Abstract
Viscoelastic testing methods such as thromboelastography (TEG) are becoming increasingly available to veterinarians in a clinical setting. TEG is useful in determining therapeutic transfusion needs and assessing global abnormalities of hemostasis of patients, given that it provides a more comprehensive assessment of coagulation than traditional tests. TEG is standardly performed at 37°C, which is considered a normal body temperature for human patients; however, 37°C is lower than normal body temperature for most canine patients. In an in vitro study, we investigated the potential effect that this difference in body temperature and test temperature might have on TEG results. Citrated blood samples were collected from clinically normal, as well as sick, dogs with various body temperatures. Samples were analyzed concurrently at the patient’s body temperature and at 37°C. There was very high correlation between TEG performed at body temperature and at 37°C for R (min) and MA (mm), high correlation for K (min) and alpha angle (deg), and moderate correlation for LY30 (%) and LY60 (%). For canine patients with normal to mildly abnormal body temperatures, performance of TEG at the standard 37°C is acceptable.
Keywords
Viscoelastic testing methods including thromboelastography (TEG) are becoming increasingly available in veterinary hospitals to assess the coagulation status of patients. Compared to traditional assays such as prothrombin time and activated partial thromboplastin time, viscoelastic methods yield a more comprehensive assessment of coagulability reflecting the cell-based rather than the traditional cascade model of coagulation. Hence, viscoelastic tests are used routinely in human medicine to predict various abnormalities in hemostasis, as well as to determine treatment course.8,9 TEG assesses the viscoelastic properties of a clot from initial formation through fibrinolysis, producing a tracing based on several variables that reflect different components of the cell-based model of coagulation, including clot formation, strength, and lysis. Clot formation is quantified by reaction time (R), kinetic time (K), and alpha angle. 7 Clot strength is described by maximum amplitude (MA). Clot lysis is reflected by the percentage return to baseline at 30 and 60 min after MA (LY30 and LY60, respectively).
Although TEG is an extremely useful tool for evaluation of hemostasis in the clinical setting, its methodology is complex, and patient and analytical variability can affect the results. In human patients, TEG is traditionally performed at a machine set temperature of 37°C, reflective of normal human body temperature. Body temperature is known to have an effect on coagulation, with hypothermia causing decreased coagulation enzymatic activity, decreased platelet function, and disordered fibrinolysis. 15 Temperature-corrected TEG has been evaluated in human patients in a variety of settings with somewhat conflicting results, but suggesting that, in the majority of clinical settings, analysis at 37°C is appropriate.4,6,10,11
In 2014, the Partnership on Rotational ViscoElastic Test Standardization (PROVETS) published evidence-based guidelines on viscoelastic assays in veterinary medicine. 7 One of the questions the guidelines attempted to answer was whether analysis at 37°C compared to analysis at patient body temperature alters TEG results. The panel found insufficient evidence in veterinary patients to recommend the use of a species-specific temperature or patient body temperature for TEG analysis. In the absence of evidence, it was suggested that routine TEG assays be performed at the standard 37°C. It was also suggested that, in patients with markedly abnormal body temperature, clinicians consider performing the tracings at both temperatures, 37°C and body temperature.
To our knowledge, the effect of TEG test temperature has not been reported in dogs, whose normal body temperature is often >37°C, the standard assay temperature. Although there are many published normal ranges of canine body temperature, several veterinary reviews on hypothermia classify mild hypothermia in dogs as temperatures including 32–37°C.1,12 Performing TEG at 37°C may not be appropriate for canine patients; therefore, we investigated the effect of test temperature on canine TEG tracings. We hypothesized that performing TEGs concurrently at the patient’s body temperature and at 37°C would yield similar results, with any differences being small and clinically irrelevant.
Materials and methods
Animals
Our study was approved by the University of Pennsylvania (Philadelphia, PA, USA) Institutional Animal Care and Use Committee and the Matthew J. Ryan Veterinary Hospital of the University of Pennsylvania (MJR-VHUP) Privately Owned Protocol Animal Committee. Dogs were enrolled in the study if they were patients at MJR-VHUP or if they were healthy patients belonging to staff members. All dogs were enrolled after informed owner consent was obtained. No attempt was made to discriminate based on signalment or underlying disease process. Patients were excluded if their rectal temperature was exactly 37°C or if their underlying condition precluded the collection of blood for the study. Demographic data were collected including patient sex, age, breed, weight, and chief complaint.
We enrolled 51 patients in the study, including 23 healthy pets and 28 sick patients representing a variety of signalments and a range of body temperatures (Table 1). Breeds included 14 mixed-breed dogs, 4 Golden Retrievers, 3 Boxers, 2 American Pitbull Terriers, 2 Labrador Retrievers, 2 Shih Tzus, 2 Weimaraners, and 1 of each of the following breeds: Bichon Frise, Boston Terrier, Brittany Spaniel, Chesapeake Retriever, Dachshund, English Cocker Spaniel, French Bulldog, Goldendoodle, Gordon Setter, Great Dane, German Shepherd, German Shorthair Pointer, Irish Setter, Miniature Schnauzer, Maltese, Norwich Terrier, Pomeranian, Rottweiler, Shetland Sheepdog, Standard Poodle, Welsh Terrier, and Yorkshire Terrier.
Demographic data of patients and temperatures.
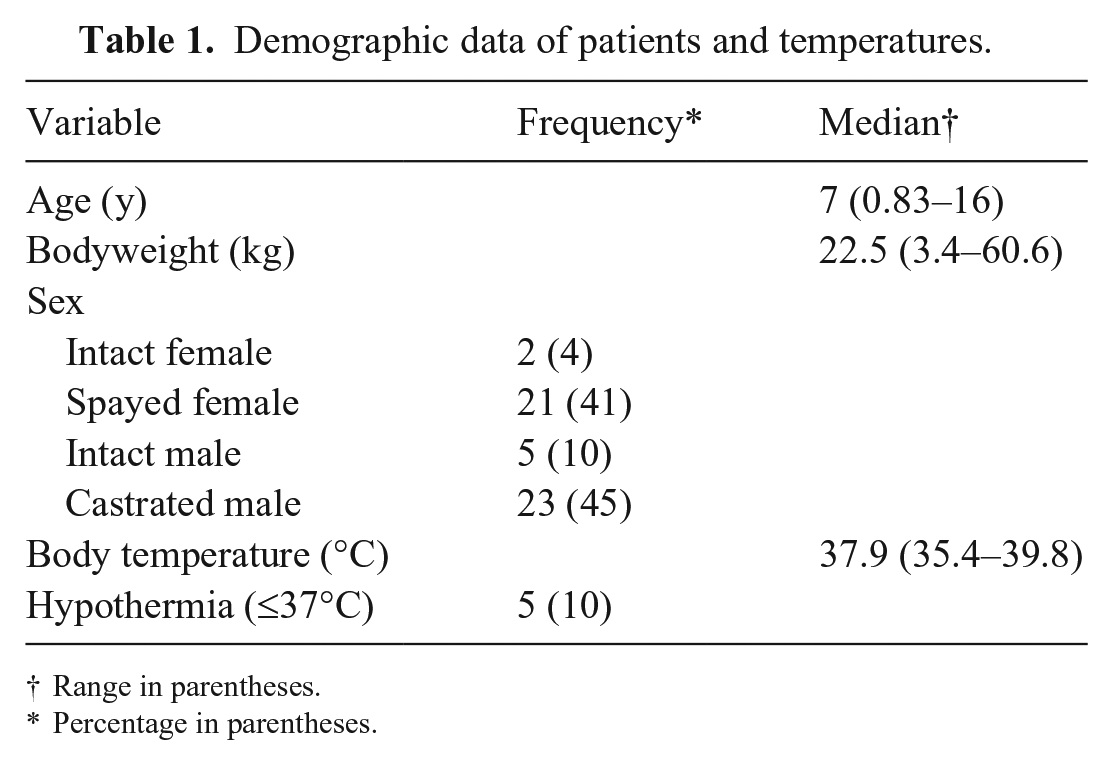
Range in parentheses.
Percentage in parentheses.
Immediately prior to blood collection, rectal body temperature was measured (Lumiscope model 2210 digital thermometer; Graham-Field) with a range of 32.0–43.9°C (accuracy ±0.1°C, 34–42°C).
Blood collection and sample handling
Sample collection and processing for TEG analysis followed the published PROVETS guidelines. 7 Blood was obtained via direct venipuncture from a jugular or peripheral vein with a 3-mL syringe attached to a 22-gauge needle. For patients with indwelling catheters capable of blood sampling, the following technique was used: the port was disconnected, and a 5 mL pre-sample was collected into two 3-mL syringes containing 0.5 mL of 0.9% NaCl with 1 IU/mL of heparin. The study sample was then collected from the port using a 3-mL syringe. The pre-sample volume was returned to the patient, and the catheter port was flushed with 2 mL of 0.9% NaCl. With either method, 1.8 mL of blood were collected and immediately transferred into a 1.8-mL tube containing 3.2% sodium citrate. The tube was inverted 8 times and allowed to rest for 30 min at room temperature (21 ± 2°C).
Thromboelastography
Analysis with TEG was performed (2-channel 5000 series TEG analyzer; Haemonetics) to acquire the parameters: R time, K time, alpha angle, MA, LY30, and LY60. Tissue factor was used as an activator at a dilution of 1:3,600 with HEPES (N-2-hydroxyethylpiperazone-N-2-ethanesulfonic acid) buffer solution (Thermo Fisher). The TEG was performed 30 min after sample collection. The channels of the analyzer were set to the corresponding temperature; samples were randomized to have channel 1 or channel 2 set to body temperature; the other channel was set to 37°C. Ten microliters of tissue factor solution and 20 µL of 0.2 M/L calcium chloride were added to each TEG cup. After rest time, the tube of blood was inverted 5 times, 330 µL of blood were pipetted into each cup, and the TEG test was initiated. The test was allowed to run until completion of all variables including R, K, alpha angle, MA, LY30, and LY60. All tests were performed by 1 of the 2 authors trained in the TEG protocol.
Statistical analysis
The Shapiro–Wilk test was used to assess continuous variables for normality. Descriptive statistics consisted of the median and range for all continuous variables given that the majority of the variables were not normally distributed. The count and percentage (%) were used to report frequency data. Variabilities and correlations between TEG parameters were assessed using the Lin concordance correlation coefficient and Bland–Altman (BA) plots. Lin concordance correlation coefficients were measured to evaluate both precision and accuracy to determine deviation of the measured data from the line of perfect concordance. Method comparison was evaluated using BA analysis to calculate both the bias with 95% CIs and the limits of agreement (bias ± 1.96SD) between TEG variables performed concurrently at the patient’s body temperature and at 37°C as referent. Agreement was classified as good when the 95% limits of agreement were narrow, and the bias was small. Pearson correlation coefficients were interpreted as follows: 0.9–1 = very high correlation, 0.70–0.89 = high correlation, 0.50–0.69 = moderate correlation, 0.30–0.49 = low correlation, and <0.30 = little, if any, correlation.
16
For all comparisons,
Results
At standard assay temperature of 37°C and using institutionally derived RIs,
2
12 patients had prolonged R time with none having shortened R; 6 had an increased K time with none having shortened K; 7 had a decreased alpha angle and none had an increased alpha angle; 2 had a low MA with 20 having increased MA. Comparisons between analyses at the 2 temperatures showed very high correlation for R and MA values, high correlation for alpha and K values, and moderate correlation for LY30 and LY60, with significance documented for all variables (Table 2; Figs. 1, 2; Suppl. Figs. 1, 2). Agreement was classified as acceptable for all variables. A sub-analysis of the 5 hypothermic patients (body temperature ≤37°C) showed very high correlation for MA and alpha angle, high correlation for R, and moderate correlation for K and LY60 (%), with significance documented for all variables except K (
Comparison of thromboelastography (TEG) variables analyzed at 37°C and patient body temperature using Lin concordance correlation coefficients and Bland–Altman (BA) plots.
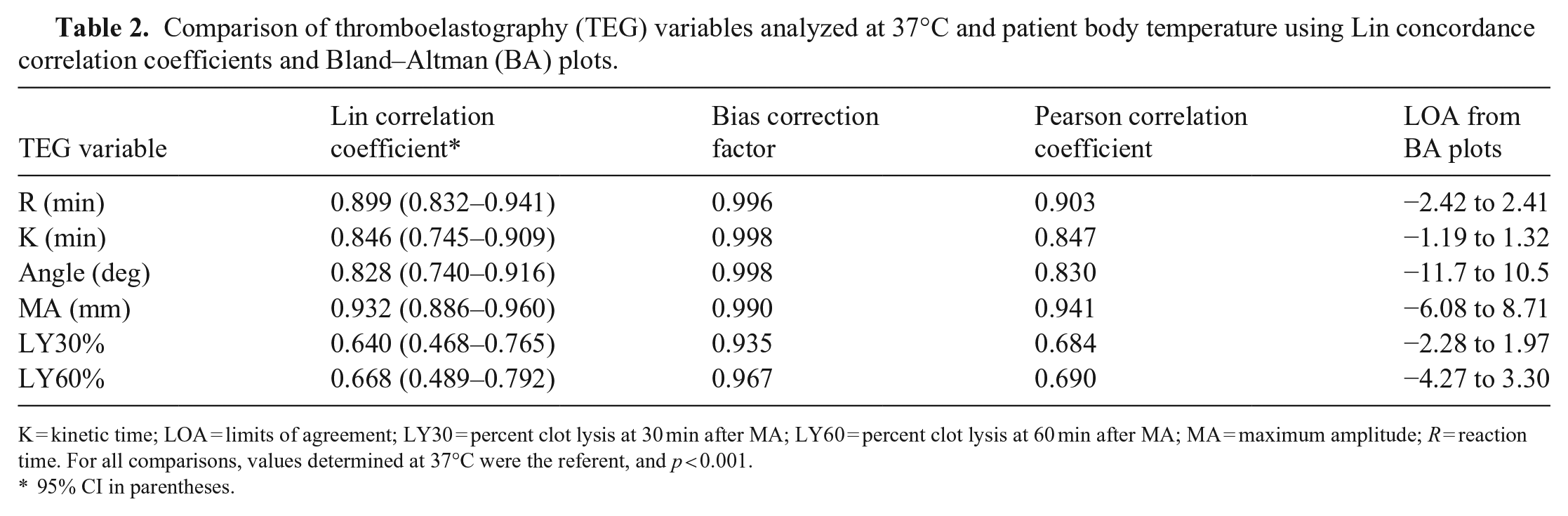
K = kinetic time; LOA = limits of agreement; LY30 = percent clot lysis at 30 min after MA; LY60 = percent clot lysis at 60 min after MA; MA = maximum amplitude;
95% CI in parentheses.
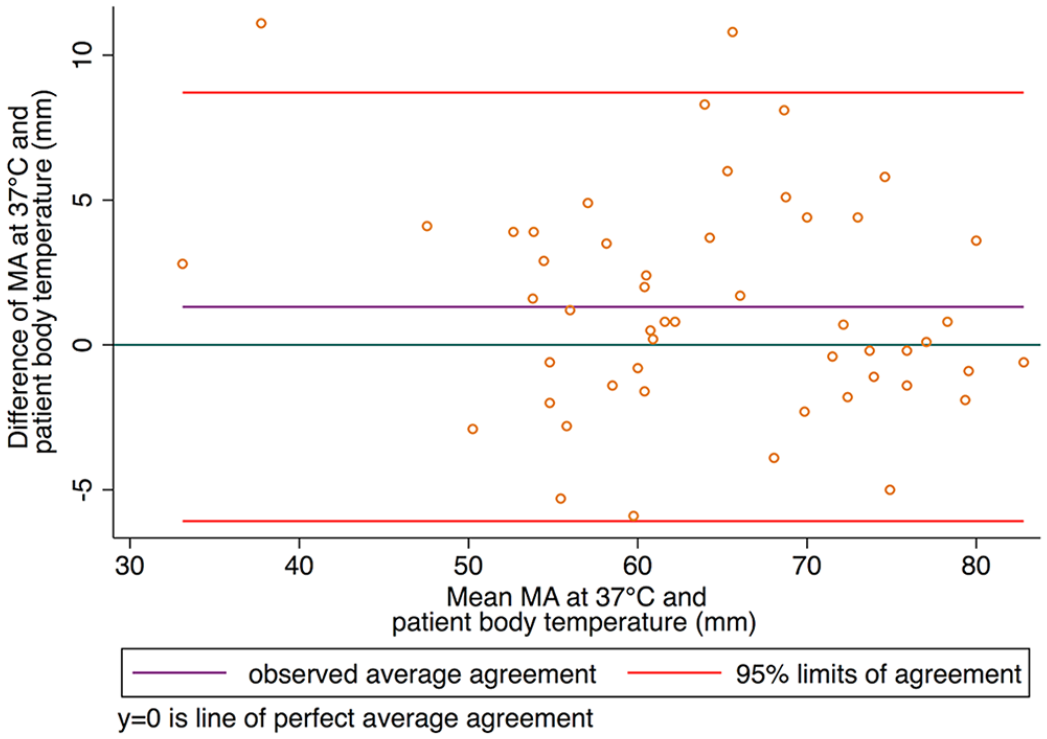
Bland–Altman plot comparing thromboelastography maximum amplitude (MA) analyzed at 37°C and patient body temperature.
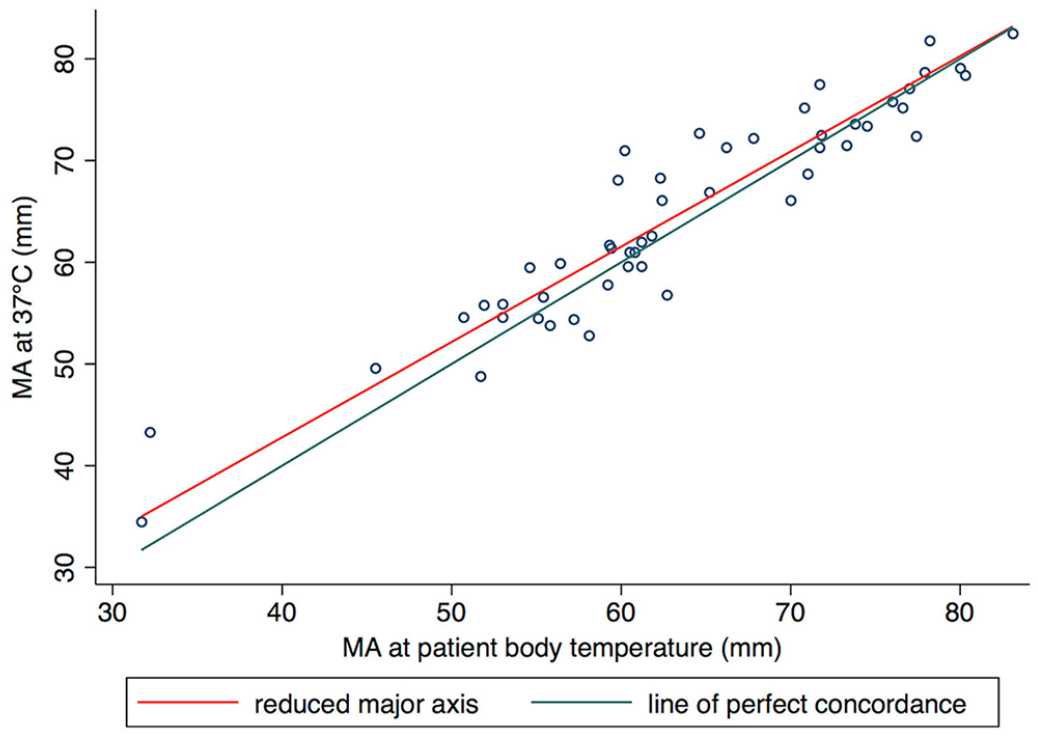
Pearson correlation plot comparing thrombo-elastography maximum amplitude (MA) analyzed at 37°C and patient body temperature.
Discussion
For all clot formation and kinetic variables (R, K, alpha, and MA) studied, we found high to very high correlation between the results obtained from performing the TEG at either 37°C or patient body temperature. Current recommendations set forth in the PROVETS guidelines suggest performing TEG at 37°C but indicate that evidence is lacking to support this. 7 Our findings provide evidence to support the current recommendation.
LY30 and LY60 showed only moderate correlation when TEGs were performed concurrently at the patient’s body temperature and at 37°C. In veterinary patients, TEG has been markedly understudied with respect to diagnostic utility for hyper- and hypofibrinolysis, and cutoff values; RIs for LY30 and LY60 are also poorly established, making this result difficult to interpret. In human trauma patients, a LY30 of 7.5% has been used traditionally as a cutoff for initiating antifibrinolytic therapy, but recent studies have advocated a lower cutoff value of 3% because patients with >3% lysis at LY30 have been demonstrated to have an increased risk of requiring massive transfusion. 3
Little is published about the use of LY60 and cutoffs. In both human and veterinary medicine, the diagnostic sensitivity of TEG values LY30 and LY60 is questioned. A 2015 study investigated hyperfibrinolysis in dogs that had been presented to emergency facilities for spontaneous hemoperitoneum. 5 Only minimal fibrinolysis was observed in any patients, and no difference was found between the LY30 or LY60 of patients with spontaneous hemoperitoneum and healthy controls. However, after the addition of 50 U/mL of tissue plasminogen activator to samples, marked fibrinolysis was noted and a significant difference was found in both LY30 and LY60 between patients with spontaneous hemoperitoneum and healthy controls. This finding suggests that TEG on native, citrated, or other activated samples without the addition of a fibrinolytic agent may have poor sensitivity for detecting hyperfibrinolysis in the absence of an extreme state of hyperfibrinolysis.
Only 5 patients in our study were classified as hypothermic, defined as body temperature ≤37°C, and no patients had a body temperature <35°C. The range of temperatures included in our study represents critically ill and hospitalized patients whose clinicians sought enrollment in the study to use the diagnostic information from a TEG. We suspect that the majority of markedly hypothermic patients are actively warmed before a TEG is requested or performed. Therapeutic hypothermia was not used as a treatment for any patients included in our study. Only a few patients had hyperthermic temperatures as well, with 4 patients having temperatures >39°C, but no patients having temperatures ≥40°C.
Various studies in human medicine have investigated the effect of analysis temperature on TEG results in different populations of patients. In a study of patients undergoing therapeutic hypothermia post-cardiac arrest, the authors found an influence of temperature adjustment on clot formation variables R and K, but wide limits of agreement made the influence of temperature-adjustment clinically irrelevant. 4 In a group of anesthetized patients undergoing intracranial surgery, patients were cooled to 36, 34, and 32°C, and samples collected and TEG performed at 37°C and at the corresponding body temperatures. 11 The results of the TEGs performed at 37°C were similar among patients in all groups; however, the same samples showed statistically significant differences in the temperature-corrected analysis, with the samples from patients cooled to 32°C having a prolonged R and K and a reduced alpha angle when compared to the other groups. However, even though these values were altered in the 32°C group, they remained within the normal range. Of interest, the TEG MA value was not different between the temperature-corrected analyses or standard 37°C analyses, which is consistent with results of other investigations, demonstrating the effects of hypothermia on clot initiation and formation but not on overall clot strength. 12 Given that all values remained within the normal range, that study suggests that temperature-corrected TEG analysis is not clinically necessary in patients undergoing controlled periods of hypothermia <4 h. 12
A population in which the effect of temperature-adjustment may be clinically relevant is neonatal humans. In neonatal human patients undergoing therapeutic hypothermia for encephalopathy, TEG results did differ significantly between the assays performed at 33.5°C versus 37°C, yielding more hypocoagulable results at the lower temperature. 6 The temperature-adjusted results also served as better predictors of clinical bleeding in these patients, suggesting that there is clinical utility of a body temperature–specific TEG analysis or that temperature-specific RIs need to be created for neonatal patients with induced hypothermia. Analysis temperature of TEG in any population of dogs has not been reported previously. Although our study indicates that standard 37°C analysis is adequate for patients with a normal to mildly abnormal body temperature, conclusions cannot be drawn regarding specific subpopulations of veterinary patients such as hypothermic neonates. However, in our hospital, TEG is rarely used for these patients. Further studies investigating temperature-related viscoelastic test alterations in various veterinary populations, and correlation of those abnormalities to clinical bleeding, are indicated.
An in vitro investigation of simulated abnormal body temperatures on blood samples taken from normal dogs but incubated at temperatures of 27–42°C found differences in clot formation kinetics, with significantly longer K and lower alpha values at the samples incubated at 27°C and 30°C compared to those at normal or elevated temperatures. 14 However, these were not in vivo temperatures, rather the resting temperatures were different between the concurrent samples analyzed. Therefore, conclusions regarding the appropriate temperature at which to perform the TEG, 37°C or body temperature, cannot be drawn from that study. The effect of rest temperature on canine TEG was examined in another study, which found significant but small and clinically irrelevant differences in MA and alpha values for samples rested at 37°C or prewarmed for 5 min at 37°C compared to those rested at room temperature. 13
Our findings are based on analysis of TEG using tissue factor as an activator, which provides an assessment of the tissue factor pathway of coagulation. It is likely that using a different activator, or no activator, may have altered the TEG values in our study; however, given that the same activation method would be used to compare the temperatures, we do not suspect that a different activator would alter our conclusions. The limited range in body temperatures in our study prevents drawing conclusions regarding those patients who are markedly hypothermic or hyperthermic. In populations of adult humans, temperature-corrected TEG in hypothermic states has been shown to differ with statistical significance but questionable clinical relevance compared with standard temperature TEG, but may be useful in hypothermic neonatal patients. Evidence is lacking regarding the influence of hyperthermia on TEG variables. Only 4 patients in our study had body temperatures >39°C, and no patients had a body temperature >40°C. Application of our findings to patients with more extreme body temperatures, both high and low, is limited, and future studies focusing on these patient populations are warranted. Based on our results, it is reasonable for clinicians to rely on standard temperature TEG results for patients with normal or mildly abnormal body temperatures.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211042922 – Supplemental material for The effect of analytical temperature on thromboelastography tracings in dogs
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211042922 for The effect of analytical temperature on thromboelastography tracings in dogs by Marie T. Keith, Nolan V. Chalifoux and Yekaterina Buriko in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Kenneth Drobatz (University of Pennsylvania) for his contribution to the generation of this study. We also thank the Extracorporeal Therapy Service at the Veterinary Hospital of the University of Pennsylvania for the loan of their space to house instruments and supplies.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
