Abstract
The eosinophilic oesophagitis (EoE) is a chronic immune/antigen disorder of the oesophagus clinically characterized by dysphagia and pathologically by mucosa eosinophilic infiltration. Th2-type allergic reactions are considered having important roles in the aetiopathogenesis of EoE. Avoidance of food allergens, administration of steroidal anti-inflammatory medications and dilation of the oesophagus are the most important treatments. ‘Systemic nickel allergy syndrome’ (SNAS) interests about 20% of patients with nickel contact allergy which could present systemic cutaneous manifestations (urticaria, oedema, etc.) and also respiratory and digestive symptoms (meteorism, abdominal pain, diarrhoea, etc.). In the literature, it is demonstrated that nickel oral immunotherapy is effective in reducing symptoms of SNAS and in modulating inflammatory parameters. We describe the case of a 48-year-old woman suffering from EoE not responsive to the topical steroid administration and diagnosis of SNAS. The patient started nickel oral desensitization according to the literature protocol continuing nickel-free diet. After 1 year from the beginning of the treatment, during the maintenance dose (500 ng three times a week), she decreased gradually the dosage of immunotherapy and reintroduced all the culprit foods. After the immunotherapy interruption, during the free diet, she repeated the oesophagogastroscopy with a complete macroscopic and histological resolution. We showed the first case of an EoE in a patient affected by SNAS responsive to the nickel-free diet and the oral immunotherapy.
Introduction
The eosinophilic oesophagitis (EoE) is a chronic immune/antigen-mediated disorder of the oesophagus clinically characterized by episodic dysphagia and pathologically by dense eosinophilic infiltration of the oesophageal mucosa. 1
Th2-type allergic reactions, including interleukin (IL)-5, IL-13, IL-15, thymic stromal protein (TSP) and eotaxin-3, are considered having important roles in the aetiopathogenesis of EoE. 2
Avoidance of food allergens, administration of steroidal anti-inflammatory medications and dilatation of the oesophagus are the mainstays of treatment. 3
‘Systemic nickel allergy syndrome’ (SNAS) interests about 20% of patients with nickel contact allergy. These patients could be affected not only by systemic cutaneous manifestations (urticaria, oedema, eczema, erythema) but also by respiratory and digestive symptoms (nausea, gastric pyrosis, abdominal pain, diarrhoea and constipation).4,5
Intestinal biopsies from SNAS patients show an inflammatory infiltrate of lymphocytes and plasma cells with oedema and vasodilatation of the lamina propria 6 and a significant increase of intercellular adhesion molecule 1 (ICAM-l) levels suggesting an inflammatory state induced by ingested nickel. 7 Furthermore, an increase in some Th2 cytokines, such as IL-13, IL-5 and eosinophilic cationic protein, was observed in SNAS. 8
More authors demonstrated that nickel oral immunotherapy is effective in reducing symptoms in SNAS and is able to modulate inflammatory parameters.
In this case report, we describe the case of a 48-year-old woman suffering from EoE and concomitantly SNAS who presented a complete regression of eosinophilic infiltration in the oesophageal epithelial layer at the histological examination after nickel desensitization.
Materials and methods
The patient gave her informed and signed consent to participate in the study.
Case report
A 48-year-old woman referred to the Endoscopic Digestive Surgery Unit of Policlinico Gemelli for severe dysphagia with cervical oesophagus stenosis confirmed by radiological exams.
She underwent an oesophagogastroscopy with transnasal endoscope that demonstrated a cricopharyngeal substenosis that was finally breached by a standard gastroscope.
The endoscopy demonstrated the presence of furrows and transient concentric rings that indicated oesophageal longitudinal muscle contraction (Figures 1 and 2); the histopathological examination of the upper oesophageal specimen demonstrated an inflammatory aspect of mucosa with at least 15 eosinophils/HPF (high-power field) (40×). These findings supported the clinical suspicion of EoE.

Endoscopy picture pre-treatment.

Another endoscopy shot pre-treatment.
Because of the high frequency of allergic diseases in EoE patients, she referred to the Allergy Unit of the same hospital and underwent a complete allergological evaluation:
Patch tests (Gruppo Italiano Ricerca Dermatiti da Contatto e Ambientali (GIRDCA) series) according to the
The food SPTs were all negative, while we had a positive result for nickel patch test permitting to make the diagnosis of nickel IV–type allergy. The patient performed also a blood count that confirmed eosinophilia (1.1 × 109/L) and the assay of total serum immunoglobulin E (IgE) that were normal (120 kUA/L).
According to SNAS guidelines, the patient started a diet without nickel-containing foods (Table 1) to evaluate gastrointestinal symptom enhancement.
Foods with high nickel content.
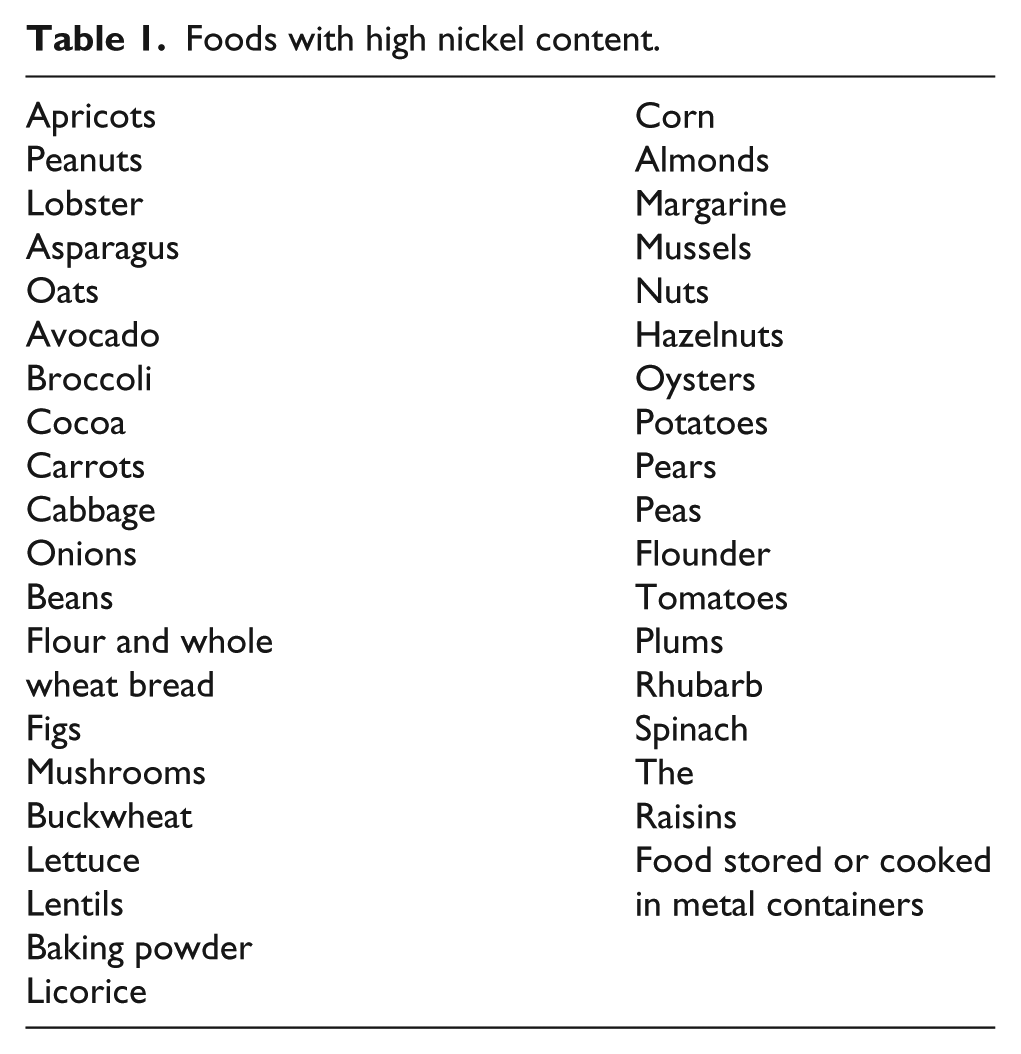
After 6 weeks of diet, the patient reported an almost complete improvement of her symptoms, and to confirm the diagnosis of SNAS, we decided to perform nickel oral challenge test starting with 2.5 mg and increasing the dose by 2.5 mg every 96 h up to 10 mg. The test was interrupted at the dose of 5 mg for the onset of gastrointestinal symptoms (the test was considered positive).
So, we confirmed the diagnosis of SNAS and, according to the literature protocol and after signing the informed consent, the patient started nickel oral immunotherapy according to the following scheme: 5
Week 1: one capsule of 0.1 ng three times a week;
Week 2: two capsules of 0.1 ng three times a week;
Week 3: one capsule of 1 ng three times a week;
Week 4: two capsules of 1 ng three times a week;
Week 5: one capsule of 10 ng three times a week;
Week 6: two capsules of 10 ng three times a week;
Week 7: one capsule of 100 ng three times a week;
Week 8: two capsules of 100 ng three times a week;
Week 9: one capsule of 500 ng three times a week.
After 1 year from the beginning of the treatment, during the maintenance dose (500 ng three times a week), the patient gradually decreased the dosage of immunotherapy and reintroduced all the culprit foods. After the immunotherapy interruption, during free diet, she repeated the oesophagogastroscopy that demonstrated the resolution of the substenosis. Furthermore, the oesophageal lumen appeared normal in all its features without macroscopic signs of oesophagitis (Figures 3 and 4). On the histological examination of the upper/middle oesophagus, there was

Endoscopy picture post-treatment.

Another endoscopy shot post-treatment.
Discussion
The EoE diagnosis is performed by the criteria shown in Table 2 with exclusion of other potential causes of oesophageal eosinophilia. 11
Diagnostic criteria and pathological characteristics of the EoE.

EoE: eosinophilic oesophagitis; HPF: high-power field (400× magnification).
About 50% of patients with EoE are affected by allergic and atopic diseases 1 and they are at higher risk for development of EoE.
In fact, EoE is considered an allergic disease caused mainly by the ingestion of food allergens including wheat, egg, milk, soy, nuts and fish/shellfish. Th2-mediated responses are important: IL-5, IL-13, IL-15, thymic stromal lymphopoietin (TSLP), transforming growth factor-β, periostin and eotaxin 3 are the major players in disease development, 2 particularly the last one is a potent highly specific mediator for eosinophil chemotaxis and activation explaining the high density of eosinophils in target tissues and damage due to their activation.
Our case seems to be the first report in the literature that demonstrates a possible aetiopathological association between EoE and SNAS and a possibility of therapeutic strategy for the resolution of these pathological conditions without side effects.
Our patient, suffering from severe dysphagia, underwent the nickel oral desensitization for a period of 12 months. Thanks to the oral immunotherapy, the patient presented clinical regression of symptoms and she could return to a free diet; contemporarily, the oesophagus histological examination demonstrated a complete disappearance of the eosinophilic mucosa infiltration and the inflammation resolution.
The literature reported that oesophagus exposure to food and aeroallergens in genetically predisposed individuals may initiate the process of EoE although the correct mechanism is currently unknown. 2
On the basis of our case, we could also hypothesize the
Gastrointestinal biopsies of patients with SNAS are characterized by an inflammatory infiltrate of lymphocytes and plasma cells with oedema and vasodilatation of the lamina propria 6 and by an increase in some Th2 cytokines, such as IL-13, IL-5 and eosinophilic cationic protein 8 similarly to that we currently observed in patients suffering from EoE. On this basis, we could speculate possible similar aetiopathogenetic mechanisms.
Topical and systemic steroids and endoscopic treatment are the principal therapies for EoE but they are characterized by important side effects. Another possibility for treatment of the EoE is represented by dietary therapy. 1
The three ways of dietary modification include the following:
Elemental diet – amino acid–based formula to remove food allergens that is expensive and unpalatable and affects patients’ quality of life;
Six-food group elimination diet (SFGED) – the most common food allergens in EoE including milk, egg, wheat, soy, peanuts/tree nuts and sea food (fish/shellfish);
Targeted or tailored elimination diet – this therapy is guided by the detection of food allergens by skin prick/patch tests and blood tests.
Although symptomatic improvement occurs after dietary therapy, recurrence is common after discontinuation of treatment. 3
According to Lucendo et al., 12 it seems that oral immunotherapy for food allergy has been associated with new cases of EoE. It seems that it is true only for patients with IgE-mediated food allergy; instead, SNAS is not IgE-mediated allergy but systemic symptoms are due to nickel ingestion through foods and coexists in almost all patients with nickel contact dermatitis (nickel IV type allergy).
In our case, the clinicopathological remission could be permitted not only by a nickel-free diet (in a first moment) excluding several nickel-rich foods, but it could also be maintained thanks to nickel oral hyposensitization. In fact, it is well known that we observe a significant reduction of interferon (IFN)-γ, IL-5 and IL-13 after nickel desensitization, whereas no changes were observed in patients that received only the elimination diet. 5
Interestingly, the same ILs are involved also in aetiopathogenesis of EoE and seem to reduce in concomitant with the clinical enhancement. 5
In conclusion, we present an innovative approach to evaluate the possible aetiopathogenetic causes of EoE and a new therapeutic strategy that could permit a lasting remission without severe diet restrictions and pharmacological therapy with side effects. Further studies are certainly needed with a wide number of patients to evaluate the relationship between EoE and SNAS and the efficacy of nickel oral hyposensitization in EoE/SNAS-affected patients.
Footnotes
Acknowledgements
E.N. and R.C. contributed equally to this work. This study will be presented in poster form at conferences. The authors are responsible for the content and the writing of this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
