies are encouraged to investigate whether this specific ultrasonographic appearance actually represents oedematous pancreatitis rather than necrosis in cats.
Supplemental Material
Table 1
Clinical pathology results
Supplemental Material
Table 2
History and physical examination findings
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
Supplementary material
The following files are available online:
Table 1: Clinical pathology results.
Table 2: History and physical examination findings.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
