Abstract
Objectives
In humans, renal aging is associated with an increased frequency of glomerulosclerosis, interstitial fibrosis, inflammation and tubular atrophy. The purpose of this study was to describe the frequency of renal histopathologic lesions in cats without kidney disease.
Methods
A cross-sectional study of archival kidney tissue from 74 cats without kidney disease (serum creatinine <1.6 mg/dl; urine specific gravity >1.035) was carried out: 0–4 years (young, n = 18); 5–9 years (mature, n = 16); 10–14 years (senior, n = 34), 15+ years (geriatric, n = 6). Glomerulosclerosis, tubular atrophy, interstitial inflammation and fibrosis, and the presence or absence of lipid in the interstitium and tubules were scored by a pathologist masked to clinical data. Statistical analyses were performed as appropriate.
Results
Geriatric cats had significantly more glomerulosclerosis than mature (P = 0.01) and young cats (P = 0.004). Senior cats had significantly more glomerulosclerosis than young cats (P = 0.006). Glomerulosclerosis was weakly positively correlated with age (r = 0.48; P <0.0001). Geriatric cats had significantly more tubular atrophy than mature (P = 0.02) and young cats (P <0.0001). Senior cats had significantly more tubular atrophy than young cats (P <0.0001). Geriatric cats had significantly more inflammation than senior cats (P = 0.02), mature cats (P = 0.01) and young cats (P <0.0001). Senior cats had significantly more inflammation than young cats (P = 0.004). Geriatric and senior cats had significantly more fibrosis than young cats (P = 0.01 and P = 0.04, respectively). Frequency of tubular lipid increased with age (young: 28%; mature: 56%; senior: 79%; geriatric: 100%) as did the frequency of interstitial lipid (young: 22%, mature: 56%, senior: 85%, geriatric: 100%).
Conclusions and relevance
Evidence of renal aging exists in cats. These changes imply that the aging kidney may be more susceptible to injury and impaired healing.
Introduction
Renal aging refers to physical and functional changes that occur in the kidney with age. Age is associated with decreases in renal parenchymal volume and the number of functioning nephrons, as well as a decline in glomerular filtration rate (GFR).1–5 In multiple species, histopathologic lesions such as tubulointerstitial fibrosis, tubular atrophy and glomerulosclerosis are seen before clinically relevant chronic kidney disease (CKD) is apparent.2,3,6 Mechanisms that contribute to renal aging include cellular senescence, pericyte loss and imbalance of pro- and antiangiogenic factors that promote capillary rarefaction. 1
A key functional outcome of renal aging is a decreased capacity to cope with normal and abnormal stressors due to an altered ability to engage in healthy repair. 7 For example, in humans, this manifests as decreased recovery from acute kidney injury in the elderly and decreased success with transplantation of kidneys from older donors.7–11 Unsurprisingly, the concept of renal aging has significant implications for the development of CKD, and in human medicine there is debate as to what constitutes renal aging vs disease.2,6
CKD is common in older cats and results in similar histopathologic lesions; thus, these questions are undoubtedly applicable to feline patients.12–15 In cats, there is some evidence of renal aging, with histopathologic lesions increasing in severity with age in cats without CKD. 14 To date, few studies have assessed the histopathology of renal aging in cats.14,16,17 Therefore, the purpose of our study was to determine the frequency of histopathologic lesions in the kidneys of cats without CKD across a range of age groups. We hypothesized that lesions such as glomerulosclerosis, tubular atrophy, inflammation and fibrosis would increase with age.
Materials and methods
Case selection
The study was performed using archived tissues from necropsy. Consent for necropsy was obtained from the owner by the institution at the time of death. A medical records search was performed at five universities for feline patients that had a necropsy, biochemistry and urinalysis performed. Medical records were reviewed and cases were further excluded if no renal tissue was available or clinicopathologic data to determine the absence of clinical renal dysfunction were inadequate. The absence of CKD was defined as a serum creatinine <1.6 mg/dl, urine specific gravity (USG) >1.035 and no clinical suspicion of CKD (based on history, physical examination and prior laboratory test results, when available). Exclusion criteria included renal disease/neoplasia, diabetes mellitus, hyperthyroidism, congestive heart failure or thromboembolism, urinary tract infection, proteinuria, administration of non-steroidal anti-inflammatory drugs, chemotherapy or cardiac medications (eg, furosemide). Cats were grouped by age into the following 5 year increment categories, which approximated the 2010 Life Stage Guidelines: young (0–4 years), mature (5–9 years), senior (10–14 years) and geriatric (⩾15 years). 18
Histologic assessment
Archival renal tissue preserved in 10% neutral buffered formalin was routinely processed and embedded in paraffin prior to sectioning at 3 µm. Three slides from each cat were separately stained with hematoxylin and eosin, periodic acid–Schiff (PAS)–hematoxylin and Masson’s trichrome. PAS–hematoxylin and Masson’s trichrome stains were employed to appropriately assess basement membranes of glomeruli, tubules and blood vessels, and the presence of fibrosis, respectively.
The presence of tubular atrophy, interstitial inflammation and fibrosis in both the outer cortex (‘cortex’) and inner cortex (‘corticomedullary junction’ [CMJ]) was assessed by a board-certified pathologist (SM) blinded to clinical information. All the cortical tissue available was examined, and the percentage of affected tissue was estimated as one of the following six categories (0%, <5%, 5–15%, 16–25%, 26–50%, 51–75% and >75%), based on a previously used assessment method. 14 To allow statistical comparison, a numerical score was assigned based on the percentage of affected tissue (0 = 0%; 1 <5%; 2 = 5–15%; 3 = 16–25%; 4 = 26–50%; 5 = 51–75%; 6 >75%). The frequency of glomerulosclerosis was determined by examining 25 glomeruli from both the cortex and CMJ, and recording those that contained sclerosis. Glomerulosclerosis was defined as obliteration and consolidation of any portion of the glomerular capillary tuft by the extracellular matrix. 19 Glomerular obsolescence was included as a subcategory within the glomerulosclerosis variable and defined as a small, shrunken glomerulus composed of matrix and few cells.
Dichotomous variables (assessed as present or not present) included lipid in the interstitium, lipid casts within tubules and the presence of vascular lesions. Lipid was not confirmed by stains, but rather identified by the historical perspective of vacuoles within the cytoplasm of the renal tubular epithelium or vacuoles within the interstitial tissues. Vascular lesions were stratified for descriptive purposes into subcategories of vascular degeneration and injury as previously described. 14 Degeneration was characterized by concentric medial smooth muscle hyperplasia (hyperplastic arteriolosclerosis), segmental or circumferential intimal proliferation with disruption of the internal elastic lamina (fibrointimal hyperplasia) or plasma insudation within the wall of arterioles (hyaline arteriolosclerosis). Histologic evidence of vascular injury (eg, fibrinoid vascular necrosis and arteritis) were recorded if present.
Statistical analysis
Statistical analyses were performed using Prism software (GraphPad). Histologic variables were compared between groups using the Kruskal–Wallis test with Dunn’s post-hoc analysis. Spearman’s rank test was used to assess the correlation between age and the number of sclerotic glomeruli. Fisher’s exact test was used to compare dichotomous variables (tubular lipid, interstitial lipid, fibrointimal hyperplasia) in cats aged <10 years vs cats aged ⩾10 years, and tubular lipid and interstitial lipid in male cats vs female cats within each age group. Statistical significance for all analyses was set at P <0.05.
Results
Cats
A total of 1271 records that met the search criteria were reviewed; 1197 cases were excluded based on the predetermined exclusion criteria or lack of available renal tissue, and 74 cases had archived samples that met the predetermined inclusion criteria (18 young, 16 mature, 34 senior, six geriatric). Signalment, serum creatinine concentration and USG for cats from each age group are detailed in Table 1. Breeds included in the study were domestic shorthair (n = 52), domestic longhair (n = 11), Siamese (n = 2), Persian (n = 2) and one each of Bengal, Bambino, Maine Coon, British Shorthair, Manx, Egyptian Mau and domestic mediumhair. Causes of euthanasia or death for cats aged 0–4 years included hepatic disease (n = 6), neoplasia (n = 2), unknown (n = 3), and one each diarrhea and weight loss, Wallerian degeneration, research cat euthanized for an unrelated study, panleukopenia, upper respiratory infection (URI), feline leukemia virus and intestinal perforation. Causes of euthanasia or death for cats aged 5–9 years included neoplasia (n = 7), hepatic disease (n = 3), unknown (n = 2), lower airway disease (n = 2), choked on food (n = 1) and URI (n = 1). Causes of euthanasia or death for cats aged 10–15 years included neoplasia (n = 16), lower airway disease (n = 4), hepatic disease (n = 3), anesthetic complications (n = 2), chylothorax (n = 2), unknown (n = 3), glossitis (n = 1), paralysis (n = 1), inflammatory bowel disease (IBD; n = 1) and pyothorax (n = 1). Causes of euthanasia or death for cats aged ⩾15 years included neoplasia (n = 4), IBD (n = 1) and unknown (n = 1).
Signalment, serum creatinine and urine specific gravity for cats in the four age groups

Data are presented as median (range)
USG = urine specific gravity; MI = male intact; MN = male neutered; FS = female spayed; FI = female intact
Histopathology
Areas of tubular atrophy, inflammation and fibrosis tended to be segmental and variably interspersed throughout the kidney tissue available for evaluation (Figures 1 and 2). The presence of glomerulosclerosis, tubular atrophy, inflammation and fibrosis increased with age (Table 2; Figures 3 and 4) in both the cortex and corticomedullary junction. The number of sclerotic glomeruli was positively correlated with age in both the cortex (r = 0.48; P <0.0001) and CMJ (r = 0.43; P = 0.0002).

Representative photomicrographs of cats from each age group (×2 magnification, hematoxylin and eosin). (a) 0–4 years; (b) 5–9 years; (c) 10–14 years; (d) ⩾15 years. Asterisks mark areas of focal lesions

Representative photomicrographs of cats from each age group (×2 magnification, trichrome). (a) 0–4 years; (b) 5–9 years; (c) 10–14 years; (d) ⩾15 years. Asterisks mark areas of focal lesions
Glomerulosclerosis, frequency of histopathologic lesion scores (0 = 0%; 1 <5%; 2 = 5–15%; 3 = 16–25%; 4 = 26–50%; 5 = 51–75%; 6 >75%) and the presence of lipid and vascular lesions in four age groups of cats without chronic kidney disease
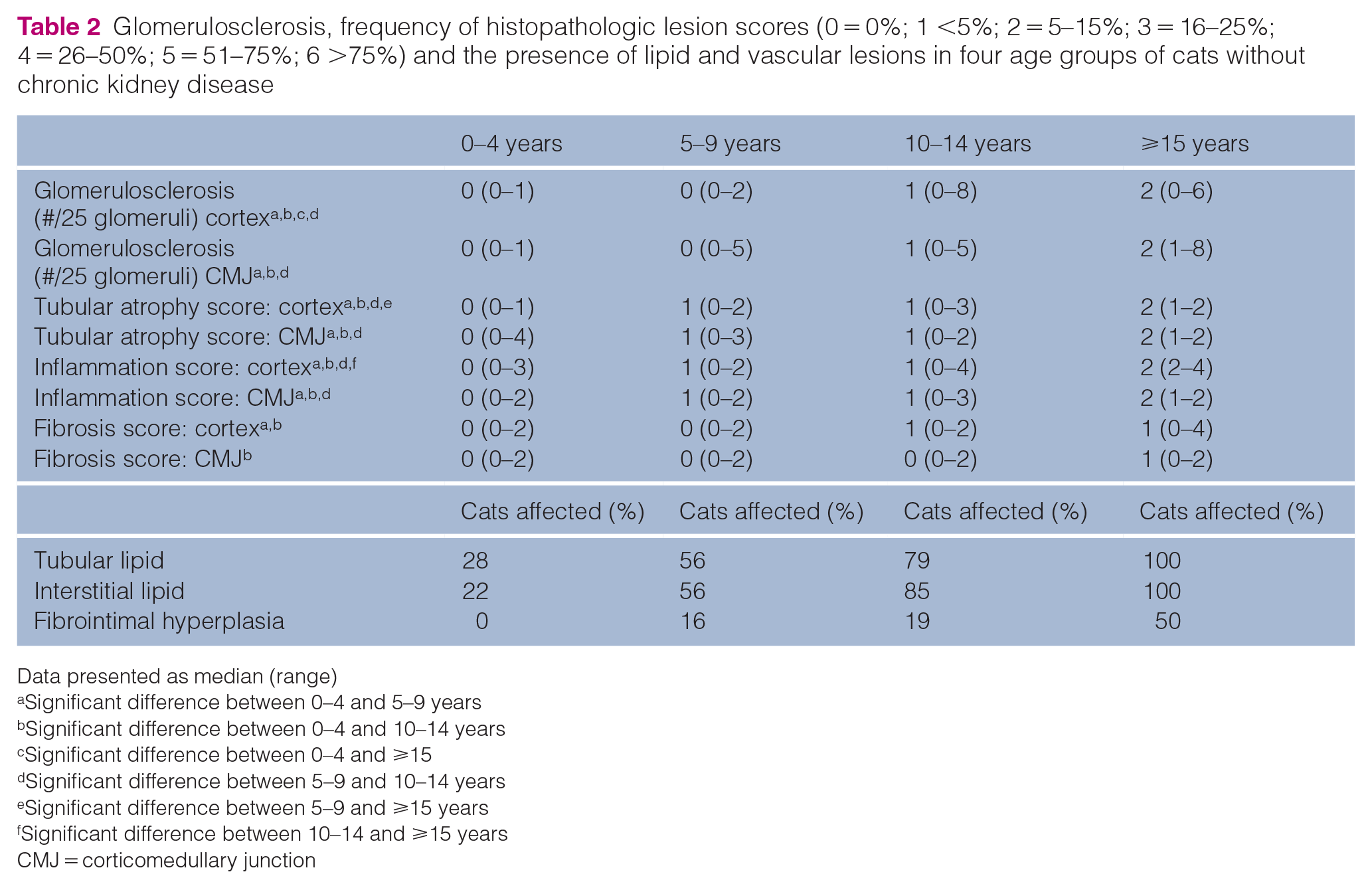
Data presented as median (range)
Significant difference between 0–4 and 5–9 years
Significant difference between 0–4 and 10–14 years
Significant difference between 0–4 and ⩾15
Significant difference between 5–9 and 10–14 years
Significant difference between 5–9 and ⩾15 years
Significant difference between 10–14 and ⩾15 years
CMJ = corticomedullary junction
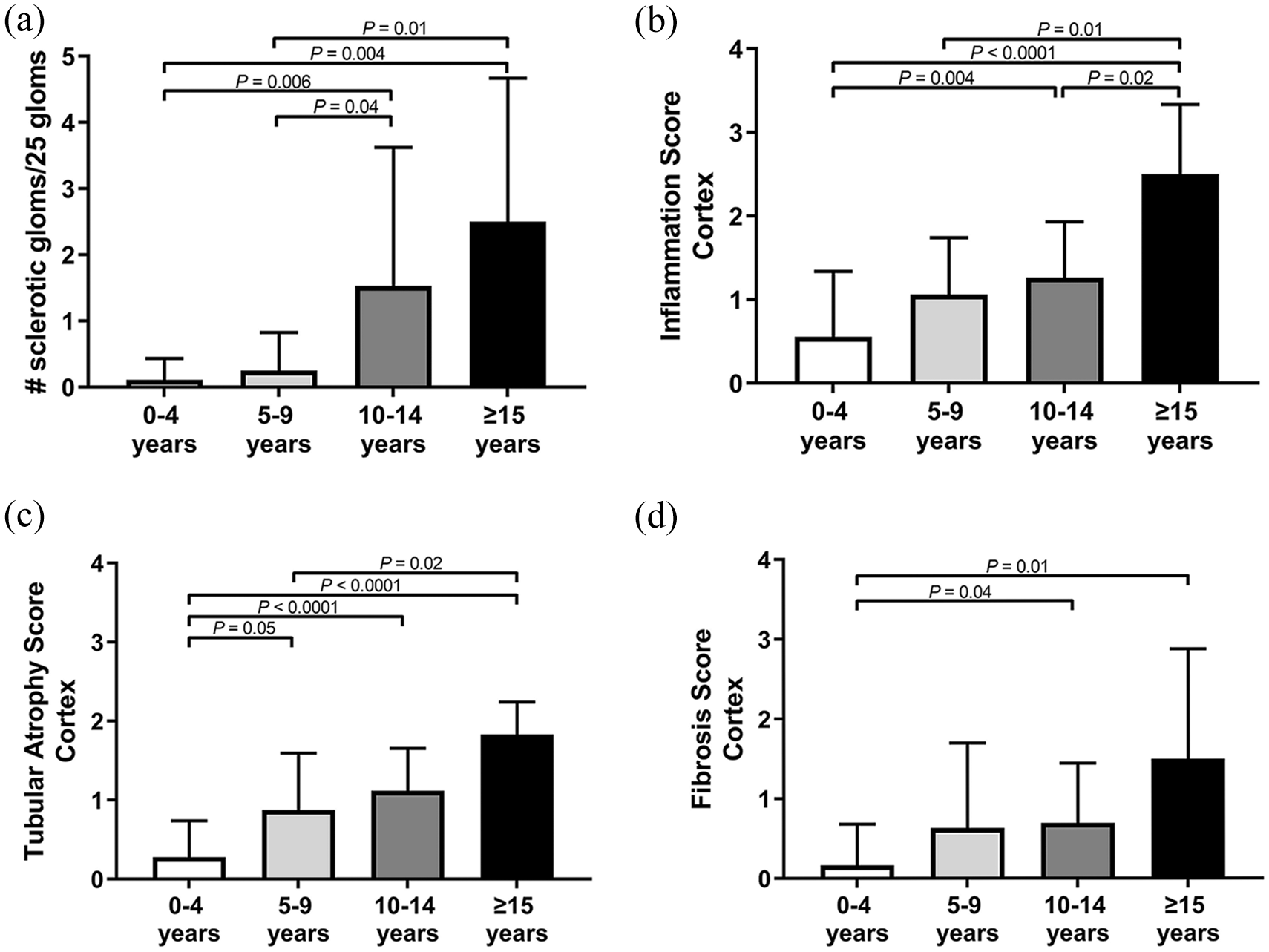
Frequency of renal histopathologic lesions (0 = 0%; 1 <5%; 2 = 5–15%; 3 = 16–25%; 4 = 26–50%; 5 = 51–75%; 6 >75%) in the renal cortex from different age groups. (a) Glomerulosclerosis, (b) inflammation, (c) tubular atrophy and (d) fibrosis increases with age in the renal cortex of cats without chronic kidney disease. Data are displayed as mean ± SD

Frequency of renal histopathologic lesions (0 = 0%; 1 <5%; 2 = 5–15%; 3 = 16–25%; 4 = 26–50%; 5 = 51–75%; 6 >75%) in the renal corticomedullary junction from different age groups. (a) Glomerulosclerosis, (b) inflammation, (c) tubular atrophy and (d) fibrosis increases with age in the renal cortex of cats without chronic kidney disease. Data are displayed as mean ± SD
Tubular and interstitial lipid (scored as a dichotomous variable) were both more common in cats aged ⩾10 years (P = 0.0003 and P <0.0001, respectively). Tubular and interstitial lipid were significantly more prevalent in male cats than females in the 5–9 years age group (P = 0.04 for both). A significant difference in prevalence of tubular or interstitial lipid was not seen between sexes in other age groups. The presence of interstitial lipid was noted to be commonly associated with focal areas of inflammation (Figure 5). There was no significant difference in the number of cats with fibrointimal hyperplasia in cats aged ⩾10 years vs younger cats.

(a) Tubular and (b) interstitial lipid in the kidney increases with age in cats without chronic kidney disease. (c) Photomicrograph demonstrating both tubular and interstitial lipid in an area of tubular loss with surrounding inflammation. Tubular lipid is apparent as multiple clear, round and variably sized cytoplasmic vacuoles that compress tubular epithelium and lumina. Interstitial lipid (asterisk) is associated with interstitial inflammation. Also pictured is a sclerotic glomerulus (thin arrow) in comparison to a more normal glomerulus (thicker arrow) (×10 magnification, trichrome)
Discussion
Histopathologic changes in the kidneys of cats without CKD increased with age and were characterized by increasing glomerulosclerosis, tubular atrophy, interstitial inflammation and fibrosis. These lesions were segmental and variable in their distribution throughout the kidney. These findings imply that such lesions increase in frequency over the lifetime of a patient and contribute to the phenotype of renal aging.2,6 Although it is unknown why CKD is so common in senior cats, currently the most accepted theory is that a combination of repeated insults over the life of the kidney result in damage that eventually culminates in clinical disease.20–23 The findings of our study are consistent with this theory. This is also supported by evidence from experimental models in cats, whereupon glomerulosclerosis, tubular atrophy, interstitial inflammation and fibrosis are seen 6 months after a 90 min unilateral renal ischemia event. 22 Similar histopathologic findings are found in rodent models where CKD develops after severe or repeated episodes of mild injury, 24 and in Mesoamerican nephropathy in humans where recurring episodes of renal damage are secondary to occupational heat stress and dehydration.25,26
The functional implications of these histologic lesions for the aging feline patient are unknown at this time. Although it is well documented in humans that GFR declines with age at a specific rate that varies based on sex and ethnicity,2,6 an extensive study has not been performed in veterinary species. In small dogs, iohexol GFR is demonstrated to decrease with age. 4 In a feline study assessing various modalities of GFR assessment, cats aged 9–12 years (n = 6) had significantly decreased GFR as measured by plasma clearance of creatinine compared with young cats aged 7–12 months (n = 6). 5 However, this effect was not appreciated when GFR was assessed by other modalities. 27 Therefore, it is unclear if an increased frequency of lesions in the kidney is correlated with decreasing GFR. USG is documented to decrease with age in apparently healthy cats, 28 and, as this can be one of the first signs of decreased renal function in cats, it is plausible this might be related to renal aging and loss of functional parenchyma. However, USG is inherently variable and has not been shown to be an independent predictor of development of azotemia in cats. 29
The clinical relevance of the aging kidney may be subtler than the onset of signs of early clinical disease. Cellular senescence is a central player in renal aging. Over the course of time, cells incur DNA damage due to various stressors (eg, oxidative stress). If injury to the DNA is irreparable, the cell either undergoes apoptosis (programmed cell death) or senescence (permanent inhibition of the cell cycle). 30 The accumulation of senescent cells with age can have a negative effect on the kidney’s ability to regenerate effectively and can also promote fibrosis.7,8,30 Senescence plays a role in aging in the feline kidney, as p16-mediated renal senescence is positively correlated with age in cats without CKD. 31 Taken together with our description of the histologic lesions associated with age, these data present evidence for renal aging in cats, and a concern for decreased ability to withstand injury and promote effective repair.
Tubular and interstitial lipid were present in a substantial portion of cats in this study and both increased significantly in frequency with age. Tubular lipid appears common in normal cats, and its relatively high prevalence in felids without disease (including lions and tigers) may be unique to Felidae.32–36 Although it is logical that interstitial lipid would increase with age due to accumulated episodes of injury and lipid release from tubules, 37 it is less clear why tubular lipid accumulation is so prevalent in normal felids and why it increases with age. Sex and neuter status likely have an effect on lipid accumulation as the current study demonstrated significantly more mature male cats with tubular lipid compared with females, but this was not seen in any other life stage. Neuter status has previously been demonstrated to affect the presence of lipid in the feline kidney, 34 but more recent studies involve patient populations that are predominantly neutered and of an older age and thus this differential is not seen.14,37 In humans, lipid accumulation is associated with obesity,38,39 and it might be presumed that neutered cats would be as prone to obesity as mature adults. Unfortunately, a correlation between body condition and the presence of lipid could not be made in our study as no consistent information on body condition score was available from retrospective medical record review, and retrospective medical record data at the time of euthanasia are unlikely to provide information about lifetime obesity trends. Additional research is needed to explore a possible relationship between obesity and tubular lipid in cats.
Tubular and interstitial lipid accumulation could be an important etiologic concept for the development of CKD as it is thought that once tubular injury occurs, disruption of the basement membrane (tubulorrhexis) results in the formation and accumulation of interstitial lipid, which acts as a nidus for inflammation.21,23 Tubular and interstitial lipid is common in cats with CKD and an increased prevalence of lipid accumulation in these patients is found with age.14,37 In experimental models of renal ischemia in cats, interstitial lipid accumulation in the vicinity of tubulorrhexis was first apparent 12 days after the ischemic event and was commonly associated with granulomatous inflammation.22,40 Similarly in the present study, interstitial lipid was associated with focal areas of inflammatory infiltrate (Figure 5), and this was already present in some cats at a young age. Additionally, recent literature indicates lipid accumulation in proximal tubular epithelial cells may have implications for cell health, even without an injury event. Evidence suggests that lipid accumulation may be pro-inflammatory and pro-fibrotic, promote the formation of reactive oxygen species, and have cytotoxic effects, possibly through impaired mitochondrial dysfunction.38,41,42 Several experimental models have provided evidence that lipid toxicity may be a novel therapeutic target.38,41,43 Therefore this subject merits additional study in the feline patient.
Limitations of this study include its retrospective nature, which could have influenced the accuracy and consistency of the clinical data collected. Blood pressure data were generally not available, so any effect of hypertension on renal histopathology could not be assessed. Additionally, information on body condition or muscle mass was also not available; therefore, in some cases serum creatinine could underestimate kidney dysfunction. In some cases, only one instance of serum creatinine or USG were available, and some cats with CKD may have a USG >1.035. As archived kidney tissues were used, and no university had a standard approach to kidney sampling, it is possible that there was bias in the selection of tissues, and the sample selected may not be representative of the entire kidney or both kidneys. Sections of kidney available for evaluation differed in size, and the entire area of cortical tissue was examined as opposed to selecting a consistent number of designated fields of view. Lesions were not scored for severity as the aim of the project was lesion frequency. It was not possible to perform lipid staining on archival tissues used in the study; however, since the original descriptions of lipid in feline kidney, other papers in the literature have assessed lipid without performing specific stains.14,22 Kidneys were only evaluated at one point in time in the lifespan of the patient and lesions could have been present at an earlier age.
Conclusions
Evidence for a phenotype of renal aging exists in cats. Clinically, this implies that the kidneys of the elderly feline patient may be more susceptible to injury and less effective repair.
Footnotes
Acknowledgements
The authors wish to thank Todd Bass for assistance in locating samples.
Correction (December 2022):
Article updated online to correct the name of the author Andrea Erickson.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Buttons Fund for Feline Chronic Kidney Disease Research.
Author note
The results of this study were presented in part, as a research abstract at the American College of Veterinary Internal Medicine Annual Forum, 2019.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
