Abstract
Previous work demonstrated renal fibrosis 70 days after a single unilateral in vivo renal ischemic event, but changes associated with a single episode of renal ischemia past this time are unknown. In this study, we evaluated renal function and structural changes 6 months after a 90-minute in vivo unilateral renal ischemic event. Six adult female cats underwent unilateral renal ischemia and renal function was followed for 6 months, at which time the kidneys were evaluated by histology and histomorphometry. Over time, there was a significant reduction in the glomerular filtration rate and an elevation of serum creatinine of 31% and 42%, respectively. All cats had tubulointerstitial lesions characterized by segmental interstitial inflammation, tubular atrophy, and interstitial fibrosis. Unlike short-term studies, ischemic kidneys had variable numbers of obsolescent glomeruli, consistent with the development of atubular glomeruli and subsequent ischemic glomerulosclerosis. Chronic changes associated with acute renal ischemia may include loss of function and glomerulosclerosis.
Establishing a model of chronic kidney disease (CKD) in cats is critically important to further our understanding of the initiation and progression of this disorder and to support preclinical trials of potential therapeutics. While clinical cases of CKD are available and should be studied, continued effort into accurately modeling CKD in the target species is also critically important. The typical logistical challenges in studying clinical CKD cases include slow, intermittent disease progression; variable owner compliance; and the significant influence of comorbid disease. However, more important, identifying and understanding the initiates of spontaneous CKD is difficult in a clinical population, as most cats present in International Renal Interest Society (IRIS) Stage 2–4, which is relatively advanced chronic disease and well beyond the initiation phase. Further, as the process appears to involve pathological changes that seem to be most severe in the deep cortex and medulla, outer cortical biopsies from cats with spontaneous CKD are unlikely to be fully illustrative. Deeper renal biopsies are dangerous and difficult to justify in these patients, particularly in the early IRIS CKD stages. Use of cats as models for feline disease is important as feline CKD is uniquely different from the proteinuric, glomerulocentric CKD typically observed in inbred strains of rodents, dogs, and people, so there is not a viable in vivo model in other species. Investigations in reductionist cell culture or co-culture systems is not currently possible, as CKD is a complex process, and understanding the complex interactions between the vasculature, interstitial cells, and nephrons is currently not possible in a cell culture or co-culture system.
We have previously demonstrated that a single episode of unilateral renal ischemia induces tubular atrophy, interstitial fibrosis, and chronic inflammation in cats, findings that support the hypothesis that renal ischemia plays a role in the pathogenesis of naturally occurring CKD in cats. 2,12 To investigate the chronic changes associated with this model and its potential to induce persistent lesions consistent with CKD, we evaluated the structural and functional changes in the kidney of cats 6 months after a 90-minute in vivo unilateral renal ischemic event. This study protocol was approved by the University of Georgia Institutional Animal Care and Use Committee (Animal Use Protocol A2015 10-043-Y2-A5). Six purpose-bred young adult (age 336 ± 18 days) female intact cats were anesthetized and underwent right renal artery and vein occlusion (day 0). Cats were fasted 12 hours prior to anesthesia; sedated with intramuscular acepromazine (0.01 mg/kg), buprenorphine (0.04 mg/kg), and ketamine (7 mg/kg); and induced and maintained on isoflurane gas and 100% oxygen delivered via facemask. Cats were moved into an adjacent, sterile surgery suite after induction and received intravenous isotonic crystalloid fluids (0.9% NaCl) at 10 mL/kg/h throughout the procedure. Body temperature was supported with a hot water blanket.
Other than the increased duration of renal ischemia, surgical procedures were identical to what has been previously described. 12 The contralateral kidney was not disturbed during the ischemic event. Postoperatively, a 25 µg/h transdermal fentanyl patch (Fentanyl Transdermal System; Noven Pharmaceuticals Inc, Weston, Florida, USA) was placed on the lateral abdomen; in cats less than 5 kg, half of the patch was folded on itself to allow only half of the absorptive area to contact the skin. Based on a discomfort score 12 assigned during monitoring, postoperative analgesia was augmented via transmucosal buprenorphine (0.03 mg/kg) as indicated in the 24 hours following fentanyl patch application.
Serum urea nitrogen (SUN) and creatinine concentrations and urine-specific gravity (USG) were assessed at days –5 (baseline), 1, 3, 7, and 30 and then monthly for 6 months. Blood pressure was determined as the average of 3 consecutive measurements obtained by Doppler sphygmomanometry on the right front leg with a #2 cuff on days –5 and 7 and then monthly for 6 months (Model 811-B; Parks Medical Electronics, Aloha, Oregon, USA). Glomerular filtration rate was estimated by iohexol clearance at baseline and 3 and 6 months, as previously described. 12 Cats were fed an adult maintenance diet daily and weighed weekly. Water was provided ad libitum.
Cats were euthanized by intravenous injection of sodium pentobarbital at 180 days after renal ischemia, and the kidneys were measured and weighed. Samples of ischemic (IK) and contralateral nonischemic kidney (CK) were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, cut at 3 μm, and stained with hematoxylin and eosin (HE), periodic acid-Schiff and hematoxylin, and picrosirius red. Histology and histomorphometry evaluation was performed without knowledge of tissue source on 10 consecutive 200× fields per kidney. The number of fields having free interstitial lipid in areas of inflammation, reflecting past tubular rupture, was determined. 12 The severity of interstitial inflammation and tubular atrophy was evaluated by 2 pathologists (CAB and DRR). Interstitial inflammation was scored as 0 (no inflammatory cells), 1 (mild with <10% of interstitium affected; Fig. 1), 2 (moderate with 10-50% of interstitium affected; Fig. 2), and 3 (severe with >50% of interstitium affected; Fig. 3). Tubular atrophy was scored as 0 (no tubular atrophy), 1 (mild with <10 scattered atrophic tubules; Fig. 4), 2 (moderate with a single linear streak of tubular atrophy; Fig. 5), and 3 (severe with 2 or more streaks of tubular atrophy; Fig. 6). The total number of glomeruli in ten 200× fields (glomerular density, including obsolescent glomeruli) and the percentage of glomeruli having periglomerular sclerosis or global sclerosis (glomerular obsolescence) were determined. The planar areas of 20 superficial nonsclerotic glomerular capillary tufts per kidney were measured (Olympus cellSens Entry 1.15; Olympus Corporation, Center Valley, Pennsylvania, USA), and the volume was calculated as previously described. 3
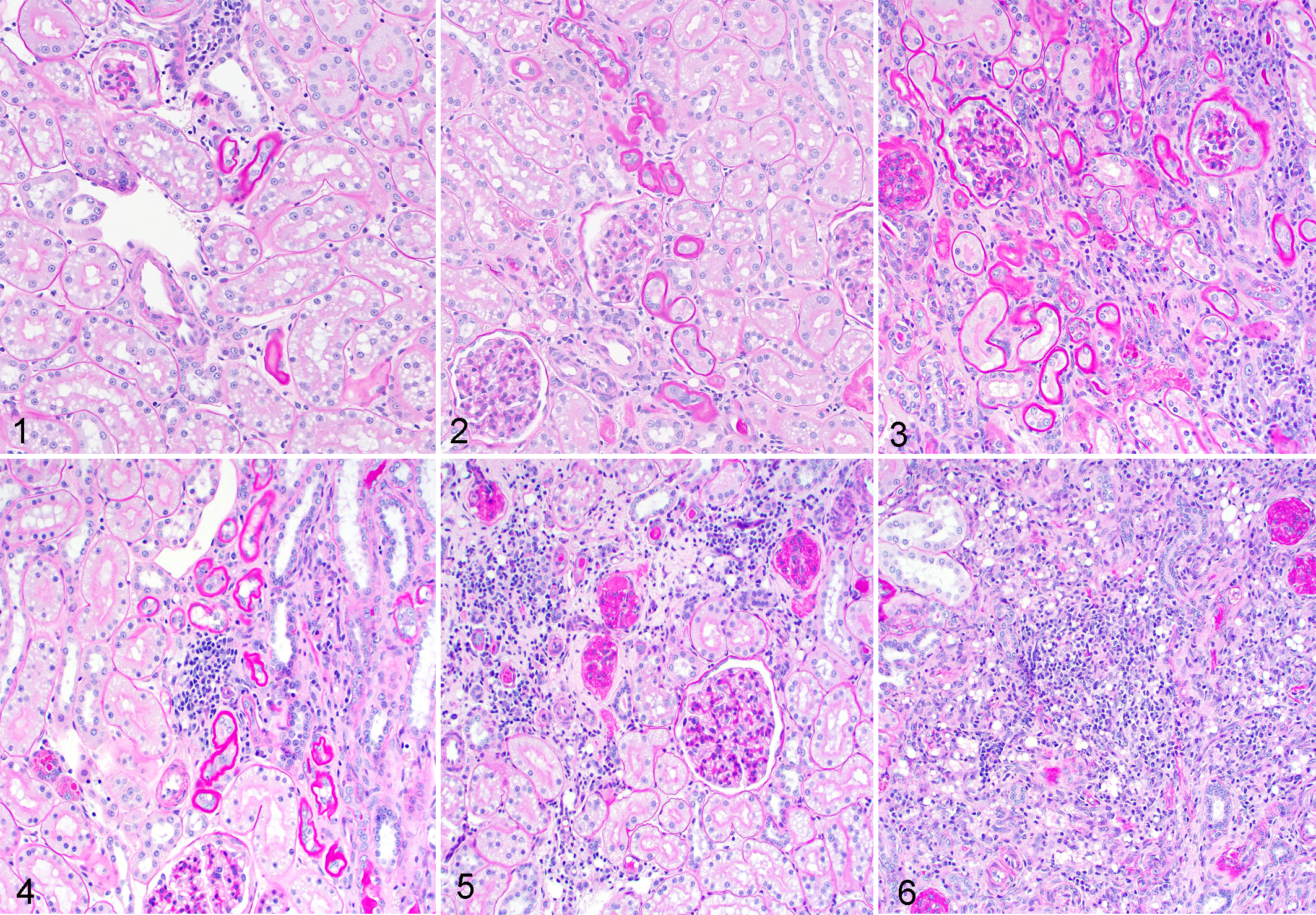
Severity scores (1–3) for cortical tubular atrophy, kidney, cat. Periodic acid-Schiff and hematoxylin.
Picrosirius red staining under polarized light 9 and immunohistochemistry (IHC) for smooth muscle actin (SMA) were used to quantify interstitial collagen and myofibroblasts, respectively. For SMA immunohistochemistry, the primary antibody was mouse monoclonal SMA (1A4) antibody (202M-98; Cell Marque, Rocklin, California, USA). The antibody was provided as a ready-to-use reagent with a staining time of 60 minutes. The primary antibody was diluted (S0809 Antibody Diluent; Dako, Carpinteria, California, USA). Antigen retrieval was performed with heat-induced epitope retrieval via citrate buffer with a pH of 6.0 (HK086-9 K; Biogenex, San Ramon, California, USA). Endogenous peroxidase was quenched with 3% hydrogen peroxide (H312-500; Fisher Scientific, Fair Lawn, New Jersey, USA). All other blocking was completed with a commercially available solution (Power Block, HK085-5 K; Biogenex). Positive tissue controls consisted of formalin-fixed, paraffin-embedded canine and feline intestine. As a negative reagent control, the primary antibody was eliminated and substituted with purified mouse immunoglobulins in buffer (NC494 H; Biocare Medical LLC, Concord, California, USA). The secondary antibody used was biotinylated equine anti-mouse IgG, rat absorbed (BA-2001; Vector Labs, Burlingame, California, USA). A streptavidin-based label was used for detection (Biocare 4Plus Streptavidin HRP Label, HP604 H; Biocare Medical LLC). The substrate chromogen system used was DAB (K3466; Dako). The tissue sections were counterstained with Gills II hematoxylin and bluing; dehydrated in alcohol levels of 70%, 95%, and 100%; cleared in xylene; and mounted with xylene-based mounting medium.
Images were captured from ten 200× fields in the cortex, corticomedullary junction, and medulla, and image analysis was performed using commercial image analysis software (Adobe Photoshop, CC 2015.5; Adobe Systems, Inc, San Jose, California, USA). Areas of positive staining were selected, and the number of positive-staining pixels were quantified and expressed as a percentage of all pixels on the image. Descriptive statistics were reported as mean and standard error of the mean, or as median and range. Normality was determined by the Kolmogorov-Smirnov test. Renal functional parameters (serum creatinine concentration, SUN, USG, urine protein creatinine ratio, and glomerular filtration rate [GFR]), as well as body weight and blood pressure, were evaluated as a change from baseline assessed with a repeated-measures analysis of variance and Dunnett’s multiple comparison test. For histological scoring, histomorphometry percentages, kidney weight, and kidney volume, a 2-tailed, unpaired Student t-test or Mann-Whitney test was used for comparison of scores from ischemic samples to contralateral samples. In samples where one kidney received only scores of 0, a Wilcoxon signed rank test was used to evaluate for a significant difference from a hypothetical value set at 0. Statistical significance was taken as P < .05. Statistics were completed using a commercially available software package (GraphPad Prism v. 5.00 for Windows; GraphPad Software, San Diego, California, USA).
All cats survived surgery and the 6-month post–renal ischemia period without complications. Cats gained a significant amount of weight over the duration of the study (baseline = 3.1 ± 0.2 kg, day 180 = 4.6 ± 0.2 kg; P < .0001, Supplemental Table S1). Parameters of renal function are summarized in Table 1. While several cats had minimal changes of renal function (Table 2, Supplemental Tables S2–S5), as a group renal function declined over time, as evidenced by a significant reduction in GFR of 31% compared to baseline and a significant elevation in serum creatinine concentration of 42% above baseline.
Renal Function Parameters Following 90 Minutes of Unilateral In Vivo Renal Ischemia in 6 Cats.a

Abbreviations: BL, baseline value obtained 6 days prior to ischemic injury; BP, Doppler blood pressure; GFR, glomerular filtration rate estimated by iohexol clearance; n/d, not done; SCr, serum creatinine concentration; SUN, serum urea; UP:C, urine protein creatinine ratio; USG, urine-specific gravity.
aFor each day, the mean ± standard error of the mean and (range) are given; n = 6.
b P < .05 compared with baseline values.
c P < .01 compared with baseline values.
d P < .001 compared with baseline values.
Renal Cortical Histopathologic Findings 6 Months After a 90-Minute Unilateral Kidney Ischemic Injury.a

Abbreviations: glom density, total number of glomeruli present; GS, global glomerulosclerosis (% of glomeruli); IC, interstitial collagen in cortex; IS, interstitial inflammation score (0–3 scale); lipid, numbers of fields with interstitial lipid; mean glom vol, mean glomerular volume of nonobsolescent glomeruli; PeriGS, nonobsolescent glomeruli with periglomerular sclerosis (% of glomeruli); TAS, mean tubular atrophy score (0–3 scale).
aData presented are mean values from 10 consecutive 20X fields from the ischemic kidney (IK)/contralateral kidney (CK).
IKs were significantly lighter (mean ± SEM IK weight = 10.7 ± 1.7 g, CK weight = 15.2 ± 1.1 g; P = .05) and smaller (mean ± SEM IK volume = 14.2 ± 2.4 cm3, CK volume = 23.7 ± 2.8 cm3; P < .05) compared to CKs (Supplemental Table S6). All 6 cats had tubulointerstitial lesions characterized by interstitial inflammation, tubular atrophy, and interstitial fibrosis as previously described at 70 days post–renal ischemia and in cats with naturally occurring CKD. 2,11,12 Interstitial inflammation and tubular atrophy were mild to moderate (Table 2), with significantly higher scores for mean inflammation and tubular atrophy in the IK compared to the CK (P < .01 and .05, respectively). In all cats, the lesions were segmental, such that fields with tubular atrophy, inflammation, and fibrosis were interspersed with fields with no or minimal lesions. Four cats (Nos. 2, 3, 4, and 6) had prominent lipid vacuoles, indicative of prior ischemic tubulorrhexis, in areas of cortical inflammation (Table 2, Fig. 7). Cortical interstitial fibrosis, measured as polarizable fibrillar collagen in picrosirius red–stained sections (Fig. 8), was present in all IKs. The median percentage of positive picrosirius red staining was significantly greater in the cortex of the IK compared to the CK (IK cortex = 5.21%, range = 2.60%–24.13%; CK cortex = 1.89%, range = 1.21%–2.82%; P = .004) (Fig. 9). Significant differences in picrosirius red staining were not observed in the corticomedullary junction (CMJ) or medulla (mean ± SEM % positive staining in the IK CMJ = 7.37% ± 1.6%, CK CMJ = 3.75% ± 1.9%; P = .08; IK medulla = 9.40% ± 1.4%, CK medulla = 7.53% ± 2.1%; P = .48) (Supplemental Table S7). SMA staining, as a marker of myofibroblasts and a measure of the profibrotic state, was significantly greater in IK CMJ compared to the CK CMJ (IK CMJ = 3.25% ± 0.52% positive, CK CMJ = 1.68% ± 0.12% positive; P < .05) (Fig. 10), but no significant differences were noted between the IK and CK cortex (IK cortex = 1.72% ± 0.43%, CK cortex = 1.09% ± 0.15%; P = .20) or medulla (IK medulla = 2.41% ± 0.23%, CK medulla = 1.99% ± 0.23%; P = .22) (Supplemental Table S8). Despite increased inflammation and interstitial collagen in the IK, glomerular density was increased compared to the CK in 5 of 6 cats (Table 2), as net loss of intervening parenchyma brought glomeruli closer together (Fig. 7).
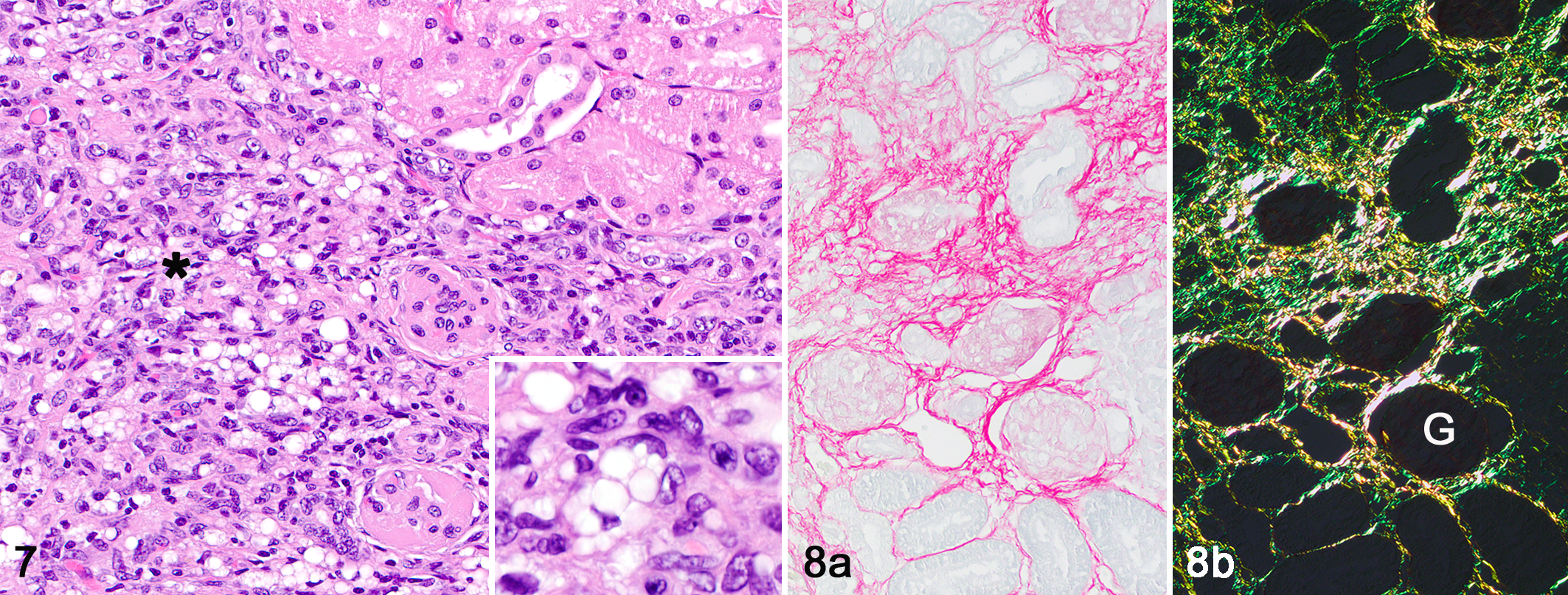
Interstitial lesions present 6 months after unilateral transient renal ischemia, kidney, cat.
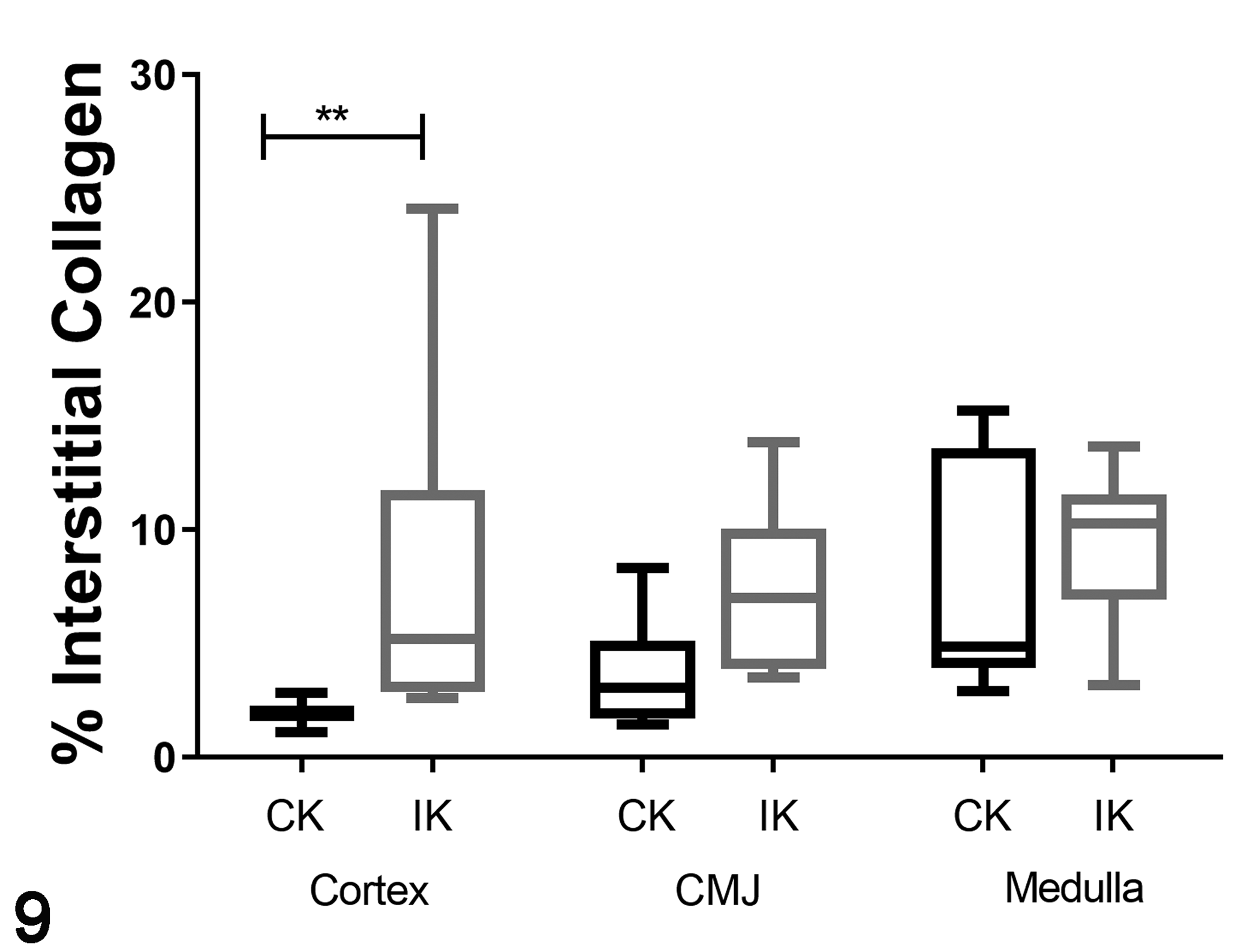
Box-whisker plots of interstitial collagen staining (Fig 9) and smooth muscle actin immunolabeling (Fig 10) of tissue from 6 cats at 6 months following 90 minutes of unilateral renal ischemia. The boxes extend from the 25th to 75th percentiles and the horizontal line is the median. The whiskers are the range of the data. The previously ischemic kidney had more cortical staining for interstitial collagen (Fig. 9; **P < .01) and immunolabeling for smooth muscle actin (Fig. 10; *P < .05.) than the contralateral control kidney. CK, contralateral kidney; CMJ, corticomedullary junction; IK, ischemic kidney.
Glomerular lesions, not present in previous studies of this model at 70 days postischemia, 12 were present in all cats at 6 months (Table 2). Periglomerular fibrosis, which has been associated with atubular glomeruli or glomeruli in which the connection is stenotic and the tubule is atrophic, 8 was present in 5 of 6 cats (Fig. 11). Proliferation of parietal and proximal epithelial cells was occasionally observed in glomeruli with periglomerular sclerosis or atrophic connecting tubules (Fig. 12). As filtration does not occur in atubular glomeruli, these glomeruli are considered nonfunctional. 8 In the IKs, 2% to 82% of glomeruli were obsolescent; glomerular obsolescence was not present in the CKs. Obsolescent glomeruli were small, with wrinkling of the glomerular basement membrane, collapse of the capillary tuft, and thickening of Bowman’s capsule, consistent with ischemic glomerulosclerosis (Fig. 13). 6
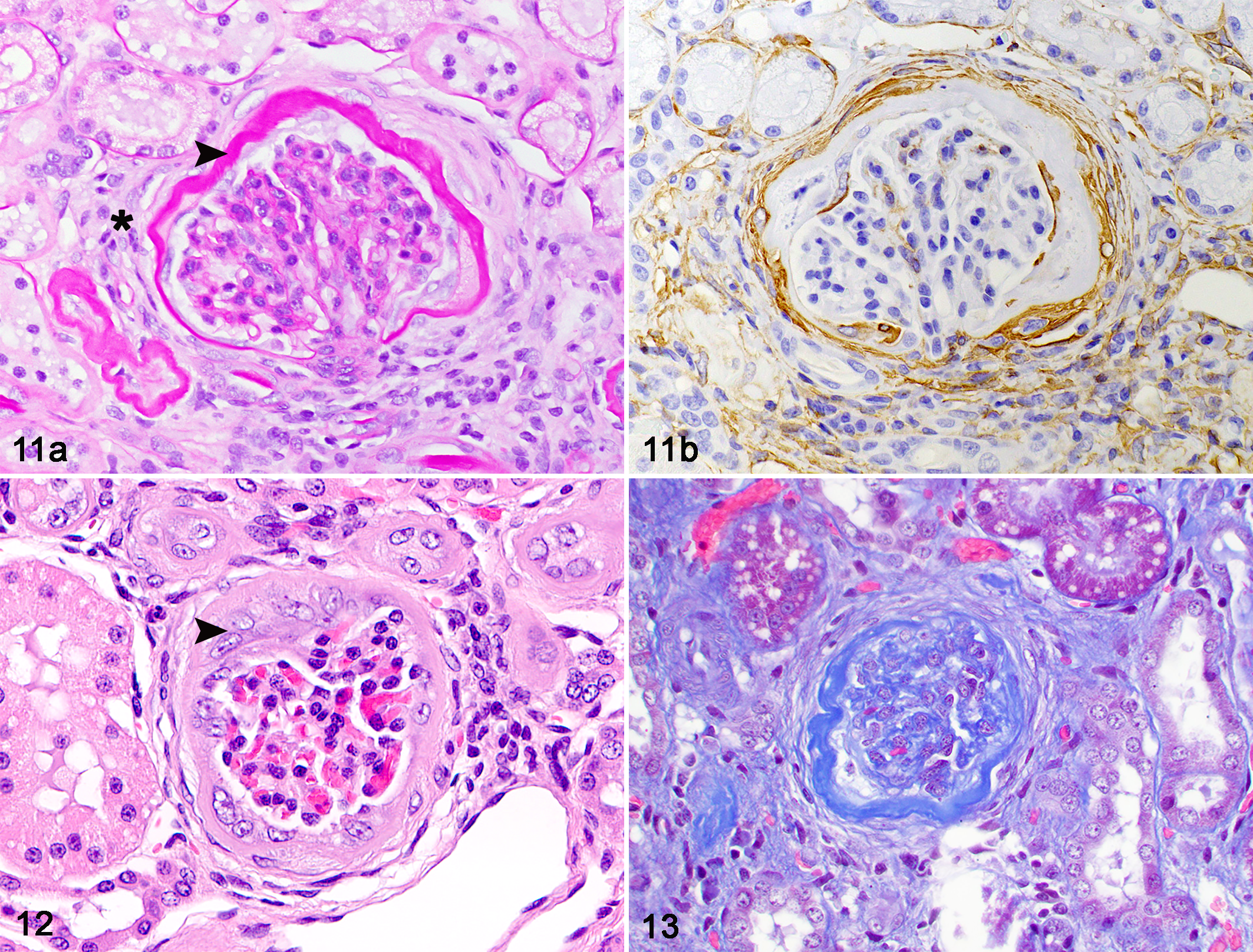
Periglomerular sclerosis and glomerular obsolescence at 6 months after unilateral renal ischemia, kidney, cat.
The intercat variability in lesion severity was marked, as has been described in a previous study in which renal ischemia was of shorter duration (60 minutes vs 90 minutes in the present study). 12 While the ischemic insult caused acute tubular necrosis of similar severity in all cats in that study, later time periods were associated with variable severity of chronic lesions and possible resolution of lesions in some cats. These observations suggest that some cats were able to maintain intact tubules and undergo epithelial regeneration, while others were unable to preserve tubular structure following ischemia, leading to atubular glomeruli that progressed to obsolescence. The cats with the highest percentage of sclerotic glomeruli in the present study also had extensive tubular rupture and dropout, evidenced by widespread interstitial lipid 10 and increased glomerular density. In contrast, cats with no or minimal interstitial lipid had low numbers of obsolescent glomeruli and lower glomerular density, indicative of tubular preservation.
Multiple episodes of injury or a single episode of severe ischemia, as in these cats, may result in CKD. In a mouse model of toxic proximal tubular injury, mild injury causes reversible fibrosis while severe injury or repeated mild injury leads to interstitial fibrosis, inflammation, glomerulosclerosis, and atubular glomeruli. 13 Severe or recurrent proximal tubular injury with inadequate repair are likely the key cellular mechanisms in the progression to CKD. 13 Our findings suggest that the obsolescent glomeruli occurring in our renal ischemia model are the result of inadequate tubular repair, subsequent tubular loss, and the development of atubular glomeruli, which subsequently become obsolescent. Ischemic changes of thickening of Bowman’s capsule, periglomerular sclerosis, and collapse of the glomerular capillary tufts were present in all cats, indicating that renal ischemia can lead to CKD in this species. A similar ischemic pathogenesis likely occurs in cats with naturally occurring CKD 2,4 and has been proposed as the cause of Mesoamerican nephropathy in humans. 14 A CKD affecting rural inhabitants of Central America, Mesoamerican nephropathy is characterized by lesions of tubulointerstitial inflammation and fibrosis, glomerulosclerosis, and chronic glomerular ischemia similar to those described in our model and in cats with naturally occurring CKD. Mesoamerican nephropathy appears to be caused by recurrent renal ischemia due to occupational heat stress and salt depletion. 14
In humans, it has long been argued that the progressive decline in renal function in CKD is caused by chronic glomerular injury with secondary tubular ischemia. 1 Global glomerulosclerosis (obsolescent glomeruli) develops as the end stage of focal segmental glomerulosclerosis following compensatory glomerular hypertrophy and hyperfunction. 7 However, the importance of injury to the proximal tubule in the progression of CKD recently has been recognized. 5,13 The development of atubular glomeruli in humans with end-stage renal disease, characterized by interstitial fibrosis and tubular atrophy, contributes to progression of CKD. 15 In feline CKD, in which glomerulosclerosis is secondary to tubular loss, preservation of renal function may be dependent on tubular repair mechanisms. We have observed substantial interanimal variability in the severity of glomerulosclerosis that developed in cats following identical ischemic insults. It is interesting to note that the rate of progression of spontaneous feline CKD is similarly variable. 5 It will be important to determine if variability in tubular repair mechanisms contributes to these interanimal differences or if therapies that minimize tubular injury slow progression.
Limitations to the model include the relatively high variability of lesions, the low number of cats, inaccuracy of available biomarkers of renal function, and the confounding effect of the CK on assessment of global renal function. The CK substantially reduces morbidity associated with renal ischemia, but chronic changes may be masked by the presence and hypertrophy of the CK. A single acute renal ischemic event can result in chronic and variable loss of renal function and segmental, variable interstitial inflammation, tubular atrophy, interstitial fibrosis, and glomerulosclerosis in cats. This model may be useful in further evaluating the role of renal ischemia in the pathogenesis and treatment of feline CKD.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819837721 - Chronic Renal Changes After a Single Ischemic Event in an Experimental Model of Feline Chronic Kidney Disease
Supplemental Material, DS1_VET_10.1177_0300985819837721 for Chronic Renal Changes After a Single Ischemic Event in an Experimental Model of Feline Chronic Kidney Disease by Cathy A. Brown, Daniel R. Rissi, Vanna M. Dickerson, Anastacia M. Davis, Scott A. Brown and Chad W. Schmiedt in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
