Abstract
Objectives
The aim of this study was to compare viscoelastic test results from samples collected from a jugular vein using a 20 G needle and a medial saphenous vein using a 22 G needle in cats presenting for elective ovariohysterectomy (OHE) or castration.
Methods
Forty apparently healthy cats (20 males and 20 females) presenting for elective OHE or castration were included in a prospective study observing viscoelastic test results from central and peripherally collected whole blood. Cats were anesthetized during blood collection with a standardized protocol including buprenorphine, ketamine, dexmedetomidine and isoflurane. Blood samples from jugular and saphenous veins were collected near simultaneously. Viscoelastic evaluations of whole blood were performed using a point-of-care device measuring clot time (CT), clot formation time (CFT), alpha angle (α), maximum clot formation (MCF), and amplitude at 10 and 20 mins (A10 and A20, respectively). Viscoelastometry continued post-clot time to determine a lysis index at 30 and 45 mins (LI30 and LI45, respectively) to assess fibrinolysis.
Results
Studied cats had a median age of 18 months (range 5 months to 5 years) and a median weight of 3.6 kg (range 2.7–5.9 kg). A total of 80 samples were available for analysis. While lysis indices were not different, viscoelastic measures of coagulation differed between sampling sites (CT, P <0.005; CFT, P = 0.01; α, P <0.05; MCF, P <0.0005; A10, P <0.0005; A20, P <0.0005).
Conclusions and relevance
Viscoelastic results from jugular venous blood samples appear to be more hypercoagulable than those collected from the medial saphenous vein, suggesting that the same site should be used consistently for serial monitoring or for collecting study data.
Introduction
Viscoelastic testing provides a global assessment of clot formation and lysis. Veterinary viscoelastic studies typically use thromboelastography (TEG) or rotational thromboelastometry (ROTEM). 1 Veterinary guidelines for these instruments recommend use of citrated whole blood, recalcified after a 30-min rest period. 2 This delay can be problematic in an emergency setting and is not required for a recently released veterinary point-of-care viscoelastic device, the VCM Vet (Entegrion), which uses a small volume of non-anticoagulated whole blood immediately after collection. The VCM Vet also differs from TEG or ROTEM in the method of coagulation activation. For both TEG and ROTEM, the cups and pins in contact with blood are manufactured from a polymer, which is not an efficient activator of the contact pathway.3,4 Veterinary guidelines recommend the use of activators (eg, tissue factor and kaolin) to reduce analytical variability, rather than induction of clotting by only recalcification of citrated samples. 5 In VCM Vet cartridges, blood is in contact with two glass slides, and unlike the polymer used for TEG and ROTEM cups, these slides are intended to induce contact pathway activation without requiring an additional activator. 6
Before this device is used clinically, it is important to define preanalytical factors that influence results. For TEG and ROTEM, veterinary guidelines suggest the use of a jugular vein and a 21 G or larger needle. 2 Similar recommendations have yet to be developed for the VCM Vet, and saphenous venipuncture may be preferable for fractious cats, 7 or patients with suspected coagulopathies. 8 In dogs, jugular and saphenous citrated samples did not produce significantly different results when analyzed by kaolin-activated TEG, 9 but site of venipuncture has not been compared in cats. Saphenous samples are often collected with smaller-diameter needles. A recent feline study reported no difference between results for 21 G or 22 G butterfly needles and syringe needles when citrated samples were collected after a discard sample from a jugular vein and analyzed using kaolin-activated TEG, 10 but the effect of needle gauge on viscoelastic testing of feline non-anticoagulated whole blood requires investigation.
The aim of this study was to determine whether there were differences in VCM Vet results between whole blood samples collected from a jugular vein using a 20 G needle and a medial saphenous vein using a 22 G needle in cats presenting for elective ovariohysterectomy (OHE) or castration.
Materials and methods
Animals
The study was reviewed and approved by the Board of Directors of the Hill Country Animal League acting as an ad hoc institutional ethical review committee. Owner consent was obtained before entering patients into the study.
Twenty male and 20 female, apparently healthy, cats presenting for elective OHE or castration were entered into the study. Cats were weighed and confirmed to be ⩾0.9 kg body weight and ⩾2 months of age, as reported by the owner/agent and confirmed by dentition. Before sample collection, cats were anesthetized with a standardized protocol, including buprenorphine, ketamine, dexmedetomidine and isoflurane. Equal volumes of ketamine (100 mg/ml; Ketamine HCl [VEDCO]) and dexmedetomidine (0.5 mg/ml; Dexmedetomidine HCl [Putney]) were mixed in a single sterile vial. Anesthesia was induced by an intramuscular injection of 0.4 ml of this mixture. The total dosage of induction drugs ranged from 3.4 mg/kg to 7.3 mg/kg for ketamine and from 0.02 mg/kg to 0.04 mg/kg for dexmedetomidine. Buprenorphine (0.01 mg/kg IM; Buprenorphine Injection PF [Roadrunner Pharmacy]) was administered after the induction of anesthesia. The cats had small animal transparent anesthesia masks placed over their faces and isoflurane was admin-istered with a vaporizer setting range of 0.5–1% (Isoflurane; MWI). Oxygen flow was 2 l/min using a non-rebreathing circuit.
Interventions and testing
Using a syringe, non-anticoagulated whole blood samples were collected from the jugular vein using a 20 G needle and the medial saphenous vein using a 22 G needle, by the same experienced technician, with the minimum possible delay between the two sites. The initial venipuncture site was randomly selected, using a block randomization technique to ensure half the samples were collected first from the jugular vein in female and male cats. All venipunctures were witnessed by an additional technician, and cats were excluded if either technician considered one or both venipunctures was not atraumatic. Samples were analyzed immediately upon collection using cartridge-based point-of-care viscoelastic instruments of the same model (VCM Vet). The following viscoelastic parameters were measured: clot time (CT), clot formation time (CFT), alpha angle (α), maximum clot formation (MCF), amplitude at 10 and 20 mins post clot time (A10 and A20, respectively), and lysis index at 30 and 45 mins post maximum clot formation (LI30 and LI45, respectively).
Statistical analysis
Parameters were compared between samples collected from the jugular and saphenous veins by paired t-test or Wilcoxon test for paired samples, depending upon data distribution, as assessed by the D’Agostino-Pearson test. For each parameter, the coefficient of variation (CV) for duplicate samples was estimated using the root mean square method. Bland–Altman plots were produced to visually summarize agreement between the two sites. Statistical significance was set at P <0.05. Statistical analyses were performed using MedCalc Statistical Software version 19.1.3 (https://www.medcalc.org).
Results
The median age of study cats was 18 months (range 5 months to 5 years). The median weight was 3.6 kg (range 2.7–5.9 kg). Seventy-five percent of cats studied were domestic shorthair, 15% were domestic longhair and 10% were Siamese. Of the 20 female cats studied, 12 (60%) were determined intraoperatively to be pregnant. Eight pregnant cats (40%) were assessed to be mid-pregnant and the other four (20%) were in late pregnant. Of the eight (40%) cats that were not pregnant, six (30%) were in estrus and two were in anestrus.
Significant differences were observed for CT, CFT, α, MCF, A10 and A20 from the two sampling sites (Table 1, Figures 1–6). Estimates of the CV are provided in Table 2. No differences were noted in the lysis indices.
Summary results of viscoelastic testing from 40 cats to compare samples collected near simultaneously from the jugular and saphenous veins

Statistically significant
CT = clot time; CFT = clot formation time; α = alpha angle; A10 and A20 = amplitude at 10 and 20 mins, respectively; MCF = maximum clot formation; LI30 and LI45 = lysis index at 30 and 45 mins, respectively
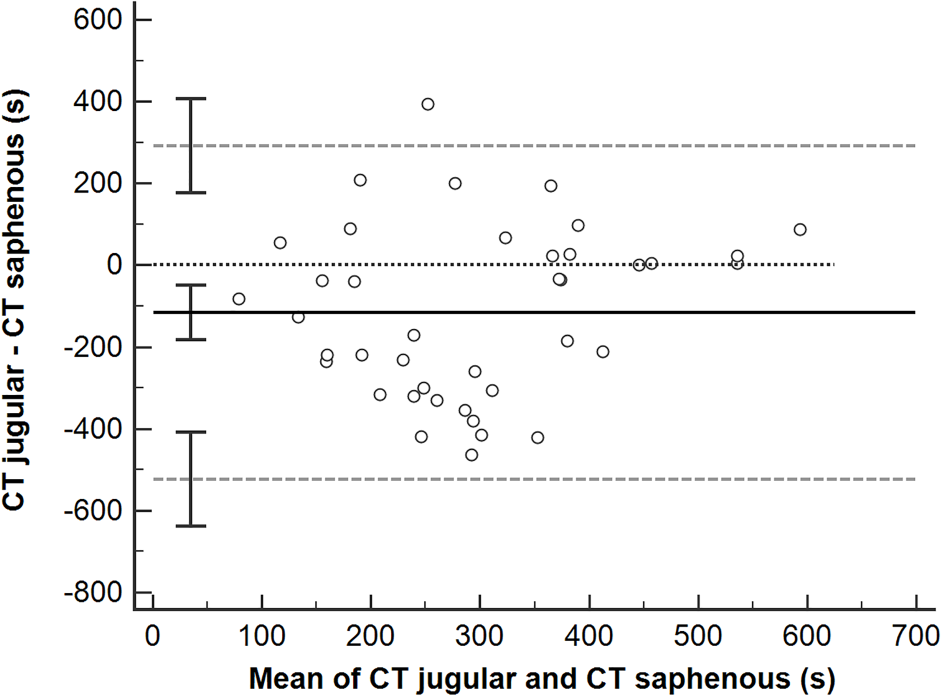
Bland–Altman plot comparing clot time (CT) for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals
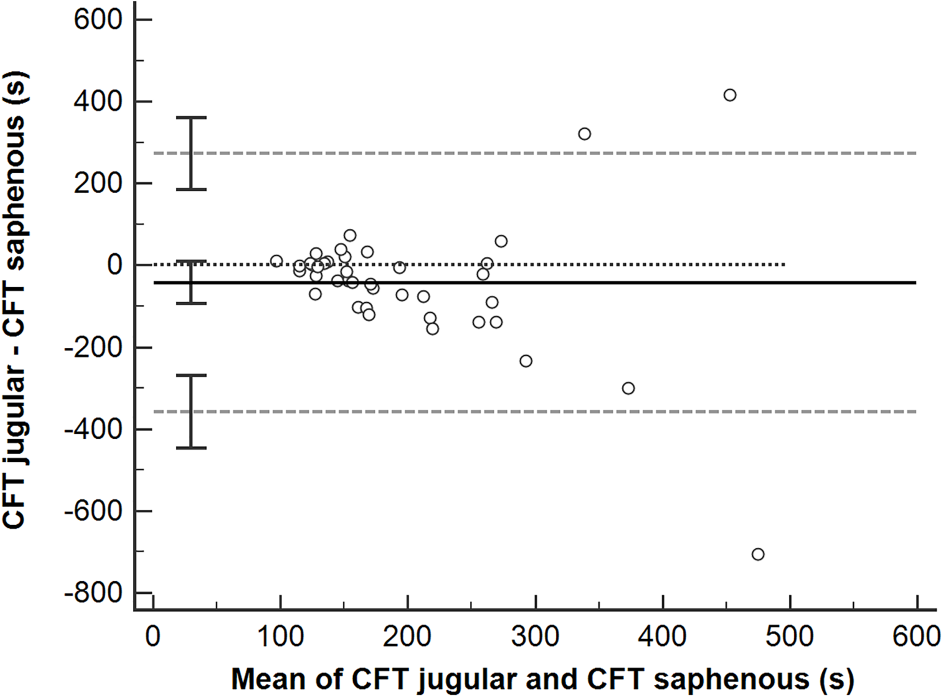
Bland–Altman plot comparing clot formation time (CFT) for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals

Bland–Altman plot comparing alpha for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals

Bland–Altman plot comparing maximum clot formation (MCF) for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals

Bland–Altman plot comparing amplitude at 10 mins (A10) for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals

Bland–Altman plot comparing amplitude at 20 mins (A20) for paired jugular and saphenous samples. Solid black line = mean difference between paired samples; black dotted line = line of identity; gray dashed line = limits of agreement; error bars = 95% confidence intervals
Coefficient of variation for paired samples collected from the jugular and saphenous veins

CT = clot time; CFT = clot formation time; α = alpha angle; A10 and A20 = amplitude at 10 and 20 mins, respectively; MCF = maximum clot formation; LI30 and LI45 = lysis index at 30 and 45 mins, respectively
Discussion
This study demonstrates that samples collected from the jugular vein using a larger bore needle are relatively hypercoagulable when compared with their matched sample collected with a smaller bore needle from the medial saphenous vein.
Both sampling sites and needle gauges have been previously used in viscoelastic studies in cats.10,11 Identification of statistical differences between the two sites suggest that the same site and needle gauge should be used consistently for serial monitoring or for research purposes. Our findings also suggest that different reference intervals (RIs) may be required for different venipuncture techniques. The extent to which we can interpret the clinical importance of the observed differences is limited because, currently, only a canine RI is supplied by the manufacturer, and data on expected results for healthy cats are available only in abstract form. 12 However, the large CV between paired samples for multiple parameters, particularly CT, suggest that the observed difference might be clinically relevant.
Previous canine and feline studies using recalcified citrated samples have not identified effects of needle or catheter gauge on viscoelastic parameters,10,13 and canine studies have not detected differences between jugular and saphenous samples. 9 The more hypercoagulable results for jugular venipuncture in the current study suggest immediate analysis of non-anticoagulated blood is more sensitive to factors such as the thicker skin overlying the jugular vein, and thus greater comparative trauma and potential for tissue factor contamination with jugular vs saphenous venipuncture. 14
Tissue factor contamination may have had greater impact on our results than those of previous studies because of differences in strength of contact pathway activation between the VCM Vet and TEG or ROTEM assays. In studies involving TEG or ROTEM, the addition of a contact pathway activator (eg, kaolin) reduces the variability of viscoelastic assays. 5 The VCM Vet cartridges do not include kaolin, and instead platelet and contact pathway activation occurs through contact with glass slides. This is not directly equivalent to analysis of samples without the addition of an activator by TEG analysis, as the cups and pins in contact with the sample during TEG analysis are manufactured from Cyrolite, a poly(methylmethacrylate)-based polymer, which, unlike glass, is a poor activator of the contact pathway. 3 Publications defining the contact pathway and platelet activation induced by the VCM Vet cartridges are not currently available, but in the current study, estimates of CV for paired measurements are broadly similar to those reported for feline samples analyzed by TEG without an activator. 15 This, together with the more hypercoagulable results for jugular venipuncture, suggests that the contact pathway activation provided by the VCM Vet cartridges may not correct for tissue factor contamination to the same extent as kaolin activation.
In the current study, a discard sample was not used, but samples were only included if venipuncture was considered atraumatic. This was based on a study of citrated canine samples analyzed by non-activated TEG, which concluded that a discard tube is not necessary when blood is collected by clean venipuncture. 16 However, our results suggest an important next step in optimizing the feline sample collection protocol for the VCM Vet is determining the extent to which discard samples can reduce tissue factor contamination, and thus variability in VCM Vet parameters.
The Bland–Altman plots identify occasional samples where differences are larger than for the majority of samples, suggesting that there may be some source of random error, such as problems with cartridge filling or clot retraction within a test cartridge during analysis. 11 An additional limitation is the collection of all samples under anesthesia. Relatively little information is available regarding the effect of anesthetic and sedative agents on viscoelastic testing. In cats, butorphanol and midazolam sedation produced statistically significant, but likely clinically insignificant, changes in kaolin-activated TEG parameters. 17 To the best of our knowledge, the effects of the specific drugs used here on feline viscoelastic testing, or the potential differential effects of these drugs on samples collected from different veins, have not been assessed. It is possible that the alterations in vascular tone induced by dexmedetomidine and isoflurane may have influenced our results. 18
Similarly, our findings do not incorporate the differential effects on jugular vs saphenous collection of patient non-compliance, and thus are not directly transferable to sample collection from conscious patients. It should also be noted that this study includes pregnant animals, and although all animals were apparently healthy, extensive pre-anesthetic health investigations were not performed. Each cat acts as its own control, so we consider it unlikely that this would have directly influenced our findings. However, since pregnancy is associated with viscoelastic changes in dogs, 19 our reported medians and ranges should not be interpreted as indicative of expected results in healthy non-pregnant cats.
Conclusions
Samples taken from the jugular vein with a larger gauge needle appeared relatively hypercoagulable as compared with the matched saphenous venous sample. This suggests that the same site and needle gauge should be used consistently for serial monitoring, or for collecting study data.
Footnotes
Acknowledgements
The authors thank Faith Northcutt, Bailey Boyanowski, Kaylie Farley, Katherine Sturgill and John Hall for their technical support, especially with monitoring blood collection technique. Further, the authors extend their gratitude to Bernadette Vogel for study administrative support and Sandra Montalbo for the table design.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Duffield Family Foundation, Maddie’s Fund (May 11, 2018).
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not necessarily required, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
