Abstract
The role of profilin as an allergen has long been questioned. The capacity of profilin to induce respiratory symptoms has recently been demonstrated; moreover, over 50% of patients sensitized to profilin experienced symptoms after the ingestion of plant-derived foods, suggesting that profilin should be considered as a clinically relevant food allergen.
We describe the cases of seven allergic patients with oral allergy syndrome and other adverse reactions after eating plant-derived food, that have been undergone to profilin desensitization treatment.
The protocol started with a drop of profilin solution (50 µg/mL) diluted 1:1018 in water until the highest dose of 10 drops of undiluted solution three times a week. At the end of the treatment we observed a decreased mean diameter of profilin wheal in skin prick test (SPT) in five of the seven participants and in profilin specific IgE values in six patients that repeated the test. Regarding basophil activation test (BAT) and the detection of IgG4, we do not have significant results because the tests have to be repeated in some patients. Regarding the double-blind placebo-controlled challenges, after about 10 months of induction phase all the patients showed tolerance to several foods that they previously did not tolerate.
Moreover, the immunotherapy with profilin has proved to be safe because no serious adverse events have been reported in our patients.
In summary, the results of this exploratory study of sublingual immunotherapy (SLIT) for profilin allergy show that it can be a promising therapeutic option that could modify the clinical reactivity of the patients to the intake of plant-derived food.
Introduction
Profilin constitutes a family of highly conserved proteins, which are present in all eukaryotic cells, including plants, fungi, protozoa, and viruses, and play a crucial role in regulating the activity in the microfilament system and intracellular calcium levels.1,2 Since profilin is an important component of essential cellular activities, it is ubiquitously spread and it can therefore be designated as a “panallergen”, responsible for a large number of allergic sensitizations clearly related to cross-reactivity and co-sensitization between inhalant, latex, and plant-derived food allergens.3,4
The first allergenic profilin was described in birch pollen and it was designated Bet v 2. 3
The prevalence of sensitization has been studied in different regions and countries. Studies conducted in European countries estimated skin sensitization to date-palm-pollen profilin to be in the range of 12.3–30% in patients with pollinosis. In vitro studies in Central Europe estimated instead that birch-pollen-profilin (Bet v 2) sensitization is responsible for 10–20% of patients with pollen allergy. A similar rate (21%) was described in a study of 370 individuals in Portugal and significant differences depending on geographical area were found in Spain. 5
The role of profilin as an allergen has long been questioned. The capacity of profilin to induce respiratory symptoms has recently been demonstrated 6 and its role as an important allergen has been confirmed in patients exposed to high quantities of grass pollens. 7 Moreover, Asero et al. 8 demonstrated that over 50% of patients sensitized to profilin experienced symptoms after the ingestion of plant-derived foods, suggesting that profilin should be considered as a clinically relevant food allergen.
Profilin is randomly distributed in pulp and peel and it is labile to heat denaturation and pepsin digestion. 9 It is therefore easy to understand that it is unable to cause sensitization via the gastrointestinal tract. In fact, the ingestion of raw fruits in patients sensitized to profilin usually determines local reaction in the oropharyngeal mucosa (oral allergy syndrome [OAS]), despite the systemic reactions to zucchini and litches reported in the literature.10,11
Moreover profilin is involved in the “birch-mugwort-celery-spice syndrome” and in adverse reactions to Apiaceae family (celery, carrot, fennel), hazelnut, Rosaceae family (apple, pear, peach, plum, apricot), tomato, pumpkin, pineapple, banana, and melon.
We describe the cases of seven allergic patients with OAS and other adverse reactions after eating plant-derived food, that have been undergone to profilin desensitization treatment.
Case presentations
The major characteristics of each patient are summarized in Tables 1 and 2.
Patient features.
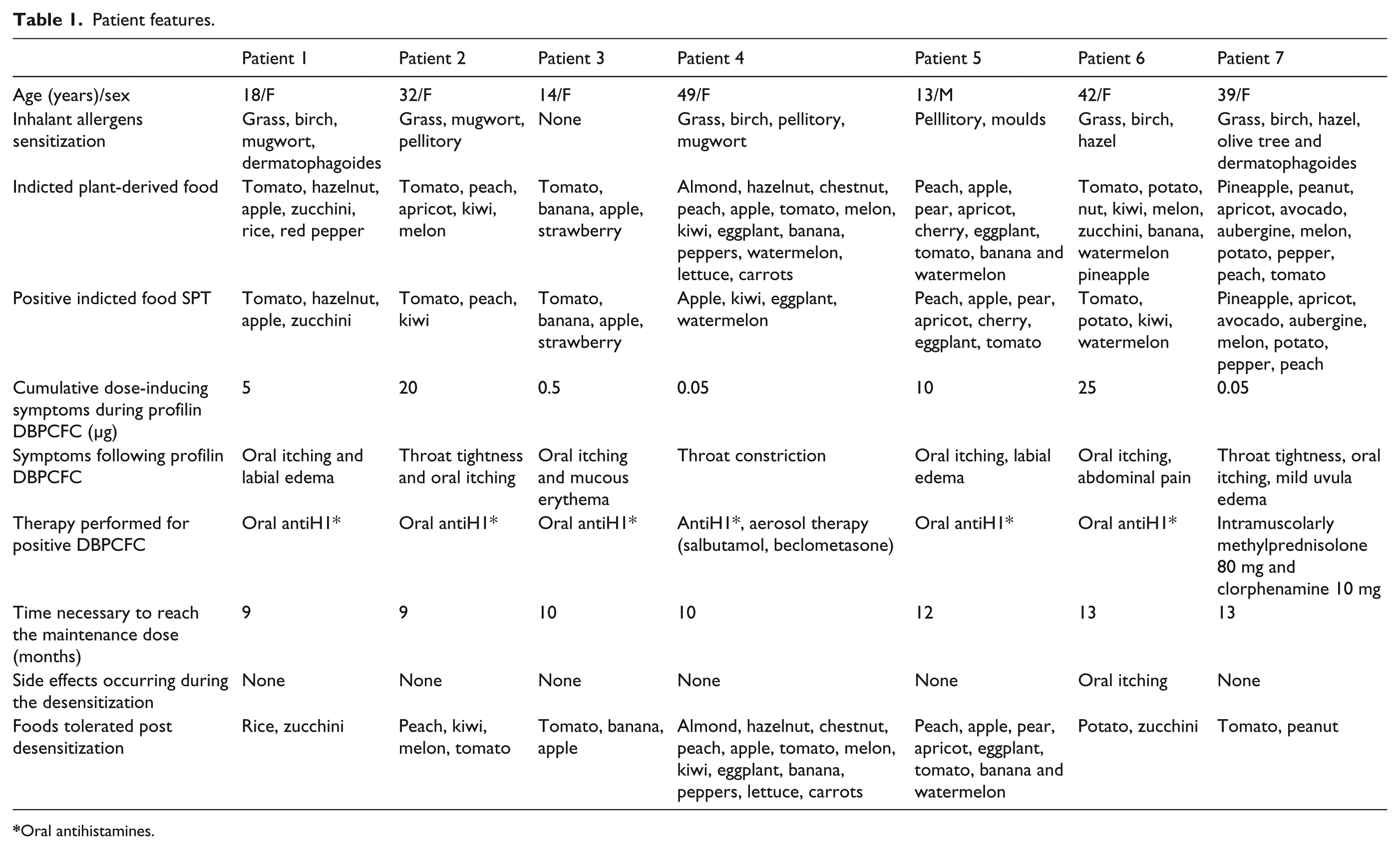
Oral antihistamines.
Patients’ allergological evaluation pre and post desensitization.

NP, not performed.
Case 1
An 18-year-old woman came to our attention with a history of oropharyngeal itching and labial edema a few minutes after the ingestion of tomato, hazelnut, apple, zucchini, rice, and red pepper (these foods were well tolerated previously). In all cases, treatment with an oral antihistamine was sufficient to reverse the reaction.
A subsequent in-depth medical history showed that the patient presented perennial oculorhinitis and asthma with worsening from April to June.
Skin testing was performed with the commercial extracts of foods involved in the reactions and, if the skin prick tests (SPTs) were negative, with fresh foods (prick-by-prick). We also did SPTs with the commercial extracts of pollens (grass pollen, parietaria, olive tree, cypress, mugwort, birch, beech tree, hazel tree) and, since we suspected a cross-reactivity between pollens and foods, with palm pollen profilin, peach non-specific lipid transfer protein (nsLTP), and apple PR-10 proteins (Alk-abellò, Milan, Italy). Specific IgE to recombinant allergens (Pru p 4 for profilin, Pru p 3 for nsLTP, and Pru p 1 for PR-10 proteins) were also detected (UniCAP, Phadia, Uppsala, Sweden). We also performed a basophil activation test (BAT) with palm profilin using fresh whole blood derived from the patient and investigated within 3 h after blood withdrawal. CD63-FITC/CD123-PE/HLA-BR-PerCP reagent mixture (BD biosciences) was used for gating activated basophils by FACSCanto flow cytometer (BD biosciences). This test was considered positive if the basophil activation percentage was >15%.
SPTs were positive to grass pollen, birch pollen, mugwort, dermatophagoides, tomato, apple, hazelnut, and zucchini. Moreover this patient showed only a positive profilin SPT (mean diameter of 10 mm), a positive BAT to profilin and a value of Pru p 4 specific IgE and IgG4 (UniCAP, Phadia, Uppsala, Sweden) of 2.3 and 0.07 kUA/L, respectively.
To confirm the diagnosis of profilin related food allergy, we performed double-blind placebo-controlled challenges (DBPCFC) administering every 30 min increasing doses of the profilin extract (Alk-Abellò) containing 50 µg/mL of palm profilin appropriately diluted hidden in a vanillin tasting preparation (Figure 1) or placebo (containing only vanillin).

Double-blind controlled to placebo challenge with profilin extract.
After a few minutes from the administration of 5 µg of profilin the patient presented oral itching and labial edema. The test was considered positive and the symptoms regressed after the administration of an oral antihistamine. DBPCFCs with the culprit foods were not performed because the patient had recent reactions.
Case 2
A 32-year-old woman experienced episodes of immediate oral and throat itching followed by generalized urticaria after eating tomato, peach, apricot, kiwi, and melon. She also had a history of seasonal allergic rhinitis since childhood.
The diagnostic SPTs were positive to grass pollen, mugwort, pellitory, tomato, peach, and kiwi.
In this case we also performed an allergological evaluation for the panallergens previously described and we obtained a wheal of 8 mm with profilin extract, a value of 3.74 kUA/L to Pru p 4 specific IgE and a positive BAT to profilin. Profilin IgG4 value was instead 0.01 kU/L.
After these results we did the DBPCFC only with profilin and the woman reacted at the dosage of 20 µg of allergen presenting throat tightness and oral itching with regression of symptoms after oral antihistamine therapy.
Case 3
Patient 3 was a 14-year-old girl with a history of atopic dermatitis, who presented with urticaria and angioedema after the ingestion of banana, apple, tomato, and strawberry.
This girl had positive SPT and specific IgE to profilin (3 mm of wheal diameter and 0.87 kUA/L, respectively) and to every plant-derived food referred in anamnesis. She also presented Pru p 4 IgG4 value of 0.11 and a positive profilin BAT.
To confirm the diagnosis, she underwent DBPCFC with profilin extract that was interrupted at 0.5 µg of allergen, because she showed oral itching and mucous erythema treated with antihistamines. Moreover she performed a DBPCFC with tomato and after 1 g she presented erythema of legs reduced with the same therapy.
Case 4
Patient 4 was a 49-year-old woman with rhinitis from January to June who referred oropharyngeal itching, lip edema, urticarial, and sometimes dyspepsia after eating almond, hazelnut, chestnut, peach, apple, tomato, melon, kiwi, eggplant, banana, peppers, watermelon, lettuce, and carrots.
She had positive SPT and specific IgE for inhalant allergens (grass, birch tree, pellitory, and mugwort) and for food allergens (apple, kiwi, eggplant, and watermelon). Profilin SPT showed a wheal with a mean diameter of 6 mm, while the serological assay showed a value of profilin specific IgE of 5.4 kUA/L, specific IgG4 of 0.09 kU/L, and CD63 upregulation of 37%, using BAT.
She underwent DBPCFC test with profilin extract, which resulted positive (throat constriction, which receded with antihistamines and aerosol therapy with salbutamol and beclometasone) at 0.05 µg. Also in this patient, DBPCFCs with all the responsible foods were not performed because she had recent reactions.
Therefore, at the end of the allergological evaluation, we made the diagnosis of allergic rhinitis and food allergy determined by a profilin sensitization.
Case 5
The patient was a 13-year-old boy with a clinical history of oral allergic syndrome (oral itching, lip edema, and throat constriction) after the ingestion of peach, apple, pear, apricot, cherry, eggplant, tomato, banana, and watermelon, and rhinitis from March to August.
The allergological work-up showed positive SPTs for inhalants (pellitory and moulds), some culprit vegetables (peach, apple, pear, apricot, cherry, eggplant, tomato) and profilin. The profilin specific IgE and IgG4 values were 12.2 and 0.9 kU/L, respectively, while BAT showed a positivity of 16%. Profilin DBPCFC was positive after the administration of 10 µg (oral allergic syndrome) and symptoms receded with oral antihistamines. No other DBPCFCs were performed.
Case 6
A 42-year-old woman with a previous diagnosis of moderate seasonal allergic rhinitis due to pollen sensitization (grass, birch, and hazel) referred to our attention for urticaria and face angioedema after eating many vegetables (tomato, potato, nut, kiwi, melon, zucchini, banana, watermelon, and pineapple). In this patient we found positive SPTs for tomato, potato, kiwi, and watermelon, and a wheal of 6 mm with profilin extract. Then we detected the Pru p 4 specific IgE value which was 3.74 kUA/L; conversely (nsLTP) and apple PR-10 proteins were negative (in SPT and specific IgE).
On the basis of these results, we did the DBPCFC only with profilin and the woman referred throat itching and abdominal pain after ingestion of 25 µg of allergen; this disorder regressed after the administration of an oral antihistamine.
Case 7
The last case was a 39-year-old woman previously followed by our Unit for allergic rhinitis due to polisensitization (grass pollen, birch pollen, hazel, olive tree, and dermatophagoides). She reported episodes of face urticaria and angioedema after the ingestion of some vegetables (pineapple, peanut, apricot, avocado, aubergine, melon, potato, red pepper, peach, and tomato).
SPTs and prick-by-prick were positive for pineapple, apricot, avocado, aubergine, melon, potato, red pepper, and peach, while the mean diameter of the wheal obtained with profilin SPT and Prup 4 specific IgE were 12 mm and 13 kUA/L, respectively. Therefore profilin BAT showed a positivity of 30%. Also in this case SPTs with nsLTP and apple PR-10 proteins were negative.
Finally she was subjected to DBPCFC with profilin and she reacted at the dosage of 0.05 µg, presenting throat tightness, oral itching, and mild uvula edema, so we administered aerosol therapy with salbutamol and beclometasone and intramuscular therapy with methylprednisolone 80 mg and clorphenamine 10 mg with regression of symptoms. This patient also performed a DBPCFC with tomato that caused labial edema, oral itching, and throat tightness after the ingestion of 20 mg; those symptoms receded after antihistamine therapy.
Desensitization
Therefore, at the end of the allergological evaluation in all seven patients, we made the diagnosis of food allergy determined by a profilin sensitization and we thus attempted to treat our patients with a sublingual desensitization treatment with profiling (Table 2).
Informed consent that included the reasonable risks of treatment was obtained from all the patients.
The protocol started with a drop of palm profilin solution (50 µg/mL) diluted 1:1018 in water until the highest dose of 10 drops of undiluted solution three times a week (Figure 2). The first dose was administered in day-hospital regimen and each patient continued the desensitization treatment at home with day-hospital visits monthly to switch to the next dilution. According to the protocol they were trained in medical treatment of allergic reactions and equipped with an emergency kit: autoinjectable epinephrine, betamethasone, and clorphenamine.

Profilin desensitization treatment.
After 9 months of the treatment without side effects, patient 1 underwent DBPCFCs with rice, zucchini, and apple, and in order to evaluate the immunological changes occurring during the immunotherapy, we performed SPTs and the detection in serum of specific IgE and IgG4 to profilin. Challenges were negative only for rice and zucchini, while the patient presented oral itching after 3 g of apple. Moreover she showed a reduction of mean diameter of SPT (3.5 mm) and a trend of decreasing specific IgE levels and BAT value for profilin, without reaching normal values, while specific IgG4 remained fairly stable.
Patient 2 underwent the same sublingual desensitization treatment ending the induction phase in about 9 months without side effects. At the end of the treatment she underwent DBPCFCs with peach, kiwi, melon, and tomato; all challenges were negative. The detection of specific plant-derived foods and profilin IgE and IgG4 in serum showed a similar trend mentioned above, while, using BAT, we observed a reduction of profilin induced CD63 expression level after the treatment.
Patient 3, who had undergone the profilin desensitization in 10 months without side effects, performed DBPCFCs with apple and tomato with negative results. During banana DBPCFC the girl presented OAS instead. The profilin SPT wheal diameter decreased slightly. The profilin specific IgG4 and specific IgE have to be repeated, while BAT results were still positive.
Patient 4 became tolerant in 10 months and, at the end of induction phase, she repeated the allergological evaluation, showing a reduction of size of profilin SPT wheal and value of profilin specific IgE, while profilin specific IgG4 slightly increased. Profilin induced CD63 expression level instead, reduced, but presented a positive value again. Moreover she performed the DBPCFCs with tomato and kiwi without side effects and reintroduced individually all the culprit foods, apart from watermelon.
In patient 5, the desensitization treatment lasted 12 months and then the allergy testing showed a reduction of profilin specific IgE value (1.99 kUA/L). No increase of profilin specific IgG4 or reduction of profilin SPT wheal size resulted. Moreover, BAT was still positive. The DBPCFCs with peach, kiwi, and apple were negative, so the boy reintroduced all the other vegetable foods without problems, except for cherry that still causes oral allergic syndrome.
During the 13 months of the induction phase, patient 6 presented oral itching in many episodes; she was the only patient who presented side effects. At the end of treatment, we repeated again the allergological evaluation and profilin specific IgE reduced to a value of 3.61 kUA/L. Profilin specific IgG4 assay and BAT were not performed either before or after. Moreover the patient autonomously reintroduced potatoes and zucchini without problems.
Patient 7 performed the desensitization in 13 months without side effects. The next profilin SPT presented a mild reduction of wheal mean diameter (11 mm) and profilin specific IgE reduced their value to 1.36 kUA/L, while profilin specific IgG increased to 0.04 kU/L. Post-treatment BAT has not been performed yet, while tomato and peanut DBPCFCs were negative.
Discussion
In 2009 we published the results 12 about profilin desensitization in two patients with clinical sensitization to a wide range of vegetables food with consequences to diet and metabolic imbalance. So in this case series we aimed to increase the patient numbers to confirm our previous discoveries.
In literature, studies about desensitization treatment with profilin in patients with OAS due to profilin sensitization have not been reported. Only Fernandez-Rivas et al. 13 and Pereira et al. 14 described two reports about non-specific lipid transfer protein (nsLTP) sublingual desensitization (SLIT) and in these studies the SLIT seems to be a promising therapeutic way.
On the basis of the principles of these studies, therefore, our goal was to develop a specific immunotherapy with the purified cross-reactive allergen to allow our patients to resume a normal diet. We relied on the methods (dose and length of the induction phase) previously used 12 and at the end of the treatment we found again the same immunological changes in allergological evaluation. In fact we observe a decreased mean diameter of profilin wheal in SPT in five of the seven patients and a decreased profilin specific IgE values in six patients that repeated the test. Regarding BAT and the detection of IgG4, we do not have significant results because the tests have to be repeated in some cases.
However, because the DBPCFC remains the gold standard tool to evaluate the clinical reactivity of patients reporting adverse reactions to foods, 15 it has been applied again in this work to evaluate the efficacy of SLIT. So after about 10 months of induction phase all the patients showed tolerance to several foods that previously did not tolerate.
Moreover, the immunotherapy with profilin has proved to be safe because no serious adverse events have been reported in our patients: in fact only patient 6 presented oral itching during the induction phase receded by coming back to previous dilution of profilin desensitization protocol and increasing more slowly the administration.
At the moment, all the patients are still under treatment, taking an average cumulative dose of profilin of 100 µg/mL weekly, because we do not have data about the optimal desensitization duration. This is a major limitation of the study.
In summary, the results of this exploratory study of SLIT for profilin allergy show that it can be a promising therapeutic option that could modify the clinical reactivity of the patients to the intake of plant-derived food. This clinical case series confirmed the results of our previous study, demonstrating that this treatment is safe, at least at a concentration that we used, since it was well tolerated and extremely effective on clinical symptoms. Moreover the patients reached a tolerance after the intake of the majority of vegetable food sources, the DBPCFC were promptly negative and an improvement of the metabolic imbalance was obtained. The decreasing SPT and the specific IgE results to profilin are in agreement with the clinical response. The patients are still under treatment and we believe that more pronounced immunological changes will be observed in future.
In conclusion, further studies are needed to refine the treatment, and to define the optimal allergen dose and duration of the therapy in a larger group of patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
