Abstract
Objectives
The aim of this study was to evaluate two-dimensional and Doppler ultrasonographic changes of the ovary and uterus during estrus and the early post-estrus period in domestic cats.
Methods
Two-dimensional and Doppler ultrasonographic evaluations of the ovaries and uterus were performed in seven queens on days 1, 3 and 5 of estrus, and 5 days after estrus (AE5).
Results
On day 1, 5.4 ± 0.5 follicles of 2.1 ± 0.1 mm were detected progressively increasing in number and size up to day 5 and then decreased on AE5 (P <0.05). A maximum follicular diameter of 4.1 ± 0.1 mm was achieved on day 5. Both during and after estrus, the uterus was generally hypoechoic compared with the surrounding tissues and delineated by a thin hyperechoic line corresponding to the serosa. In some animals, the uterine layers were distinguished during and after estrus. The blood flow waveforms of the intraovarian and uterine arteries were characterized by a systolic peak and diastolic flow extending throughout the remainder of the cardiac cycle to the next systole. In the uterine artery waveforms, the early diastolic notch was mild or absent during most estrous observations. The resistance index of the intraovarian arteries decreased up to day 5, and then increased on AE5 (P <0.05). The resistance index of the uterine arteries was lower during estrus than in the post-estrus period (P <0.05).
Conclusions and relevance
It is concluded that in the domestic cat, follicular number and diameter as well as ovarian and uterine blood flow changed during and immediately after estrus. Doppler ultrasound proved suitable to evaluate the hemodynamic changes involved in the cyclic remodeling of ovarian and uterine tissues that occurs during and after follicular growth in domestic cats.
Introduction
Ultrasonography is an imaging diagnostic method that allows non-invasive visualization of the internal reproductive organs. 1 In females of domestic species, the capability of this technique for data acquisition and statistical testing of hypotheses has enlightened reproductive research.2,3 In assisted reproductive technologies, ultrasound is essential to monitor ovarian activity prior to artificial insemination or follicular puncture.4,5 In female cats, B-mode ultrasound evaluation of the reproductive tract has been mainly carried out to assess physiological and pathological uterine conditions.6–8 Conversely, ultrasonographic ovarian changes have been scarcely documented in this species.9–11 During the feline estrous cycle, the ovaries and uterus undergo morphological changes under hormonal influences. 12 Studies including both ovarian and uterine ultrasonographic evaluations throughout the estrous cycle are missing, probably due to the small size of these organs in cats. 10 Only one feline study carried out a two-dimensional follow-up of the ovaries during estrus, but uterine examination was not performed. 9 The accurate identification of a two-dimensional uterine image during the estrous cycle is essential to differentiate physiological changes from uterine diseases.
In mammalian species, cyclic remodeling of ovarian structures and vasculature occurs during each reproductive cycle.13,14 The development of Doppler ultrasound extended the scope of ultrasonographic imaging from an anatomical to a physiological basis. During the past few years, Doppler ultrasonography has been carried out in theriogenology to provide valuable information about organ perfusion.15,16 This technique is routinely used for both ovarian and uterine examination in cows,17,18 mares19,20 and sows, 21 improving reproductive management. Changes in the uterine artery resistance index (RI) and waveform morphology during the various stages of the estrous cycle have been described in the dog using the Doppler technique. 22 In queens, the uterine blood flow has been assessed during normal pregnancy, puerperium, abortion and pyometra.7,23–26 Doppler ultrasonographic characterization of domestic cat ovaries has been carried out in pubertal cats, but information on post-pubertal queens is lacking. 27 It is hypothesized that during estrus and shortly after it, feline ovaries and uterus develop structural modifications associated with vascular changes. Thus, the aim of this study was to evaluate two-dimensional and Doppler ultrasonographic changes of the ovary and uterus during estrus and the early post-estrus period in feline species.
Materials and methods
Animals
Seven female domestic shorthair cats, 10.3 ± 0.9 months old, weighing 2.6 ± 0.08 kg, were included in this study. All the queens had a history of at least two previous estrous cycles. The cats were kept and reared free in indoor enriched catteries (two rooms 4 × 3 m with a 14L:10D photoperiod) and fed with dry commercial premium food and water ad libitum.
This study was reviewed and approved by the Animal Care and Use Committee of the Faculty of Veterinary Sciences of the National University of La Plata, Argentina (IACUC number 56-2-16T) and all experiments were conducted under the guidelines established in The Guide for The Care and Use of Laboratory Animals, USA.
Estrus detection
The animals were observed daily for typical estrous behavior (ie, rubbing on objects, rolling, lordosis, tail lateralization and vocalizing) and vaginal cytology was carried out three times a week, or whenever estrous signs appeared. The onset of estrus (day 1) was diagnosed by the first appearance of more than 80% keratinized vaginal epithelial cells and a clean background in vaginal smears for at least three successive days. 28 During estrus, vaginal cytology was performed daily. The end of estrus was determined by the first appearance of more than 50% intermediate epithelial cells in the vaginal smears and the absence of sexual behavior. 12
Ovarian and uterine ultrasonographic evaluation
Two-dimensional ultrasonography, color-coded and pulsed-wave Doppler evaluations of the ovaries and uterus (Nemio XG, 14 MHz linear-array transducer; Toshiba, Japan) were performed on days 1, 3 and 5 of estrus, and 5 days after estrus (AE5). The female cats were positioned in dorsal recumbency or seated upright on the operator’s lap without sedation and the hair of the ventral skin was clipped after 5 mins of acclimatization. Acoustic gel was applied to the transducer and coupled directly to the skin. The ovaries were identified near the caudal pole of the ipsilateral kidney. 29 The operator was trained to recognize ovarian structures.
Longitudinal and transverse diameters (mm) of the ovaries were measured. All anechoic >1 mm spherical structures were considered to be follicles. The number and mean outer diameter of these ovarian structures per queen were recorded. The maximum follicular diameter (mm) was also registered. The presence or absence of layering (visualization of different uterine layers) and the uterine body transverse diameter (mm) were documented.
For ovarian Doppler assessment, the intraovarian arteries of the right and left ovaries were detected within the ovarian tissue using color Doppler and pulsed-wave Doppler was performed to obtain the waveforms. 27 For uterine Doppler assessment, two-dimensional ultrasonography was used to identify the uterine body in a transversal axis. The main right and left uterine arteries adjacent to the uterine body were localized using color Doppler and pulsed-wave Doppler was performed to obtain the waveforms. 24 To minimize variability, three uniform consecutive waveforms were recorded. Standardized presets (frequency of 14 MHz, depth of 3–5 cm, angle of insonation 0–20°, pulse repetition frequency of 4.7–5.8 kHz, color gain 15–18) were used throughout the evaluations. Peak systolic velocity (PSV, cm/s) and end diastolic velocity (EDV, cm/s) of right and left intraovarian and uterine arteries were recorded to automatically calculate the RI. 30
Statistical analysis
Normal distribution of the data was determined by the Shapiro–Wilk test. PSV, EDV and RI of the left and right intraovarian and uterine arteries were compared using the Student’s t-test. As no differences were found (P >0.1), values were averaged. Repeated measures ANOVA followed by the Tukey’s test were carried out to evaluate the two-dimensional and Doppler ultrasonographic parameters, which included the average of PSV, EDV and RI of the left and right intraovarian and uterine arteries. The mathematical model included the main effect of time (SPSS 18.0; SPSS). All the variables were expressed as mean ± SEM and the level of significance was set at P <0.05.
Results
In the seven queens in this study, estrus lasted 4.7 ± 0.1 days. On AE5, no corpora lutea were ultrasonographically detected in the ovaries.
In all two-dimensional ultrasonographic images, the ovaries appeared ovoid and hypoechoic compared with the surrounding tissues (Figure 1). Although the right ovary was more difficult to identify than the left one, it could be assessed in all cases. In these animals, neither longitudinal nor transverse ovarian diameters changed during the study period (P >0.1; Table 1). On day 5, the mean maximal ovarian longitudinal diameter was 11.8 ± 1.3 mm and the largest ovary was 15.2 mm long. Multiple submillimetric anechoic spherical structures were variably detected throughout the course of the experiment. On days 1, 3, 5 and AE5, the total number of follicles in each queen were 5.4 ± 0.5, 5.7 ± 0.6, 5.6 ± 0.3 and 3.5 ± 0.6, respectively (P <0.05). The mean and maximum follicular diameter gradually increased throughout estrus and then decreased on day AE5 (P <0.05).
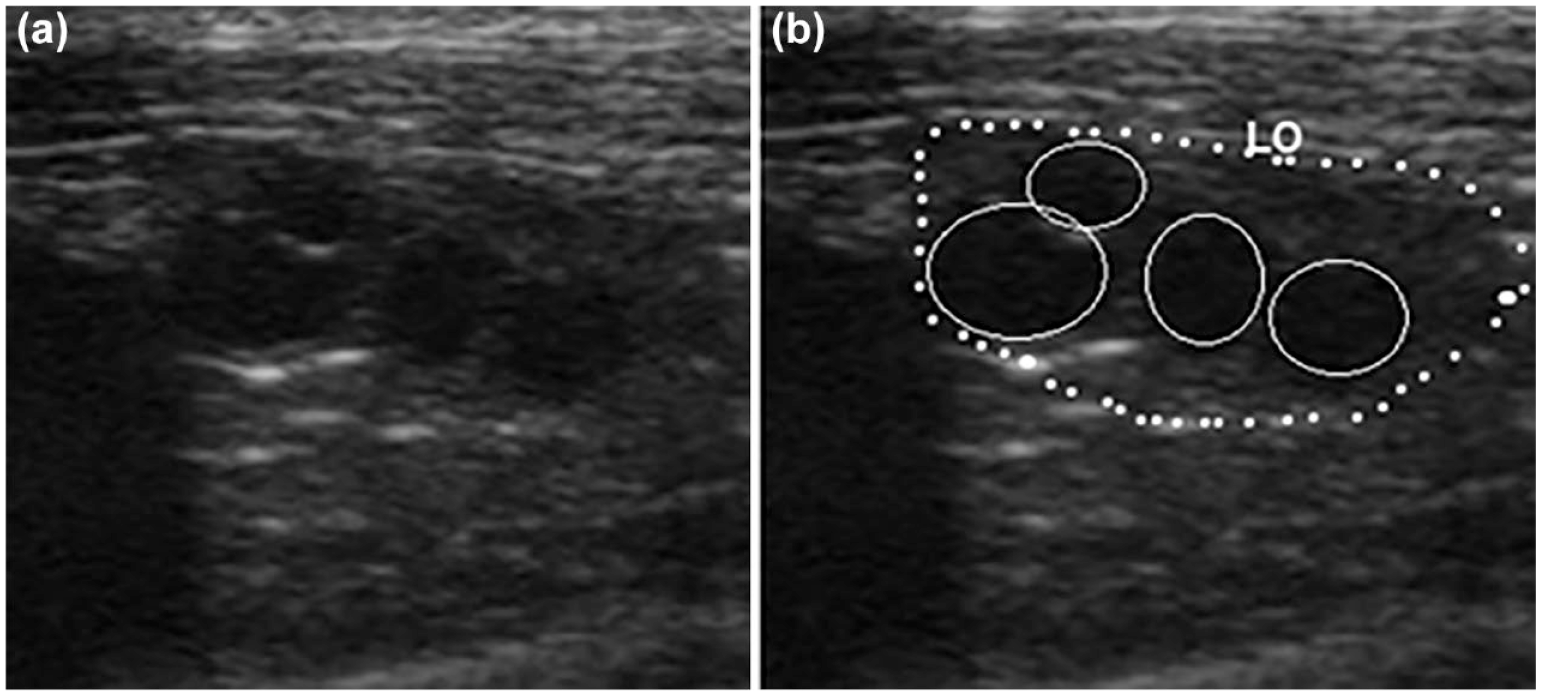
Longitudinal image of the left ovary (LO) in a selected female cat on day 3 of estrus. The ovary is outlined with a dotted white line on (b). (a) and (b): four anechoic structures >1 mm (follicles; solid white line)
Ultrasonographic ovarian measurements (mean ± SEM) during and after estrus in seven healthy queens

Different letters within a column indicate differences (P <0.05) among days
SEM = Standard error of the mean: AE5 = 5 days afrer estrus
Both during and after estrus, in 5/7 queens (71.4%) the uterus was hypoechoic compared with the surrounding tissues and delineated by a thin hyperechoic line. In 2/7 animals (28.6%), the uterine layers were distinguished during and after estrus. When observed, two layers were detected: the endometrium, which was more hypoechoic than the myometrium, which was echoic. No uterine morphological differences between the estrus and interestrus period were observed. The uterine body transverse diameter remained unchanged throughout the time points (P >0.1).
The arterial blood flow waveforms in the ovaries presented a systolic peak and diastolic flow extending throughout the remainder of the cardiac cycle to the next systole (Figure 2). The morphology of the waveforms did not change throughout the course of the experiment. End diastolic velocity increased on day 5 and then decreased after estrus (P <0.05), while PSV remained unchanged (P >0.1). The RI of the intraovarian arteries decreased up to day 5 and rose on AE5 (P <0.05; Figure 3).

Pulsed-wave Doppler ultrasound recording of intraovarian arteries of a female cat during estrus.

Resistance index of the intraovarian arteries of seven queens evaluated on days 1, 3 and 5 of estrus and 5 days after estrus (AE5). Different letters indicate differences (P <0.05) among days
The uterine artery waveforms were also characterized by a systolic peak and diastolic flow extending throughout the remainder of the cardiac cycle to the next systole (Figure 4). This pattern was detected in all the queens and time points; nevertheless, an early diastolic notch was mild or absent during most estrous observations. End diastolic velocity decreased after estrus (P <0.05), while PSV did not change (P >0.1). The RI of the uterine arteries was lower during estrus than in post-estrus (0.53 ± 0.01 vs 0.59 ± 0.00, respectively; P <0.05).

Uterine artery waveforms of a female cat (a) during estrus and (b) 5 days after estrus. The early diastolic notch was mild or absent during most estrous observations
Discussion
To the best of our knowledge, the results of this article provide the first ultrasonographic report of the structural and blood flow changes of the feline ovaries and uterus during and after estrus in an anovulatory cycle.
The sizes of the ovaries and uterine body observed in this study were similar to those reported in previous ultrasonographic studies in pubertal and adult queens.11,27 Despite the architecture differences found in these cycles (ie, follicular growing, thickening of uterine layers), no dimension changes were found in the organs using this equipment and settings. Interestingly, the maximal ovarian diameter was registered on day 5, probably due to the largest follicles being detected on that day. Ultrasound variations of ovarian size were described during the follicular and luteal phases of canine estrous cycle. 31 One limitation of this feline study could be the size of the organs in this species, which are smaller than in bitches.
In these cats, the number of antral follicles detected at the beginning of estrus was similar than that previously reported in domestic cats for the same stage of the estrous cycle. 9 This follicular quantity slightly increased throughout estrus and then decreased after it, evidencing the initial recruitment of a cohort of follicles, some of which may have undergone atresia.32,33 Consistent with previous reports, in these cats estrus was characterized by a cohort of three to seven dominant follicles. 12
Follicular size also increased during estrus, reaching the largest diameter on day 5. This result agrees with a previous ultrasonographic report in the feline ovary, where follicular diameter sharply increased during the first 4 days of estrus and then reached a plateau for 1–2 days and decreased onwards. 9 The follicular size measured for these queens is consistent with previous histologic and ultrasonographic reports in domestic cats.9,11,34,35 In the present study, the maximum follicular size detected was slightly higher that previously reported by ultrasound (4.3 mm vs 4.1 mm). 9 This might be explained by the different observers, equipment and probes. Importantly, oocytes collected from larger follicles show higher maturational ability in vitro in sexually mature cats. 5
Numerous submillimetric anechoic structures were observed in the ovaries throughout the study. These structures may correspond to early antral follicles, which have been histologically described in the feline ovary throughout the estrous cycle.36,37 In all these queens, >1 mm antral follicles were ultrasonographically detected on AE5.
Although the ultrasonographic appearance of the uterus has been described in the domestic cat,10,11 no differentiation between estrus and interestrus images has been reported. In the present study, uterine wall layering was observed during and after estrus probably due to the estrogen influence. 12 The distinction between uterine layers in this species may be challenging due to the small size of the organ.
The waveform morphology of the intraovarian arteries was constant throughout the experiment and always showed a low resistance pattern, which may be explained by the proximity of these vessels with the capillary area of the ovarian tissue. 15 An increase in EDV was detected on day 5, probably due to the reduced resistance in ovarian vessels. Importantly, the angle of insonation used in this study was below 20°. When the angle between the ultrasound beam and the blood flow is close to 0°, a correct estimation of the real value of the blood velocity is obtained. 38
In this study, the increase in follicular number and sizes was associated with a decline in the RI of the intraovarian arteries, probably caused by the increase in EDV with constant PSV. The diastolic component of the cardiac cycle seems to be more accurate in reflecting RI changes than the systolic portion.15,39 This finding indicates that ovarian blood flow resistance decreases in follicular phase. Similar results were found in pubertal estrus in queens. 27 This phenomenon is also similar to that observed in bitches, where intraovarian perfusion increases gradually from proestrus to the preovulatory period. 40 According to previous reports on the three-dimensional architecture of ovarian vessels in murine models, follicular development is accompanied by an increase in vasculature branching. 13 In cows, the ovarian antral follicles blood supply is also closely related to the follicular growth.17,41
In all the queens and time points, the uterine artery waveforms were characterized by a continuous diastolic component. This pattern corresponds to type C waveforms, defined by Dickey (1997), being indicative of low vascular resistance. 15 This type of waveform was observed during most of the stages of the canine estrous cycle, exhibiting an early diastolic notch. 22 Unlike in dogs, uterine waveforms found during estrus in these cats were mostly characterized by a slight or absent early diastolic notch. This result is consistent with a previous report in pregnant queens, where this pattern was also detected in uterine arteries with decreased resistance. 24
These cats presented a lower uterine arteries resistance during estrus than in post-estrus. Conversely to the follicular phase, an early diastolic notch was detected in most of the uterine waveforms on AE5 indicating an increased resistance in this vessel. This finding is consistent with studies in cows, mares and sows and may be due to estrogen influence, among other molecules, on uterine vascular smooth muscle.16,42,43 This estrogenic effect was also postulated in bitches, where an increase in the uterine RI was observed 3 days prior to ovulation at the time of declining estrogens and increasing progesterone concentrations. 22
Conclusions
In the domestic cat, follicular number and diameter, as well as ovarian and uterine blood flow, changed during and immediately after estrus in an anovulatory cycle. Both two-dimensional ultrasound and Doppler ultrasound proved suitable to evaluate the morphological and hemodynamic changes involved in the cyclic remodeling of ovarian and uterine tissues that occur during and after follicular growth in domestic cats.
Footnotes
Conflict of interest
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was partially supported by the Teaching Incentive Program of NULP (grant number 11/V269). The authors are career scientists (PG Blanco and C Gobello) and research fellows (R Vercellini and A Arizmendi) of the National Research Council (CONICET) of Argentina.
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
