Abstract
Summary
Airway access is needed for a number of experimental animal models, and the majority of animal research is based on mouse models. Anatomical conditions in mice are small, and the narrow glottic opening allows intubation only with a subtle technique. We therefore developed a microscopic endotracheal intubation method with a wire guide technique in mice anaesthetized with halothane in oxygen. The mouse is hung perpendicularly with its incisors on a thread fixed on a vertical plate. The tongue is placed with a pair of forceps between the left hand's thumb and forefinger and slightly pulled, while the neck and thorax are positioned using the third and fourth fingers. By doing so, the neck can be slightly stretched, which allows optimal visualization of the larynx and the vocal cords. To ensure a safe intubation, a fine wire guide is placed under vision between the vocal cords and advanced about 5 mm into the trachea. An intravenous 22G × 1 in. plastic or Teflon catheter is guided over this wire. In a series of 41 mice, between 21 and 38 g, the success rate for the first intubation attempt was >95%. Certainty of the judgement procedure was 100% and success rate was higher using the described method when compared with a transillumination method in a further series. The technique is safe, less invasive than tracheostomy and suitable for controlled ventilation and pulmonary substance application.
Intubation is a standard airway management procedure that provides security during anaesthesia due to controlled ventilation and protected airways. It is less invasive than tracheostomy and also allows the administration of substances into the lung. Mice are the most commonly used laboratory animal species because of the opportunities they offer for studying immunological and genetic variations. During the past years, there has been an increasing awareness for mouse models in targeting lung disease. Especially, long-term models for infectious disease, chronic obstructive pulmonary disease or asthma have become more important. Repeated, safe and precise intratracheal substance application (Su et al. 2004) or lung function measurement may therefore be of growing interest and relevance. Furthermore, in a wide range of mouse models, controlled ventilation is helpful or even necessary, particularly if deep anaesthesia is required or spontaneous ventilation is limited, e.g. with pneumoperitoneum (Moehrlen et al. 2005). Moreover, orotracheal intubation is much less traumatic and can be performed faster than tracheostomy.
To our knowledge, a safe endotracheal intubation method for mice has not been published and evaluated. We therefore describe and evaluate a stereomicroscopic technique that has several advantages: it is feasible, safe, non-traumatic, reproducible in the same mouse and requires minimal equipment.
Materials and methods
Animals and housing
Three different mouse strains were used (specific pathogen-free C3HeB/FeJ, BALB/c and complement receptor 2 -/-). The experiments were performed according to the Helsinki Convention for the use and care of animals and conformed to the German animal protection laws. All experimental protocols were approved by the local review boards of Freiburg im Breisgau, Germany and of Homburg/Saar, Germany for animal care.
Environment
All racks were kept in the same animal room at a room temperature of 22.1°C, with 55–60% relative humidity, a 12/12 h light/dark cycle and a light intensity of 80–120 lux. Radio sound was present during the whole light period.
Housing
Four animals were usually kept together in specific pathogen-free type II Makrolon cages.
Food and water
Tap water in drinking bottles and pelleted food (Altromin No. 1314, Altromin GmbH, Lage, Germany) were given ad libitum.
Bedding
Wood shavings (40–50 g) were used for bedding. Cages, bedding and metal sheets were changed every fifth day.
Description
We used a surgical microscope (Wild M650, Heerbrugg, Switzerland, with 10× magnification). An intravenous 22G × 1 in. catheter (Terumo® Surflo, Emergency Medical Products Inc, Waukesha, WI, USA) serving as an endotracheal tube, appropriate wire guide (e.g. Seldicath 2F, Laboratoire Plastimed, Saint-Leu-La-Forêt Cedex, France) as well as a pair of non-traumatic microsurgical forceps (Figure 1; Fehling Ceramo DeBakey 18 cm microforceps, Fehling Instruments, Karlstein, Germany) were prepared. A vertical Plexiglas plate, with a rigid thread, allowed the mouse to be fixed in a hanging position for the intubation procedure (Figure 1). As intubation requires both hands, the microscope was pre-focused approximately 15 mm below the thread to the putative working field, the vocal cords.

For intubation, the mouse was anaesthetized with 4% halothane (Sigma-Aldrich, Deisenhofen, Germany) in oxygen (Penlon Oxford miniature vaporizer, Abingdon, UK), at a flow rate of 1 L/min into a transparent chamber (200 mL volume; Figure 1). The mouse was observed for anaesthetic effects such as sedation, immobilization and decrease in respiratory rate; when the rate dropped to 60 breaths/min, the mouse was taken out and hung perpendicularly by its two front upper teeth (incisors) with the thread of the vertical plate.
Using the forceps in the right hand, the tongue was carefully pulled out, fixed between the left hand thumb and medio-dorsal forefinger, and slightly pulled with those two fingers. The mouse's neck and thorax were kept between the third and fourth fingers, so that the neck could be slightly stretched, which allowed the optimal microscopic vision of the vocal cords (without the use of a laryngoscope; Figure 2a, video clip). This optimal positioning was accomplished with the operator's left hand.
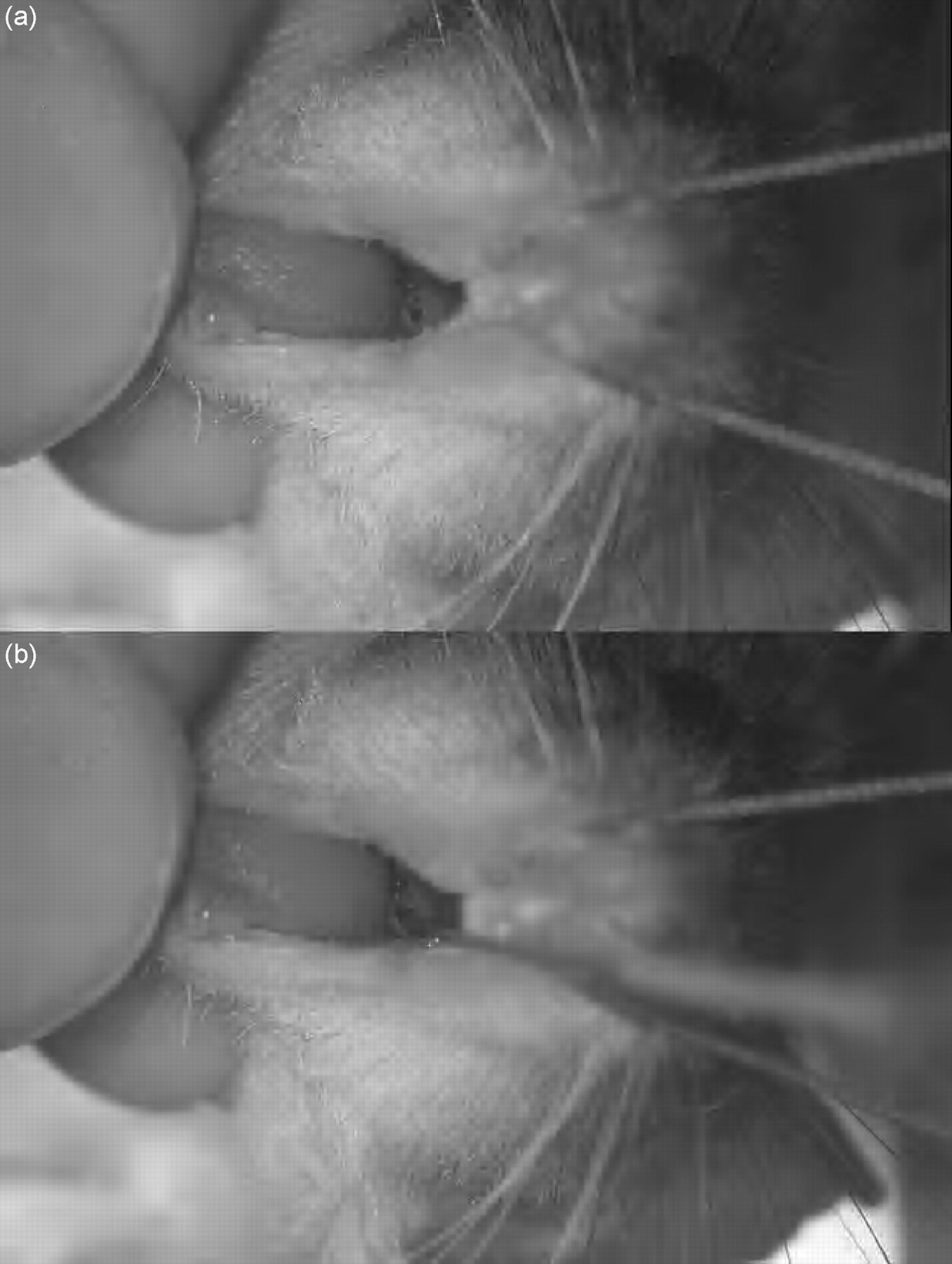
In order to continuously observe the wire guide tip during the insertion manoeuvre, the wire guide was firmly held in the right hand slightly backwards between the fingertips of the thumb and index finger so that vision was not obscured (video clip). The wire guide was then placed under continuous vision of both tip and anatomical structures between the vocal cords (Figure 2b, video clip), as certainty about the wire's correct intratracheal position is mandatory for safe airway management. The wire was then gently pushed approximately 5 mm into the trachea. The endotracheal catheter was introduced over the wire approximately 5 mm into the trachea. An adequate intubation depth was obtained when the end of the thin part of the catheter reached the incisors.
A corresponding didactic video is available at
Evaluation of the microscopic wire guide-based transoral intubation method
We measured the time from the moment the mouse was placed in the anaesthesia chamber until the respiration rate dropped to 60 breaths/min (Table 1). We also measured the time between opening the anaesthesia chamber and correct intratracheal intubation, as well as the time between opening the anaesthesia chamber and correctly connecting the tube to the mouse ventilator (Hugo Sachs-Harvard Apparatus 845 MiniVent, March-Hugstetten, Germany). Every mouse was breathing spontaneously and adequately at the time the ventilator was connected and tolerated the tube and ventilation (set at 8 µL/g body weight tidal volume at a rate of 160/min). Such ventilation further confirmed that tube placement was correct, as thoracic cage movement was symmetrical and synchronous with the ventilator. The mouse was disconnected from the respirator and extubated when spontaneous breathing was judged sufficient.
Data are given as mean (standard deviation)
*P < 0.05 compared with C3HeB/FeJ mice
Comparison of a published transillumination method with the microscopic wire guide-based transoral intubation method
In a recently published method, transillumination of the thorax and neck of the mouse allows the larynx to become visible when a laryngoscope is used (Brown et al. 1999). We compared that method with the one we describe here. In order to eliminate bias, both investigators used this method during a series of intubations during routine experiments that were not evaluated. Then at the specific working place, each investigator intubated mice that had been killed at the end of experiments for a total of 80 intubations using the transillumination method before the measurement series was started.
Both methods were prospectively compared with the same anaesthesia as before and with the identical type of venular cannula serving as the tracheal tube. Each mouse was intubated a total of four times, always in sets to assess both methods of intubation. Animals were not allowed to completely wake up, but immediately after weaning were again put in halothane/oxygen to directly re-intubate them, so that intubations were performed at time intervals of about 3–5 min each. Animals were assigned randomly to the first method and the second investigator performed the two techniques in inverse order immediately afterwards. Gradings were recorded by the investigators for each intubation: resistance during tube insertion was graded as ‘low’; ‘slightly or moderately higher resistance’; ‘much higher resistance’; ‘very much higher resistance’, whereas the certainty of successful intubation immediately after tube insertion was graded as ‘100%’; ‘75–99%’; ‘50–74%’; ‘25–49%’, ‘ <25%’.
If any intubation was unsuccessful as assessed by a short period on the mouse ventilator, the investigator immediately attempted to intubate the animal with the alternate method using inhalation anaesthesia again.
Learning process assessment of the intubation method
In order to assess the feasibility of the intubation method, three students and one physician who needed to learn the method and who were without intubation experience were taught the procedure by explanation, demonstration of the teaching video and supervision of their attempts. Teaching was considered as accomplished when 20 consecutive intubations were successful within 60 s each.
Statistical analysis
Data are presented as mean (±standard deviation). Linear regression was used for continuous variables. Two-sided t-tests or analysis of variance were performed using Bonferroni post-test, or Wilcoxon signed rank tests for categorical variables, with P < 0.05 considered as significant.
Results
Method evaluation
We used three mouse strains (41 mice [9 female] with a weight range from 20.9 to 37.8 g, specific pathogen-free C3HeB/FeJ, BALB/c and complement receptor 2 -/-) (Table 1).
Mice were randomly intubated. Mean intubation time was 31.3 (8.7) s with one investigator and 23.3 (5.4) s with the other. The time between opening of the anaesthesia chamber and the correct connection of the mouse with the respirator was 37.2 (9.3) s in one investigation, 27.6 (5.6) s in the other. The strain-specific results are shown in Table 1. The mice were disconnected within 20–60 s from the respirator. No complication occurred with the extubation procedure.
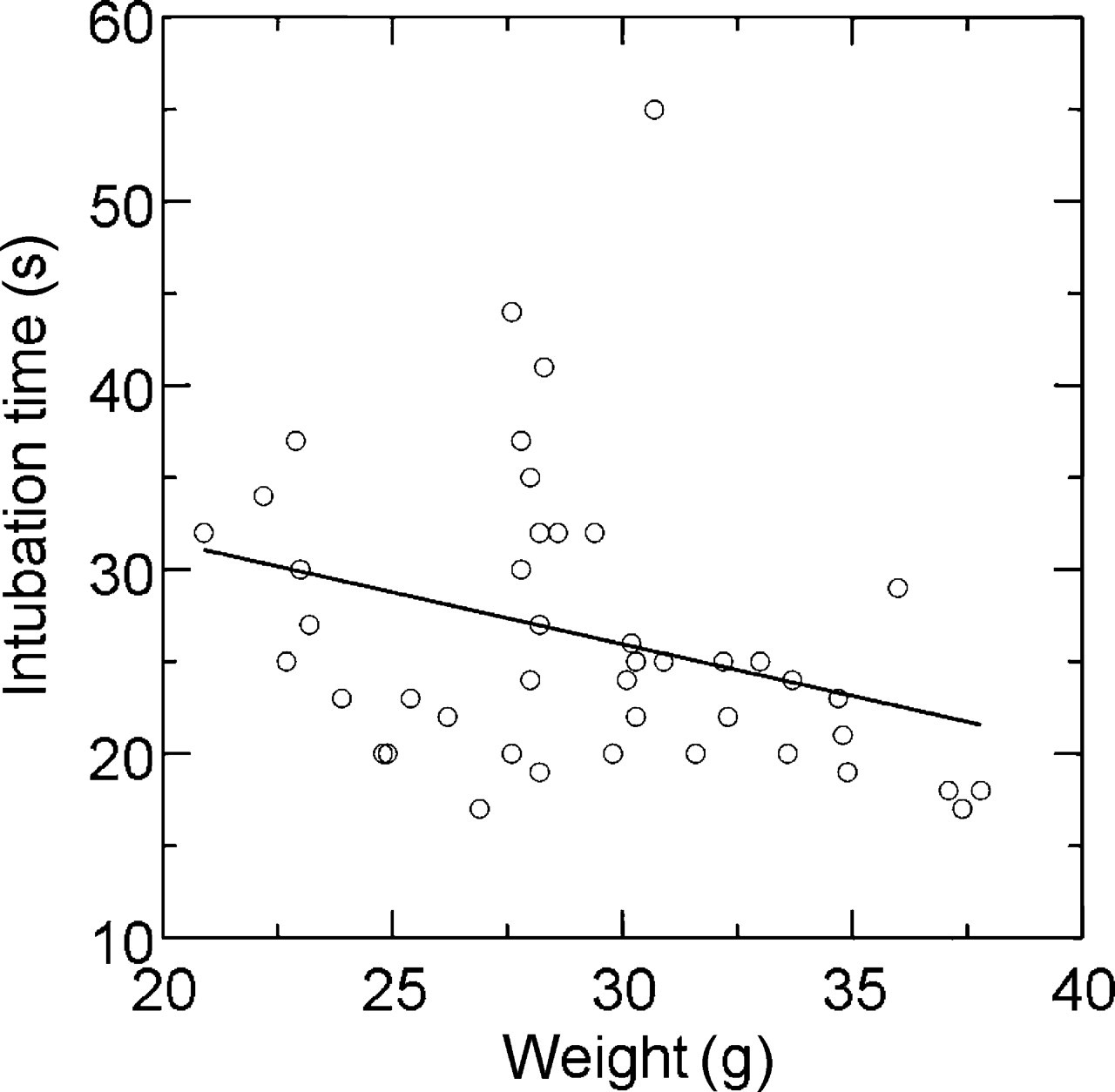
There was a significant negative linear correlation between intubation time and mouse weight, as shown in Figure 3 (r = −0.33, P = 0.04). The same was true with time for connecting of the mouse to the respirator (r = −0.34, P = 0.03).
One mouse (2.2%) could not be intubated the first time, possibly because the microscope was initially unfocused; that was the first in the series for one investigator.
Vascular 22G × 1 in. catheters (Figure 1) turned out to be appropriate endotracheal tubes for the successful ventilation of a wide range of outbred or inbred mice between 8 and >40 g (not shown). None of the venous catheters showed any trace of blood after intubation, and none of the animals had physical or behavioural evidence of injury or blood trace the following day; thus all animals survived.
Direct comparison of a transillumination method with the microscopic wire guide-based transoral intubation method
A set of 28 animals, totalling 56 intubations of each technique were performed and analysed. Mice of the same strain weighing 20.2 ± 9.1 g were used.
The transillumination was performed successfully in 51 of 56 instances (91%); five intubations were false intubations. The median of the ‘certainty of successful intubation’ was graded as ‘75–99%’ and ranged from 100% in only eight of 56 cases (14%) to below 50% in 11 of 56 instances (20% of cases). Intubation time was 38.6 ± 10.4 s. The investigators could not predict their own intubation success with the transillumination method, as their certainty grading did not differ between the successfully intubated mice and those not successfully intubated (P = 0.30).
The microscopic wire guide-based intubation was successful in all 56 attempts. The difference in success was significant (P = 0.013) when compared with the transillumination method. Grading of certainty was always 100%, indicating that clear vision was attained in all intubations by both investigators. The difference in certainty grading between the transillumination and the microscopic method was significant (P < 0.0005). Resistance was graded as low in all cases, and the difference in resistance grading between the transillumination and the microscopic method was also significant (P < 0.0005).
There was no indication of any learning effect or ‘learning curve’, e.g. in terms of intubation times with the alternate method, when analysing the first versus the last quartile or the first versus the second half (P = 0.54 or P = 0.91, respectively). Least importantly, intubation time was shorter and less variable with the microscopic technique (20.7 ± 2.8 s) than with the transillumination technique (38.6 ± 10.4 s; P < 0.0005). After intubation had failed with the transillumination method, the microscopic wire guide intubation method was successful in all five cases with an intubation time between 19 and 25 s and was always feasible with 100% certainty.
Learning process assessment
The procedure was taught to four students. Twenty consecutive successful intubations within 60 s each were achieved within 20–48 intubations.
Discussion
We describe a novel, safe and efficient method of transoral mouse intubation. On one hand, we intended to ventilate the animal. On the other hand, it was necessary to safely monitor the depth of anaesthesia so that the mouse did not require any ventilator support, as intubation is uniquely aimed at applying a particular drug into the air spaces.
This method provides certain advantages over other methods of airway management or intrapulmonary substance application. When compared with tracheostomy or direct tracheal puncture (Su et al. 2004), this method is faster, less invasive, not mutilating and probably less painful for the animal, thus representing an example of ‘refined’ airway management. As also recently shown with a radioactive tracer (Su et al. 2004), the certainty of dosage and substance application is higher than in most alternatives, including: direct ‘blind’ transoral intratracheal injection (Vernooy et al. 2001); or external surgical tracheal preparation and injection (Kujime & Natelson 1981) or non-surgical injection (Su et al. 2004); intranasal application (Szarka et al. 1997, Su et al. 2004); aerosol chamber exposition (Mizoguchi et al. 1986), otoscopic (DeLeonardis et al. 1995, Hastings & Summers-Torres 1999), direct (Spoelstra et al. 2007) or fibreoptic laryngoscopic (Costa et al. 1986), or arthroscopic transoral tracheal application (Vergari et al. 2003). The intraoral substance application is also used (Vlad et al. 1970). As in our technique, most investigators use intravenous catheters (Schwartz et al. 2001), polyethylene tubes (Brown et al. 1999, Vergari et al. 2003), angiocaths (DeLeonardis et al. 1995) or feeding needles (Nick et al. 2000). Feeding needles have also been used for selective left lung intubation (Kitamura et al. 2001).
To our knowledge, few reports exist on tracheal intubation in mice. Quite comparably with our technique, Guilbault et al. (2005) used a microscopic orotracheal intubation technique, but without the advantages of a wire guide use. Brown et al. (1999) described another method that would allow readers to duplicate the procedure. They used a tracheal transillumination method with an external fibreoptic light source that was adjusted at the neck just below the vocal cords, with mice hanging on their front upper teeth. Their main differences are the use of a custom-made mouse laryngoscope, the transillumination for non-microscopic vocal cord visualization and their renouncement on a wire guide. A modification with continuous inhalational anaesthesia application and modified laryngoscopes has recently been described (Spoelstra et al. 2007).
There are clear advantages in the use of a stereomicroscope. Approximately 1 mm in diameter, the mouse larynx is small. Both magnification and the three-dimensional view of the larynx allow the operator to control the movement much better with the wire guide during the procedure. As the wire guide's diameter is comparatively smaller than the cannula diameter, vision is preserved when it is introduced into the larynx. However, a major part of vision will disappear as soon as a cannula is in front of the larynx, which may be an important drawback of a recently described technique of Guilbault et al. (2005). Whereas the wire guide easily leads the cannula under vision during insertion, a ‘final blindness’ occurs with any non-wire guide method (Brown et al. 1999, Su et al. 2004) just when the operator should theoretically lead the cannula and clearly find his aim. In addition, the transillumination method requires the optimal three-dimensional alignment of all three – the visual axis, the mouse and the light source. This requires time and adds a novel variation to be managed with the two hands of the operator.
The prime issue in airway management is safety. The 100% certainty attained for the judgement procedure observed with our method contrasts with inferior certainty and missing failure prediction by the transillumination method by Brown et al. (1999). Also, the success rate was higher, and less importantly, the intubation time was at least 40% shorter by the microscopic method. A video-assisted orotracheal intubation, as also described in rats (Clary et al. 2004), using a small fibreoptic arthroscope and a wire guide in mice, may be an alternative (Vergari et al. 2003). The more difficult handling of a mouse lying supine may have translated to long intubation times of 3 ± 0.4 min that virtually exclude inhalation anaesthesia.
Whereas animals anaesthetized with injected drugs can be successfully intubated by our technique, we specifically aimed to assess the inhalation anaesthetics using this method. Both the induction and recovery times are much shorter than that of most injected drugs, and dose adjusting is easier. Isoflurane may be a valid alternative, e.g. using a lower cut-off than 60 breaths/min for induction, in order to have enough anaesthesia lag time. Our use of a non-cuffed tube to efficiently ventilate animals, e.g. during situations of abdominal overpressure (Moehrlen et al. 2005), or during thoracotomy (Stammberger et al. 2002), is consistent with its use in human clinical settings such as rigid bronchoscopy (Wain 2001, Tomaske et al. 2006).
The study was performed in mice with wide-ranging weights. The negative correlation of intubation time and mouse weight may reflect the slightly more demanding positioning and wire guide insertion when rather small mice are intubated. Small individual variation coefficients observed for the required intubation time suggest modest variation, supporting the easy reproducibility of the technique.
In conclusion, the presented microscopic orotracheal mouse intubation method is atraumatic, bears both a very high success rate and certainty for an intubation attempt, is rather easy to learn as shown in this paper and is validated in comparison with the transillumination method, where, with the presented readouts, it has shown superiority and thus may be regarded as a refined method. It is less invasive than tracheostomy, allows both a controlled anaesthesia (Moehrlen et al. 2005) and pulmonary substance application, and is repeatable in individual mice.
Footnotes
Acknowledgements
The authors acknowledge Mrs Rebekka Munke and Mrs Anke Huber, University of Konstanz, Germany, for technical assistance; Prof Albrecht Wendel, University of Konstanz, Germany, Prof Rudolf Lucas, Technical University of Krems, Austria and Prof Sami I Said, Stony Brook University, New York, USA, for critically reading the manuscript.
The work of J Hamacher was supported by a grant from the Deutsche Forschungsgemeinschaft (FOR 321/2-1; research group ‘Endogenous tissue injury: Mechanisms of autodestruction’) and by the Herrmann Josef Schieffer Prize of the ‘Freunde des Universitätsklinikums Homburg eV’.
