Abstract
Objectives
The aim of this study was to describe the endoscopic appearance of gastroduodenal ulcers (GDUs), and to assess the clinical, ultrasonographic and histological data, as well as long-term follow-up, in cats.
Methods
The medical record databases of five veterinary endoscopists were evaluated between January 2016 and 2020, in a retrospective study. Cats with at least one gastric or duodenal ulcer detected by endoscopic examination were included. All the medical records of the selected cats were reviewed and information was collected regarding breed, age, sex, neuter status, medical history, clinical signs, and ultrasonographic, endoscopic and histological findings. The cats were evaluated at 6, 12 and 18 months.
Results
Sixty-one cats with a median age of 9.0 years (range 2.0–16.0) were included in the study. The most common complaints were vomiting (n = 55; 90%) and hyporexia (n = 40; 66%); haematemesis was reported in 12 (20%) cats. Endoscopy showed GDUs in the following locations: gastric body in 28 cats (46%), antropyloric area in 34 cats (56%), fundus in 13 cats (21%) and duodenum in eight cats (13%). A single GDU was found in 42 cats (69%) and multiple GDUs were seen in 19 cats (31%). Histopathological evaluation revealed benign lesions in 33 (54%) cats and malignant lesions in 28 (46%; 24 high-grade lymphoma, one low-grade lymphoma and three carcinoma). High-grade lymphoma was detected only in the stomach. Cats diagnosed with malignant GDUs (median 10.5, range 4–16) were significantly older than cats with benign lesions (P = 0.002).
Conclusions and relevance
GDUs are common and were detected in 5.1% of cats undergoing an upper gastrointestinal endoscopy. The risk of a malignant ulcer increases proportionally with each year of increasing age. GDU location, number and morphological appearance do not provide any indication of the nature of the ulcer; however, duodenal ulcers are frequently benign. Endoscopic examination facilitates the early and minimally invasive detection of GDUs in cats.
Introduction
Gastroduodenal ulcers (GDUs) are rarely found in cats.1–3 They may be caused by multiple factors that compromise the normal physiological defence mechanism of the gastroduodenal mucosa exposing the submucosa or deeper layers of the gastric and duodenal wall to gastric acid.1–3
In cats, GDUs are often associated with neoplasia, with lymphoma being over-represented.3–5 Less frequently, the following have been associated with feline GDUs: inflammatory bowel disease (IBD), 6 hypereosinophilic syndrome,7–9 non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroid administration,3,5,10 feline gastrointestinal eosinophilic sclerosing fibroplasia 11 and Actinomyces species infections. 12
Cats with GDUs present for hyporexia, vomiting, lethargy, mental dullness, melaena, haematemesis and weight loss.3,5,13 GDU is often diagnosed late, upon surgery for gastroduoenal perforation.13–16 In some cases, GDU may cause death and is diagnosed post mortem.3,5
Endoscopy, a minimally invasive and minimally traumatic procedure that allows direct visualisation of the gastric and internal wall, is considered the gold standard for the diagnosis of GDUs in cats and dogs. It is associated with low morbidity and mortality. 17 The endoscopic appearance of GDUs in cats has rarely been described, however, and is less frequently reported than in dogs.13,18,19
The aim of this study was to describe the endoscopic appearance of GDUs, the clinical, ultrasonographic and histological data and follow-up of 61 cats with GDUs.
Materials and methods
Case selection criteria
A multicentre retrospective study was performed. The medical record databases of five veterinary endoscopists (EB, EB, PR, DC, AC) from the Endovet Association were searched for cats that had undergone upper gastrointestinal endoscopy. Cats that had at least one gastric or duodenal ulcer detected by the endoscopic examination were included in the study.
Medical records review
All the medical records of the selected cats were reviewed and information was collected regarding breed, age, sex, neuter status, medical history, clinical signs, and ultrasonographic, endoscopic and histological findings. Upper gastrointestinal endoscopies were routinely performed by video endoscopes (Fujinon EG 270 N- 5 diameter 5.9 mm, working channel 2 mm or Silver Scope Karl Storz 60719 PKS/NKS diameter 7.9 mm, working channel 2.8 mm) and usually at least eight gastric and eight duodenal biopsies were collected.
A lesion of at least 5 mm in diameter was considered to be an ulcer (Figure 1). 3 GDU size was measured approximately with 1.8 mm biopsy forceps with open valves (6 mm) as the dimensional marker. Anatomical location of the ulcers was defined by endoscopy as gastric body, fundus, antrum (including pylorus) or duodenum. A single GDU location (single presentation) was defined if only one anatomical site was affected; multiple GDUs (multiple presentation) were defined when more anatomical sites were affected.

Endoscopic evaluation of gastroduodenal ulcers. Endoscopic appearance of a (a) gastric ulcer (lymphoma) and (b) duodenal ulcer (inflammatory)
Histological diagnoses were divided into two categories: benign and malignant. Malignant lesions were also divided into four subgroups: lymphoma, carcinoma, sarcoma or other tumours. For the benign ulcers, the type of inflammation was characterised according to one of the following categories: lymphoplasmacytic inflammation, lymphoplasmacytic and eosinophilic inflammation, lymphoplasmacytic and neutrophilic inflammation, and eosinophilic inflammation. Information regarding treatment and follow-up at 6 (T6), 12 (T12) and 18 (T18) months was also included. General health status was recorded in the clinic at T6, and clinical or telephone check-ups were also performed at T12 and T18.
The cats were evaluated as follows: alive without vomiting and/or hyporexia, alive with vomiting and/or hyporexia (once or twice a week) and dead.
Statistical analysis
Data were presented as median (range) for continuous variables, and as absolute (relative) frequencies for categorical variables. Predictors included sex, neuter status, breed, age, anatomical location, single/multiple presentation and dichotomous histological diagnosis. Survival (alive/dead) at T18 was the outcome. Before developing multivariable models, each potential predictor was evaluated for its association with the outcome using univariable logistic regression models. Multivariable logistic regression models were used to identify potential predictors that were significant (P <0.05) in the univariable analysis. No pairwise interactions or multicollinearity between final model variables were found (P >0.05, variance inflation factors <5).
To test the goodness-of-fit of the model, predicted probabilities were computed from the final model variables. A receiver operating characteristic (ROC) analysis was then performed and the area under the curve (AUC) and 95% confidence intervals (CIs) were calculated. Sex, neuter status, breed, age, location and single/multiple presentation were associated with histological diagnosis using Fisher’s test (when there were fewer than five cases in at least one cell in a 2×2 table) or χ2.
Results
Cats
Between January 2016 and January 2020, GDUs were reported in 63 cats (5.1%) from the 1224 upper gastrointestinal endoscopic examinations performed in the same period. Two cats were excluded from the study due to incomplete information: a final diagnosis was not reached in one cat, and the other cat was lost to follow-up. Sixty-one cats were therefore included in the study.
Median age was 9.0 years (range 2.0–16.0). Thirty-one (51%) cats were female (29 spayed) and 30 cats (49%) were male (28 castrated). Forty-six cats (75%) were domestic shorthair (DSH). Purebred cats included: three Maine Coons (5%), three Persians (5%), three Siamese (5%), two Chartreux (3%), two Ragdolls (3%), one Birman (2%) and one Himalayan (2%).
Medical history, clinical signs and diagnostic results
Six cats (10%) had been treated with NSAIDs, 13 (21%) with corticosteroids and one with both NSAIDs and corticosteroids for non-gastrointestinal clinical reasons, with variable dosages and duration in the 2 months prior to the endoscopic examination. In all the cats included, no steroids and/or NSAIDs had been administered in the 7 days prior to the endoscopic examination.
The most common complaints were vomiting (n = 55; 90%) and hyporexia (n = 40; 66%). Clinical signs occurred at least twice a week for 3 months, 2 months or 1 month in 22 (36%), 30 (49%) and nine (15%) cats, respectively. Haematemesis was reported in 12 (20%) cats.
Abdominal ultrasound reports were available for 46 cats (75%), and changes were present in 38/46 cats (83%; 15 consistent with gastric ulcers, four duodenal ulcers, four with peritoneal reactivity or abdominal fluid, and 23 with duodenal thickening).
Endoscopy and histological evaluation
Endoscopic examination located GDUs as follows: gastric body in 28 cats (46%; Figure 1a), antropyloric area in 34 (56%), fundus in 13 (21%) and duodenum in eight (13%; Figure 1b). A single GDU was found in 42 cats (69%), and multiple GDUs were observed in 19 cats (31%; Table 1). A single duodenal GDU was observed in all eight cats. When associated with multiple presentations, gastric GDUs were observed in different gastric sites in 19/53 cats (10/19: antropyloric area + gastric body; 4/19: gastric body + fundus; 3/19: antropyloric area + gastric body + fundus; 2/19: antropyloric area + fundus).
Information relevant to the population and the endoscopic and histopathologic evaluation of gastroduodenal ulcers (GDUs) in 61 cats
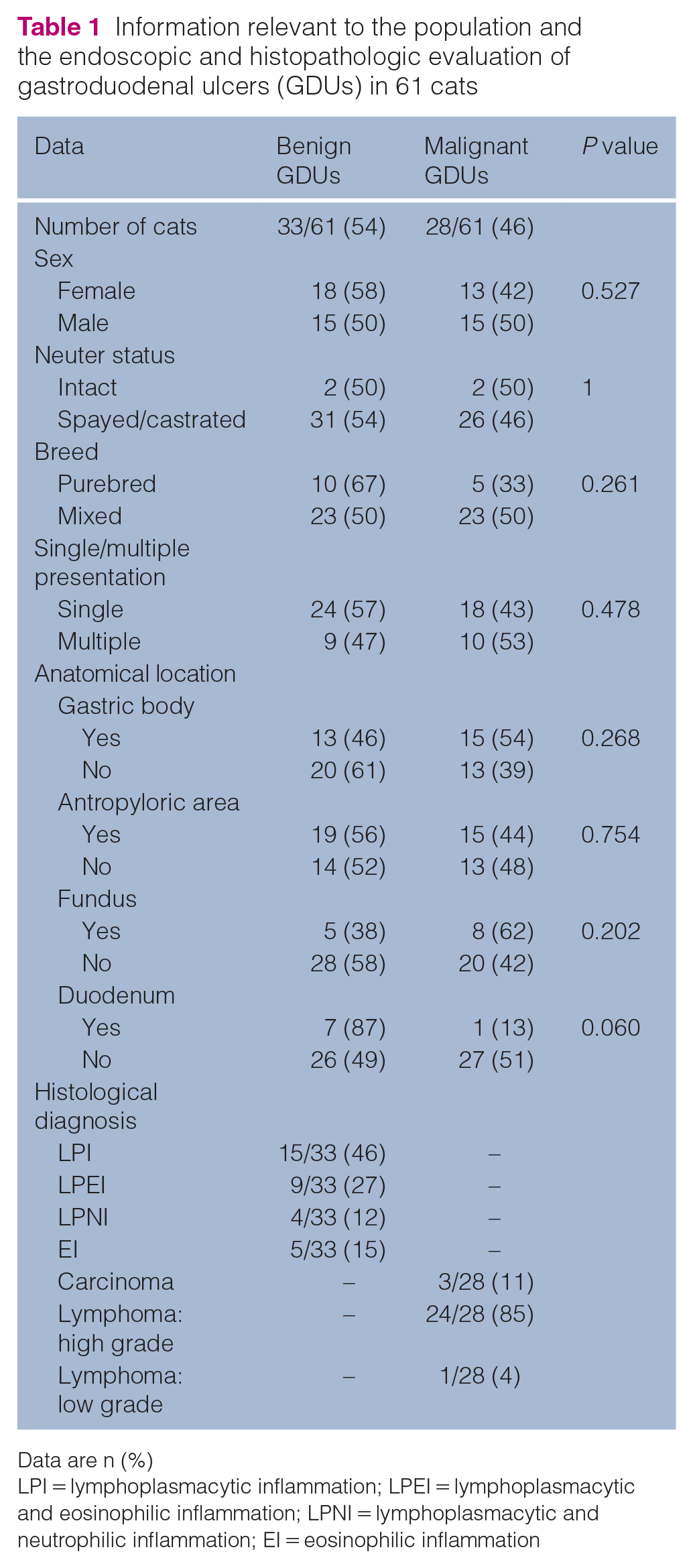
Data are n (%)
LPI = lymphoplasmacytic inflammation; LPEI = lymphoplasmacytic and eosinophilic inflammation; LPNI = lymphoplasmacytic and neutrophilic inflammation; EI = eosinophilic inflammation
A histopathological evaluation of endoscopic biopsies was performed in all patients, which showed benign lesions in 33 (54%) cats and malignant lesions in 28 (46%). Benign lesions were histologically classified as follows: lymphoplasmacytic inflammation in 15 cats, lymphoplasmacytic and eosinophilic inflammation in nine, lymphoplasmacytic and neutrophilic inflammation in four and eosinophilic inflammation in five. Among the malignant GDUs, 24 cats (86%) were diagnosed with high-grade lymphoma, one cat (4%) with low-grade lymphoma and three cats (11%) with carcinoma (Table 1). High-grade lymphomas were present only in the stomach. Among the duodenal ulcers, only one cat (13%) was diagnosed with a malignant lesion (low-grade lymphoma). Cats diagnosed with malignant lesions were significantly older (median 10.5 years [range 4–16]; P = 0.002) than cats diagnosed with benign lesions (median 7 years [range 2–15]). Associations between sex, neuter status, breed, location and single/multiple presentation with histological diagnosis are shown in Table 1.
Briefly, sex, neuter status, breed, gastric location and presence of single or multiple ulcerative lesions were not associated with the dichotomous (benign/malignant) histological diagnosis. Duodenal location tended to be associated with benign lesions; however, this was not statistically significant (P = 0.060).
Follow-up and prognosis
One cat received both surgery and medical therapy; the other 60 cats received only medical therapy. No cats diagnosed with malignant lesions were alive at T18, while only seven cats (11.5%) diagnosed with benign lesions had died at the same time point. Follow-up data are shown in Table 2.
Evaluation of follow-up at 6 (T6), 12 (T12) and 18 (T18) months in 61 cats with gastroduodenal ulcers (GDUs)

A = alive and in good condition; B = alive with gastrointestinal signs; C = dead
Both age (continuous) and histological diagnosis (benign/malignant) were independently associated with the outcome (survivor/non-survivor at T18) of univariate (OR 1.44, 95% CI 1.11–1.87; P = 0.006) and multivariate (OR 92, 95% CI 9–934; P <0.001) analyses. The ROC analysis of predicted probabilities confirmed the goodness-of-fit of the model (AUC 0.955, 95% CI 0.9–1; P <0.001).
Discussion
Gastroduodenal ulcerations in cats are not commonly reported and GDUs have been defined as rare.3,5,18 In this retrospective multicentre study, GDUs were detected in 5.1% of cats undergoing an upper gastrointestinal endoscopy in a 4-year time period. Analysing over 1200 endoscopy examination reports, our data showed that GDUs are common in cats.
Gastroduodenal ulcerations and perforation have previously been reported to mainly affect middle-aged to older cats, without any breed predisposition.3,13 Similarly, in the present study GDUs occurred mainly in older cats, and there was no breed predisposition. However, our data showed a significant relationship between elderly cats and the malignant origin of the GDU. To the best of our knowledge, no previous study has investigated the relationship between the age and histological diagnosis of GDUs in cats.3,5,13
Most gastric ulcers in cats have been reported to have a neoplastic origin.3–5 This was not confirmed by our study, in which almost half (49%) of the gastric ulcers found were benign. This important finding is related to the retrospective nature of the present study and to the authors’ database. In previous studies, GDUs were detected in cats with serious clinical conditions resulting in surgical or post-mortem biopsy examination.3,5 In our study, cats with chronic and presumably milder clinical signs were endoscopically investigated, making it difficult to make a direct comparison with the literature. Moreover, in the present study the diagnosis was performed with endoscopic biopsy, resulting in a possible underestimation of the disease, especially for submucosal tumours. However, almost 90% of the cats diagnosed with benign lesions were alive 18 months after the diagnosis, thus making a misdiagnosis unlikely.
We can therefore assert that, when considered together, gastric and duodenal ulcers in cats are more common than expected, and GDUs are associated with benign lesions in half of the cases. Endoscopic examinations enabled biopsies of ulcerated lesions to be performed under direct visual guidance, we believe, resulting in an early diagnosis of the GDUs.
Our data failed to show a statistical association between the location and number of lesions and histopathological diagnosis of GDUs. Non-neoplastic ulcers have almost always been reported in the antropyloric or fundic regions of the stomach.3,10
In our study, GDUs were found in all the gastric regions, with no associations with histological diagnosis. We believe that this finding sheds light on the origin of GDUs, thus suggesting that clinicians should be careful not to overestimate the diagnosis of malignancy in the detection of GDUs.
Furthermore, in our case series, duodenal ulcers were benign in 7/8 cats, and the remaining cat was diagnosed with a low-grade lymphoma. In a previous study, a benign duodenal ulcer associated with perforation secondary to carprofen administration was described in one cat. 10 In our study, six cats had been treated with NSAIDs within the month prior to presentation and one cat had been treated with both NSAIDs and steroids.
NSAIDs induce gastrointestinal tract perforation through direct contact injury to gastric epithelial cells and inhibition of cyclooxygenases.5,20 Recently, gastric ulcerations associated with NSAID and glucocorticoid administration have been described in dogs. 21 The risk of GDUs in dogs receiving NSAIDs and glucocorticoids has long been known, but there are few data available in cats. 21 Owing to the study design and retrospective nature of this work, we were not able to conclude whether or not the benign GDUs that we observed were the consequence of NSAID or steroid administration.
In our study, in 33 cases of benign GDUs there was no statistical association between the histological inflammatory type and the location of the ulcers or follow-up. Interestingly, in these patients the histological examination of the gastric and enteric mucosa that was not ulcerated always showed the same type of inflammation detected at the ulcer level.
IBD has rarely been reported as being a cause of gastroduodenal erosion and ulceration in cats,5,13,6 and, as described by Liptak et al 3 and also in our patients with non-neoplastic ulcers, a definitive aetiology was not identified. 5 Assuming that a unique and specific aetiology for gastrointestinal inflammatory diseases is not known, ulceration may be a manifestation of the inflammatory pathology, although it is not possible to assign any diagnostic or prognostic significance to its presence.
In our work, in 15% of cats with benign GDUs, eosinophilic inflammation was present. None of these cats was affected by feline gastrointestinal eosinophilic sclerosing fibroplasia (FGESF) or hypereosinophilic syndrome (HES).8,9,11 Both of these pathologies are rare. During FGESF, ultrasound changes generally lead to a histological diagnosis by surgical sampling. In relation to HES, clinical pathological changes or systemic involvement would have been essential for a diagnosis, but these were not found in our cases.8,9,11 None of the cats with eosinophilic inflammation and ulceration included in our study has, to date, manifested evolution to FGESF or to HES; however, it would be interesting to follow them up in the long term.
In our study, high-grade lymphoma was the most common malignant tumour, which in all cases was located in the stomach. Cats with high-grade lymphoma show a more acute onset and rapidly progressive clinical course. In Gustafson et al, 22 the prognosis was evaluated in 16 cats with gastric lymphoma. The median overall clinical remission duration was 108 days and median overall survival was 171 days. 22 Also, in our cases, the cats with gastric lymphoma had a poor prognosis and short follow-up.
In our work, the duodenal ulcers were all inflammatory except for one with low-grade intestinal lymphoma. To the best of our knowledge, duodenal ulcers in low-grade enteric lymphoma have not been reported before,23,24 and it cannot be ruled out that different factors (eg, drugs or transit of foreign bodies) induced the formation of ulcers in the neoplastic context. However, this possible endoscopic finding in low-grade lymphoma merits further investigation.
Interestingly, in our study three gastric carcinomas presented with an ulcerated appearance. In dogs, the gastric ulcer has frequently been associated with gastric carcinoma; however, in cats this macroscopic appearance is not considered typical.14,21,25,26
We believe that the ulcerative appearance could be included among the possible manifestations of gastric carcinoma in cats and that it merits further investigation.
The limitations of our study are mainly due to its retrospective nature, which prevented us from collecting the same laboratory and diagnostic imaging information for all the cases and therefore being able to make more significant statistical correlations.
Conclusions
In the present study, GDUs were frequently observed during upper gastrointestinal endoscopy but were not always of a neoplastic nature. Endoscopic examination of these cats proved to be an important part of the diagnostic effort, even when the gross endoscopic appearance of the lesion did not provide direct diagnostic or prognostic information. In cats, the risk of a malignant ulcer increases proportionally with each year of increasing age. The location, number and morphological appearance do not provide any indication of the nature of the ulcer; however, duodenal ulcers are frequently benign.
Gastroduodenal endoscopic examinations facilitate an early and minimally invasive diagnosis.
Footnotes
Author note
Parts of this paper were presented at the 2020 ECVIM-CA conference.
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
