Abstract
The TEG 6s (Haemonetics) point-of-care viscoelastic analyzer is portable, compact, simple to use, and has the potential for rapid viscoelastic analysis that can guide the treatment of veterinary patients at the site of care. Although approved for use in people, the TEG 6s has yet to be evaluated for hemostatic analysis in veterinary medicine. Citrated whole blood (CWB) was collected from 27 healthy dogs. An aliquot of CWB from each dog was diluted by 33% with an isotonic crystalloid, representing an in vitro model of hemodilution. Unaltered and diluted CWB samples were analyzed using 2 TEG 6s and 6 TEG 5000 (Haemonetics) analyzers. The 6 TEG 5000 analyzers ran duplicate analyses of either unaltered or diluted samples using 1 of 3 reagents (Haemonetics): Kaolin TEG, RapidTEG, or TEG Functional Fibrinogen. Duplicate TEG 5000 analyses were averaged and compared with a single TEG 6s analysis. Lin concordance correlation coefficient and Bland–Altman plots were used to evaluate agreement of reaction time, kinetic time, alpha angle, maximum amplitude (MA), and G value (G) for samples activated with Kaolin TEG, and agreement of MA for samples activated with RapidTEG between the 2 machines. Overall, agreement between the TEG 6s and TEG 5000 analyzers was poor. Viscoelastic measurements by the TEG 6s and TEG 5000 in healthy dogs were not all interchangeable. Agreement was satisfactory only for MA and G measurements of diluted blood samples activated with Kaolin TEG, and MA measurements for both unaltered and diluted blood samples activated with RapidTEG.
Viscoelastic testing enables global assessment of hemostasis, measuring the dynamics of fibrin formation, clot strength, platelet function, and fibrinolysis. 27 Compared to standard plasma-based coagulation tests, such as prothrombin time and activated partial thromboplastin time, viscoelastic tests reflect more closely the in vivo cell-based model of hemostasis, 21 allowing for more accurate understanding of a patient’s current coagulation status.24,27 Viscoelastic tests serve as a valuable adjunct for individualized clinical decision-making in veterinary patients with conditions accompanied by altered hemostasis, such as trauma, 16 spontaneous hemoperitoneum, 12 disseminated intravascular coagulation, 2 sepsis, 37 immune-mediated hemolytic anemia, 15 neoplasia, 25 renal disease, 10 intestinal disease, 8 hepatic disease, 13 iatrogenic anticoagulation,1,26,28 pyometra, 9 and pancreatitis. 32
Two of the most common benchtop viscoelastic analyzers used in veterinary medicine are the TEG 5000 Thrombelastograph (Haemonetics) and the ROTEM delta (Werfen).6,30 A major limitation of benchtop viscoelastic analyzers is the requirement for blood samples to be processed shortly after sample collection. Whole blood samples for the TEG 5000 should be analyzed within 4–6 min of collection. 40 Citrated blood samples for the TEG 5000 should be analyzed after a 30-min holding period at room temperature, but any delay in processing by >30 min after the holding period may significantly alter TEG results.11,40 Another major limitation of benchtop viscoelastic analyzers is the need for operators specially trained in sample handling and manual pipetting of reagents and blood to mitigate significant pre-analytical error.7,11,30 Additionally, benchtop analyzers should be housed in a controlled environment because analysis is vulnerable to vibration.11,30 Because of these limitations and the cost of the machines, consumables, reagents, quality control, and servicing, viscoelastic testing is typically performed only at the largest veterinary referral centers with clinical pathology laboratories, which are often located distant to the site of care on the clinic floor.5,29
The TEG 6s hemostasis analyzer (Haemonetics) is a relatively new, commercial, point-of-care, viscoelastic analyzer designed to mitigate many of the limitations of benchtop viscoelastic analyzers. 17 The TEG 6s has been validated for use in humans, 17 but has not been evaluated for use in veterinary patients, to our knowledge. The unit employs a single-use, disposable cartridge (TEG global hemostasis assay; Haemonetics) and several milliliters of citrated whole blood (CWB), which is transferred into the loaded cartridge. It takes only 1–2 min to setup and start the analysis. Immediately upon starting the analysis, viscoelastic data from 4 simultaneously performed TEG assays are displayed in near real-time.
The TEG 6s global hemostasis cartridge includes the traditional intrinsic pathway–activated Kaolin TEG assay, as well as 3 nontraditional assays: Kaolin TEG with Heparinase, RapidTEG, and TEG Functional Fibrinogen. 4 The Kaolin TEG with Heparinase assay eliminates any effect of heparin in the patient sample, allowing for identification of systemic heparin or heparinoids when used in conjunction with the Kaolin TEG assay. 20 The RapidTEG assay uses an activator that consists of kaolin and tissue factor, which generates TEG results more rapidly. 22 The RapidTEG assay can be used to assess hemostasis of veterinary species (dog, cat, horse) in ~15 min. 4 Studies evaluating the TEG 5000 analyzer with the RapidTEG activator in healthy dogs have shown mixed results using RapidTEG to monitor rivaroxaban therapy,1,26 as well as results supporting using RapidTEG for monitoring unfractionated heparin therapy. 28 The TEG Functional Fibrinogen assay uses an activator that consists of tissue factor and abciximab, which binds to human platelet glycoprotein (GP) IIb/IIIa receptors, inhibiting platelet aggregation.33,38 In people, the TEG Functional Fibrinogen assay excludes the platelet contribution to clot strength, allowing for estimation of fibrinogen contribution to the clot when used in conjunction with the Kaolin TEG assay. 4 We found no reports of evaluation of the TEG Functional Fibrinogen activator in canine TEG analysis.
Similar to the TEG 5000, the TEG 6s requires that citrated blood samples be tested 15 min to 2 h post-draw. 18 Therefore, both the TEG 5000 and TEG 6s are limited to “near-patient testing.” However, compared to the TEG 5000, the TEG 6s has many advantages. The TEG 6s analyzer is more compact and portable, which may make the TEG 6s more appealing to veterinary hospitals with limited laboratory space or to veterinary personnel caring for working dogs at field hospitals or combat support hospitals. The TEG 6s employs simple-to-use, cartridge-based technology, which may allow for easier training of veterinary personnel, preventing the need to call-in specially trained operators after hours. The cartridge-based technology eliminates the need for manual pipetting of blood and reagents, which may reduce pre-analytical error. Also, the cartridge-based technology allows for the simultaneous generation of 4 separate TEG assays for each analyzed sample; the TEG 5000 generates only 1 TEG assay for each analyzed sample.
The TEG 5000 and the TEG 6s rely on different technologies for measuring clot viscoelasticity: the cup-and-pin method versus the resonance-frequency method, respectively. The TEG 5000 consists of 2 mechanical parts: an oscillating cup, which holds the activated blood sample, and a pin, which is suspended in the sample via a torsion wire. 27 Initially, the liquid blood sample in the oscillating cup does not affect the pin. 27 However, as coagulation proceeds and fibrin strands “tether” the oscillating cup to the pin, torsional forces are transmitted through the pin to be recorded by a non-contact rotation sensor, resulting in a computer-generated TEG trace.17,27 In contrast, the TEG 6s assay uses a disposable, 4-channel, microfluidic cartridge that requires only the addition of an unmetered CWB sample (~0.4 mL). Under fully automated instrument control, dry TEG reagents are reconstituted, and prepared samples are delivered to 4 separate test cells, in which coagulation is monitored. Individual test cells consist of a short vertical tube open at the top and bottom ends, in which blood is held by surface tension. An optical sensor focuses on the convex meniscus that is formed by the sample hanging from the bottom opening of the test cell. The optical sensor measures the up-and-down motion of the sample meniscus as a vibration frequency is applied to the sample. The frequency that causes resonance is identified and converted into a computer-generated TEG trace. As clotting occurs, resonant frequencies increase.17,19 Both the TEG 6s and TEG 5000 generate a qualitative tracing and quantitative measures describing global hemostasis.
To our knowledge, the use of the TEG 6s has not been reported previously in veterinary patients. Our objective was to determine agreement of the TEG 6s measurements with those of the TEG 5000 in healthy dogs. We hypothesized that results from the TEG 6s would agree with its predecessor, the TEG 5000, and produce clinically relevant data regarding the coagulation status of dogs.
Materials and methods
Animals
Our study was approved by the Department of Defense Military Working Dog Veterinary Service’s Institutional Animal Care and Use Committee (DODMWDVS IACUC). All animals used in our study were active government-owned canines (GOCs)—either military working dogs (MWDs) or Transportation Security Administration (TSA) working dogs. Dogs were included in the study if they were in good health with no detectable inflammatory, debilitating, or systemic disease based on history, physical examination, routine hematology, and serum biochemistry analysis. Dogs were excluded from our study if they were being given medication known to interfere with coagulation.
Sample size
An initial pilot phase of the study included 10 GOCs to establish the variability of data collected from the TEG 6s and TEG 5000 analyzers. A power and sample size calculation was then performed using these data to determine the total number of animals necessary to obtain meaningful results based on a clinically relevant effect size, desired power of 80%, and 95% CIs. Once the study sample size was estimated, an addendum to the protocol was submitted to the DODMWDVS IACUC requesting the use of 27 GOCs.
Blood sample collection
Citrated whole blood samples were obtained from 27 GOCs. We collected 10 mL of whole blood via single, atraumatic venipuncture of either the lateral saphenous, jugular, or cephalic vein using a butterfly needle (20 or 18 gauge) with blood collection set inserted directly into vacuum tubes (two 5-mL plastic tubes containing 3.2% buffered sodium-citrate; Vacutainer citrate tubes, Becton Dickinson). Blood tubes were filled by vacuum. All blood samples were obtained from GOCs that were undergoing blood collection for wellness or pre-anesthetic screening, and were collected using a DODMWDVS routine procedure without sedation or tranquilization.
Sample dilution
Blood samples obtained from each GOC were divided into 2 groups: normal blood (
TEG analysis
Blood sample collection and processing for TEG analyses followed the Partnership on Rotational ViscoElastic Test Standardization recommendations for veterinary viscoelastic testing. 14 At 30 min following blood pooling and sample dilution, unaltered and diluted blood samples were tested simultaneously using 8 TEG analyzers: 2 TEG 6s systems (using the global hemostasis cartridge) and six 2-channel TEG 5000 systems. The 6 TEG 5000 systems performed duplicate analyses of either unaltered or diluted blood samples using 1 of 3 reagents: Kaolin TEG, RapidTEG, or TEG Functional Fibrinogen. The TEG 5000 assays were run until the analyzer returned results for the following parameters: reaction time (R), kinetic time (K), alpha angle (A), and maximum amplitude (MA). The G value (G) with the unit dynes/cm² (d/sc) was calculated using the MA for each trace as described previously. 23 Clot lysis at 30 min (LY 30) and 60 min (LY 60) were not measured on the TEG 5000 given that these parameters are not available for testing with the TEG 6s. Data were collected through TEG analytical software, downloaded, and entered into an Excel spreadsheet (v.16.58; Microsoft). All TEG assays were performed by experienced research personnel following the manufacturer’s instructions for the given analyzers and activators. All TEG 6s assays were performed by one individual, and all TEG 5000 assays were performed by another individual.
Using the TEG 6s, 162 analyses were performed: 1 unaltered blood sample from each dog and 1 diluted blood sample from each dog, using either the Kaolin TEG assay (U.T6.KT and D.T6.KT, respectively), RapidTEG assay (U.T6.RT and D.T6.RT, respectively), or TEG Functional Fibrinogen assay (U.T6.FFT and D.T6.FFT, respectively; Fig. 1). For the Kaolin TEG assays, R, K, A, MA, and G were measured. Only MA was measured for the TEG 6s RapidTEG and TEG Functional Fibrinogen assays. Using the TEG 5000, 324 analyses were performed: duplicate analyses of 1 unaltered blood sample from each dog and 1 diluted blood sample from each dog, activated with either Kaolin TEG (U.T5.KT and D.T5.KT, respectively), RapidTEG (U.T5.RT and D.T5.RT, respectively), or TEG Functional Fibrinogen (U.T5.FFT and D.T5.FFT, respectively). For all TEG 5000 analyses, R, K, A, MA, and G were measured, regardless of reagent.
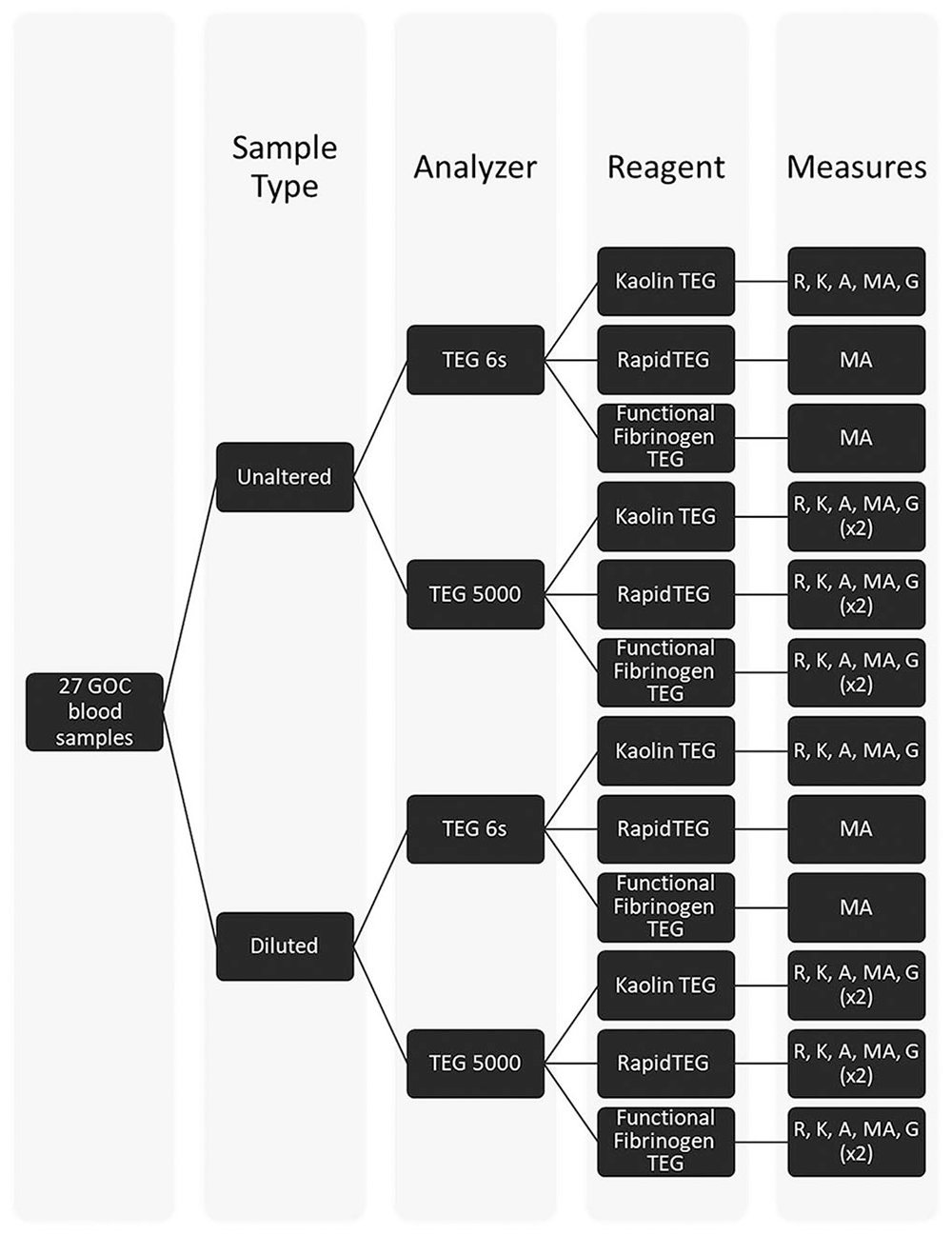
Processing and TEG analysis of government-owned canine blood samples. A = alpha angle; G = G value; GOC = government-owned canine; K = kinetic time; MA = maximum amplitude; R = reaction time.
Agreement analysis
Our statistical analysis focused on the agreement between a single analysis using the TEG 6s machines and the mean of 2 replicate analyses using the TEG 5000 machines. A single analysis from the TEG 6s machines was determined a priori to be sufficient because the TEG 6s is an automated, cartridge-based test and is less subject to pre-analytical error. The mean of the 2 TEG 5000 replicates provides a more reasonable value for comparison because the TEG 5000 measurement is more subject to pre-analytical error. The averaged results from the TEG 5000 were considered the “gold standard” measurements for comparison. Measurements less than the lower bound of the machine-specific reportable value were assumed to be equal to the lower limit of the analytic range (
Pre-analytical error
In 9 dogs, 1 of the TEG 5000 duplicate analyses showed an inappropriate tracing that was neither representative of a physiologic nor pathologic process. Therefore, averages were not calculated for these 9 dogs, and only the normal duplicate value was included in the statistical analysis. This pre-analytical error occurred in 2 U.T5.KT, 2 U.T5.RT, 1 U.T5.FFT, 2 D.T5.KT, and 2 D.T5.FFT analyses. In another dog, both of the duplicate U.T5.RT analyses had an inappropriate tracing that did not represent physiologic or pathologic processes. These values and the corresponding U.T6.RT values were excluded from the CCC and Bland–Altman analyses. Finally, the TEG 6s analysis of one of the diluted samples from an additional dog was performed after the predetermined 30-min holding period. This value and the average value of the corresponding TEG 5000 analyses were also excluded from the CCC and Bland–Altman analyses.
Results
No dogs were excluded for health reasons or laboratory abnormalities. Dogs were 1–8-y-old (x̄ = 2.7 y). The breed distribution was as follows: 12 German Shepherds, 8 German Shorthaired or Wirehaired Pointers, 4 Belgian Malinois, and 3 Labrador Retrievers. There were 15 intact males, 7 castrated males, 3 intact females, and 2 spayed females.
For unaltered and diluted blood samples activated with TEG Functional Fibrinogen and analyzed using the TEG 6s, 19 and 7, respectively, had MA measurements greater than the upper bound of the machine-specific reportable value (i.e., 52 mm; Table 1). The minimum MA measurements for the remaining unaltered and diluted samples were 37.2 mm and 32.3 mm, respectively. For samples analyzed using the TEG 5000 and activated with TEG Functional Fibrinogen, 51 of the 54 duplicate analyses of unaltered blood samples and 38 of the 54 duplicate analyses of diluted blood samples had a MA >52 mm, with minimum values of 48.3 mm and 37.8 mm, respectively.
Descriptive statistics for TEG analytic measures of coagulation in dogs, by blood sample type.
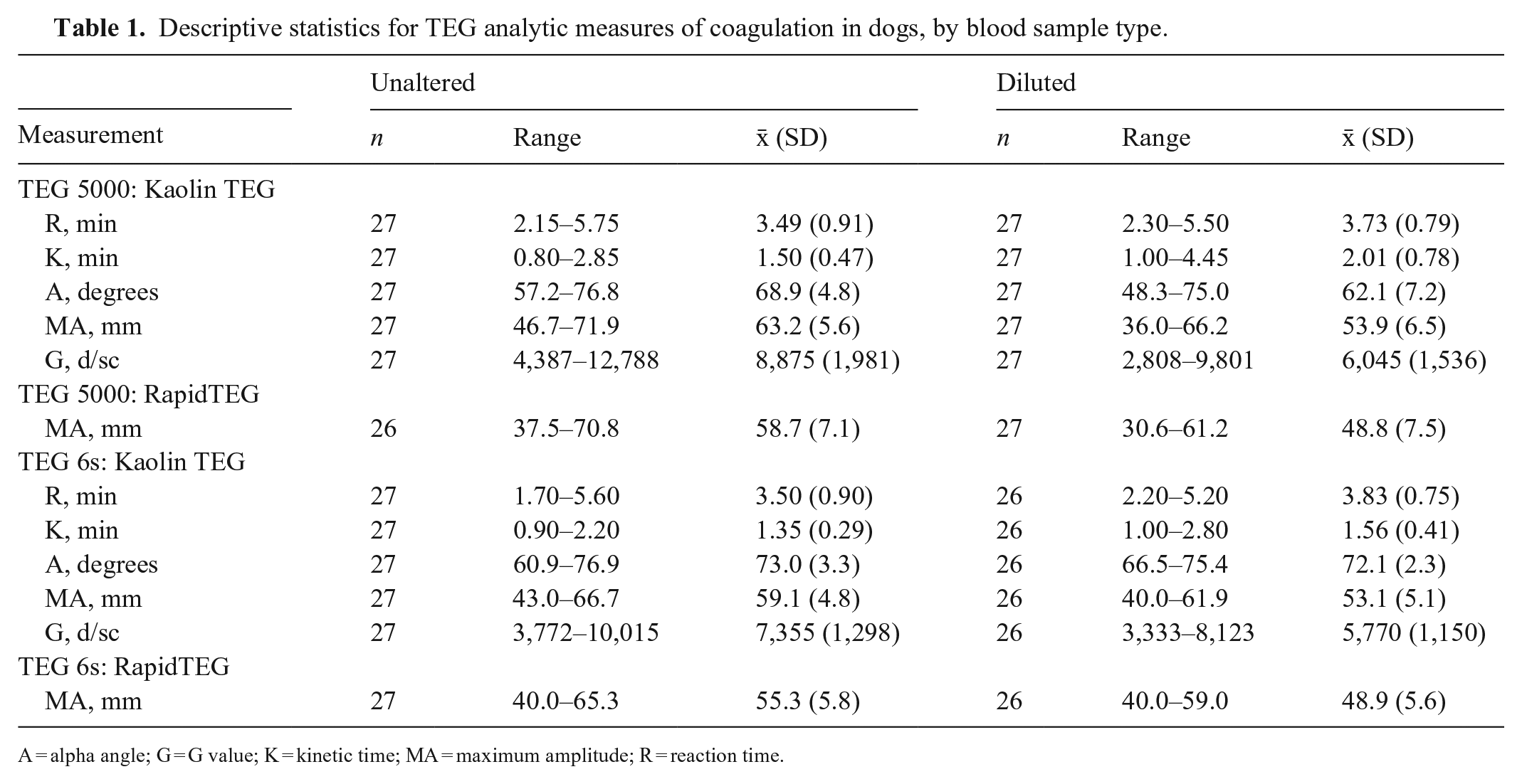
A = alpha angle; G = G value; K = kinetic time; MA = maximum amplitude; R = reaction time.
Generally, agreement between the TEG 6s and TEG 5000 analyzers, as assessed by the CCC LCI (Figs. 2, 3) and Bland–Altman plots (Suppl. Figs. 1, 2), was poor. Agreement was acceptable only for the MA and G measurements for diluted blood samples activated with Kaolin TEG and the MA measurement for unaltered and diluted blood samples activated with RapidTEG. In addition, significant fixed bias (mean difference between the analyzers) was found in several measurements.
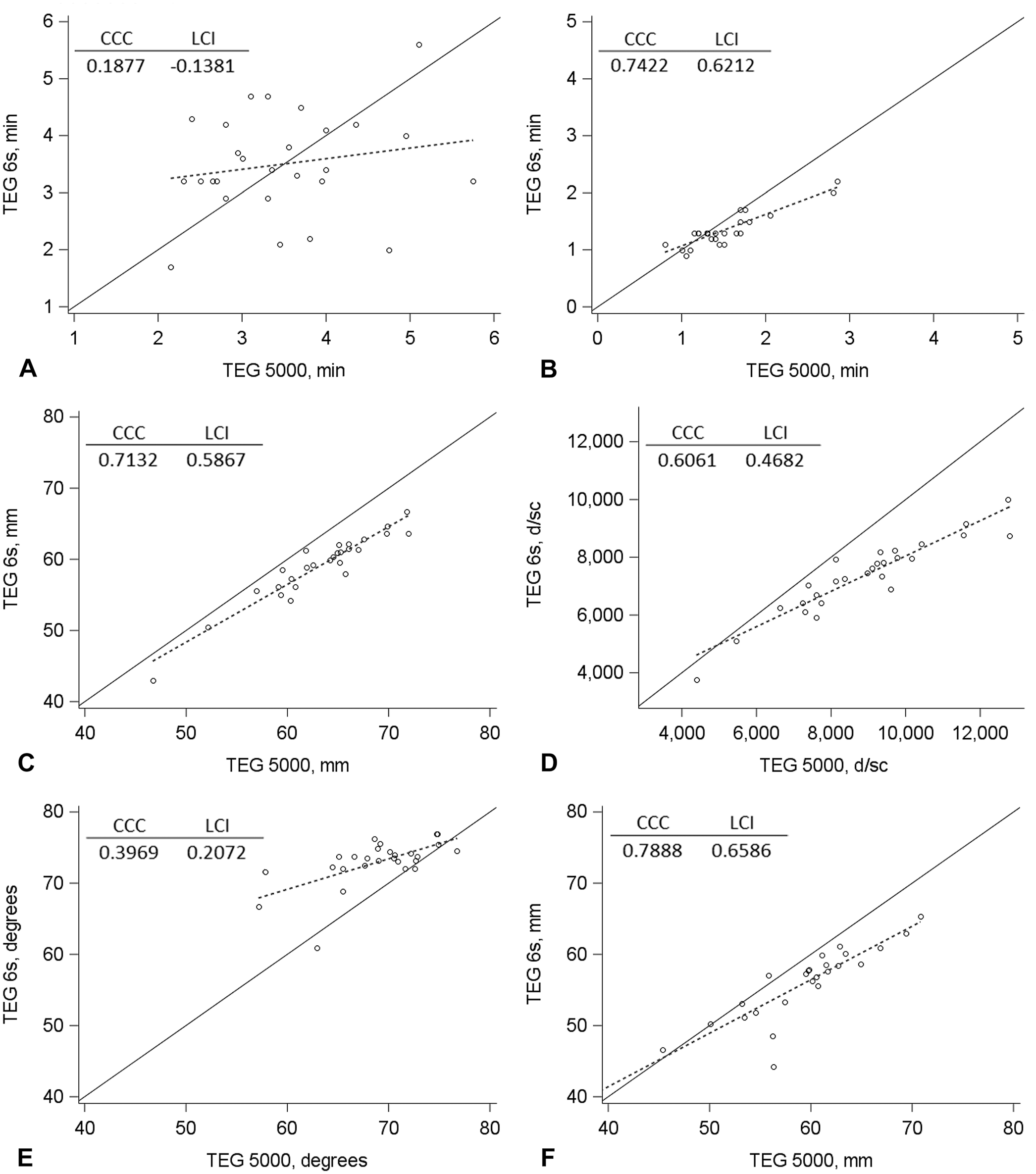
Concordance plots of
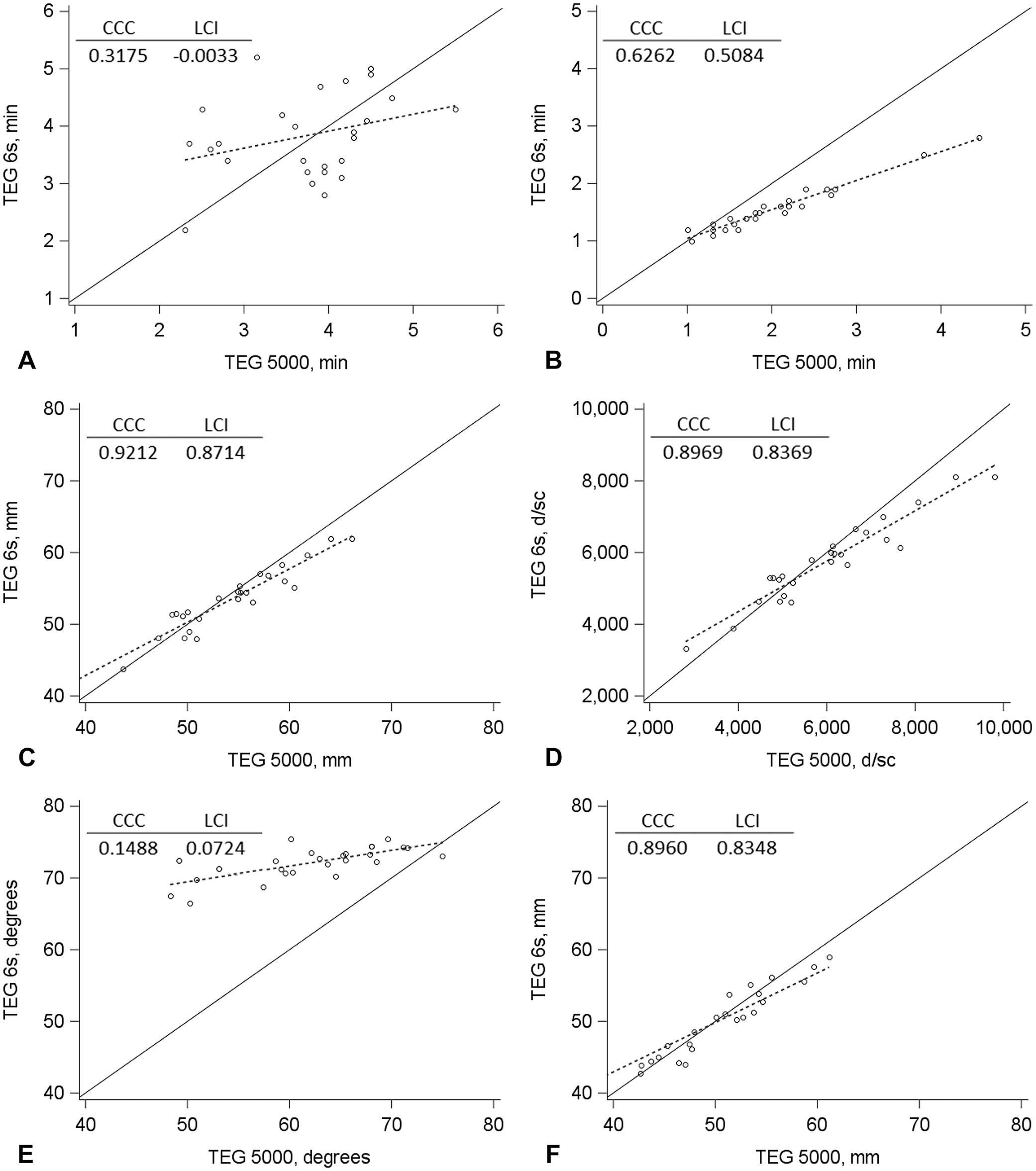
Concordance plots of
Discussion
Overall agreement was poor between TEG 6s and TEG 5000 measurements when using blood samples from healthy GOCs. Our findings differ from those of studies that have shown agreement between the TEG 6s and TEG 5000 analyzers for standard hemostasis tests in various populations of human patients.17,31
Studies have demonstrated divergent results between different viscoelastic analyzers (e.g., between the TEG 5000 and ROTEM analyzers), concluding that viscoelastic measures are not interchangeable between different analyzers.35,36,38 One possible explanation for a lack of interchangeability of results from the TEG 5000 and ROTEM analyzers is the use of different reagents and, thus, different coagulation triggers. Given that the TEG 5000 and TEG 6s analyzers use the same reagents, differences in coagulation triggers are unlikely the cause for non-agreement of results from these 2 analyzers. Possible explanations for a lack of interchangeability between results from the TEG 5000 and TEG 6s analyzers include different technologies (resonance-frequency method vs. cup-and-pin method) used by the 2 systems, different physical properties of the TEG 6s cartridge and TEG 5000 cup and pin, different volumes of blood and reagent required for each assay, and wide coefficient of variance of viscoelastic analyzers (the coefficient of variability for TEG and ROTEM is commonly reported as 30%).35,38
Neuter status in female dogs has been shown to significantly influence viscoelastic analysis using the ROTEM delta analyzer.
34
However, given the small number of female dogs (
To our knowledge, the use of the TEG Functional Fibrinogen reagent for canine viscoelastic analysis has not been reported previously. All TEG Functional Fibrinogen MA values measured by the TEG 6s exceeded the upper limit of the analytic range (15–32 mm). Both the TEG 6s and TEG 5000 Functional Fibrinogen assays showed considerable alteration in TEG tracings when compared to Kaolin TEG, which corroborated results from a study that evaluated TEG analysis of canine blood using abciximab, an active ingredient in the TEG Functional Fibrinogen reagent; abciximab had no effect on TEG tracings but caused profound inhibition of platelet aggregation as measured in the PFA-100 platelet function analyzer (Siemens). 3 Abciximab is a monoclonal Fab-fragment that binds with strong affinity to the human platelet GP IIb/IIIa receptor, inhibiting platelet-to-platelet binding via fibrin bridges and, thus, inhibiting the formation of a strong clot under high-shear conditions. 39 Speculated reasons as to why abciximab’s profound inhibition of platelet aggregation is not reflected in TEG analysis of dog blood include lower shear stress in TEG assays compared to the systemic circulation, species differences in abciximab affinity for canine platelet GP IIb/IIIa receptors, species differences for abciximab affinity for other cellular components involved in hemostasis, and species differences in abciximab kinetics. 3 Based on the observations from our study and findings of other studies, the recommendation stands that the TEG Functional Fibrinogen assay should not be used to determine functional fibrinogen concentrations in dogs. 4
Although we observed overall poor agreement between the TEG 6s and TEG 5000, this should not be interpreted as the TEG 6s being an unreliable assay for evaluation of coagulation status of dogs. Rather, the TEG 6s provides different values for coagulation status compared to the TEG 5000, and these values may indeed be useful to assess a dog’s coagulation status. Furthermore, the ability to perform multiple tracings simultaneously may lead to a more robust understanding of a dog’s coagulation status. However, further studies are needed to establish TEG 6s RIs for healthy dogs and to evaluate the TEG 6s in dogs with clinical coagulopathies. Establishment of cutoff values on the TEG 6s for each variable will enable clinicians to utilize this device for decision-making and possibly therapeutic interventions.
Some of the limitations of our study are inherent to the assays themselves, including the wide coefficient of variance and possible variations as a result of patient signalment. Given differences in patient physiology, RIs for each TEG 6s variable could vary depending on a given patient’s age, sex, or breed. Another limitation is the relatively small sample size of a somewhat homogeneous sample of dogs. The dogs that we tested are relevant to MWDs and civilian working dogs, which have a high risk of traumatic injuries leading to altered hemostasis, and thus may not fully represent the larger population of dogs.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221112638 – Supplemental material for Comparison of assessment of coagulation in healthy dogs by the TEG 6s and TEG 5000 viscoelastic analyzers
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221112638 for Comparison of assessment of coagulation in healthy dogs by the TEG 6s and TEG 5000 viscoelastic analyzers by Lance R. Wheeler, Thomas H. Edwards, Justin A. Heinz, Laura L. F. Scott, Lonnie E. Grantham, Jeffrey D. Keesee, Alice F. Henderson, Angelina C. Gerardo, Guillaume Hoareau and James A. Bynum in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
DoD disclaimer: the views expressed in our manuscript are those of the authors and do not reflect the official policy or position of the U.S. Army Medical Department, the Department of the Army, DoD, or the U.S. Government.
Our research was supported in part by an appointment to the Research Participation Program at the U.S. Army Institute of Surgical Research (USAISR) administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and USAISR.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, or publication of this article.
Funding
Funding for this study was provided by the Department of Defense.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
