Abstract
The diagnosis of pancreatic disease in small animal veterinary patients is complicated by nonspecific clinical signs and the limitations of diagnostic testing. Pancreatic cytology is a potential diagnostic tool, but safety and diagnostic yield are not well characterized in large patient cohorts. We hypothesized that pancreatic fine-needle aspiration (FNA) in dogs would frequently generate diagnostic-quality samples and subsequent adverse medical events would be uncommon. Ninety-two client-owned dogs undergoing pancreatic FNA for clinical diagnostic evaluation were identified retrospectively by a computer search for pancreatic cytology submissions. Archived slides were reviewed by a single board-certified clinical pathologist using a predetermined descriptive scheme. Medical records were reviewed for adverse events 48 hr following FNA, for concurrent procedures and diagnosis in patients with adverse events and for histology results. Diagnostic yield was calculated as the % cases in which a cytologic diagnosis could be achieved; correlation with histology or other confirmatory testing was determined when possible. Diagnostic yield was 73.5%, and the major pathologic process identified cytologically correlated with confirmatory testing in 10 out of 11 cases. There were 7 adverse events, all in dogs with significant comorbidities or undergoing other invasive procedures. Pancreatic FNA in dogs has a good diagnostic yield and a low rate of clinical complications in a large case series of dogs. Correlation of cytology and histology results was high in a limited number of cases.
The diagnosis of pancreatic disease in dogs and cats is clinically challenging due to variable presenting clinical signs and the lack of highly sensitive and specific diagnostic tests. Several diagnostic methods have been reported to detect canine pancreatitis with varying sensitivities, including serum amylase and lipase (54–73%), radiography (24%), and ultrasonography (68%). 10 Canine-specific pancreatic lipase (cPL) has been reported to have high sensitivity and specificity; however, there is the potential for a false-positive diagnosis in up to 40% of dogs presenting with acute abdominal signs. 9 Laparoscopy and contrast enhancing ultrasonography techniques may improve visualization of lesions.11,16 Even histologic diagnosis is problematic in canine pancreatitis because focal and randomly distributed lesions can be present in apparently healthy dogs.2,13,14
Pancreatic cytology is another relatively noninvasive diagnostic tool, although concerns about iatrogenic injury and lack of data regarding the probability of obtaining a diagnostic sample are currently limiting. A retrospective case control study, published in 2014, of 73 cats undergoing ultrasound-guided fine-needle aspiration (FNA) of the pancreas, found no difference between study and control cats in the rate of complications within 48 hr of the procedure, survival to discharge, or length of hospital stay. 6 Another study of 27 healthy Beagle dogs documented that ultrasound-guided FNA of the pancreas did not cause immediate adverse effects, and FNA and biopsy did not cause a change in cPL values with short-term followup. 5 Endoscopic ultrasound-guided FNA also appears to be safe in healthy dogs. 12 Pancreatic FNA has been used anecdotally to diagnose pancreatic pseudocysts, exocrine pancreatic neoplasia, and islet cell tumors.2,7,8,17 While initial data suggests that pancreatic FNA is safe, more information is needed to establish diagnostic yield, normal and abnormal cytologic findings, complications, and diagnostic accuracy in larger scale studies including a variety of pancreatic diseases. Objectives of the current study were 1) to report the diagnostic yield and cytologic findings of pancreatic FNA in sick dogs and 2) to report postprocedure complications after ultrasound-guided FNA of the pancreas in a large group of dogs.
A retrospective review of the electronic medical record database was conducted at the University of Minnesota Veterinary Medical Center (St. Paul, Minnesota) over a 5-year period (September 2004 to September 2009) to identify client-owned dogs for which pancreatic cytology samples were submitted. Most were acquired by percutaneous ultrasound-guided FNA but 2 were obtained intraoperatively. Cases were excluded when cytology slides were not available for review. Date of presentation, signalment, recorded diagnoses, histologic findings (when available), and adverse events within 48 hr of pancreatic aspirate procedure (death, new onset of fever, blood transfusion, diagnostic peritoneal lavage, or recheck ultrasound) were noted. Two to 3 ultrasound-guided samples are typically obtained as previously described with a 20- or 22-gauge hypodermic or spinal needle using either aspiration or fenestration techniques, or a combination. 3
Pancreatic cytology samples were evaluated by a single board-certified veterinary clinical pathologist (LCS) with the previously described healthy Beagle dogs as a reference population. 5 Cellularity was characterized as poor, adequate, or optimal, and blood contamination as none, mild to moderate, or marked. Greater than 80% neutrophils indicated suppurative inflammation and <80% neutrophils with other cells indicated mixed inflammation. The presence of mesothelial cells, bacteria, and background material including zymogen granules, proteinaceous fluid, mineralization, and necrotic debris was described. Low nucleated cellularity and abundant fluid indicated a cystic lesion. The presence of exocrine and endocrine pancreatic cells was noted. Previously described diagnostic criteria for exocrine and endocrine pancreatic neoplasia were used. 1 Mild to moderate cytologic atypia with concurrent inflammation was considered most likely reactive unless marked, although it is recognized that tumor-associated inflammation is common in many malignancies. Each submission was assigned a single categorical predominant cytologic diagnosis: 1) normal exocrine pancreas, 2) exocrine pancreatic hyperplasia, 3) inflammation, 4) malignant neoplasia, 5) cystic lesion, or 6) necrosis. Slides were designated as inconclusive if diagnostic interpretation could not be achieved. Diagnostic yield was defined as the proportion of cases that were of sufficient quality to reach a cytologic conclusion.
Ninety-four submissions were identified from 92 dogs with 2 repeat aspirates 3 and 7 days apart, respectively. One to 10 slides were available for review for each submission. Median patient age was 9.8 years (range: 2–17 years) with 48 spayed females, 41 neutered males, 2 intact males, and 1 intact female. Breed distribution was Cocker Spaniel (11), Labrador Retriever (10), Shetland Sheepdog (6), Miniature Schnauzer (5), Golden Retriever (4), Pug (4), West Highland White Terrier (4), German Shepherd Dog (3), Jack Russell Terrier (3), 2 each of Bichon Frise, English Springer Spaniel, Keeshond, Miniature Pinscher, Pomeranian, Vizsla, Yorkshire Terrier, and mixed breed, and 1 each of 26 other breeds.
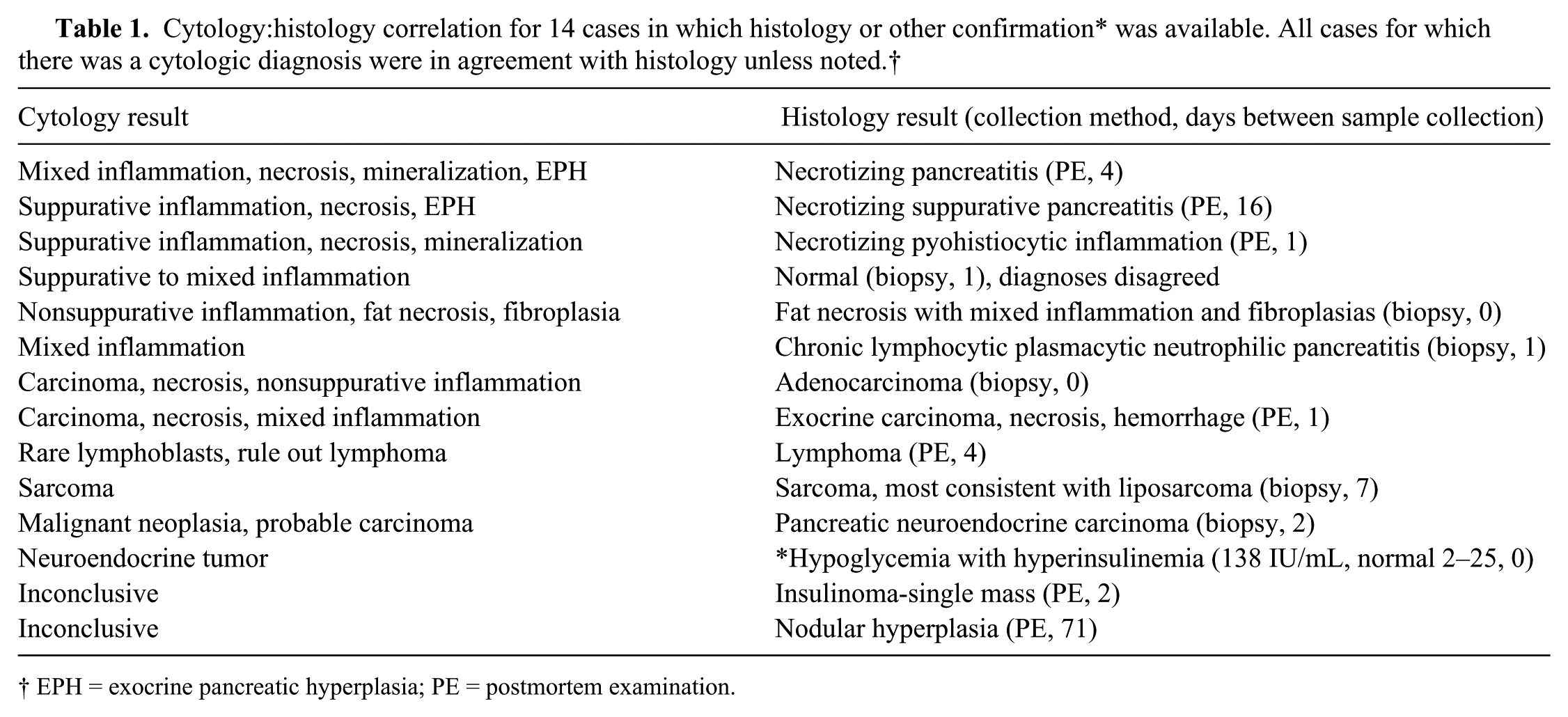
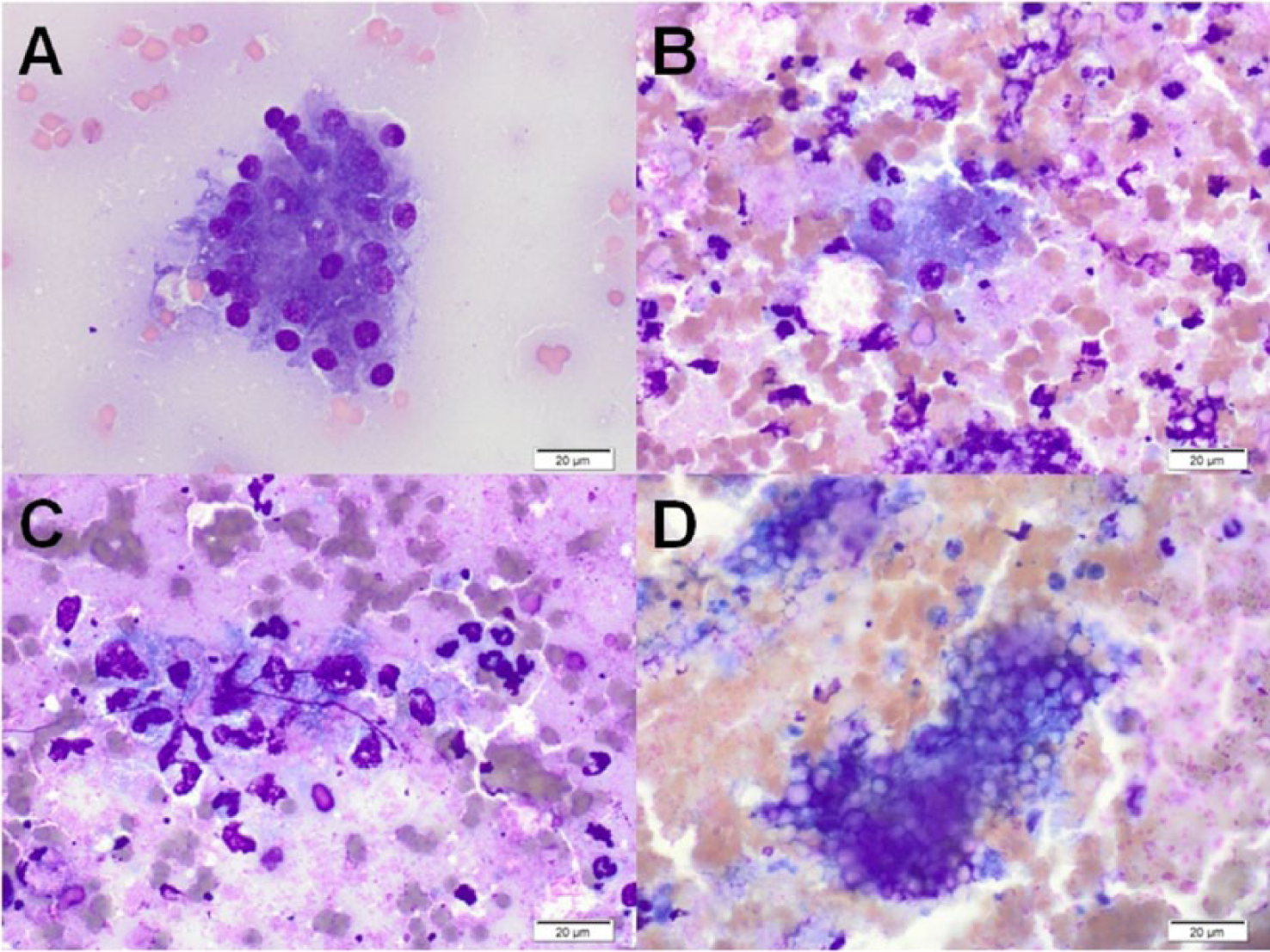
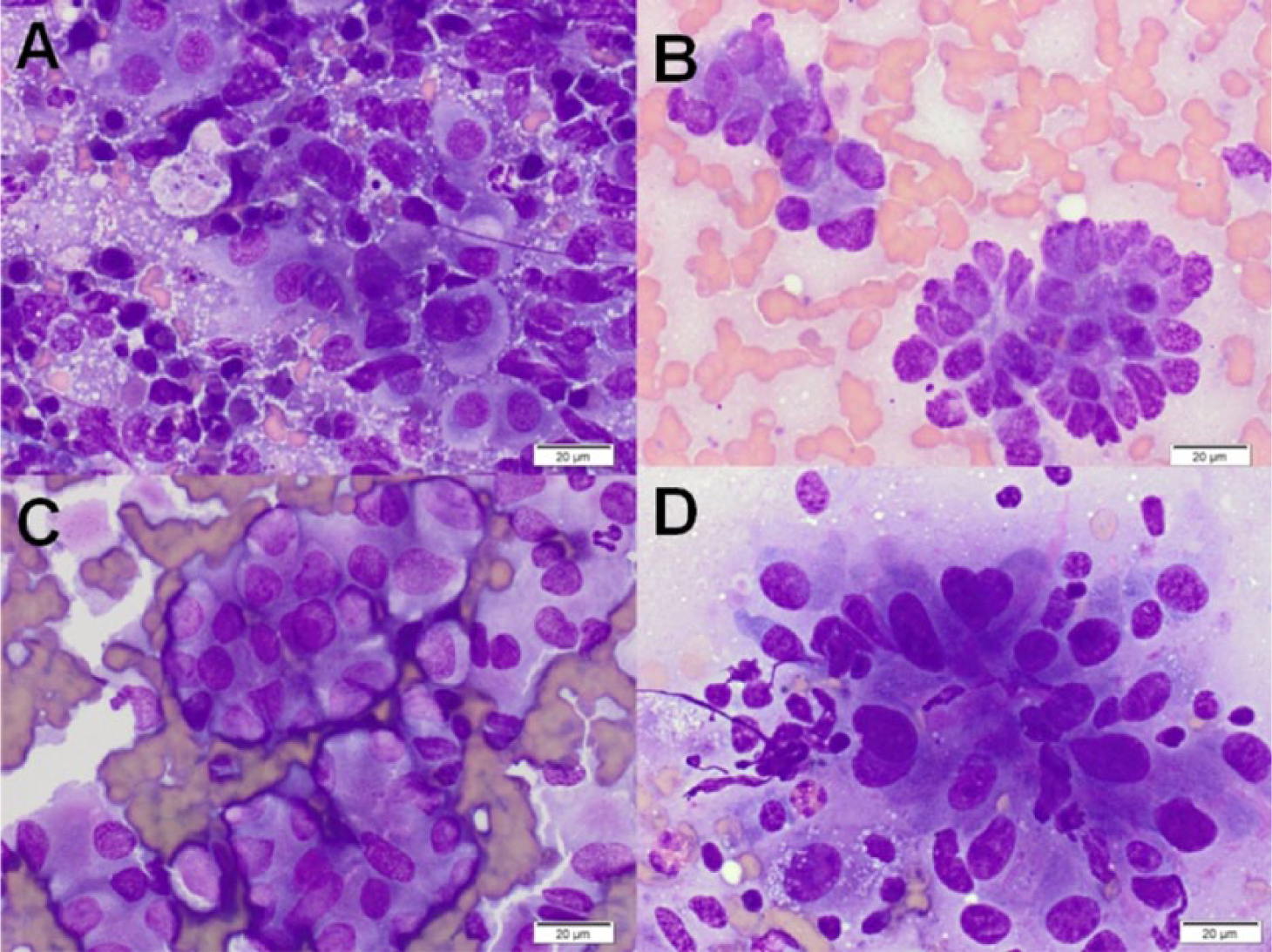
Forty-six samples (48.9%) were optimally cellular, 25 (26.6%) were adequately cellular, and 23 (24.4%) were poorly cellular. Twenty-five samples (26.5%) were nondiagnostic. In 3 cases considered adequately cellular, a diagnostic conclusion could not be reached due to poor cell preservation or other artifacts, resulting in an overall diagnostic yield of 73.5%. Blood contamination was common (82%). Mesothelial cells were observed in 7 cases, and hepatocytes in 1. Background material included proteinaceous fluid (37 cases), necrotic debris (20 cases), lipid (18 cases), mineralization (14 cases; 8 had both necrotic debris and mineral), free zymogen granules (6 cases), mucin (5 cases), and cytoplasmic fragments (1 case of lymphoma). The predominant pathologic processes identified cytologically were: inflammation (31; 15 mixed, 11 neutrophilic, 1 of which was septic, 5 nonsuppurative), neoplasia (14; 8 carcinoma, 3 lymphoma, 2 neuroendocrine, 1 sarcoma), normal pancreatic tissue (10), exocrine pancreatic hyperplasia (9), cystic lesion (4), and necrosis (1). Histology was available for 13 dogs (see Table 1), but 2 had inconclusive cytology. In 1 additional case, a cytologic diagnosis of neuroendocrine neoplasia was considered confirmed by hypoglycemia and concurrent hyperinsulinemia. Cytologic and histologic findings (or endocrine testing) agreed in all but 1 case (10/11 cases, 91%). Figure 1 shows representative cytologic findings from histologically confirmed cases of pancreatitis, and Figure 2 shows representative images from histologically confirmed pancreatic neoplasia.
Cytology:histology correlation for 14 cases in which histology or other confirmation* was available. All cases for which there was a cytologic diagnosis were in agreement with histology unless noted.†
EPH = exocrine pancreatic hyperplasia; PE = postmortem examination.
Representative cytology images from histologically confirmed pancreatitis. Wright–Giemsa stain. Bar = 20 μm.
Representative cytology images from histologically confirmed neoplasms of the pancreas. Wright–Giemsa stain. Bar = 20 μm.
Of the 94 total aspiration procedures, no subsequent adverse effects were noted in the record for 87 (92.6%). The details of the adverse events recorded for 7 dogs are detailed in Table 2. Six of these 7 dogs had additional diagnostic procedures performed concurrently, and all had significant comorbidities. Four did not survive to discharge. None of the 7 dogs that underwent surgical biopsy of the pancreas nor the 2 dogs undergoing repeat aspirates had adverse events described in the medical record.
Adverse events after pancreatic fine needle aspirate procedure.*
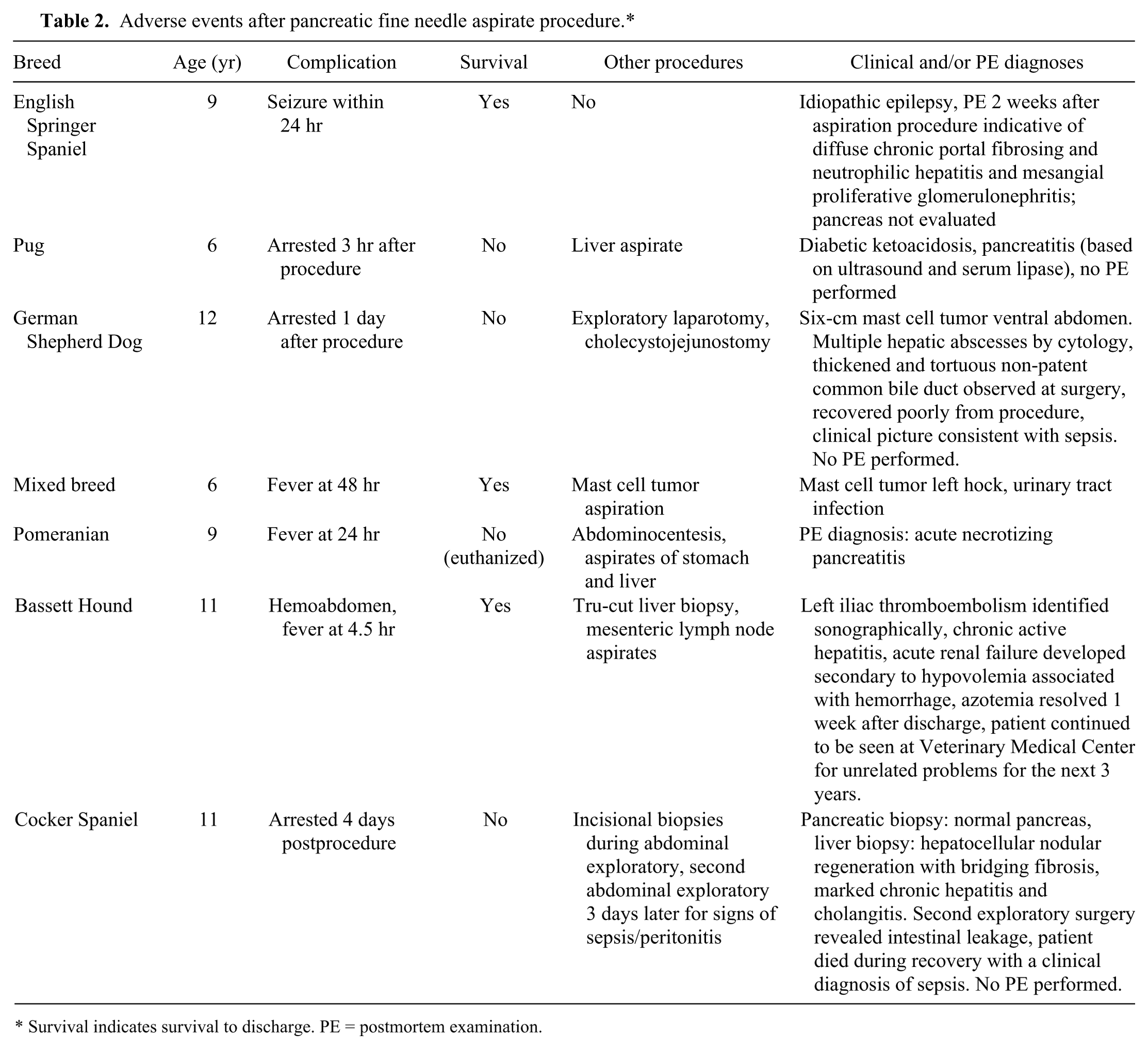
Survival indicates survival to discharge. PE = postmortem examination.
In conclusion, diagnostic yield in this series of dogs was good, with 73.5% adequate to support a specific cytologic interpretation, much higher than ultrasound-guided samples (0%) but similar to intraoperative samples in healthy Beagles. 5 The markedly improved yield in sick dogs could be due to inhibition of exfoliation by extensive fibrovascular stroma in the healthy pancreas. 5 This yield is slightly higher than the 67% reported for cats. 6 The major pathologic process agreed with confirmatory testing in 11 out of 12 cases with diagnostic cytology samples. A bias toward having histology results for more severe cases could have contributed to the very high correlation, and more systematic prospective studies are needed to evaluate the diagnostic accuracy of pancreatic cytology. Ideally, these studies would incorporate long-term clinical followup to help address the challenges of histologic diagnoses, including safety.13,14 No clinical complications were noted in a retrospective study of laparoscopic pancreatic biopsy in 20 cats and dogs; however, a study of surgical pancreatic biopsy in 44 dogs and cats reported complications in 10 animals, including 5 cases of postsurgical pancreatitis.15,18
Clinical complications of pancreatic FNA in this case series were very low at 6.3%. The high rate (6/7) of concurrent diagnostic procedures, such as percutaneous hepatic FNA or biopsy in dogs experiencing complications made it difficult to definitively ascertain the contribution of the pancreatic FNA to the adverse events recorded. Other invasive procedures and progression of comorbidities or pancreatic conditions were most likely the primary causes of complications in most, if not all, cases. Comorbidities are expected based on a large retrospective case series, demonstrating that dogs with pancreatitis were more likely to have concurrent hepatobiliary, endocrine, or neurologic disease compared with a reference necropsy population. 4 Dogs experiencing complications in our study had a similar spectrum of concurrent conditions that were likely to have complicated the clinical situation. Limitations of this study include the retrospective nature and the limited confirmatory testing. While we report a low rate of complications, this is not a systematic case-controlled study, so results should be interpreted cautiously. This study corroborates smaller case series in the literature suggesting that cytology can be a safe and effective diagnostic tool for the evaluation of canine pancreatic disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
