Abstract
Objectives
The objective of this study was to evaluate the time to decreased reactivity of the arytenoid cartilages in cats after application of topical lidocaine.
Methods
One hundred and ten mixed-breed cats were randomly assigned to one of five groups based on the time between application of lidocaine and stimulation of the larynx: 5 (T5), 15 (T15), 30 (T30), 45 (T45) or 60 (T60) s. Cats were premedicated with dexmedetomidine, ketamine and buprenorphine. Anesthesia was induced with propofol to effect. Lidocaine 2% (2 mg/kg) was applied topically to the vocal cords using a catheter attached to a syringe under direct laryngoscopy. After lidocaine application, the designated time elapsed and the vocal cords were stimulated with the patient end of an endotracheal tube. Severity of reaction was reported as none, mild, moderate or severe. All cats were intubated after the reactivity score was recorded. Anesthesia was maintained with isoflurane and 100% oxygen while cats were spayed or castrated. Cats were monitored in recovery for signs of respiratory complications and pain.
Results
There was a significant difference in overall reactivity score between T5 and T45 (P = 0.0038). Also, there was a significant difference in the number of cats with no reaction compared with cats with any reaction between T5 and T30 (P = 0.03), as well as between T5 and T45 (P = 0.0028). No cat had a severe reactivity score at T45 or T60. All cats were successfully intubated. There were no complications during intubation, maintenance of anesthesia or recovery.
Conclusions and relevance
As the lowest overall reactivity score occurred at T45, it is recommended to wait at least 45 s after application of topical lidocaine before attempting tracheal intubation.
Introduction
A patent and protected airway is beneficial to provide oxygen and inhalant anesthetic, and to protect against regurgitation and aspiration during general anesthesia. Laryngospasm has been reported in cats following mechanical stimulation from attempts at endotracheal intubation.1–3 Strategies to reduce laryngospasm facilitate successful intubation. 4 Lidocaine 2%, 5% and 10% topically desensitized the larynx allowing easy intubation in cats.2,4 Toxicity from systemic absorption of lidocaine may be of concern in cats as reduced cardiac index and heart rate have been reported after intravenous (IV) injection. 5 Plasma concentration of lidocaine remained within safe limits when cats received 2 and 10 mg of lidocaine topically on the larynx without signs of toxicity. 3
Time to desensitization of the larynx after topical lidocaine application has been reported to be 90–120 s.2,4 Clinical studies and guidelines suggest 60 s as a minimum time.3,6 Clinically, most veterinary professionals may not wait 60 s before an attempt to intubate owing to time constraints and concerns about respiratory depression from induction agents and risk of aspiration. However, there is no evidence that suggests shorter times are effective at reducing laryngeal sensitivity to intubation.
The objective of this study was to assess the number of cats exhibiting reduced laryngeal reactivity in response to stimulation after topical lidocaine administration with longer wait times. We hypothesized that there would be reduced reactivity scores 15–45 s after application of lidocaine.
Materials and methods
Ethical approval was granted from the Midwestern University Institutional Animal Care and Use of Committee (IACUC) (IACUC 2829). All cats enrolled in the study were shelter owned and admitted for sterilization procedures as part of a student surgery laboratory. Written permission was obtained from each shelter.
A total of 110 mixed-breed cats were enrolled. Cats were admitted the day before the procedure and food was withheld overnight. Cats were allowed access to water and a litter box until premedication. All were healthy based on physical examination, packed cell volume, total protein, blood glucose and blood urea nitrogen (Azostix; Siemens). Any cat with any illness was excluded from the study and laboratory. Cats were randomly assigned to one of five treatment groups (22 cats per group) using randomly and blindly drawn pieces of paper from an envelope. Treatment groups were based on the number of seconds between application of lidocaine and stimulation of the larynx: 5 (T5), 15 (T15), 30 (T30), 45 (T45) and 60 (T60) s.
All cats were premedicated with dexmedetomidine 0.008 mg/kg (Dexdomitor; Zoetis), buprenorphine 0.02 mg/kg (Buprenorphine HCl; Par Pharmaceutical) and ketamine (Zetamine; VetOne) 5 mg/kg given by the intramuscular route. Once adequately sedated, cats were brought into the surgery preparation room where they were placed on a conductive heating device (Hot Dog Patient Warming Device; Augustine Biomedical + Design) to maintain body heat. A pulse oximeter probe, electrocardiogram, rectal thermistor and oscillometric blood pressure cuff measuring 30–40% of the circumference of the opposite front leg to the IV catheter were attached to the patient (Bionet; Bionet). Heart rate, blood pressure, pulse oximetry and rectal temperature were monitored and recorded every 5 mins. An oxygen mask was placed over the muzzle of the cat and oxygen flow at 4 l/min was administered until the patient was intubated. A 22 G 1.0 inch over-the-needle catheter (BD Insyte; Becton Dickinson Infusion Therapy Systems) was aseptically placed into either cephalic vein.
Approved study personnel were two board-certified veterinary anesthesiologists and two veterinary technicians (TJ, AdC, KB or KC). With one of these personnel present, depth of sedation was assessed. If the cat’s mouth could be opened, tongue extruded and larynx visualized without resistance, no propofol was administered. If there was resistance, anesthesia was induced with propofol (Propofol; Hospira) given by the IV route to effect to achieve visualization of the larynx. The dose of propofol required was recorded. Time from premedication to topical administration of lidocaine was recorded. A Miller laryngoscope blade was used to visualize the larynx and injectable lidocaine 2 mg/kg (Lidocaine 2%; VetOne) was applied onto the arytenoid cartilages using a 22 G 1.0 inch over-the-needle catheter with the stylet removed attached to a syringe. When the appropriate amount of time elapsed, TJ, AdC, KB or KC used the patient tip of the endotracheal tube (Endotracheal Tube; VetOne) to touch the arytenoids and score the severity of reaction (Table 1). This was recorded and the cat’s trachea was intubated using the lubed and cuffed endotracheal tube by a veterinary student. The endotracheal tube was connected to a non-rebreathing circuit and the cuff was inflated. Anesthesia was maintained with isoflurane and 100% oxygen. Vital parameters were monitored and recorded every 5 mins. Any cardiovascular or respiratory complications were treated as deemed clinically appropriate by the attending anesthesiologist. The surgery was completed by a student surgeon with supervision by a veterinarian.
Description of the reactivity score of arytenoid cartilages to stimulation using an endotracheal tube after topical lidocaine application

Once surgery was complete, the cat was moved into its recovery cage in sternal or lateral recumbency. When the cat swallowed, the endotracheal tube cuff was deflated and the cat was extubated. Cats were monitored in recovery for 45–60 mins for any signs of respiratory distress and pain. Signs of dyspnea, tachypnea or reduced oxygen saturation indicated by cyanotic membrane color would be considered respiratory distress. The Short Form Glasgow Pain Scale was used to assess pain. Any cat with a score ⩾5/20 was given additional buprenorphine 0.02 mg/kg IV and the pain scoring was repeated. Signs of nausea including lip licking or salivation would be treated with maropitant (Cerenia; Zoetis) 1 mg/kg IV or subcutaneously at the discretion of the anesthesiologist.
Statistical analysis
Data are reported as mean ± SD for normally distributed data and median and range for non-normally distributed data. Normality was tested using the D’Agostino–Pearson test. An ANOVA was used to determine differences in weight, sex, dose of propofol required for induction, time from premedication to IV catheterization and induction between groups. A Kruskal–Wallis test was used to assess differences in age and reactivity score between groups. Subjects were designated as no reaction or any reaction, and a χ2 test, with post-hoc Fischer’s exact test was run. A χ2 test was used to compare sex distribution between groups. All statistical tests were completed using GraphPad Prism (GraphPad Prism Version 8.3.0). A power calculation was completed prior to the study based on the results of Dyson, 2 using an online calculator. 7 To detect a difference of 25% in the number of cats which responded at each time point with an alpha of 0.05 and beta of 0.2, 22 cats per group were needed. Statistically significant results were considered as having a P value of <0.05.
Results
Weight, sex and time from premedication to IV catheterization and topical administration of lidocaine were normally distributed; age, dose of propofol and reactivity score were non-normally distributed.
Cats enrolled in this study were 0.5 years of age (range 0.19–6 years) and weighed 2.4 ± 0.9 kg. There were 59 male and 51 female cats. The dose of propofol administered to achieve intubation was 1.0 mg/kg (range 0–5.3 mg/kg). Thirty-nine cats did not receive propofol. The mean time to IV catheterization was 18.7 ± 7.8 mins and time to topical administration of lidocaine was 22.8 ± 8.8 mins. There were no statistically significant differences with regard to age (P = 0.55), weight (P = 0.71), propofol dose (P = 0.28), distribution of sex (P = 0.88) or time from premedication to IV catheterization (P = 0.38) or time from premedication to topical administration of lidocaine (P = 0.19) between the groups. There was no significant difference in overall reactivity score in cats that received propofol compared with those that did not (P = 0.93).
Severity of reaction to stimulation of arytenoid cartilages is presented in Figure 1. There was a significant difference in overall reactivity score between T5 and T45 (P = 0.0038). There was a significant difference in the number of cats that had no reaction compared with any reaction based on time, specifically, between T5 and T30 (P = 0.03) and between T5 and 45 (P = 0.0028). All cats were easily intubated in one attempt even with a severe reactivity score. No cats had a severe reactivity score at T45 or T60. There were no respiratory complications in recovery. Seven cats were administered buprenorphine intraoperatively. Twenty-six cats received buprenorphine within 60 mins of extubation, one of which also was administered maropitant
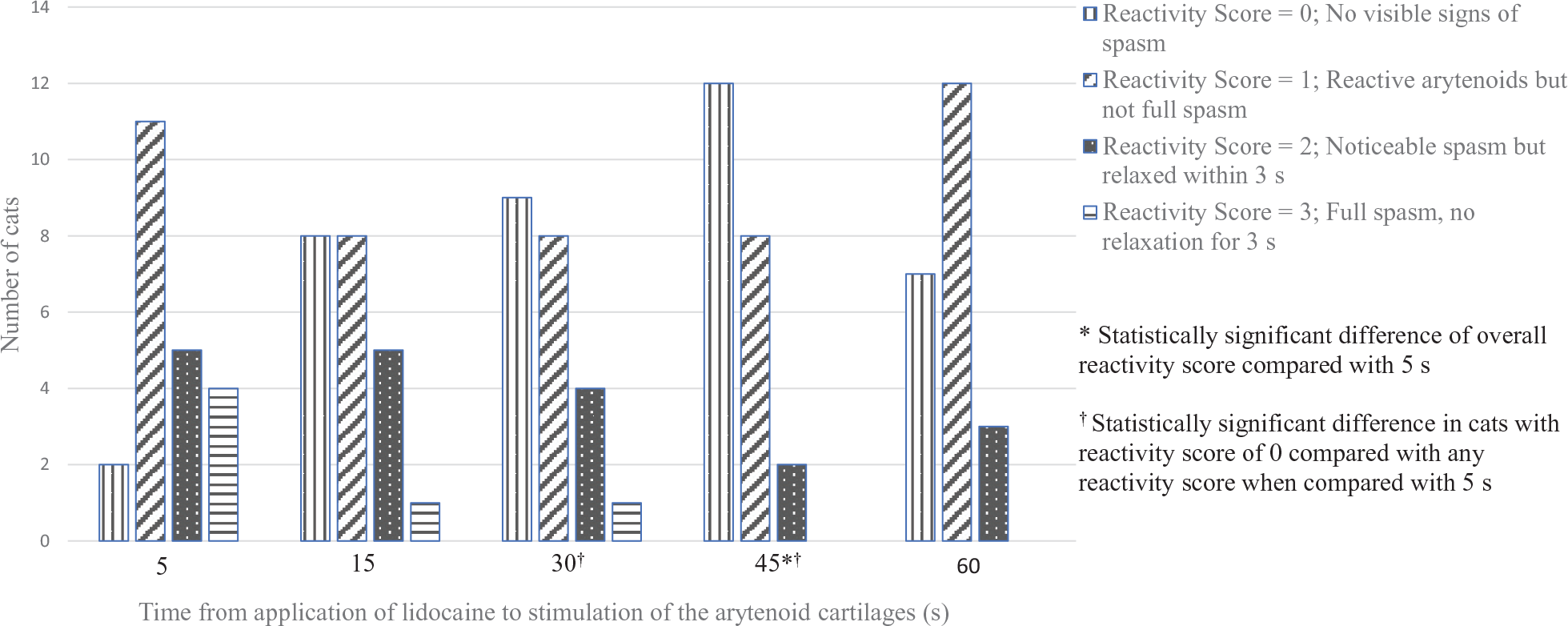
Number of cats with each reactivity score in response to stimulation of the arytenoid cartilages
Discussion
The lowest reactivity score was recorded at 45 s after application of lidocaine. All cats were intubated easily without signs of respiratory obstruction, hypoxemia or postoperative complications. As lidocaine has been shown to be effective at improving the ease of intubation and our goal was to assess the time to minimal reactivity, we did not include a control group. The results are compared with a 5 s baseline, which clinically would be equivalent to application of lidocaine and immediate intubation. However, without the inclusion of a control group the effect of application of lidocaine to the larynx on the reactivity score or baseline reactivity without lidocaine cannot be determined.
Previous research reports that topical lidocaine reduces the laryngeal reflex in response to mechanical stimulation.2,4,8,9 This study reported increased laryngeal relaxation at 45 s when compared with 5 s after lidocaine application. Mucosal mechanoreceptors are responsible for eliciting the laryngeal adductor response. 10 Application of lidocaine topically blunted the response of the laryngeal mucosal neuroreceptors and mechanoreceptors in piglets. 11 Topical and IV lidocaine has blunted the incidence of laryngospasm and cough response in people and dogs, respectively.12,13
Abolition of laryngeal response based on electromyographic studies was recorded 90–120 s after topical application of 2% lidocaine in another study. 4 Intubation was easily performed 1.5 mins after application of 2% lidocaine and loss of response to mechanical stimulation after 1.95 mins. 4 A 60–90 s desensitization time has also been used without complications.2,3,6,14 As this was a clinical study, it was elected to complete the evaluation using a standardized score at the time of application of lidocaine by a trained group of veterinary professionals. However, this resulted in the evaluations being completed by personnel not blinded to the treatment group. Evaluation of video recordings of laryngeal reaction could have removed this limitation, although there are difficulties acquiring a video in focus with enough detail for evaluation. Clinically, it is common to have minimal delays from application of lidocaine to first attempt to intubate which may increase the risk of spasm or laryngeal damage. Forty-five seconds was determined to have the lowest overall reactivity score in this study. Evaluation of reactivity scores at 90 and 120 s would have determined whether maximal reactivity was at 45 s, as well as what the trend of reactivity scores after 60 s would be. In spite of the reaction in cats in the earlier time points, endotracheal intubation was successfully performed in all cats without additional interventions.
Despite a difference in overall reactivity scores at T45 and a difference in the number of cats with no reaction at T30 and T45, there was no difference at T60 compared with T5. However, no cats had a severe reactivity score at T45 or T60. The predicted sample size detected an overall statistical difference in reactivity scores; a larger sample size may have distinguished differences between each reactivity score at the various time points.
Conclusions
The lowest reactivity scores in this study occurred 45 s after application of lidocaine. Therefore, anesthetists should wait at least 45 s before attempting endotracheal intubation. Intubation immediately after topical application of lidocaine is likely to elicit a laryngeal reaction.
Research Data
Study_data – for Evaluation of the time to desensitization of the larynx of cats following topical lidocaine application
Supplemental material, Study_data for Evaluation of the time to desensitization of the larynx of cats following topical lidocaine application by Teela L Jones, Kyrissa Boyer, Kelly Chapman, Brea Craigen, Anderson da Cunha and Erik H Hofmeister in Journal of Feline Medicine and Surgery
Footnotes
Supplementary material
The following file is available online:
Study data
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedures(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
