Abstract
Although regulatory T cells (Tregs) play an integral role in immunologic tolerance and the maintenance of intestinal homeostasis, their involvement in canine gastrointestinal diseases, including idiopathic inflammatory bowel disease (IBD) and intestinal lymphoma, remains unclear. Here we show altered numbers of forkhead box P3 (Foxp3)–positive Tregs in the intestine of dogs with IBD and intestinal lymphoma. IBD was diagnosed in 48 dogs; small cell intestinal lymphoma was diagnosed in 46 dogs; large cell intestinal lymphoma was diagnosed in 30 dogs; and 25 healthy beagles were used as normal controls. Foxp3-positive Tregs in the duodenal mucosa were examined by immunohistochemistry and immunofluorescence. Duodenal expression of interleukin-10 mRNA was quantified by real-time reverse transcription polymerase chain reaction. The number of Foxp3-positive lamina propria cells and the expression of interleukin-10 mRNA were significantly lower in dogs with IBD than in healthy dogs and dogs with intestinal lymphoma. The number of Foxp3-positive intraepithelial cells was higher in dogs with small cell intestinal lymphoma. Some large cell intestinal lymphoma cases had high numbers of Foxp3-positive cells, but the increase was not statistically significant. Double-labeling immunofluorescence showed that CD3-positive granzyme B–negative helper T cells expressed Foxp3. In small cell intestinal lymphoma cases, the overall survival of dogs with a high Treg density was significantly worse than that of dogs with a normal Treg density. These results suggest that a change in the number of Foxp3-positive Tregs contributes to the pathogenesis of canine IBD and intestinal lymphoma by disrupting mucosal tolerance and suppressing antitumor immunity, respectively.
Keywords
Regulatory T cells (Tregs) are a subset of CD4-positive T lymphocytes that inhibits the activation of immune responses. Tregs are necessary for maintaining peripheral tolerance. 8,13,52 The primary function of Tregs is to suppress effector T cells and antigen-presenting cells through direct contact and the secretion of immunosuppressive cytokines such as interleukin 10 (IL-10) and transforming growth factor β (TGF-β). 8,52 The majority of Tregs express high levels of the IL-2 receptor α chain (CD25). The transcription factor forkhead box P3 (Foxp3) is critical for the development and function of the majority of Tregs. 64 Thus, Foxp3 is used as a specific nuclear marker of Tregs that allows these cells to be distinguished from other T cell subpopulations. Mice and humans with mutations in the foxp3 gene develop multisystem autoimmune disease associated with an absence of Tregs, suggesting the importance of Tregs in maintaining self-tolerance. 3,7,63
Inadequate suppression of immune responses to commensal bacteria resulting from abnormalities in Treg number or function is thought to contribute to the pathogenesis of inflammatory bowel disease (IBD). The evidence supporting this hypothesis comes from experiments demonstrating the development of gastrointestinal inflammation in T cell–deficient mice injected with naive CD4-positive CD25-negative T cells, a population that includes effector T cells but lacks CD4-positive CD25-positive Tregs. 51 Furthermore, IL-10-deficient mice spontaneously develop enterocolitis when maintained in conventional environments but exhibit no evidence of intestinal inflammation when kept in germ-free conditions. 32,55 Intermittent depletion of Foxp3-positive Tregs leads to intestinal inflammation characterized by severe weight loss and death in mice. 6 While all these findings suggest that Tregs maintain intestinal homeostasis and that a lack of Tregs is associated with enterocolitis in mice, it is unclear whether Treg defects underlie the pathogenesis of IBD in humans and dogs.
In mice and humans, Tregs play a role not only in the regulation of immune homeostasis but also in the progression of cancer. In tumor-bearing mice, depletion of Tregs by administration of anti-CD25 monoclonal antibody (mAb) enhances antitumor immunity and results in tumor eradication. 45,56 In humans, Treg infiltration is found in certain tumors, and the number of Tregs is increased in tumor-bearing individuals. 18,42 The high infiltration of Tregs into tumors correlates with poor prognosis in patients with ovarian 12,53 and breast cancers 2,23 and hepatocellular carcinoma. 19,21 As reported in humans, a number of studies have shown that the number of peripherally circulating and tumor-infiltrating Tregs is increased in dogs with various types of cancer.* However, little is known about the role of Tregs in canine intestinal lymphomas.
In the present study, we investigated the number of Tregs in the duodenal mucosa of dogs with IBD and intestinal lymphoma, using Foxp3 as a marker of Tregs, and we evaluated the association between Treg number and prognosis to clarify the contribution of Tregs to the pathogenesis of canine IBD and intestinal lymphoma.
Materials and Methods
Study Population
The study included 124 dogs with clinical signs of chronic gastrointestinal disease that underwent endoscopic examination in the Veterinary Medical Center of the University of Tokyo between 2009 and 2012. Informed consent was obtained from all owners, and the study protocol was approved by the animal care committee of the Veterinary Medical Center of the University of Tokyo. The case selection criteria for IBD and intestinal lymphoma have been described. 34,35 Briefly, IBD diagnosis was based on histopathologic evidence of lymphocytic-plasmacytic enteritis, the absence of clonal gene rearrangement of IgH or TCRγ in polymerase chain reaction for antigen receptor gene rearrangement (PARR), and exclusion of food- and antibiotic-responsive enteropathies. Intestinal lymphoma diagnosis was based on histopathologic examination, the presence of clonal IgH or TCRγ gene rearrangement, and immunohistochemistry (IHC) for CD3 and CD20 as described previously. 34 Dogs that had been treated with corticosteroids in the 2 weeks before the study were excluded. All cases were scored for severity according to the canine chronic enteropathy clinical activity index (CCECAI). 1
Twenty-five beagles were used as healthy controls. The group included 9 females (1 intact and 8 spayed) and 16 males (all intact), with a median age of 25.5 months (range, 19–80 months) and a median body weight of 11 kg (range, 8.6–17 kg). The dogs were healthy with no clinical signs of gastrointestinal disease, and they had received no drugs. No abnormalities were observed in urinalysis and blood examinations, including complete blood count and measurements of blood urea nitrogen and creatinine concentrations as well as alanine aminotransferase and alkaline phosphatase enzyme activity. Moreover, there were no abnormalities in the parasitic and bacterial analyses of the fecal samples. The use of dogs in this study was approved by the animal care committee of the University of Tokyo.
Endoscopy
In preparation for endoscopy, dogs were fasted for 12 to 18 hours. Gastroduodenoscopy and/or ileocolonoscopy was performed under general anesthesia. Multiple mucosal biopsies (6 in each area) were collected from the stomach, duodenum, ileum, and colon for histopathology. At least 1 biopsy specimen from each site was used for PARR. 20 Duodenal samples for RNA extraction were immediately submerged in RNAlater (Qiagen, Valencia, CA, USA) and stored at −20°C.
Histopathology
The mucosal biopsy specimens from each site were fixed in 10% formalin for 48 hours, embedded in paraffin, and stained with hematoxylin and eosin. A histopathologic diagnosis of gastrointestinal inflammation was made according to the World Small Animal Veterinary Association criteria. 14,33,35 A histopathologic diagnosis of intestinal lymphoma was made from hematoxylin and eosin–stained sections, PARR results, and IHC. Lymphocyte epitheliotropism, heterogeneity, and nuclear size were evaluated to differentiate between intestinal lymphoma and inflammation. 9, 30 The intestinal T cell lymphomas are recently classified into enteropathy associated T cell lymphoma (EATL) type 1 (small cell lymphoma) and type 2 (large cell lymphoma) in humans and canine cases. 9 In this study, the lymphomas are classified into 2 categories; small and large cell lymphomas according to the morphological characteristics of the proliferating lymphocytes.
Immunohistochemistry
IHC for Foxp3 was conducted on 4-µm-thick paraffin-embedded sections of the duodenal samples obtained from endoscopic biopsy. Previous immunohistochemical study showed that canine Foxp3-positive cells in the duodenum were higher than those in the jejunum, ileum, and colon. 27 Therefore, we chose the duodenum to examine intestinal Foxp3-positive cells. Heat-induced antigen retrieval was performed by autoclaving the sections for 5 minutes at 121°C in 10mM sodium citrate buffer (pH 6.0). Endogenous peroxidase activity was blocked by incubation with REAL Peroxidase-Blocking Solution (Dako, Glostrup, Denmark) at room temperature for 10 minutes. The sections were blocked with 5% skim milk in Tris-buffered saline with 0.1% Tween 20 (TBST) at room temperature for 60 minutes and then incubated with a rat anti-mouse Foxp3 mAb (1:400; clone FJK-16s; eBioscience, San Diego, CA, USA) at 4°C overnight. The cross-reactivity of this mAb with canine Foxp3 was confirmed in a previous study. 40 The slides were washed with TBST and then incubated with a biotin-labeled anti-rat IgG antibody (Vector Laboratories, Burlingame, CA, USA) at 37°C for 30 minutes. The sections were washed and incubated with HRP-labeled streptavidin (Dako) at room temperature for 30 minutes. The reaction products were visualized with 3,3′-diaminobenzidine. In place of the primary antibody, an isotype-matched antibody (normal rat IgG; Santa Cruz Biotechnology, Santa Cruz, CA, USA) was placed on each slide as a negative control.
Cells with clear lymphocyte morphology, distinct nuclear staining for Foxp3, and an unstained cytoplasm were classified as Tregs. Foxp3-positive Tregs were quantified in different compartments (lamina propria and the intraepithelial area) via image analysis software (ImageJ). 54 Ten appropriate sites were chosen for each compartment, and positively stained cells were counted with a ×40 objective, a ×10 eyepiece, and a square eyepiece graticule (10 × 10 squares, with a total area of 62 500 μm2) according to the published paper. 27 The results were expressed as total Foxp3-positive cells per 62 500 μm2, Foxp3-positive lamina propria cells per 62 500 μm2, and Foxp3-positive intraepithelial cells (IELs) per 100 enterocytes.
Quantitative Real-time Polymerase Chain Reaction
Total RNA was extracted from the duodenal samples using an RNAspin Mini RNA Isolation Kit (GE Healthcare, Buckinghamshire, England). Genomic DNA was eliminated from the samples using the TURBO DNA-free Kit (Applied Biosystems, Foster City, CA), and the samples were then stored at −80°C for later use. The expression of IL-10 mRNA was quantified by 2-step real-time reverse transcription polymerase chain reaction (Thermal Cycler Dice Real Time System; Takara Bio, Shiga, Japan), as described previously, 33,34 with the following primer pair: 5′-CGA CCC AGA CAT CAA GAA CC-3′ (forward) and 5′-CAC AGG GAA GAA ATC GGT GA-3′ (reverse; product size, 101 base pairs). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), TATA-box binding protein (TBP), and succinate dehydrogenase complex subunit A (SDHA) were used as reference genes. 46 The primer sequences for these 3 housekeeping genes have been described. 36
Double-labeling Immunofluorescence
The 4-µm-thick paraffin-embedded duodenal tissue sections were used for immunofluorescence. Heat-induced antigen retrieval was performed by autoclaving the sections for 10 minutes at 121°C in 10mM sodium citrate buffer (pH 6.0; for CD20, granzyme B, and Ki-67) or Target Retrieval Solution, High pH (Dako; for CD3). The sections were blocked with 8% skim milk in TBST at 37°C for 40 minutes and then immunostained using primary antibodies against Foxp3 (1:100, clone FJK-16s, rat; eBioscience), CD3 (undiluted, rabbit; Dako), CD20 (1:100, rabbit; Thermo Fisher Scientific, Waltham, MA, USA), granzyme B (1:50, rabbit; Spring Bioscience, Pleasanton, CA, USA), and Ki-67 (undiluted, clone MIB-1, mouse; Dako) at 4°C overnight. The sections were washed and incubated with secondary fluorescent antibodies at 37°C for 60 minutes. The secondary antibodies included Alexa Fluor 594 goat anti-rat IgG (1:200; Life Technologies, Carlsbad, CA, USA), Alexa Fluor 488 goat anti-rabbit IgG (1:200; Invitrogen, Carlsbad, CA, USA), and Alexa Fluor 488 donkey anti-mouse IgG (1:200; Invitrogen). Sections were mounted with Vectashield HardSet with DAPI (Vector Laboratories, Burlingame, CA, USA). Images were captured with an Eclipse E800 fluorescence microscope (Nikon, Tokyo, Japan).
Statistics
Statistical analyses were performed using JMP 9 (SAS Institute, Cary, NC, USA). Fisher exact test was used to compare the sex distribution among dogs with IBD and dogs with intestinal lymphomas. The Kruskal–Wallis test was used to test for overall differences. The Steel–Dwass test was used to analyze between-group differences. Survival curves were generated with the Kaplan–Meier method, and survival rates were compared using the log-rank test. The relationships were evaluated using the Spearman rank correlation coefficient. Statistical significance was defined as P < .05.
Results
Case Evaluation
A summary of the characteristics of dogs diagnosed with IBD and intestinal lymphoma is shown in Table 1. Forty-eight dogs with IBD were included in this study. All of the dogs had evidence of inflammation within the intestinal mucosa and a histopathologic diagnosis of lymphocytic-plasmacytic enteritis (Figs. 1–6; see Figs. 1, 4). The breeds were as follows: Miniature Dachshund (n = 5), Yorkshire Terrier (n = 5), Chihuahua (n = 4), Pembroke Welsh Corgi (n = 3), Cavalier King Charles Spaniel (n = 3), Toy Poodle (n = 3), Pug (n = 3), French Bulldog (n = 3), Boston Terrier (n = 3), Shiba Inu (n = 3), Shetland Sheepdog (n = 2), Jack Russell Terrier (n = 2), Maltese (n = 2), Cairn Terrier (n = 1), West Highland White Terrier (n = 1), German Shepherd Dog (n = 1), Beagle (n = 1), Bichon Frise (n = 1), and Pomeranian (n = 1). All dogs received prednisolone (0.5–2 mg/kg) after endoscopy.
Comparison of Dogs With IBD and Intestinal Lymphoma.a

Abbreviation: IBD, inflammatory bowel disease.
aValues expressed as median (range), unless noted otherwise.
bNo. of castrated in parentheses.
cNo. of spayed in parentheses.
dCanine chronic enteropathy clinical activity index.
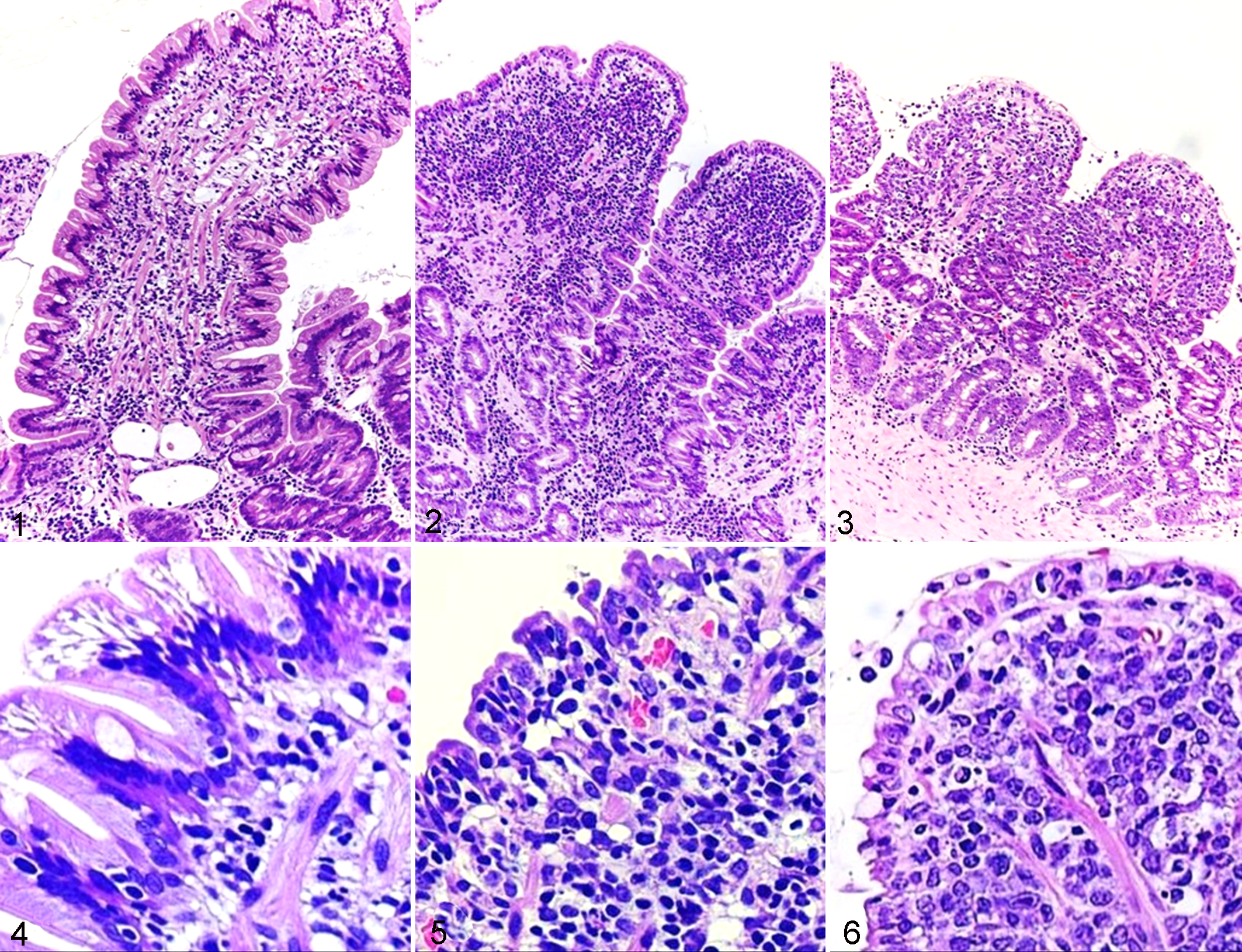
Forty-six dogs were diagnosed with small cell intestinal lymphoma (Figs. 2, 5). Of these, 42 (91.3%) neoplasms were classified as T-cell lymphoma, and 4 (8.7%) were classified as B-cell lymphoma by IHC and PARR results. The breeds were as follows: Shiba Inu (n = 8), Miniature Dachshund (n = 7), Cairn Terrier (n = 3), Chihuahua (n = 3), French Bulldog (n = 3), mongrel (n = 2), Pembroke Welsh Corgi (n = 2), Boston Terrier (n = 2), Maltese (n = 2), Labrador Retriever (n = 2), Golden Retriever (n = 1), Shetland Sheepdog (n = 1), German Shepherd Dog (n = 1), Tibetan Terrier (n = 1), Toy Manchester Terrier (n = 1), Newfoundland (n = 1), Pug (n = 1), Papillon (n = 1), Beagle (n = 1), Flat-coated Retriever (n = 1), Miniature Pinscher (n = 1), and Yorkshire Terrier (n = 1). All dogs received prednisolone (1–2 mg/kg), and 31 were treated with chlorambucil (2 mg/m2) in combination with prednisolone.
Thirty dogs had large cell intestinal lymphoma (Figs. 3, 6). Of these, 22 (73.3%) neoplasms were classified as T-cell lymphoma and 8 (26.7%) as B-cell lymphoma. The breeds were as follows: Shih Tzu (n = 4), Pug (n = 4), Shiba Inu (n = 4), Miniature Dachshund (n = 4), Labrador Retriever (n = 3), Maltese (n = 2), American Cocker Spaniel (n = 1), mongrel (n = 1), Italian Greyhound (n = 1), West Highland White Terrier (n = 1), Jack Russell Terrier (n = 1), Toy Manchester Terrier (n = 1), Papillon (n = 1), Beagle (n = 1), and Border Collie (n = 1). Nine dogs with large cell intestinal lymphoma received prednisolone (2 mg/kg), and 21 were treated with a 6-month modified version of the Wisconsin-Madison chemotherapy protocol (UW-25). 22
Comparison of Clinical Findings
There were no significant differences in sex, body weight, and serum albumin concentration among dogs with IBD and dogs with intestinal lymphomas (Table 1). Significant differences in age, clinical score, and overall survival were observed among the 3 groups (Table 1). Dogs with IBD were significantly younger than dogs with large cell lymphoma (P = .008). Dogs with small cell lymphoma had significantly higher CCECAI scores than dogs with IBD (P = .0115). Dogs with large cell lymphoma had significantly higher CCECAI scores than dogs with IBD (P < .0001) and dogs with small cell lymphoma (P = .0002). The overall survival was significantly shorter for dogs with intestinal lymphoma than for dogs with IBD (IBD vs small cell lymphoma, P = .0027; IBD vs large cell lymphoma, P < .0001; small cell lymphoma vs large cell lymphoma, P = .0124).
Foxp3 Expression in the Duodenal Mucosa
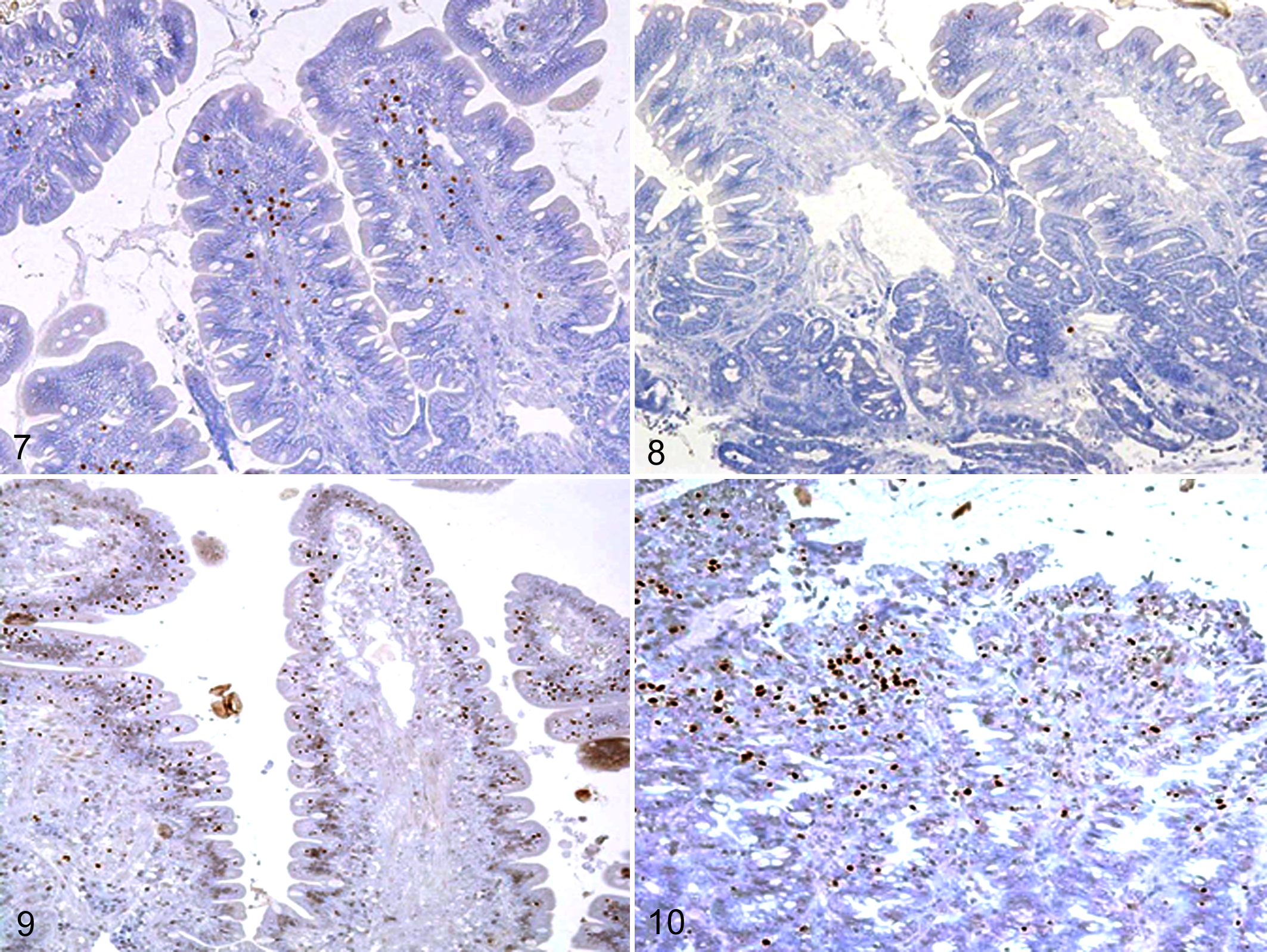
IHC was performed to investigate the localization and number of Foxp3-positive cells in the intestine of dogs with IBD and intestinal lymphoma. The isotype-matched negative controls indicated no nonspecific staining (data not shown). In healthy dogs, IHC showed nuclear expression of Foxp3 in lymphocytes scattered in the villous lamina propria, with no staining of epithelial cells (Fig. 7). Some IELs in healthy dogs expressed Foxp3. In dogs with IBD, the number of Foxp3-positive cells was reduced (Fig. 8). Conversely, Foxp3-positive cells accumulated in dogs with intestinal lymphoma (Figs. 9, 10). Interestingly, the number of Foxp3-expressing IELs was increased in dogs with small cell lymphoma (Fig. 9).
The number of Foxp3-expressing lamina propria cells, IELs, and total cells was counted in healthy dogs, dogs with IBD, and dogs with intestinal lymphomas (Fig. 11). There were significantly fewer total Foxp3-positive cells and Foxp3-positive lamina propria cells in dogs with IBD than in healthy controls or dogs with intestinal lymphomas (Fig. 11a, b). The numbers of total Foxp3-positive cells and Foxp3-positive IELs were higher in dogs with small cell lymphoma than in healthy dogs and dogs with IBD (Fig. 11a, c). Although increased numbers of Foxp3-positive cells were observed in some cases of large cell lymphoma, there was no significant difference when compared to the number in healthy controls. Further analysis showed that the number of total Foxp3-positive cells in the intestine correlated with the CCECAI score in dogs with small cell lymphoma (rs = 0.4157, P = .0041), although there was no significant correlation in dogs with IBD (rs = –0.0399, P = .7984) and dogs with large cell lymphoma (rs = 0.0446, P = .8252).
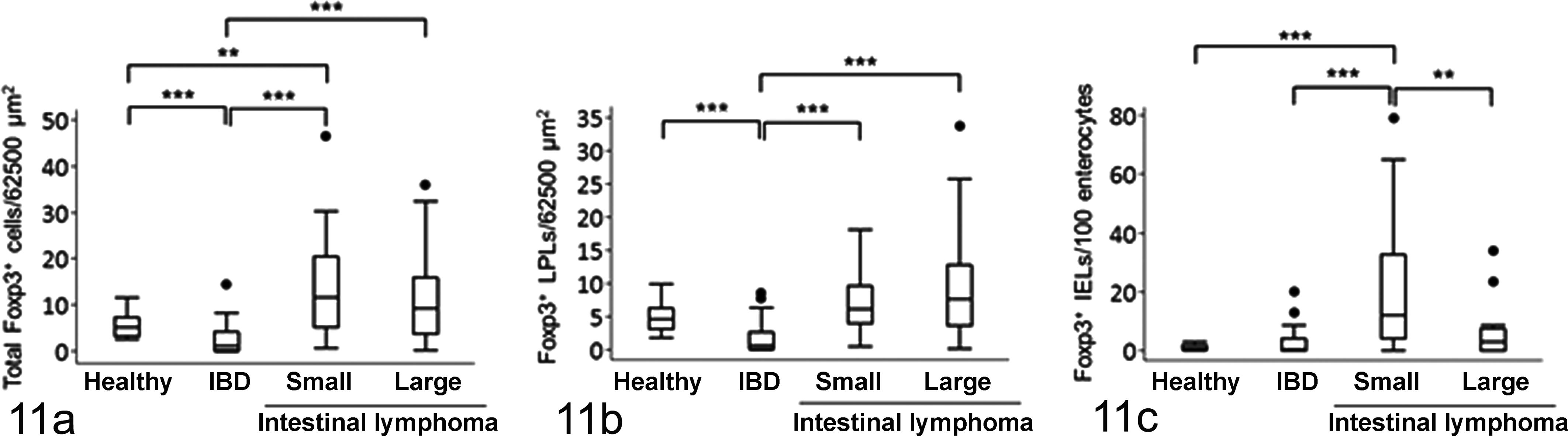
The number of forkhead box P3 (Foxp3)–positive (a) total cells, (b) lamina propria lymphocytes (LPLs), and (c) intraepithelial lymphocytes (IELs) in the duodenal mucosa of healthy dogs (n = 25) and dogs with idiopathic inflammatory bowel disease (IBD; n = 48), small cell lymphoma (n = 46), and large cell lymphoma (n = 30). Data are presented as the median, with the 25th and 75th quartiles in each box plot. The whiskers indicate the highest and lowest data points within 1.5 times the length of the quartiles. The circles represent outliers. **P < .01; ***P < .001.
IL-10 mRNA Expression in the Duodenal Mucosa
Duodenal IL-10 mRNA expression was significantly lower in dogs with IBD than in healthy control dogs or dogs with intestinal lymphomas (Suppl. Fig. 1). Significant positive correlations were observed between IL-10 mRNA expression and the number of total Foxp3-positive cells (rs = 0.3590, P = .0001), Foxp3-positive lamina propria cells (rs = 0.2415, P = .0019), and Foxp3-positive IELs (rs = 0.4538, P = .0001). However, there was no significant correlation between intestinal IL-10 expression and the CCECAI score (rs = 0.1163, P = .1078).
Characterization of Foxp3-positive Cells
To characterize the phenotype of Foxp3-positive cells in the canine intestine, surface antigens were examined in healthy dogs by double-labeling immunofluorescence (Figs. 12–17). As expected, some CD3-positive T cells expressed Foxp3 (Fig. 12) in the healthy canine intestine, while CD20-positive B cells did not (Fig. 14). Granzyme B-positive cytotoxic T cells did not express Foxp3 (Fig. 13), indicating that Foxp3-expressing cells are a subset of helper T cells. We evaluated CD3, CD20, or granzyme B expression in Foxp3-positive cells of 8 dogs with IBD and 16 dogs with intestinal lymphoma, although we did not examine all cases. The Foxp3-positive cells expressed CD3 but not CD20 and granzyme B in all cases examined, whether they had IBD or lymphoma (data not shown). In dogs with intestinal lymphoma, CD3-positive Foxp3-positive Tregs accumulated in the lesion (Fig. 15). We next investigated whether the accumulated Foxp3-positive cells were neoplastic cells. Proliferating neoplastic cells were detected by Ki-67 staining. In dogs with small cell lymphoma, Ki-67-positive proliferating cells did not express Foxp3 (Fig. 16). Yet, some Ki-67-positive cells expressed Foxp3 in large cell lymphoma (Fig. 17).
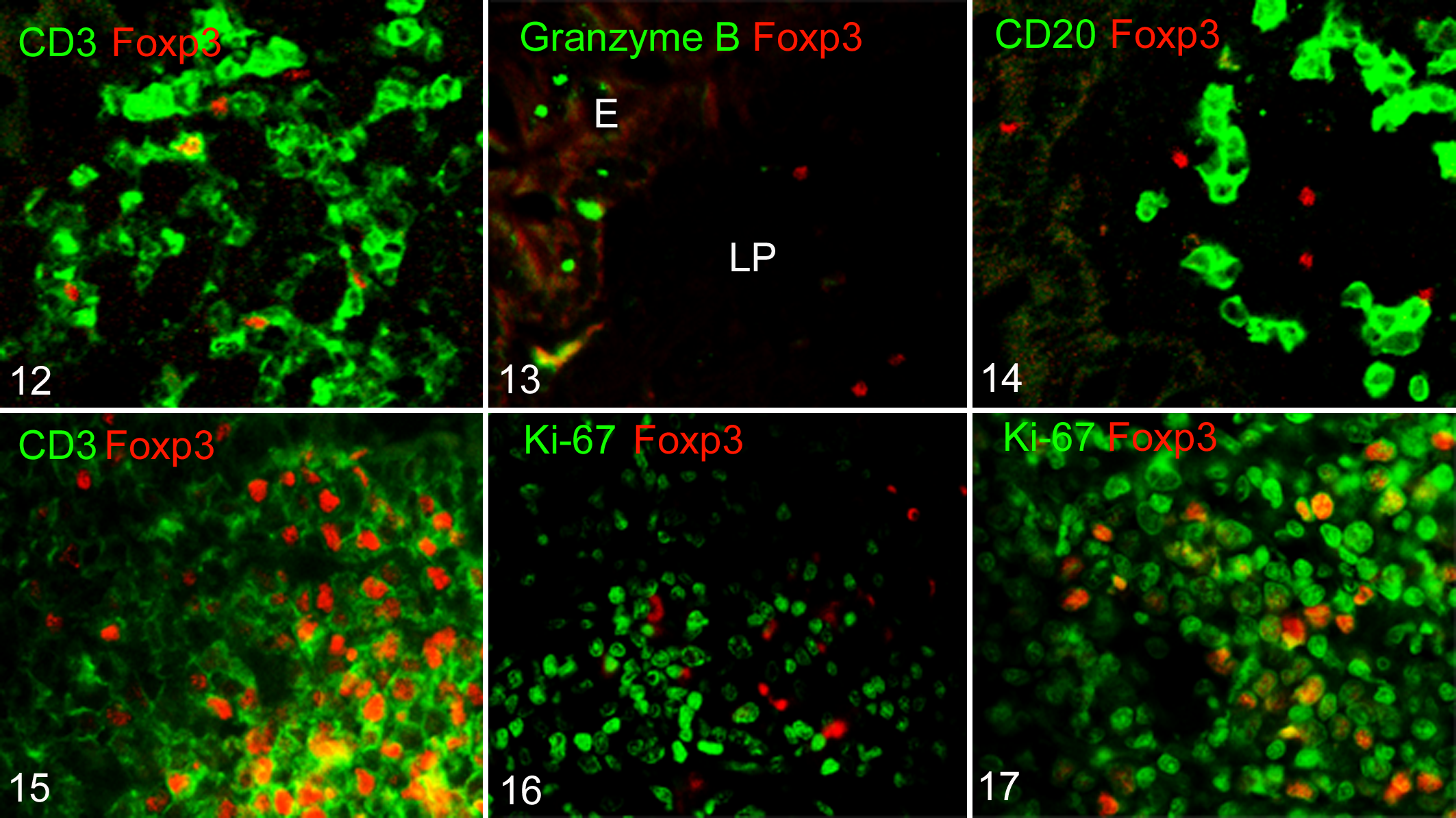
Phenotyping of forkhead box P3 (Foxp3)–positive cells in the canine intestine.
Association Between Foxp3-positive Cell Number and Prognosis
Finally, we analyzed the correlation between the Foxp3-positive cell number and prognosis in dogs with IBD and intestinal lymphoma (Fig. 18). For survival analysis, each case was classified as having a high, low, or reference density of Foxp3-positive Tregs relative to the mean ± 2SD in healthy dogs (reference, total Tregs: 0.5531–10.6775 cells/62 500 μm2). In dogs with IBD, the Foxp3-positive Treg number was not significantly related to the prognosis (Fig. 18a). In dogs with small cell lymphoma, the overall survival for cases with a high Foxp3-positive Treg number was significantly shorter than for cases with a reference number of Foxp3-positive Tregs (P = .019632; Fig. 18b). In dogs with large cell lymphoma, the Foxp3-positive Treg number was not significantly related to the prognosis (Fig. 18c).
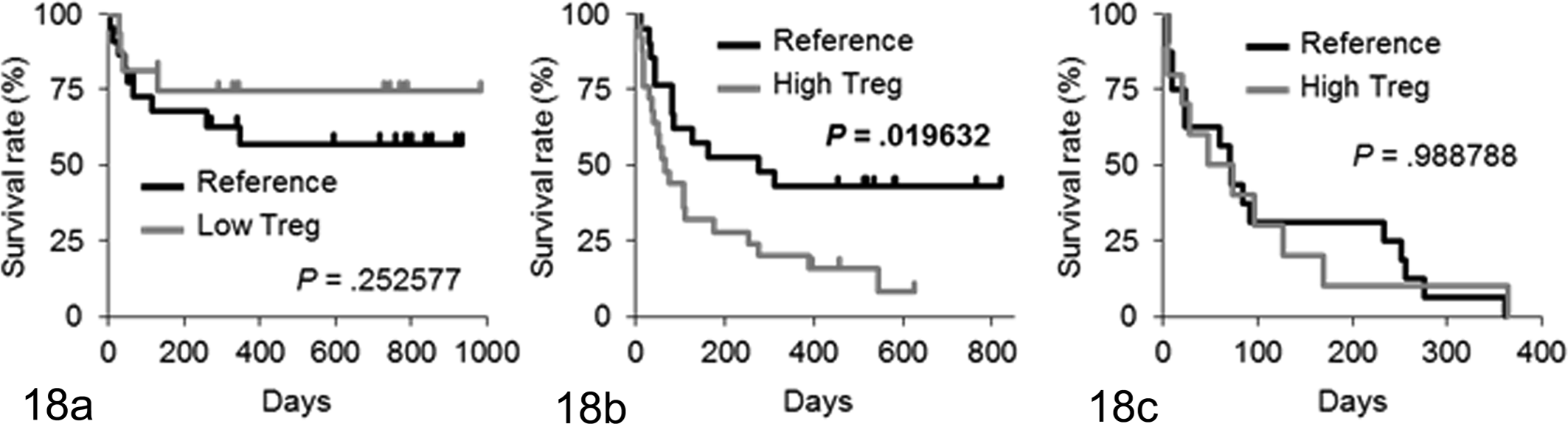
Kaplan-Meier analyses of overall survival according to the number of regulatory T cells (Tregs) in (a) dogs with idiopathic inflammatory bowel disease, (b) dogs with intestinal small cell lymphoma, and (c) dogs with intestinal large cell lymphoma. Cases were classified as having a high, low, or reference density of forkhead box P3–positive Tregs relative to the mean ± 2SD in healthy dogs (reference total Tregs, 0.5531–10.6775 cells per 62 500 μm2). In dogs with inflammatory bowel disease, 27 dogs were classified into the reference Treg group, and 21 were classified into the low Treg group. In dogs with small cell lymphoma, 21 dogs were classified into the reference Treg group, and 25 were classified into the high Treg group. In dogs with large cell lymphoma, 18 dogs were classified into the reference Treg group, and 12 were classified into the high Treg group.
Discussion
In this study, we observed altered numbers of Foxp3-positive Tregs in dogs with IBD and intestinal lymphoma. The number of Foxp3-positive cells in the duodenal mucosa of dogs with IBD was decreased. This observation is consistent with the results of a previous immunohistochemical study. 27 In addition, the expression of the Treg-related immunosuppressive cytokine IL-10 in dogs with IBD was significantly lower than in healthy dogs and dogs with intestinal lymphoma. A previous study also reported that IL-10 expression was decreased in the small intestine of dogs with IBD. 26 These results suggest that a decrease in the Treg number in the gut plays an important role in the pathogenesis of canine IBD via the disruption of mucosal tolerance. In humans, the number of Tregs in peripheral blood is lower in IBD patients than in healthy donors, whereas the Treg number is increased in mucosal IBD lesions. 37,58,60,62 A redistribution of circulating Tregs into the inflamed mucosa in response to active inflammation could explain this discrepancy. However, canine Tregs are decreased not only in peripheral blood 61 but also in intestinal lesions. Therefore, the underlying pathogenesis of canine IBD might be different from that of human IBD.
Unlike IBD, intestinal lymphoma in dogs, especially the small cell type, was characterized by an increased number of Foxp3-positive cells. A significant positive correlation was observed between the number of Foxp3-positive cells and the clinical severity in intestinal small cell lymphoma cases. Furthermore, poor prognosis was associated with a high density of Foxp3-positive Tregs in small cell lymphoma. Immunofluorescence showed that Foxp3-positive cells in small cell lymphoma did not express Ki-67, indicating that the Foxp3-positive cells were not neoplastic cells but infiltrated Tregs. In dogs with intestinal large cell lymphoma, some Ki-67-positive cells expressed Foxp3. Given that the Treg number did not correlate with prognosis in large cell lymphoma, the neoplastic cells might have originated from Tregs and lost their Foxp3 expression and immunosuppressive functions. Another possible explanation is that the overall survival for large cell lymphoma was too short to detect correlation between the prognosis and lesional Foxp3-positive Treg number. Accumulating evidence indicates that Tregs suppress immune responses to tumors and worsen the prognosis in dogs as well as in mice and humans. Studies have shown increased percentages of peripheral blood Tregs in dogs with cancer. 4,24,25,43 Tumor-infiltrating Tregs have been reported in dogs with osteosarcoma, 5 melanoma, 59 mammary carcinoma, 29,44 seminoma, 28 and multicentric B-cell lymphoma. 39,47 To our knowledge, this is the first study to describe tumor-infiltrating Tregs and their negative prognostic impact in intestinal T-cell lymphoma. Recent studies have also shown that a high Foxp3-positive Treg number is a negative prognostic factor in various types of canine tumors. 5,29,44,47 Phenotypic and functional analyses have characterized Foxp3-positive Tregs in dogs and demonstrated that they have properties similar to those of human Foxp3-positive Tregs and a potent ability to suppress the proliferation of responder T cells. 31,48 Collectively, these observations suggest that Tregs that migrate into the tumor microenvironment play a significant role in the prognosis of canine cancers, including intestinal small cell lymphoma, by suppressing local antitumor immune responses.
Contrary to our expectation that a low Treg number would be a negative prognostic factor in canine IBD, we failed to identify a significant correlation between intestinal Treg number and clinical severity or prognosis in dogs with IBD. The possible explanations for the lack of correlation are as follows: (1) a low Treg number in the IBD lesion might not correlate with clinical severity; (2) the sample size might be too small to detect a significant correlation; or (3) treatment with immunosuppressive drugs might restore the Treg number. A recent study suggested that the number of intestinal Foxp3-positive cells increases in dogs with IBD after treatment with a multistrain probiotic product. 50 However, combination therapy with metronidazole and prednisolone did not restore the number of Foxp3-positive cells in dogs with IBD, although the number of TGF-β-positive cells increased after the combination treatment. 50 Additional studies are needed to clarify the association between Tregs and prognosis and determine the effect of prednisolone on the number of Tregs in dogs with IBD.
Although the mechanism responsible for the decreased Treg number in dogs with IBD remains unknown, our previous study showed that TGF-β expression was decreased in canine IBD lesions. 35 Stimulation of cultured naive T cells with anti-T cell receptor antibodies and TGF-β induced the development of Foxp3-positive Tregs in vitro. 10,16 Furthermore, in vivo TGF-β neutralization leads to a deficiency in functional Tregs, uncontrolled leukocyte activation, and systemic inflammatory disease in mice. 11,17,38,41 These data suggest that TGF-β is essential for Treg differentiation and that impaired TGF-β signaling in canine IBD lesions contributes to the decrease in Tregs. In dogs with intestinal lymphoma, an increased Treg number was observed in the lesions. As a likely mechanism by which Tregs infiltrate tumor tissues, it has been proposed that neoplastic cells and/or tumor-infiltrating macrophages produce Treg-attractive chemokines. 42 The chemokines CCL22 and CCL28 promote the migration of Tregs into tumor tissues through their receptors, CCR4 and CCR10, respectively. 12,15,57 Other chemokine CXCR3 ligands, such as CXCL9, CXCL10, and CXCL11, also induce Treg infiltration. 49 Further research investigating the role of these chemokines in Treg migration in dogs with intestinal lymphoma is necessary.
Because differentiating between lymphocytic-plasmacytic enteritis and intestinal lymphoma, especially of the small lymphocytic type, is often difficult via hematoxylin and eosin–stained sections alone, IHC and PARR were used with histopathologic examination to assess the clonal expansion of the lymphocytes. 9,30 In this study, 48, 46, and 30 dogs had IBD, small cell lymphoma, and large cell lymphoma, respectively. The clinical severity was significantly higher and the overall survival, significantly shorter, in the order of large cell lymphoma, small cell lymphoma, and IBD. Therefore, we think that the diagnosis was valid.
One limitation of this study is that the dogs in each group were not well matched by age. There was a significant difference in age distribution among dogs with IBD, small cell lymphoma, and large cell lymphoma, although the sex, body weight, and serum albumin concentration did not differ significantly among the 3 groups. A previous study observed age-related changes in duodenal Foxp3-positive cells: the number of Tregs was higher in middle-aged dogs than in juveniles and older individuals. 27 In this study, the dogs with IBD were younger than the dogs with intestinal large cell lymphoma. However, the age difference might not matter in this study because many IBD dogs were middle-aged—the age when the number of Tregs is greatest. Another limitation of this study is that IHC for Foxp3 was the sole means by which intestinal Tregs were identified. Although Foxp3 has been established as a specific Treg marker, further characterization of the expression of CD4 and CD25 allows for accurate identification of Tregs. 48 However, clear nuclear staining of Foxp3 was observed in the canine intestine. In addition, significant positive correlations were observed between Treg number and IL-10 mRNA expression. Moreover, double-labeling immunofluorescence revealed that canine intestinal Foxp3-expressing cells were CD3-positive, CD20-negative, and granzyme B–negative cells—thus, a subset of helper T cells. Therefore, we think that the intestinal Foxp3-positive cells were Tregs. Additional studies, based on flow cytometry or multilabeling immunofluorescence of frozen sections, will be performed to confirm that the cells are Tregs.
Tregs constitutively express the high-affinity IL-2 receptor CD25, and some studies have shown that administration of anti-CD25 mAb depletes Tregs and eradicates tumors in murine models. 45,56 However, in humans, the antitumor effects of Treg depletion via an anti-CD25 mAb remain controversial because the number of activated effector T cells that also express CD25 decreases. The chemokine receptor CCR4, which is expressed in tumor-infiltrating effector Tregs, is another candidate for Treg depletion. A recent study showed that depletion of effector-type Tregs from cancer patients using an anti-CCR4 mAb evokes potent antitumor immune responses in vitro and in vivo. 57 These data suggest that depletion of Tregs or manipulation of their suppressive functions is a potential therapeutic strategy for tumor eradication.
Conclusions
We detected a decrease in the number of Foxp3-positive Tregs in the lesions of dogs with IBD. In dogs with intestinal small cell lymphoma, we found increased numbers of Foxp3-positive Tregs, which was associated with a negative prognosis. These results indicate that IHC for Foxp3 can distinguish lymphocytic-plasmacytic enteritis from small cell lymphoma. Additional studies are required to investigate the accuracy of this method in distinguishing between inflammation and lymphoma and to elucidate the molecular mechanisms underlying these findings.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant-in-Aid for Science Research from the Japan Society for the Promotion of Science (KAKENHI 23380182).
