Abstract
Feline morbillivirus (FeMV) is an emerging RNA virus in the Paramyxoviridae family that was recently discovered in domestic cats (Felis catus). To date, 2 genotypes (FeMV-1 and FeMV-2) have been detected in cats from various countries, and FeMV-1 is recognized as a pathogen associated with nephritis. However, information regarding the pathological roles and potential transmission to other felids is limited. In this article, we describe the identification of FeMV in 2 black leopards (Panthera pardus) in Thailand that showed severe azotemia and tubulointerstitial nephritis. Molecular analysis of the partial coding sequence of the L gene revealed that these leopard FeMV strains were genetically close to the FeMV-1 isolate from domestic Thai cats. Immunohistochemistry and immunofluorescence analyses using polyclonal IgG antibodies against the FeMV matrix (M) protein showed FeMV-M antigen in renal tubular epithelial cells. These analyses also showed infiltrating lymphocytes in the renal parenchymal lesions and in the cytoplasm of lymphoid cells residing in the spleen, suggesting viral tropism and a possible pathological role. These findings are the first evidence that indicates that the black leopard could be a possible host for FeMV infection. As for other cats, the role of FeMV as a potential cause of renal disease remains to be established. The pathogenesis of FeMV infection in black leopards, or in other wild felids, through a viral transmission mechanism warrants further investigation.
Keywords
Morbillivirus, a negative-sense single-stranded RNA virus in the Mononegavirales order, Paramyxoviridae family, contains 6 core genes that encode for the matrix protein (M), the hemagglutinin (H) and fusion (F) glycoproteins, 2 RNA-polymerase-associated proteins of phosphoprotein (P) and large protein (L), and a nucleocapsid (N) protein. 23 This virus family potentially causes serious disease in a wide host range of mammals, such as measles virus in humans, rinderpest virus and peste des petits ruminants virus in ruminants, phocine distemper virus in porpoises, and canine morbillivirus (initially canine distemper virus [CDV]) in various species of carnivores. 20 These viruses preferentially infect the host cells through specific signaling molecules such as the signaling lymphocyte activation molecule 31 and nectin-4, 19 which are common cellular receptors in various mammalian hosts and so suggest the potential for cross-species infection between the different hosts.
As well as from domestic and wild canids, CDV has been identified as a primary cause of fatal illness in wild felids, including tigers, lions, and leopards, 1,10,21,24,28 while there are no reports of natural CDV infections in domestic cats despite frequent contact with dogs. 2 Thus, investigation and monitoring of morbillivirus infection in domestic felids has been widely conducted worldwide, resulting in the identification of a novel morbillivirus, namely, feline morbillivirus (FeMV), in cats in Hong Kong in 2012. 35 Since then, FeMV has been identified in cats showing tubulointerstitial nephritis and detected in urine samples from cats in various countries throughout Asia, Europe, and the Americas, 4,6,9,13,25,27,36 suggesting a widespread geographical distribution of the virus.
Even though FeMV is classified as a member of the Morbillivirus genus, it has distinct genetic and biological characteristics, such as a relative phylogenetic distinction, from other morbilliviruses and an association with kidney disease. 4,29,35 Several studies have evaluated the relationship between kidney disease and FeMV infection, but the results are inconclusive. 9,11,15,29 At present, FeMV biology, including its pathogenicity and the range of susceptible hosts, is still unclear.
Furthermore, genetic analysis of the present FeMV isolates showed a genomic diversity that supports their division into 2 genotypic lineages, FeMV-1 and FeMV-2. 26,27 The FeMV-2 lineage has been shown to infect a wider cellular target range than FeMV-1, which infects only renal epithelial cells. 26 In addition, natural recombination of FeMV-1 has been reported. 14 Together, this raises the possibility that FeMV might be more diverse in its pathogenicity to other hosts.
Due to the fact that morbilliviruses may have a wide host range, FeMV infection in wild felids, besides domestic cats, needs to be investigated to identify its origin and characterize its genetic background and so give a better understanding of FeMV evolution and pathogenicity. In this study, we identified FeMV infection associated with fatal tubulointerstitial nephritis in 2 captive black leopards (Panthera pardus) in Thailand using reverse transcriptase polymerase chain reaction (RT-PCR), immunohistochemistry (IHC), and immunofluorescence (IF) analyses of the tissues.
Materials and Methods
Cases and Samples
In November 2016, a 5-year-old male black leopard, designated as case 1, raised in The Zoological Park Organization under The Royal Patronage of H.M. The King, Bangkok, showed clinical signs of anorexia, vomiting, polyuria, and polydipsia. The clinical disease worsened with bloody diarrhea, anemia, and markedly elevated serum creatinine and urea levels. Another 6-year-old male black leopard, designated as case 2, caged separately, presented similar clinical signs at the beginning of December 2016. After supportive care and treatment, both black leopards died, in late December 2016 (case 1) and at the beginning of January 2017 (case 2).
Initially, renal failure was suggested as a cause of death of these 2 black leopards, without a specific infection or underlying diseases. They were then subjected to postmortem examinations at the Department of Pathology, Faculty of Veterinary Science, Chulalongkorn University. All vital organs including brain, heart, liver, lung, lymph nodes, kidneys, and spleen were collected and preserved in 10% neutral buffered formalin for 2 weeks and subsequently embedded in paraffin for histopathology. Microscopic evaluation was performed on hematoxylin and eosin (HE)-stained sections by the veterinary pathologist (WB). Fresh kidney tissues and urine samples were kept at −80 °C for molecular analysis of viral pathogens. Moreover, samples from 2 other Felidae species—8 tigers (Panthera tigris) and 3 fishing cats (Prionailurus viverrinus)—were also included in this study.
Virologic Investigation
Viral DNA and RNA were extracted from 200 µl of homogenized renal tissues and urine samples using the Viral Nucleic Acid Extraction Kit II (GeneAid) according to the manufacturer’s recommendation. The extracted nucleic acid was then quantified and qualified by spectrophotometric analysis with a Nanodrop Lite (Thermo Fisher Scientific Inc) and kept at −80 °C until use. Nucleic acids were subjected to routine laboratory viral investigation. One-step Pan-RT-PCR and PCR assays were performed using a separate specific primer set for each of a broad range of morbilliviruses (targeting the 155-bp fragment in the L gene of morbilliviruses), 35 parvovirus, 12 herpesvirus, 34 feline leukemia virus (FeLV), 30 feline immunodeficiency virus (FIV), 3 calicivirus, 17 and coronavirus, 7 as described previously. Samples previously known to be positive for CDV, feline parvovirus (FPV), feline herpesvirus-1 (FHV-1), FeLV, FIV, feline calicivirus (FCV), and feline coronavirus (FCoV) were used as PCR positive controls while distilled water instead of DNA template was used as a negative control.
In addition, an IHC assay to detect CDV antigens was performed on formalin-fixed paraffin-embedded (FFPE) sections of the above-mentioned tissues using a monoclonal mouse antibody against the CDV envelope protein (Monotope; ViroStat) as previously described. 32 The lung section was obtained from a CDV-infected dog as a positive control and the primary antibody was omitted as a negative control.
Molecular Analysis of the FeMV L Gene
Positive samples from the pan-morbillivirus RT-PCR were then subjected to a specific pan-paramyxovirus RT-PCR (PAR-RT-PCR), amplifying the 612-bp fragment of the L gene of paramyxoviruses, as described previously, 33 with minor modifications by changing the annealing temperature to 49.6 °C. Subsequently, PCR products of the partial L gene were run on the QIAxcel capillary electrophoresis (QCES) platform as described previously. 18 Briefly, the PCR-amplified fragments were analyzed based on high-throughput capillary electrophoresis using the QIAxcel DNA High Resolution Kit (Qiagen). A custom alignment marker of 15 to 1000 bp was simultaneously run with the samples. The QIAxcel DNA size marker (50–800 bp) was used for size estimation. The samples were analyzed using the default OM500 method at 5 kV of separation voltage, 10 seconds sample injection time, and 500 seconds separation time, using QIAxcel technology. The QCES platform automated the process of detecting and measuring the size and quantity of the PCR-amplified DNA products. The positive PAR-RT-PCR samples were further resolved by 1.5% (w/v) agarose gel electrophoresis and then purified using a NucleoSpin Extract II kit (Macherey-Nagel). The purified amplicons were submitted for commercial Sanger sequencing at Macrogen Inc.
The obtained partial FeMV L gene sequences derived from both black leopards were aligned with the FeMV sequences available in GenBank using the MEGA7 software and MAFFT version 7 (http://maf.cbrc.jp/alignment/server/). The output alignments were then used as a template for phylogenetic tree construction and pairwise nucleotide distance matrix analysis. The phylogram was constructed using the maximum likelihood (ML) method with the generalized time reversible (GTR) model, as selected by analysis in the “find-best-fit” model algorithm in the Mega 7 software according to the Bayesian information criterion (BIC). The tree was bootstrapped with 1000 replicates.
To confirm further the presence of FeMV nucleic acids in other tissues derived from the 2 black leopards, viral nucleic acids were extracted from collected FFPE tissues of both leopards as described previously, using an AllPrep DNA/RNA FFPE kit (Qiagen), following the manufacturer’s recommendations. The extracted nucleic acids were then subjected to the pan-morbillivirus RT-PCR assay as mentioned above.
Antibody Production and Analysis of FeMV-M Protein
Construction of the Recombinant (r)FEMV-M Encoding Plasmid
The full-length nucleotide sequence of the FeMV strain Thai-U16M protein was amplified from the genomic RNA by RT-PCR (accession number: MF627832) using the FeMVMatrix1011_F (5′-ATA
Next, FeMV-M was further subcloned into the pet24a(+) expression vector (Novagen), transformed into E. coli BL21 (DE3) cells (Novagen) and plated on LB agar plates containing kanamycin (50 μg/ml). After enrichment of the selected colony in kanamycin containing LB medium, the derived pet24_FeMV-M expression plasmid was verified by EcoRI and XhoI digestions and confirmed by Sanger sequencing. The rFeMV-M protein was tagged with a 6-histidine residue on the C-terminus after subcloning in the pet 24a(+) expression vector and was designated as His-rFeMV-M.
Expression and Enrichment of His-rFeMV-M
The expression of His-rFeMV-M was induced by adding isopropyl-β-D-thiogalactopyranoside (Sigma-Aldrich) into the selected transformant culture for 10 hours at 28 °C with shaking at 250 rpm. After harvesting the E. coli by centrifugation at 5000g for 15 minutes at 4 °C, the expression of His-rFeMV-M was analyzed using 12% (v/v) sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). The gels were stained with Coomassie brilliant blue to verify the presence of His-rFeMV-M at 39 kDa molecular weight (MW). Induced cells were pelleted, triple-washed with phosphate buffered saline (PBS), then lysed by sonication in an ice water bath for 30 minutes at 50% duty cycle. After centrifugation, the lysed-cell suspension was clarified by centrifugation at 10 000g for 30 minutes at 4 °C and the resulting precipitate was dissolved in 6 ml binding buffer containing 6 M urea on ice and incubated for 1 hour to dissolve the protein completely. The residual insoluble materials were discarded by centrifugation at 16 000g for 30 minutes at 4 °C, while the supernatants were harvested, filtered through a 0.45 µm membrane, and then enriched through affinity chromatography using a His Bind Kit (Novagen) according to the manufacturer’s instructions. The enriched His-rFeMV-M protein was subjected to SDS-PAGE analysis and the final concentration was determined using a Nanodrop ND-1000 spectrophotometer (Thermo Fisher Scientific).
Western Blot Analysis
The enriched (to apparent homogeneity) His-rFeMV-M protein was mixed with an equal volume of sample loading buffer, boiled at 70 °C for 10 minutes and separated on a 5% (w/v) stacking/12% (w/v) separating SDS-PAGE apparatus in a Tris–glycine buffer (0.025 M Tris base, 0.25 M glycine, 0.1% [w/v] SDS). The gel was prepared for western blotting as follows. The resolved His-rFeMV-M protein was electrically transferred onto a polyvinylidene fluoride (PVDF) membrane with a Transblot apparatus (Bio-Rad). After triple washing, the PVDF membrane with 0.1% (v/v) Tween-20 in PBS (PBST), the membrane was blocked overnight at 4 °C with PBST containing 5% (w/v) skimmed milk powder. The membrane was then incubated overnight at 37 °C with a Ni–NTA–horseradish peroxidase (HRP) conjugate (Qiagen), then washed 3 times with PBST and the final color reaction was developed with a solution of 3,3′-diaminobenzidine tetrahydrochloride (DAB) to visualize the specific protein band at 39 kDa MW (His-rFeMV-M). The total lysate of E. coli with empty vector was used as a negative control.
Production of Polyclonal Antibody Against the His-rFeMV-M Protein
A 3-month-old New Zealand White rabbit was used in this study (license number IACUC EA-010/59 (R)). Five milliliters of blood was collected from the middle ear vein of the rabbit on day 0 for preparing pre-immunized serum as a negative control and the rabbit was then inoculated by the subcutaneous route with 150 µg His-rFeMV-M in 1 ml mixed with complete Freund’s adjuvant (Sigma-Aldrich) at a 1:1 (v/v) ratio. The rabbit was then injected as above on days 14, 28, and 42, but with incomplete Freund’s adjuvant (Sigma-Aldrich) replacing the complete adjuvant. On day 56, the rabbit was humanely euthanized, and then blood was collected from the seventh to eighth left intercostal area of the heart and subjected to serum separation by centrifugation at 3000g for 15 minutes at 4 °C. The immunoglobulin G (IgG) fraction in the serum was subsequently purified using an Econo-Pac Protein A Kit (Bio-Rad) and stored at −20 °C until use. Finally, the resulting IgG polyclonal antibody against His-rFeMV-M was verified by western blotting to confirm its size and immunogenicity.
In Situ FeMV Antigen Detection by IHC and IF
The IHC was performed using the HRP-conjugated envision polymer method to confirm the presence of the FeMV antigen in the FFPE tissues. Sections of collected tissues were cut at 4 µm thickness and placed on positive slides. After deparaffinization and hydration, the slides were pretreated with distilled water and autoclaved at 121 °C for 5 minutes and then endogenous peroxidase and nonspecific activities were blocked by incubating first with 3% (v/v) hydrogen peroxide solution at room temperature for 30 minutes and then with 5% (w/v) skimmed milk powder in PBS at 37 °C for 45 minutes, respectively. Sections were subsequently incubated overnight at 4 °C with the generated rabbit polyclonal antibody against His-rFeMV-M protein at a dilution of 1:500. After triple washing, sections were incubated in the anti-rabbit Dako REAL EnVision Detection system (Dako) at room temperature for 45 minutes. Finally, after washing as above, the positive antigen–antibody reaction was detected by labelling with DAB and counterstained with Mayer’s hematoxylin before mounting. A positive signal appeared as a brown color in the infected cells.
The sets of lung, brain, lymph node, and kidney sections from a FeMV-1 infected cat and a CDV-infected dog were immunostained in parallel as positive controls. The negative control was kidney sections of case 2 incubated as above except with rabbit polyclonal antibody against herpes simplex virus type 1 (HSV-1; code B 0114, Dako) in place of that against the His-rFeMV-M protein.
To confirm further the presence of the FeMV antigen in tissues, 4-μm-thick FFPE sections of kidney and spleen of both cases were deparaffinized, rehydrated, and pretreated as mentioned above ready for IHC analyses. For antigen retrieval, the slides were enzymatically treated with 0.05% (w/v) trypsin at 37 °C for 30 minutes. The endogenous peroxidase and nonspecific activities were blocked and then incubated with the generated rabbit polyclonal antibody against His-rFeMV-M as detailed for the IHC analysis. After triple washing with PBS, sections were incubated with FITC-conjugated goat anti-rabbit IgG F(ab′)2 secondary antibody (Invitrogen) at room temperature for 1 hours. The slides were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen), mounted with a cover slip and visualized under a fluorescence microscope. A positive signal appeared as a bright green color in infected cells. As a negative control, a kidney section of case 2 was incubated with rabbit polyclonal antibody against HSV-1 (code B 0114).
Retrospective Study of FeMV in Other Wild Felids
In order to investigate the potential role of FeMV in other wild felids in the recent past, fresh-frozen tissue samples of kidneys and various organs from 8 tigers (Panthera tigris) and 3 fishing cats (Prionailurus viverrinus) dating back to 2012 were also included in this study. All animals had been raised in the same zoo as the 2 black leopards and died without specific conditions. Nucleic acid was extracted from samples and then tested with the pan-morbillivirus RT-PCR and the PAR-RT-PCR assays, as described above.
Results
Pathological Changes
Postmortem examination of both black leopards showed moderately poor body condition with evidence of dehydration and pale mucous membranes. Generally, the prominent gross findings of both black leopards were similar with moderately contracted kidneys that were pale brown, and had multifocal areas of light tan discoloration of the renal parenchyma and some areas of cortical depression. There was a greater severity of kidney abnormalities in case 2 (Fig. 1), which also had mineralization in various organs including the lungs, blood vessels, gastric mucosa, tongue and muscles.
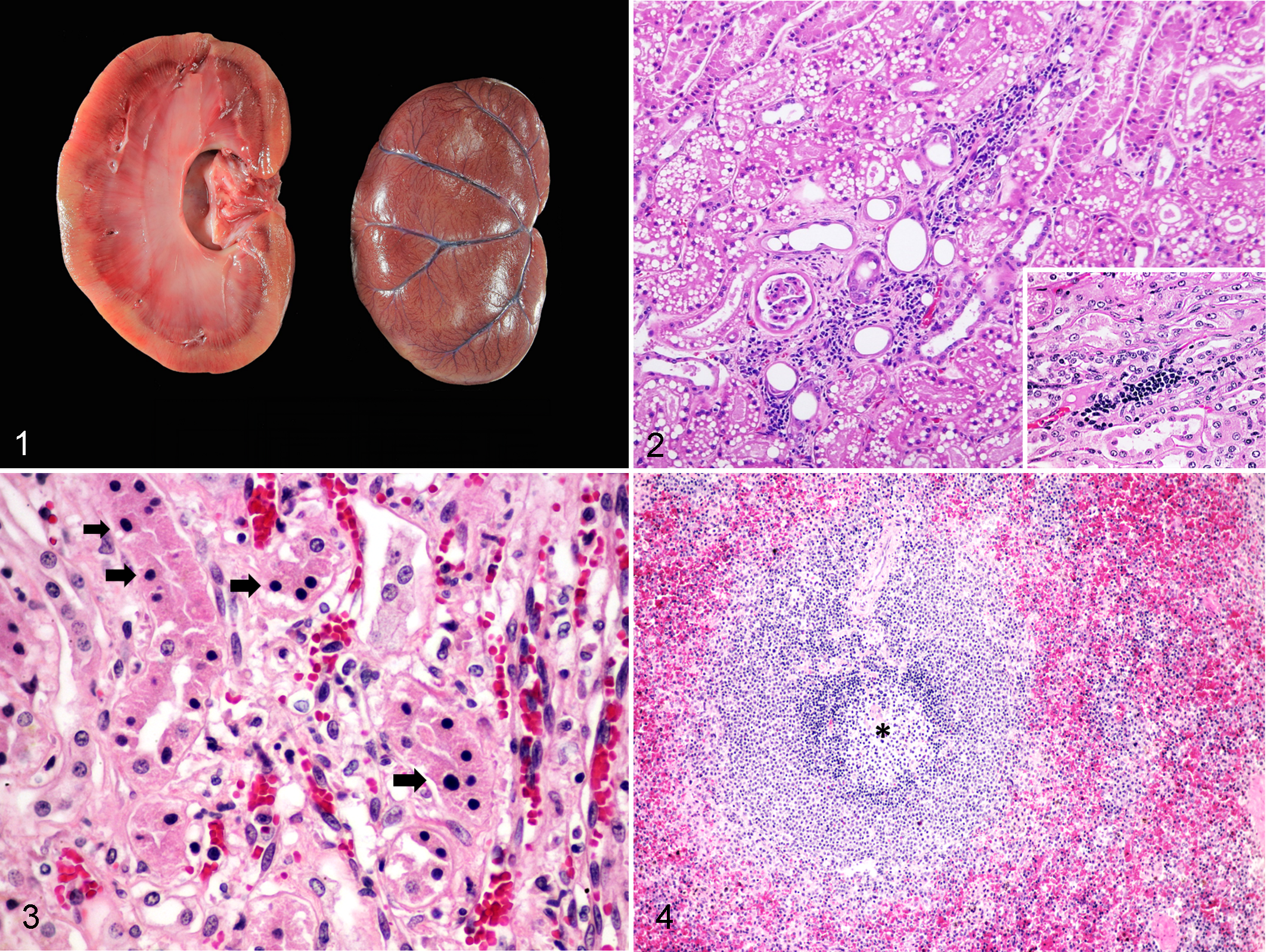
Feline morbillivirus infection, kidney, black leopard, case 2.
The histological findings were broadly similar among the 2 black leopards with moderate infiltration of lymphocytes and plasma cells in the renal tubular interstitium (Fig. 2). Severe diffuse tubular necrosis and moderate renal tubular degeneration were also observed (Fig. 3). These findings were interpreted as chronic tubulointerstitial nephritis, although the severity varied between the 2 black leopards. Moderate lymphoid depletion, severe splenic congestion, and moderate histiocytic and plasmacytic infiltration were noted in case 2 (Fig. 4), while case 1 had less severe lesions. The other examined organs were histologically normal.
Virology Investigation and Identification of FeMV
To identify potential viral causative agents, extracted viral nucleic acids from the urine and kidney samples of these 2 leopards were screened using several pan-viral PCRs for morbillivirus, parvovirus, herpesvirus, FeLV, FIV, calicivirus, and coronavirus, with the same FFPE tissues used for IHC detection of CDV antigen. While the samples showed negative results for all the other tests, the pan-morbillivirus RT-PCR was positive. The 155-bp amplicon obtained from the pan-morbillivirus RT-PCR was sequenced and aligned to the homologous sequences obtained from GenBank (BLASTn search), which showed an approximately 98% RNA (as cDNA) sequence similarity to FeMV-annotated sequences. This prompted us to investigate the presence of FeMV nucleic acid in other extracted FFPE tissues using the pan-morbillivirus RT-PCR. The 155-bp specific FeMV amplicon was detected in the kidneys of both black leopards and in the spleen of black leopard case 2, but the other tissues were negative including brain, heart, liver, lung, and lymph node.
Molecular Analysis of the FeMV L Gene
Since kidney samples of both black leopards showed positive results in the pan-morbillivirus RT-PCR, the presence of FeMV nucleic acid was confirmed using pan-specific RT-PCR for paramyxovirus, targeting the partial L gene of FeMV. The 612-bp FeMV L fragment was amplified, sequenced, and found to share 98.9% genetic similarity among leopard samples and 98.2% genetic similarity to the FeMV-1 strain CTL43 (accession number MN164532) derived from the urine of a domestic cat in Thailand (Supplemental Figs. S1 and S2). Due to the limitations of fresh tissue samples, we were not able to evaluate viral replication in cell culture nor to amplify the whole genome of the black leopard FeMV. The partial genome sequences of black leopard FeMV were deposited in GenBank with accession numbers MN295672 and MN295673 for cases 1 and 2, respectively. The ML-based phylogenetic analysis of the FeMV L gene showed the black leopard FeMVs grouped in the FeMV-1 genotype and in the Asian FeMV-1 cluster, forming a monophyletic branch close to FeMV strains CTL43, CTL16, and U16, all from Thailand (Fig. 5).

Phylogeny of feline morbillivirus genome sequences compared with reference FeMV viruses in the NCBI database. Tree construction was performed by ML using the GTR nucleotide substitution, invariable sites and gamma distribution models (GTR + G + I) for tree construction, as determined by BIC, with 1000 bootstrapping replicates. Bootstrap values (%) are shown at each node. GenBank accession numbers are shown on the tree. The FeMV genomes detected in the leopard case 1 (ZPO1) and 2 (ZPO2) are indicated by red triangles. The bar indicates the estimated numbers of nucleotide substitutions per site.
Tissue Localization of FeMV
The presence of the FeMV antigen in the brain, heart, liver, lung, lymph node, kidney, and spleen sections was evaluated by IHC analysis using a newly developed rabbit polyclonal antibody against rFeMV-M. The FeMV antigen showed diffuse and strong signals in the cytoplasm of the renal tubular epithelial cells in both cases (Fig. 6) and in infiltrating lymphocytes in conjunction with tubulointerstitial nephritis of case 2 (Fig. 7). Furthermore, the FeMV antigen was strongly labelled in the cytoplasm of infiltrating lymphocytes and histiocytes in the spleen of case 2, while case 1 showed weak staining. No FeMV antigen was detected in the other tissue sections examined (ie, brain, heart, liver, lung, lymph node). To confirm the positive results of the IHC, IF analysis was additionally performed on the kidney and spleen sections using the same polyclonal antibody against rFeMV-M. Strong rFeMV-M signals were evident in the cytoplasm of the renal tubular epithelial cells, similar to that stained with IHC (Fig. 8), while the signals were diffusely labelled in mononuclear cells infiltrating the red pulp of the spleen (Fig. 9).
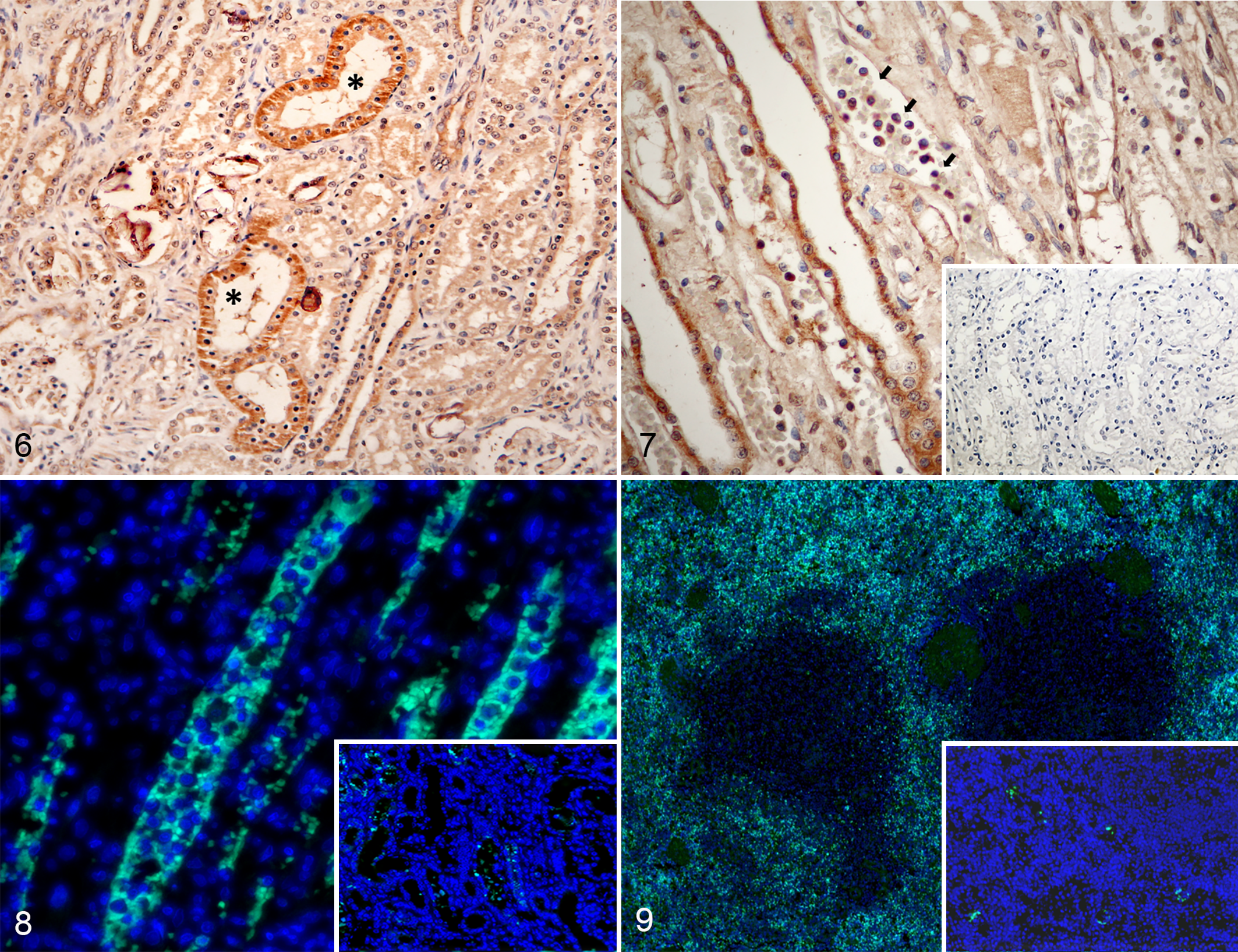
Feline morbillivirus infection, kidney, black leopard.
Retrospective Study of FeMV in Other Wild Felids
Extracted viral nucleic acids from fresh frozen tissues derived from 8 captive tigers and 3 fishing cats were tested with the pan-morbillivirus and PAR-RT-PCR assays. There was no evidence of FeMV nucleic acids in those samples.
Discussion
In recent years, FeMV isolates have been discovered in domestic cats in association with renal disease. 35 It has been proposed that FeMV might be a cause of renal disease in domestic cats but this is yet to be confirmed due to inconsistent results in recent publications, 4,15,36 the evidence of FeMV localization in renal tubular epithelial cells in cats with renal disease warrants further clarification as to whether FeMV isolates are associated with the disease. The CDV, as the FeMV counterpart, has been reported as a pathogen causing fatal outbreaks in various carnivores including wild felids, 8,16 supporting the possible role of morbillivirus infection across a diverse range of hosts. Thus, we were interested in investigating whether FeMV was associated with disease in wild felids.
In this study, we describe the detection and characterization of FeMV, which was detected in the urine, kidney, and spleen samples of 2 black leopards showing evidence of tubulointerstitial nephritis. Although the postmortem examinations revealed evidence of renal failure without specific infection, both the genetic detection, using several pan-conventional PCRs in combination with genetic sequencing, and IHC analyses of the tissue samples of both black leopards revealed no other viruses except for FeMV. Moreover, the FeMV antigen was also found in the kidney and spleen FFPE tissues by PCR, IHC, and IF analyses.
Since the black leopard FeMVs were genetically similar to the FeMV isolated from cats in Thailand, the possible role of FeMV transmission between domestic cats and leopards should be considered and needs further investigation. In this study, the whole genome of the black leopard FeMV was not determined because of the limited amount of sample. The identification of FeMV antigen in the tissues supports an association of FeMV infection with tubulointerstitial nephritis. A retrospective study of FeMV nucleic acid detection in tissue samples of 2 other wild felid species showed no evidence of FeMV infection, suggesting that FeMV infection is not common in these species. However, there were no FFPE sections available from these animals to examine the kidney lesions and only a relatively small number of animals were tested and from different temporal periods and different species. Therefore, definitive conclusions on the association of FeMV infection and tubulointerstitial nephritis in black leopards cannot be drawn.
To understand the role of FeMV-associated renal disease in these black leopards, viral antigen was identified in tissues with histological lesions of fatal tubulointerstitial nephritis. Here, the FeMV antigens were generally localized in the cytoplasm of renal tubular epithelial cells and in the mononuclear cells that had infiltrated the renal interstitial lesions. Thus, this study demonstrated FeMV localization in the tubular epithelial cells, where the most pathological changes (ie, tubular degeneration) were observed. This finding is similar to those in FeMV-infected domestic cats. 15,29 Furthermore, FeMV antigen was identified in the lymphocytes, which corresponds to the lymphotropic nature of other morbilliviruses and evidence from in vitro studies of FeMV infection. 5,22 However, the incongruous results for FeMV identification using PCR and IHC of the spleen of case 1 may result from low amounts of extracted FeMV-1 RNA from FFPE tissue that can impact subsequent ancillary diagnostics.
Recently, CDV has been detected in large felids and associated with fatal diseases in large groups of felines. 1,21,24,28 The discovery of FeMV in 2 black leopards, which is listed as a vulnerable species by the International Union for Conservation of Nature Red List raises concerns about a new, potentially fatal disease that can develop in other endangered Felidae species. The transmission and pathogenesis of FeMV infection in leopards and other nondomestic cats warrants further investigation.
In this study, we identified the presence of FeMV at postmortem in 2 black leopards in Thailand that showed clinical evidence of chronic kidney disease. This is the first report of FeMV in a nondomestic felid species. Additional testing to further characterize the virus and determine if it is identical to FeMV-1 is needed. The molecular identification of FeMV localization in tissues was confirmed by IHC and IF analyses, in conjunction with pathological changes in the kidney and spleen, suggesting a possible role of FeMV in this disease. A causative association between FeMV and fatal tubulointerstitial nephritis in black leopards could not be determined but remains possible.
Supplemental Material
Combined_supplemental_materials-Piewbang_et_al - Feline Morbillivirus Infection Associated With Tubulointerstitial Nephritis in Black Leopards (Panthera pardus)
Combined_supplemental_materials-Piewbang_et_al for Feline Morbillivirus Infection Associated With Tubulointerstitial Nephritis in Black Leopards (Panthera pardus) by Chutchai Piewbang, Surangkanang Chaiyasak, Piyaporn Kongmakee, Saowaphang Sanannu, Pornsuda Khotapat, Jadsada Ratthanophart, Wijit Banlunara and Somporn Techangamsuwan in Veterinary Pathology
Footnotes
Acknowledgements
Chutchai Piewbang was funded by a grant from the Ratchadaphisek Somphot Fund for Postdoctoral Fellowship, Chulalongkorn University. Surangkanang Chaiyasak was supported by the 100th Anniversary Chulalongkorn University Fund for Doctoral Scholarship. We are grateful to Asst Prof Dr Theerawat Tharasanit and Mr Rattapoom Thaiwong for technical assistance. The Veterinary Pathogen Bank and Animal Virome and Diagnostic Development Research Group, Faculty of Veterinary Science, Chulalongkorn University, is also acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the 90th Anniversary of Chulalongkorn University Fund (Ratchadaphisek Somphot Endowment Fund) to SC; the Thailand Research Fund (RSA6180034) to ST; a grant for joint funding of external research projects; the Ratchadaphisek Somphot Endowment Fund and Veterinary Science Research Fund (RES_61_364_31_037) and funding from Chulalongkorn University to ST.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
