Abstract
A 52-year-old woman presented with erythema, papules, pain, and itching on the buttocks and trunk that had persisted for 1 week. Twenty-two days before admission, the patient received three intramuscular injections of vitamin K1 in the gluteal muscles. Two weeks after the last intramuscular injection edematous hard erythema developed bilaterally on the gluteal muscles, accompanied by itching, pain, and a burning sensation. Afterward, the erythema and papules gradually spread to the trunk, with notable itching. Dermatological examination revealed a large area of well-defined edematous hard erythema, and blisters around the injection sites on both hips. The skin temperature was high, with mild tenderness. Dense distributions of edematous erythema and papules were observed on the trunk, some of which were fused into pieces. The patient received a diagnosis of a skin reaction induced by intramuscular injections of vitamin K1. Therefore, she was administered glycyrrhizin, calcium, vitamin C, antihistamines, topical polysulfonic mucopolysaccharide cream, and glucocorticoid cream, and the lesions subsided after 2 weeks.
Introduction
Vitamin K1, also known as chlorophyll methylnaphthoquinone, is a fat-soluble vitamin that is widely distributed in natural green plants. It promotes the transformation of prothrombin precursors into prothrombin, which is essential for the synthesis of prothrombins II, VII, IX, and X in the liver. Vitamin K1 deficiency causes hypoprothrombinemia and coagulation disorders; therefore, vitamin K1 is mainly used to prevent and cure bleeding caused by vitamin K deficiency. The adverse reactions of vitamin K1 injections, commonly used in clinics, include allergic reactions. Parenteral vitamin K1 causes delayed hypersensitivity skin reactions, which may be eczematous or erythema multiforme-like and surround the puncture sites or exhibit a diffuse patchy distribution.1–3 In addition, an excessively fast intravenous injection speed of >5 mg/min causes facial flushing, sweating, bronchospasm, tachycardia, and hypotension. Intramuscular injections also cause local swelling and pain.
Case report
A 52-year-old woman visited our department because of erythema, papules, pain, and itching on her hips and trunk that had persisted for 1 week on 20 February 2019. Twenty-two days before admission, the patient received three intramuscular injections of vitamin K1 (10 mg once a day). Two weeks after the last intramuscular injection, a large area of edematous erythema and sclerosis developed bilaterally around the injection sites on the buttocks, accompanied by itching, mild pain, and a burning sensation. Thereafter, the rash gradually expanded, pain aggravated, and erythema and itching were noted, which affected her sleep. The patient received oral administration of “desloratadine and ebastine” and external application of “desonide cream and compound calamine lotion,” but the rash did not significantly improve. Moreover, a few blisters of mung bean to soybean size developed, in addition to the hip erythema, with no fever or other discomfort. Therefore, the patient was hospitalized for further diagnosis and treatment.
Past history: Facial allergies occurred after an intravenous drip of acetylcysteine injections more than 20 days prior, which subsided after the application of diphenhydramine. Right hemihepatectomy combined with cholecystectomy was performed 22 days prior for a “liver mass” and “gallbladder stone.” Pathological results: The patient received a pathological diagnosis of hepatic cholangiocarcinoma and hepatic cavernous hemangioma. Vitamin K1 injections were administered intramuscularly three times after the surgery. Furthermore, the patient had a history of viral hepatitis B for 25 years, with no history of hypertension, diabetes, or heart disease.
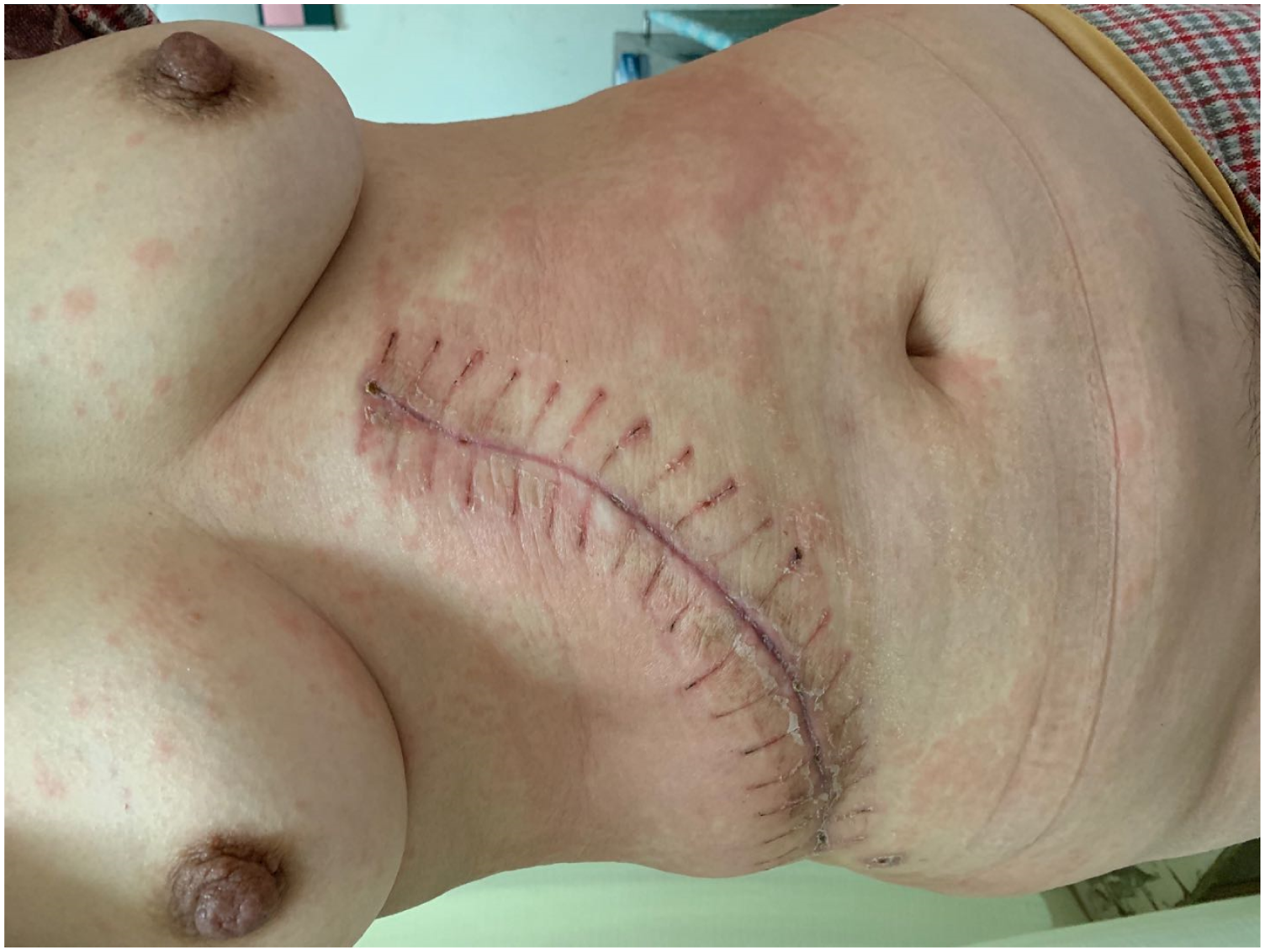
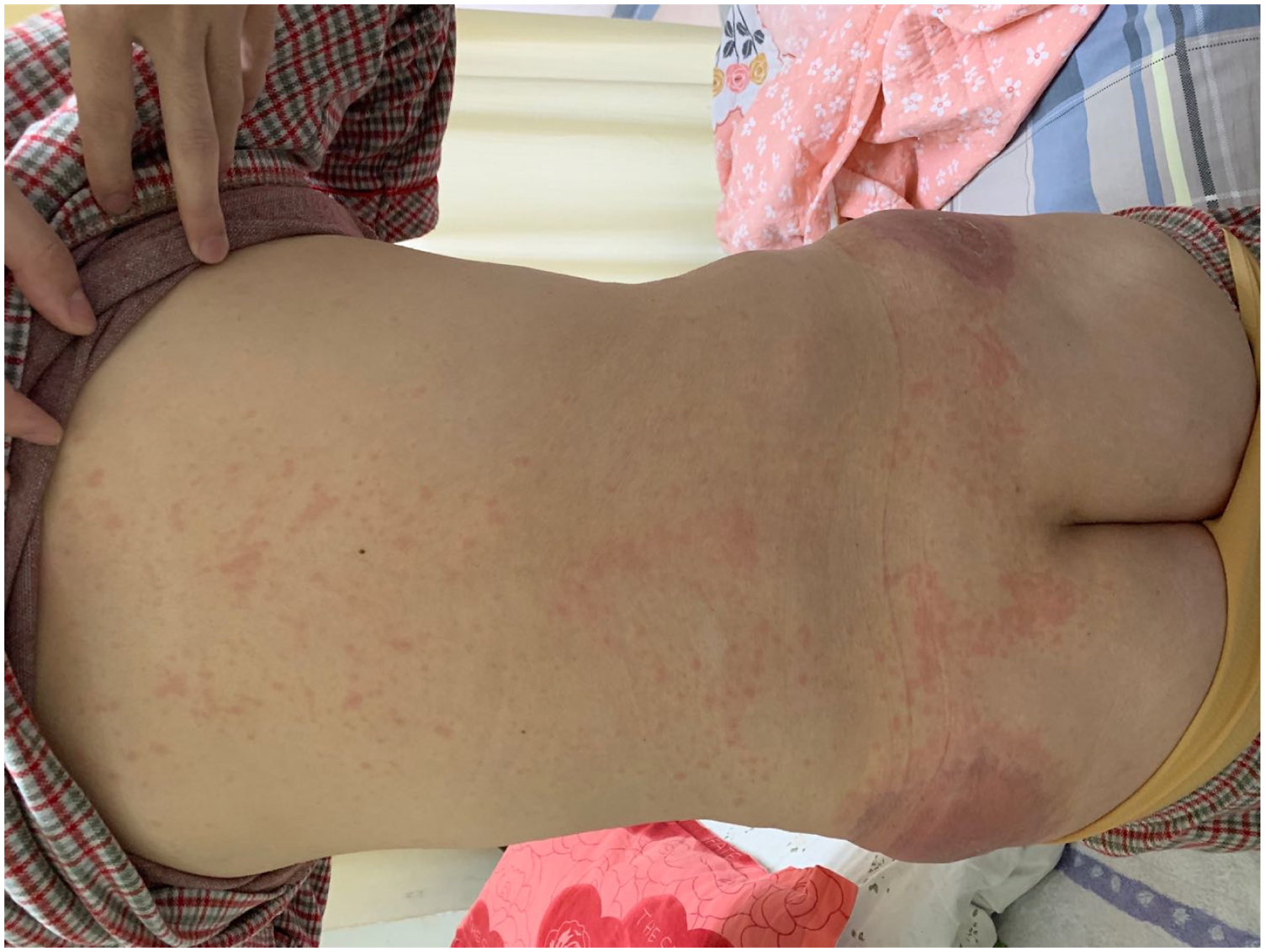
Systematic examination: No obvious abnormalities were found on cardiopulmonary examination. An arc incision approximately 30 cm in length was observed in the right abdomen. A soft abdomen, mild tenderness, and the lack of rebound pain and muscle tension were also observed. Dermatological examination: A large area of well-defined round edematous erythema and sclerosis was observed bilaterally around the injection sites on the hips, which was dominant on the left side and surrounded by a purple-red halo. In addition, a few blisters 0.5 cm in diameter were also observed, among which, some burst, with slight exudation. The local skin temperature was high, with tenderness. Dense erythema was observed in the chest, abdomen, and back, and some of the lesions fused into large patches without mucosal damage (Figures 1–3). A large area of well-defined round edematous erythema and sclerosis was seen around the injection sites on hips, which surrounded by purple-red halo. Dense erythema in the chest and abdomen, and some of them fused into large patches. Dense erythema in the back.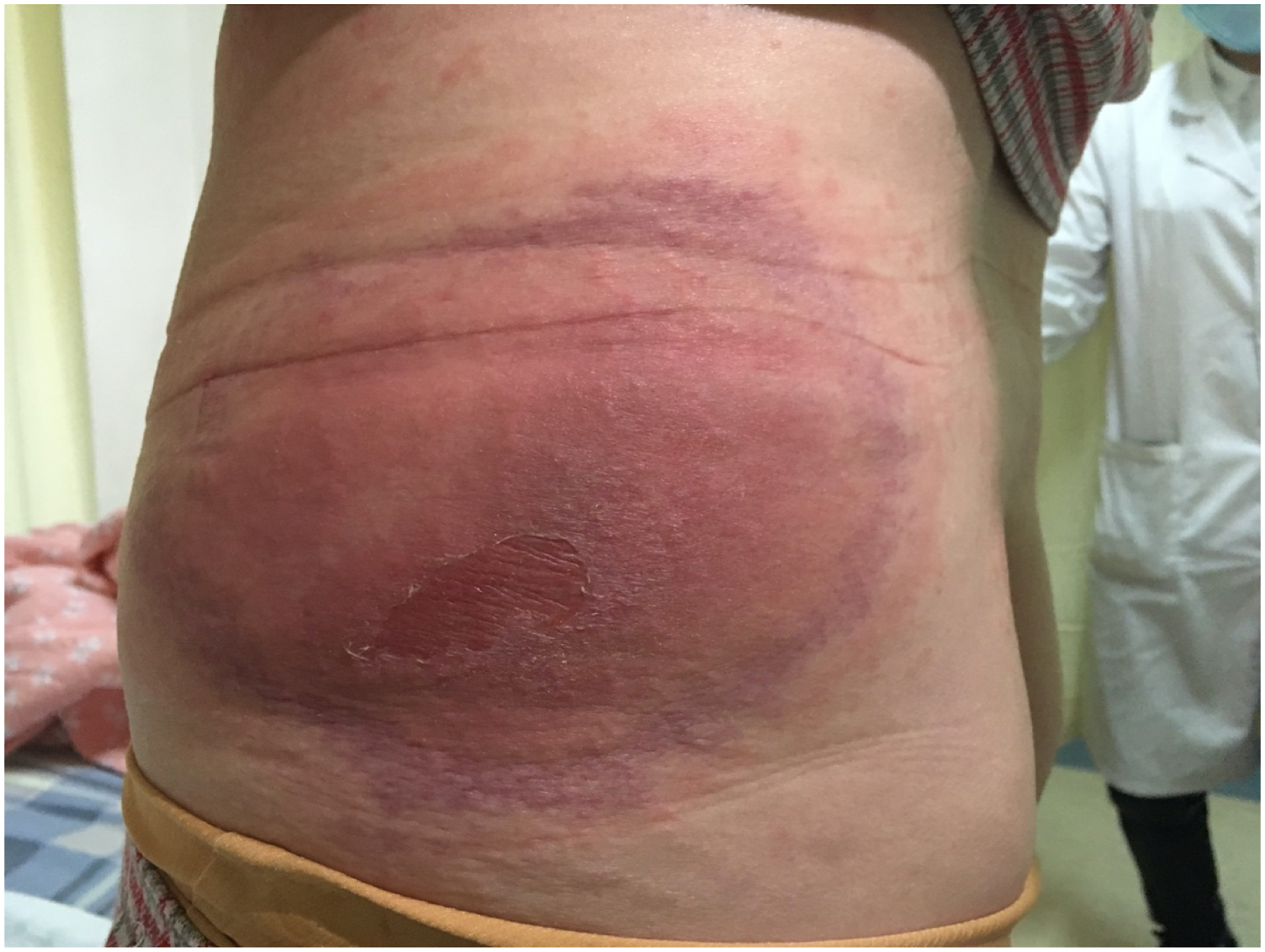
Laboratory examination: The patient presented with the following levels: white blood cells, 3.49 × 109/L; neutrophil proportion, 42%; neutrophils, 1.46 × 109/L; red blood cells, 3.42 × 1012/L; and C-reactive protein, 1.35 mg/L (0–6 mg/L). No obvious abnormality was found in the four items corresponding to the liver tumor and clotting factor values. Thoracic and abdominal CT revealed a blurred abdominal fat space after hepatectomy, a small amount of pelvic effusion, multiple low-density lesions in the liver that were suspected to be cysts, segmental atelectasis in the right lower lobe, and a few strings in the right middle lobe. A skin biopsy was performed. The epidermis showed acanthosis and spongiosis, and the dermis was edematous with a dense perivascular lymphocytic infiltrate with mononuclear cells and a few eosinophils (Figures 4 and 5). The epidermis showed spongiosis and the dermis was edematous with a dense perivascular lymphocytic infiltrate with mononuclear cells. Hematoxylin and eosin(H&E) biopsy of skin(original magnification ×100). A few eosinophils in the dermis. Hematoxylin and eosin(H&E) biopsy of skin(original magnification ×200).

Diagnosis and treatment: Based on the clinical manifestations, medication history, and drug eruption latency characteristics, the patient received a diagnosis of a skin reaction induced by intramuscular injections of vitamin K1. After admission, the patient was administered an intravenous drip of glycyrrhizin, calcium, and vitamin C; intramuscular injection of diphenhydramine; oral administration of loratadine citrate and ebastine; external application of compound calamine lotion on the trunk; and external application of fluticasone propionate cream and polysulfonate mucopolysaccharide cream on both buttocks after wet compression. After 5 days of treatment, the skin lesions on the buttocks alleviated, but the trunk lesions persisted. The skin rash aggravated with itching. Considering the wound healing and history of viral hepatitis B after surgery, the patient was treated for another 10 days without systemic use of glucocorticoids. The skin lesions on both buttocks subsided, edema, and induration improved, erythema turned pale brown, skin temperature was normal with no tenderness, and skin lesions on the trunk subsided, leaving pigmentation (Figures 6 and 7). The erythema of trunk had subsided after treatment. Skin lesion subsided after treatment.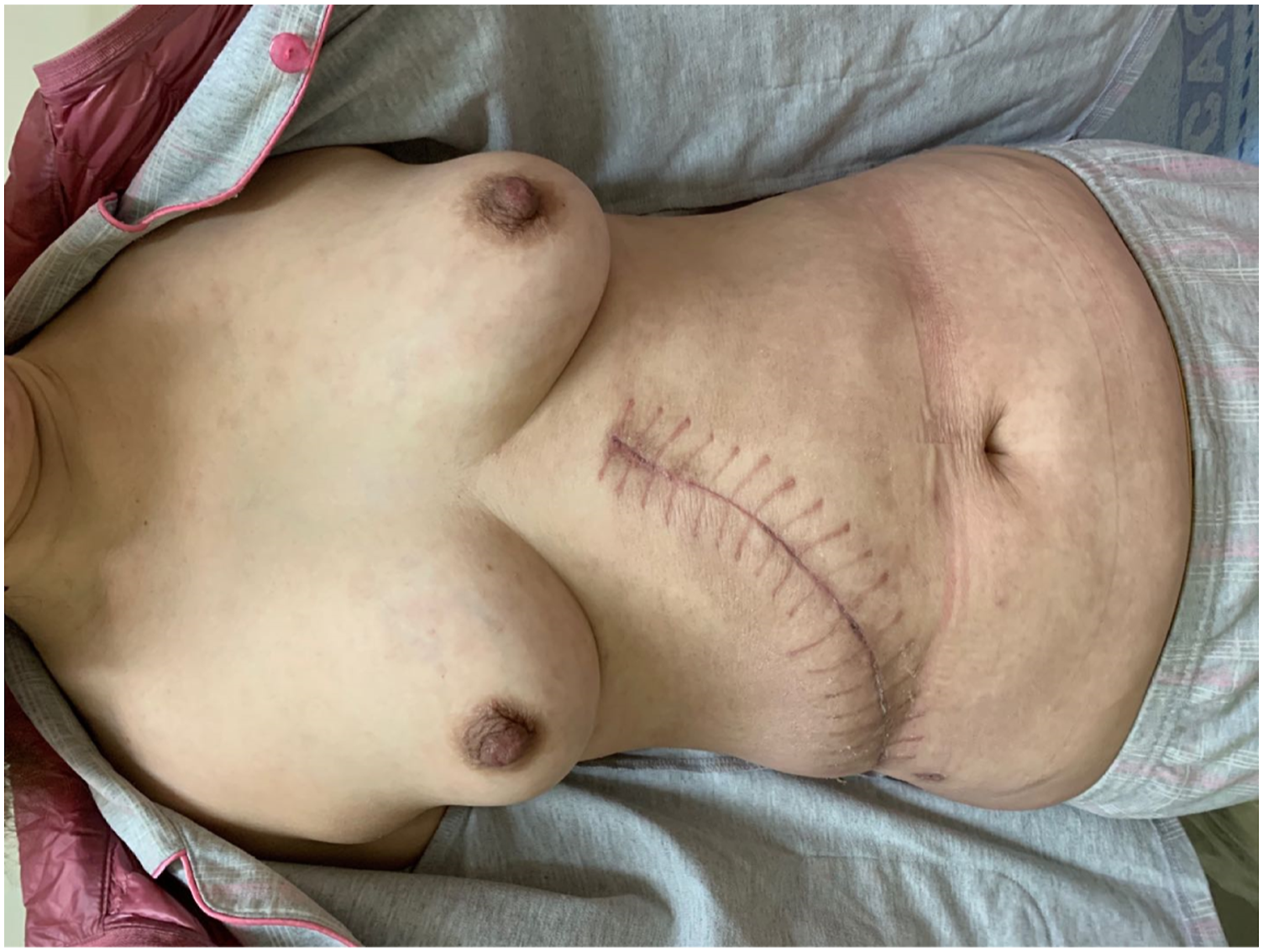

Discussion
Skin adverse reactions induced by vitamin K1 are rare and are considered to be delayed hypersensitivity phenomena related to the body, drugs, and administration methods. At present, the mechanism underlying these reactions remains unclear, and it is generally believed that it is related to immunity. 4 Vitamin K1 is mostly administered through intravenous and intramuscular injections, but rarely through oral administration; therefore, allergies are rare and no routine skin test is required. 5 Skin lesions are confined to the injection site or spread throughout the body. In most cases of generalized lesions, the lesions also form at the injection site and then gradually extend to other sites. According to the skin rash characteristics, the skin reactions induced by vitamin K1 injections are classified into three types: localized eczema type, localized scleroderma type, and eruptive disseminated macular type. 2 Among them, the eczema type is the most common, followed by the scleroderma type, while the eruptive disseminated macular papular type is the least common. Eczema usually occurs 7–14 days after injection, which may persist for 2–4 weeks and recover after several weeks or months. In one reported case, a 27-year-old woman with normal liver function developed a relapsing and remitting eczematous reaction 1 day after intramuscular injection of vitamin K1 (phytomenadione) into her thigh localized to the injection site. 6 The clinical manifestations of this type are distinct, such as erythema, plaques, and contact dermatitis. Typical lesions are red eczema-like plaques, which are hard with a high skin temperature and blisters on some plaques. Scleroplaque-like lesions are rare, occurring later (1–2 years or later), and may persist for several years. In addition, there are four common scleroderma-like lesions, including erythema, red pigmentation, and marked and degenerated scleroderma. 7 There may not be an erythematous phase before the sclerotic process, and the skin lesions of scleroderma remain unchanged 1 year after drug withdrawal. The sclerosing edema reaction may be due to hypersensitivity, as in the more common eczematous reaction. 7 The serum eosinophil chemokine levels in patients with vitamin K1-induced sclerosis are 3-fold higher than those in patients with idiopathic sclerosis. 8 Eruptive disseminated macular lesions may manifest as erythematous and urticarial lesions. No reliable data are available regarding the correlation between lesion severity and the number and dose of vitamin K1 injections. However, a single low dose of vitamin K1 also causes skin lesions.
According to a review of clinical cases, women are more susceptible to vitamin K1-induced skin allergies than men. This may be because women have more subcutaneous fat; vitamin K1 is a fat-soluble vitamin that is injected subcutaneously, and the drug thereby remains in fat, which slows down the absorption and diffusion of vitamin K1 in the body, eventually leading to an immune response between T cells entering cells and the remaining vitamin K1 as an antigen. However, the specific mechanism remains unclear at present. 9 In our case, the injection sites of the skin lesions were characterized by a large area of surrounding scleroma, followed by multiple erythematous lesions on the trunk, with typical features.
Patients with liver diseases were previously thought to be more likely to develop adverse skin reactions to vitamin K1, which may be because such patients are susceptible to hypothrombinemia and require more vitamin K1. Now, many cases of adverse reactions have been reported, such as leukemia, nephrotic syndrome, obstructive jaundice, and biliary hemorrhage.
Vitamin K1-induced skin lesions show no specific pathological changes. To be specific, the pathological changes of eczema include epidermal sponge edema, infiltration of monocytes around the dermal vessels, eosinophils between the dermal vessels, and rare infiltration of neutrophils, which may lead to the disappearance of peripheral monocyte infiltration. Scleroderma-like lesions may manifest as sclerosis of the dermis and subcutaneous tissues, thinning of the epithelial structure, decreased skin appendages, increased collagen bundles, and changes in pathological results with inflammatory progression over time. 10
In terms of treatment, no specific drug is available to prevent the occurrence of adverse reactions. Usually, the application of vitamin K1 should be withdrawn immediately upon the occurrence of skin lesions. Typically, local skin reactions at the injection site may be treated with hormone ointment, and wet compression should be applied first in the presence of exudation. In addition, antibiotics should be applied locally when secondary infection occurs, and hormone therapy may be applied systematically for extensive lesions, while the specific dosages depend on the disease severity. At the same time, oral antihistamines and calamine lotion may relieve itching symptoms. Some experts suggest that vitamin K1 in the diet may be related to the persistent exacerbation of skin rashes, but there is no relevant literature to further support this possibility. This is mainly because it is impossible for vitamin K1 to remain completely excluded from the daily diet; instead, vitamin K1 exists in small doses in many foods and in large quantities in green vegetables. Notably, foods that are rich in vitamin K include cabbage, lettuce, broccoli, mustard, coriander, lotus root, black tea, and green tea. Nonetheless, it remains unclear whether vitamin K1 may be used as an effective treatment in food intake. As an inhibitor of calcineurin, tacrolimus effectively inhibits the activation of interleukin-2 and T cells, while alleviating local inflammatory responses, and the effective external treatment of tacrolimus has been reported.
Conclusion
The adverse reactions of vitamin K1 are difficult to avoid completely or accurately due to the variations in patient characteristics, routes of administration, and role of the drug itself. It remains difficult to diagnose these lesions due to the delayed onset of skin lesions, diverse clinical manifestations, and treatment continuation after interruption, even though different methods may be used for years. Therefore, we hope that more similar reports provide relevant information for the clinical use of drugs, so that clinicians may identify and treat adverse skin reactions early, avoid misdiagnosis, and increase the continuous use of drugs, which has a certain role in the treatment and prognosis of patients with bleeding caused by primary diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
