Abstract
Objectives
While thromboelastography (TEG) has helped define a complex state of hemostasis in dogs and humans with hepatobiliary disease, it has not been explored in cats with cholestatic liver disease (CLD). The objective of this study was to describe TEG parameters in cats with CLD and to compare these parameters with conventional plasma-based coagulation tests, white blood cell (WBC) count and biochemical indicators of liver disease grade and severity.
Methods
Eighteen cats with CLD, defined by a serum bilirubin ⩾3 mg/dl and a greater than two-fold increase in serum alanine aminotransferase (ALT) and/or alkaline phosphatase (ALP) activity, were prospectively enrolled. All cats received vitamin K1 subcutaneously for 24–36 h prior to acquisition of blood for kaolin-activated TEG analysis, prothrombin time (PT) and activated partial thromboplastin time (aPTT). Patient total solids, packed cell volume, platelet count, WBC count, and serum liver enzymes and bilirubin were extracted from the medical record and correlated with coagulation test results.
Results
TEG global clot strength (TEG G) values defined 9/18 (50%), 5/18 (28%) and 4/18 (22%) cats as hypercoagulable, normocoagulable or hypocoagulable, respectively. TEG G was significantly negatively correlated with PT, aPTT and serum ALP activity and positively correlated with total solids. Five cats (5/18, 28%) were hyperfibrinolytic with clot lysis at 60 mins (LY 60) >15.3%. LY 60 was significantly positively correlated with PT.
Conclusions and relevance
By TEG analysis, cholestatic cats replete with vitamin K1 display a variety of coagulation profiles. Indications of synthetic failure (prolonged PT and aPTT) were associated with hypocoagulable and hyperfibrinolytic TEG parameters. High disease activity (serum ALP) was associated with a hypocoagulable state.
Introduction
A complex state of hemostasis accompanies hepatobiliary disease in dogs, cats and humans.1–3 Hepatobiliary disease leads to a rebalanced hemostatic system with concomitant coagulation abnormalities in primary hemostasis (platelet number and function, endothelial integrity), secondary hemostasis (synthesis and activation of pro- and anticoagulants) and fibrinolysis that can lead to both bleeding and thrombotic tendencies.2–5 Less is known about the hemostatic balance in cats with cholestatic liver disease (CLD) such as cholangitis and hepatic lipidosis. Previous studies to describe coagulation in these cats have primarily assessed conventional serum-based coagulation tests including prothrombin time (PT), activated partial thromboplastin time (aPTT) and platelet count.6,7 These studies show that platelet counts are typically normal, but many cats (40–82%) have either an abnormal PT or aPTT.6,8–10 In the majority of these cats, PT prolongation is accompanied by low factor VII activity and reflects a vitamin K1-responsive coagulopathy.6,7 A limited number of studies have looked beyond conventional coagulation testing and have shown that additional hemostatic testing abnormalities might be associated with both bleeding (decreased fibrinogen or factor XIII activity) or thrombotic tendencies (increased fibrinogen or D-dimers; decreased protein C or antithrombin activity) in cats with various types of hepatic disease.6,8,9
Both thrombotic and hemorrhagic complications have been reported in cats with CLD.4,6,8,11 Cats with CLD can develop macrothrombosis, such as in the portal vasculature. 4 Bleeding diathesis also occurs in these cats following provocative procedures.8,11–13 Despite preoperative management with blood products and/or parenteral vitamin K1, normal platelet counts and relatively mild aberrations in PT and aPTT, 57–68% of cats with extrahepatic biliary duct obstruction that undergo surgery require a red blood cell transfusion.12,13 Likewise, in a recent study of ultrasound-guided percutaneous hepatic biopsy in cats without major derangements in PT, aPTT and platelet count, major bleeding (packed cell volume [PCV] drop of 6% or greater) was seen in 57% of cats, while 16.7% of the cats had bleeding complications that necessitated therapeutic intervention. 11 Many cats with CLD have low factor XIII activity, a condition that might accelerate fibrinolysis and predispose cats to delayed bleeding after provocative procedures. 9 Conventional plasma-based coagulation testing would fail to detect factor XIII deficiency. Additional studies that describe and offer complete assessment of hemostasis in cats with CLD are needed to optimize diagnostic decisions (hepatic biopsy, surgical intervention) and therapeutic strategies (administration of blood products, antifibrinolytic agents or anticoagulants).
Thromboelastography (TEG) is a whole blood assay that can evaluate clot formation as a dynamic process measuring clot strength as well as the kinetics of clot formation and lysis. In humans, TEG has documented hypercoagulability in biliary disease, identified hypocoagulability in cirrhosis and helped to guide blood product therapy in cirrhotic patients undergoing liver transplantation.14–19 In dogs with liver disease, TEG parameters consistent with hypercoagulable, hypocoagulable and hyperfibrinolytic states have been demonstrated.20–22 TEG analysis has been performed in healthy cats,23–25 cats with obesity, 26 acute trauma,27,28 hyperthyroidism 29 and infection 30 and has been used to monitor feline anticoagulant therapy.24,31 However, no studies have been conducted in cats with CLD.
The aim of the current study is to describe TEG analysis in cats with CLD. We hypothesize that these cats will have a variety of hemostatic abnormalities reflecting the complex coagulopathy that is known to accompany liver disease in other species.
Materials and methods
Eighteen client-owned cats presenting to The Henry and Lois Foster Hospital for Small Animals at Cummings School of Veterinary Medicine at Tufts University were prospectively enrolled from February 2013 to June 2014. The study was reviewed and approved by the Clinical Studies Review Committee and Institutional Animal Care and Use Committee, and informed consent was obtained from owners.
Complete blood count (CBC) and serum biochemistry panels were either performed on the day of admission to the hospital or by the referring veterinarian within the 24 h prior to referral. Inclusion criteria included a serum bilirubin ⩾3 mg/dl and an increase in serum liver enzymes (alanine aminotransferase [ALT] and/or alkaline phosphatase [ALP]) >2 times the upper limit of normal. Cats were excluded if they had a PCV <28%, required sedation for venipuncture or were receiving drugs that could alter hemostasis (corticosteroids, non-steroidal anti-inflammatory medications, free fatty acids, or anticoagulant medications). In addition, cats with disease conditions known to be associated with coagulation abnormalities (previously diagnosed hypertrophic cardiomyopathy, protein-losing enteropathy or nephropathy, hyperadrenocorticism or non-hepatic neoplasia) were excluded after expert review (CRLW) of the medical record. Patient medical records were reviewed for signalment, weight, sex, clinical signs, platelet count, PT, aPTT, PCV, total solids (TS), white blood cell (WBC) count, serum liver enzymes (ALT, ALP), serum bilirubin, ultrasound findings and final histopathologic diagnosis, if available.
Hemostatic analysis
Kaolin-activated TEG (TEG 500 Thromboelastograph; Haemonetics Corp), PT and aPTT (ACL Elite Analyzer; Beckman Coulter) were performed in the Clinical Coagulation Laboratory at The Henry and Lois Foster Hospital for Small Animals. Blood was collected from cats after 24–36 h of parenteral vitamin K1 therapy in order to avoid the detection of hypocoagulability associated with vitamin K1 deficiency.6,7,9 Each patient received 0.5–1 mg/kg of vitamin K1 subcutaneously once or twice daily with a minimum of two doses, except for one patient that only received one dose. A single operator performed all TEGs using an institutional standardized protocol based on international guidelines.32–34 The following TEG variables were generated: R (reaction time), K (clot formation time), angle, maximum amplitude (MA) and clot lysis at 60 mins (LY 60). G value, a mathematical manipulation of MA, was calculated and used to define the state of coagulation. Based on non-parametric estimations of reference intervals, cats were labeled as hypocoagulable (G value <4.24 Kd/s), normocoagulable (G value 4.24–10.32 Kd/s), or hypercoagulable (G value >10.32 Kd/s). Hyperfibrinolysis was defined as an LY 60 >15.2%.
The cause of CLD was determined by expert analysis of the medical record (CRLW) incorporating clinicopathologic data, abdominal ultrasound performed by a board-certified radiologist or radiology resident, and hepatic cytology/histopathology and bacterial culture of the liver and/or bile. Cats were stratified by the major pathologic process that was occurring.
Statistical analysis
The reference interval for TEG was generated by analysis carried out on 31 normal, healthy cats. 33 Non-parametric estimations of reference intervals were determined by calculating the 2.5th and 97.5th percentiles using IBM SPSS Statistics software.
Data were tested for normality by histogram analysis and determination of skew and kurtosis. Subgroup analysis with one-way analysis of variance (with log transformation of data if non-parametric) was performed to compare cats with normal, hypercoagulable and hypocoagulable G values in terms of their coagulation parameters (PCV, platelet count, PT and aPTT), indices of liver disease severity and function (serum liver enzymes and serum bilirubin) and a marker of inflammation (WBC count). Correlation between TEG variables and WBC and biochemical indicators of hepatic disease grade and function, as well as conventional coagulation tests, were performed with Pearson’s correlation coefficient. Selected clinical pathology parameters and TEG variables were compared in hyperfibrinolytic and non-hyperfibrinolytic cats using the Mann–Whitney test. For all statistical analyses, a P value <0.05 was considered significant. Bonferroni correction was applied for multiple comparisons. Statistical analyses were performed using SAS 9.4 software (SAS Institute Inc, Cary, NC) or with online software (https://www.socscistatistics.com/).
Results
Eighteen cats fulfilled the inclusion criteria. Population characteristics are described in Table 1. Clinical signs included hyporexia (17/18, 94%), weight loss (4/18, 22%) and chronic vomiting (1/18, 5.5%).
Population parameters of cats with cholestatic liver disease (n = 18)

A CBC and serum biochemistry panel were available for all cats. Selected parameters are summarized in Table 2. All 18 cats had increased serum bilirubin, while 17/18 (94.4%) had increased serum ALT and 15/18 (83%) had increased serum ALP. The PCV was decreased in 2/18 (11%) cats and WBC count was increased in 4/18 (22%) cats.
Selected clinical pathology parameters in cats with cholestatic liver disease (n = 18)

ALT = alanine aminotransferase; ALP = alkaline phosphatase; PCV = packed cell volume; TS = total solids; WBC = white blood cell count
An abdominal ultrasound was performed in all cats. Hepatobiliary abnormalities included hepatomegaly with hyperechoic parenchyma (10/18, 55%), gall bladder wall thickening (8/18, 44%), dilated common bile duct (4/18, 22%), gall bladder sludge (3/18, 16.6%), extrahepatic bile duct obstruction (EHBDO; 3/18, 16.6%) and cholelithiasis (1/18, 5.5%). Other abnormalities included trace to mild peritoneal effusion (7/18, 39%) and enlarged hypoechoic pancreas surrounded by hyperechoic fat (6/18, 33.3%).
Conventional plasma-based coagulation tests (PT, aPTT and platelet count) were conducted in all cats and the results are summarized in Table 3. The results showed that 7/18 (39%) cats had abnormalities in these tests. The PT was prolonged in 4/18 (22.2%), of which two cats had severe prolongation (>1.5-fold). The aPTT was prolonged in 2/18 (11.1%) cats. In one of these cats, the aPTT (as well as the PT) was prolonged two-fold. The platelet count was increased in one cat and decreased in the other.
Coagulation testing in cats with cholestatic liver disease (n = 18)
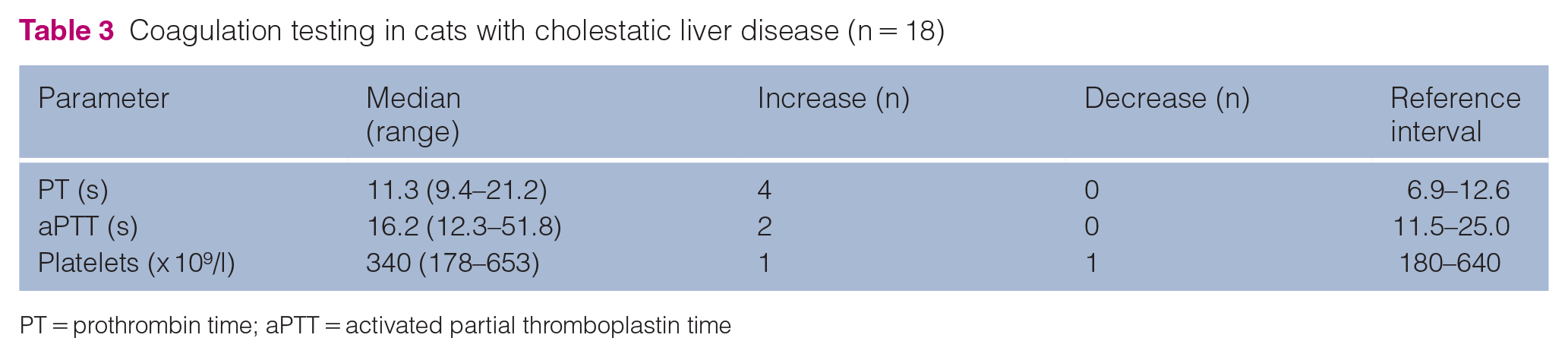
PT = prothrombin time; aPTT = activated partial thromboplastin time
TEG analysis was performed in all cats (Figure 1). Median values for all the TEG parameters were within the normal range, although 13/18 (73%) cats had abnormalities in one or more parameters. The association between TEG variables and conventional coagulation tests was explored. TEG K value showed strong positive correlation with PT (r = 0.75, P <0.001) and moderate positive correlation with platelet count (r = 0.59, P = 0.04). The R value had moderate positive correlation with PCV (r = 0.55, P = 0.018). TEG G and MA showed strong negative correlation with PT (−0.629, P = 0.007; −0.79, P <0.001, respectively) and TEG G showed a moderate negative correlation with aPTT (−0.473, P = 0.047) (Table 4). TEG angle showed strong negative correlation with PT (r = −0.68, P = 0.001), and moderate positive correlation with platelet count (r = 0.52, P = 0.03).
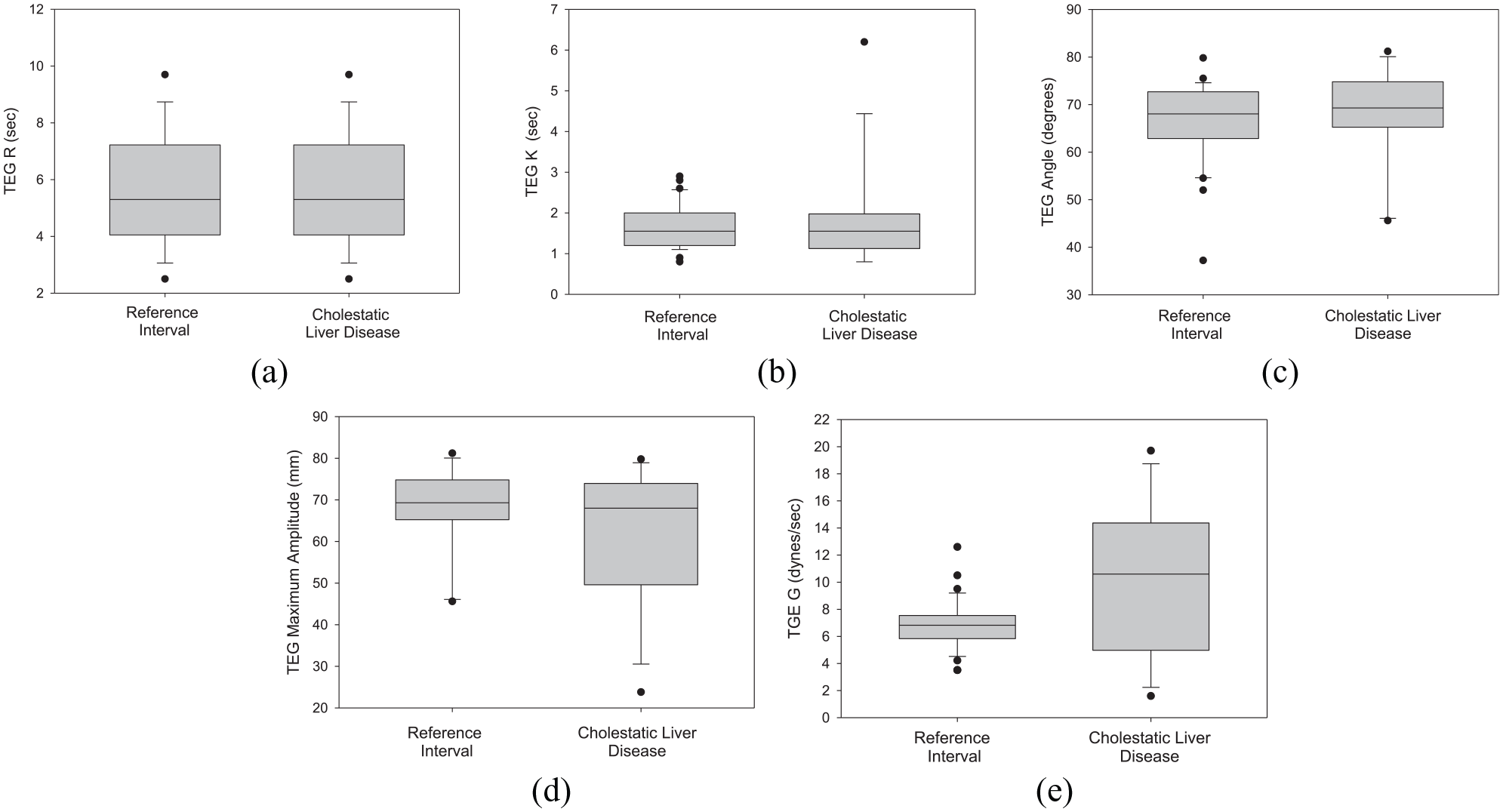
Thromboelastography (TEG) parameters in cats with cholestatic liver disease. Box and whisker plots depicting TEG results for 18 cats with cholestatic liver disease and normal cats (reference interval). Results are displayed according to the TEG variable: (a) R = reaction time; (b) K = clot formation time; (c) angle; (d) maximum amplitude; and (e) G = global clot strength. The box plots demonstrate the median (line) and interquartile range (box) and the whiskers span all data points between the 10th and 90th percentiles. Outliers are indicated by a circle
Correlation between thromboelastography global clot strength value and plasma-based coagulation tests and selected biochemical parameters

PT = prothrombin time; aPTT = activated partial thromboplastin time; PCV = packed cell volume (on presentation); TS= total solids; WBC = white blood cell count; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase
Cats were stratified by TEG G value into three categories: hypercoagulable, hypocoagulable and normocoagulable. Nine of 18 (50%), 5/18 (27.7%) and 4/18 (22.2%) cats were hypercoagulable, normocoagulable and hypocoagulable, respectively. Of the four hypocoagulable cats, three had other indices of hypocoagulability including PT (3/4) or aPTT (1/4) prolongations. Three out of four hypocoagulable cats had increased LY 60 and were considered hyperfibrinolytic. In the nine hypercoagulable cats, all plasma-based coagulation parameters were normal except for one cat with increased platelet count and one cat with increased LY 60. All of the normocoagulable cats had normal coagulation parameters except for one cat with a prolonged PT and LY 60.
Coagulation state (TEG G) was compared with measures of hepatic disease severity (and function ALT, ALP and bilirubin) or systemic inflammation (WBC count). TEG G value had significant, moderate negative correlation with serum ALP (r = −0.443, P = 0.006) and a had strong positive correlation with TS (r = 0.628, P = 0.005) (Table 4).
Five out of eighteen (28%) cats were identified as hyperfibrinolytic with LY 60 >15.28%, which was consistent with the LY 30 results. Selected clinical pathology parameters and TEG variables in hyperfibrinolytic and non-hyperfibrinolytic cats are described in Table 5. The PT and aPTT were significantly prolonged in hyperfibrinolytic cats. LY 60 had strong positive correlation with PT (r = 0.89, P <0.001), but not TEG G, aPTT, PCV, platelet count, TS, an index of systemic inflammation (WBC count) or hepatic disease activity (ALT) or function (bilirubin).
Selected clinical pathology parameters and thromboelastography variables in hyperfibrinolytic and non-hyperfibrinolytic cats
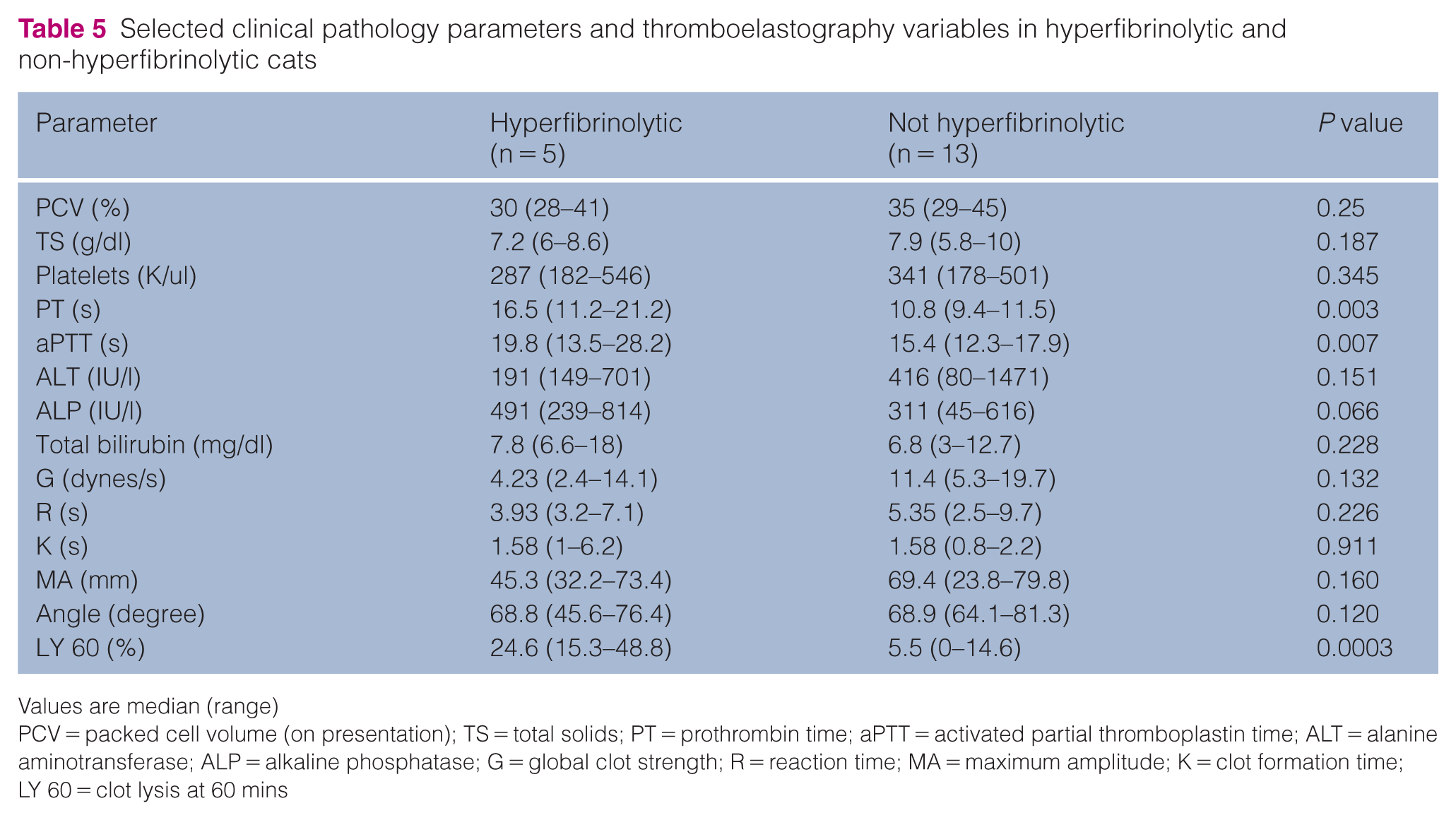
Values are median (range)
PCV = packed cell volume (on presentation); TS = total solids; PT = prothrombin time; aPTT = activated partial thromboplastin time; ALT = alanine aminotransferase; ALP = alkaline phosphatase; G = global clot strength; R = reaction time; MA = maximum amplitude; K = clot formation time; LY 60 = clot lysis at 60 mins
Twelve cats had hepatic cytology (n = 4) and/or histopathology with either ultrasound-guided percutaneous needle biopsy (4/12), surgical biopsy (4/12) or biopsy at necropsy (3/12). The primary diagnoses based on tissue sampling were inflammatory (cholangitis ± pancreatitis, 5/12, 42%), lipidosis (4/12, 33.3%) and neoplasia (3/12, 25%). All four cats with lipidosis had no other pathologic changes on tissue sampling. Two cats with neoplasia (cholangiocarcinoma and pancreatic neuroendocrine tumor) had concurrent chronic cholangitis and EHBDO. Of the five cats with cholangitis, one cat had an EHBDO with an obstructing choledocholith. Eight out of 18 (44.4%) cats had a culture of the liver and/or bile submitted. Two of eight (25%) were positive for Enterococcus species (one from the liver and one from bile) and the remaining cats were negative. One cat that cultured positive had chronic cholangitis and cholelithiasis, and the other had cholangiocarcinoma and EHBDO.
The coagulation state, as determined by TEG G in cats with various diagnoses, was variable. Three out of five cats with inflammatory disease were hypercoagulable and 2/5 cats were normocoagulable. Cats with neoplasia were either hypercoagulable (2/3) or normocoagulable (1/3). Two out of four cats with hepatic lipidosis were hypocoagulable, one was normocoagulable and one hypercoagulable. Only 4/5 cats with hyperfibrinolysis had a cytologic or histopathologic diagnosis, 2/4 cats had inflammatory disease and 2/4 cats had lipidosis. All three cats with EHBDO had TEG G values compatible with a hypercoagulable state.
Discussion
The results of this prospective study revealed that cats with CLD have variable changes in conventional plasma-based and TEG coagulation parameters that indicate tendencies for both hypocoagulable and hypercoagulable states. Coagulation status, as determined by TEG G value, showed that 50% of cats were classified as hypercoagulable, 28% as normocoagulable and 22% as hypocoagulable. In addition, the TEG LY 60 defined 28% of cats as hyperfibrinolytic. When comparing TEG and conventional coagulation testing (PT, aPTT, platelet count), TEG detected coagulation abnormalities more frequently (13/18 cats, 72%) than conventional tests (3/18 17%). This discrepancy is primarily related to the ability of TEG analysis to demonstrate hypercoagulable tendencies and to further evaluate for hyperfibrinolysis.
The finding of variable coagulation states in cats with CLD is consistent with the concept of rebalanced hemostasis in liver disease as described in human patients and dogs with hepatobiliary disease. 5 Owing to the liver’s role in the synthesis, activation, degradation and clearance of most procoagulants, anticoagulants and regulators of fibrinolysis, there is a precarious hemostatic balance in these patients that can be tipped toward bleeding or thrombosis by a variety of underlying factors.1,5,35 In people, these factors include the nature of the hepatic disease as well as the presence of concurrent infections, inflammation, renal disease and portal hypertension. 35 In the current study, cats with inflammatory and neoplastic disease tended to be hypercoagulable (5/8) or normocoagulable (3/8), while 50% of the cats with hepatic lipidosis were hypocoagulable (2/4). However, the circumstances that affect hemostatic balance in cats with CLD will need to be defined in larger studies.
TEG G was negatively correlated with PT, aPTT and serum ALP and positively correlated with TS, but there was no association of the G value with platelet count or indicators of systemic inflammation (WBC count) or liver function (serum bilirubin). The negative association of G with ALP may suggest that higher disease grade activity is reflected by a more hypocoagulable state (prolonged PT and low TEG G value). A separate study has previously demonstrated that cats with high serum ALP were more likely to have abnormal coagulation values. 6 Since the goal of the study was to evaluate TEGs in cats with CLD and not acute hepatic failure, additional indices of liver function (blood urea nitrogen, glucose, cholesterol and albumin) were not evaluated. The negative association of G with PT and aPTT could reflect the association with hypocoagulability, the presence of disseminated intravascular coagulation (DIC), 36 the persistence of vitamin K deficiency or the presence of severe hepatic disease. The lack of thrombocytopenia or red blood cell (RBC) fragmentation in these cats however makes DIC less likely. The association of TS with TEG G is harder to reconcile, but a higher TS might indicate the presence of more acute phase pro-inflammatory proteins in circulation or, alternatively, the presence of dehydration and/or hypovolemia, both of which could affect coagulation balance.
The current study identified a population of cats with CLD that were hyperfibrinolytic. Hyperfibrinolysis has been previously reported in cats with acute trauma, hemorrhagic effusion in the abdomen and thorax and with snake envenomation,37,38 but this is the first report of this phenomenon in cats with liver disease. In the current study, hyperfibrinolysis (increased LY 60) did not correlate with disease grade (liver enzymes), an indicator of systemic inflammation (WBC count) or serum bilirubin. The LY 60, however, was strongly positively associated with PT and aPTT, suggesting that it may be associated with advanced disease and loss of synthetic function, DIC or persistent vitamin K deficiency.
The presence of hyperfibrinolysis could predispose cats with CLD to delayed bleeding after provocative procedures or be associated with bleeding from mucosal surfaces. 2 The current study was not designed to look at bleeding tendencies and indeed only 50% of all cats in the study underwent provocative procedures. The five hyperfibrinolytic cats in this study did have a greater overall prolongation in PT and aPTT compared with cats without hyperfibrinolysis. In addition, 3/5 of the hyperfibrinolytic cats were considered hypocoagulable on evaluation of TEG G value, while only 1/11 of the cats without hyperfibrinolysis had a TEG consistent with hypocoagulability. Two of the five cats that were hyperfibrinolytic had normal PT, aPTT and platelet counts on conventional coagulation testing and would not have been considered a bleeding risk. Future studies are needed, however, to determine if bleeding tendencies actually occur in cats with hyperfibrinolysis.
Hyperfibrinolysis can be primary or secondary.3,5 Primary hyperfibrinolysis is associated with quantitative or qualitative abnormalities of the proteins involved in the regulation of the fibrinolytic pathways. Secondary hyperfibrinolysis is due to altered fibrin susceptibility to lysis caused by systemic activation of coagulation, such as is seen with systemic inflammatory response syndrome (SIRS), sepsis or DIC. None of the cats in the current study met the full diagnostic criteria for DIC (prolongation of PT or aPTT with thrombocytopenia and RBC fragmentation), but it is possible some of the cats could have met the criteria for SIRS or sepsis. A previous study demonstrated that many cats with liver disease (up to 70%) have low factor XIII levels. 9 The latter is involved in stabilization of the fibrin clot; a deficiency could lead to premature clot lysis and account for the hyperfibrinolysis observed in this study. Defining the cause and significance of hyperfibrinolysis in cats with CLD will require larger prospective studies that investigate bleeding tendencies and additional hemostatic parameters such as coagulation factors (factor XIII, fibrinogen, antithrombin), regulators and byproducts of fibrinolysis (D-dimers, plasminogen, tissue plasminogen activator, plasminogen activator inhibitor).
It should be noted that hyperfibrinolysis has been noted in healthy control cats in some studies.25,29,39 It is unclear if this represents true hyperfibrinolysis or occurs secondary to rapid clot retraction and separation from the test cup in this species. Cats are known to have hyperactive platelets, which might accelerate clot retraction speed. The inherent tendency for hyperactive platelets in this species may therefore contribute to the hyperfibrinolytic-appearing TEGs in healthy cats and potentially in some cats in our study. 40
The study had several limitations. This was a pilot study to describe TEG parameters in cats with CLD, so the sample size was small. The study thus may have been underpowered to find associations between changes in TEG parameters and markers of disease severity or conventional coagulation parameters. There was also some overlap in the etiologic stratification based on tissue sampling, such that the circumstances that affect hemostatic balance in cats with various causes of CLD will need to be defined in larger studies. Although all cats received vitamin K1 prior to coagulation testing, not all cats received the same dose of vitamin K1 and it was impossible to determine that the cats were truly replete in vitamin K1 prior to TEG analysis. A definitive etiologic diagnosis was not an inclusion criterion for the study, so the sample size in each disease category was too small to attempt statistical analysis. Not all cats underwent provocative procedures, and in those that did, monitoring for bleeding was not carried out in a systematic way, thus we could not examine the relationship between TEG parameters and bleeding. This analysis, however, would be especially useful since previous studies have suggested that cats with CLD (particularly hepatic lipidosis) are at an increased risk for bleeding from provocative procedures.7,11,38 Body condition scores were not consistently recorded in the medical record and thus the role of obesity, which has been shown to contribute to hypercoagulability in cats, could not be determined. 26 The current study was carried out prior to a recent publication which recognized that cats with hyperthyroidism can be hypercoagulable and thus knowledge of thyroid status was not considered in the enrollment criteria. 29 Evaluation of coagulation in cats with CLD was limited to PT, aPTT, platelet count and TEG analysis and future studies should look at a broader range of coagulation parameters. It is possible that hyperfibrinolysis was missed in some cats. Defects of the fibrinolytic system can be more accurately evaluated with tissue plasminogen activator modified TEG analysis, 41 which can be considered in future studies. Finally, infectious etiologies frequently complicate CLD in cats and can lead to the development of endothelial stress and the release of endogenous heparinoids that can affect coagulation status, so future prospective studies should carefully document if infectious disease is present by conducting bile cytology and culture on all cats. 42
Conclusions
TEG analysis in cats with CLD treated with vitamin K1 for 24–36 h is consistent with a variety of coagulation profiles with hypercoagulable, hypocoagulable and hyperfibrinolytic states. Alterations in coagulation status were identified more frequently with TEG analysis than with conventional plasma-based tests. PT correlated best with TEG parameters and thus may be the best traditional coagulation parameter to predict coagulation state in cats with CLD, but further studies are needed. Preliminary evidence from this study suggests that loss of synthetic function (as indicated by prolonged PT and aPTT) and higher disease activity (as indicated by higher serum ALP) may be associated with hypocoagulable and hyperfibrinolytic tendencies. Larger studies that evaluate a greater range of coagulation parameters in cats will be necessary to further delineate the nature of the abnormalities that define hypercoagulable, hypocoagulable and hyperfibrinolytic states. This could have important implications for planning therapeutic interventions and in minimizing morbidity following provocative procedures (feeding tube placement, liver tissue sampling, biliary surgery) in cats with CLD.
Footnotes
Author note
The findings of this study were presented in part at the 2015 American College of Veterinary Internal Medicine Forum, Indianapolis, Indiana.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the Intramural Companion Animal Health Fund at Cummings School of Veterinary Medicine at Tufts University.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
