Abstract
In this report, we present the case of vanishing bile duct syndrome in the setting of classical Hodgkin lymphoma. Vanishing bile duct syndrome was diagnosed retrospectively in this patient with Hodgkin lymphoma, who initially presented with a hepatic abnormality presumed to be drug induced. Vanishing bile duct syndrome is characterized by the disappearance of bile ducts, with the progressive damage resulting in cholestasis. Thus, nivolumab therapy was initiated for Hodgkin lymphoma, in place of the standard ABVD (Doxorubicin, bleomycin, vinblastine, dacarbazine) regimen, which resulted in autoimmune hemolytic anemia. Alternatively, GDP (gemcitabine, dexamethasone, and carboplatin) chemotherapy with protocol modification resulted in better tolerance and remission of Hodgkin lymphoma. Granulocyte colony-stimulating factor support and romiplostim supplement were provided to prevent chemotherapy-induced neutropenia and thrombocytopenia, respectively. Due to the deranged liver function in our case, we initially suspected the etiology as drug-induced cholestatic injury. While hepatic failure is the leading cause of mortality among patients with Hodgkin lymphoma-related vanishing bile duct syndrome, our case report suggests a complete remission of vanishing bile duct syndrome following an adequate treatment of Hodgkin lymphoma and an improvement in the hepatic function. To conclude, our report describes the rare case of vanishing bile duct syndrome which heralded the diagnosis of Hodgkin lymphoma, and the effective management of Hodgkin lymphoma which precedes the improvement of hepatic abnormality.
Introduction
Vanishing bile duct syndrome (VBDS) is a rare outcome but a severe complication of acute drug-induced liver injury. The other risk factors include chronic alcohol use, hepatitis-causing viruses, hemophagocytic lymphohistiocytosis involving the liver secondary to malignancy, autoimmune disease, and organ transplant recipients with graft versus host disease. It is marked clinically by chronic cholestasis without evidence of accompanying hepatitis, and histologically by the loss of intrahepatic bile ducts (ductopenia).1,2 The loss of interlobular bile ducts in at least 50% of the small portal tracts is called ductopenia. 3 The pathogenesis of VBDS is uncertain. 2 Drugs such as ibuprofen, allopurinol, ampicillin, amoxicillin-clavulinic acid, erythromycin, and meropenem have been accused of causing VBDS secondary to cholestasis.
VBDS related to Hodgkin lymphoma (HL) is a rare paraneoplastic syndrome and typically presents with jaundice, pruritis, hyperbilirubinemia, and weight loss. 4 Until recently, cases of HL with VBDS were postulated to sequel with poor survival outcomes. Given the low regenerative capacity of the injured bile ducts, 5 the irreversible damage to the bile ducts will lead to hepatic failure and death. Recent evidence indicates the potential reversibility of this disease and subsequent survival among cases who respond to cancer treatment. Our case report provides evidence of VBDS which augured the subsequent diagnosis of HL, and successful treatment with modified protocols of GDP (gemcitabine, dexamethasone, and carboplatin) chemotherapy resulting in a reversion to normal liver function and disease remission. The treatment modalities include discontinuation of nivolumab due to the induced autoimmune hemolytic anemia, as well as the initiation of granulocyte colony-stimulating factor (G-CSF) for myelosuppression following the treatment of HL.
Case report
In June 2019, a 25-year-old female patient of Asian-Indian ethnicity, presented with complaints of jaundice at our tertiary care center. The only significant history comprised consumption of ciprofloxacin antibiotics in the recent past. The patient was evaluated for various causes of acute liver injury with an ultrasound of the abdomen, liver biopsy and blood tests. The laboratory tests showed significant levels of total bilirubin level of 17 mg/dL (normal: 0.1 to 1.2 mg/dL), direct bilirubin level of 11 mg/dL (normal: <0.3 mg/dL), alkaline phosphatase (ALP) of 464 U/L (normal: 41–133 U/L), and alanine aminotransferase (ALT)/aspartate aminotransferase (AST) of 61/115 U/L (normal: 7–56 U/L/0–35 U/L). The ultrasound scan of the abdomen revealed borderline hepatomegaly with no altered echotexture. The results of liver biopsy were negative for HL immunohistochemistry (IHC) markers (CD30, CD15, PAX5, and EBV). 6 The patient was placed on supportive medication (Ursodeoxycholic acid 300 mg BD which was continued for 1 year), considering the condition as cholestatic jaundice. Following a 2-month observation period (August 2019), the patient did not show resolution of hyperbilirubinemia.
During September 2019 (3 months from presentation), results of a repeat liver biopsy were suggestive of lobular inflammation with prominence of Kupffer cells and expansion of portal tracts due to inflammation. The differential diagnosis includes a possibility of mixed portal inflammation secondary to drug-induced cholestatic injury. She was further placed on supportive medication and observation for 6 months, with intermittent follow-up examination. Subsequent evaluation with a biochemical profile and ultrasound of the abdomen during January 2020 (7 months from presentation), did not show any further improvement.
During April 2020 (10 months from presentation), the patient complained of loss of weight and was evaluated again for the possibility of a systemic disorder. A baseline fluorodeoxyglucose (FDG)-positron emission tomography-computerized tomography (PET-CT) scan was performed, which suggested lymphadenopathy (axillary, cervical, and mediastinal) along with lesions of the bone and manubrium, as well as deposits in the lung. There was no evidence of any mass lesion in the liver. The morphological impression following excision biopsy of the axillary lymph node, was suggestive of classical HL or anaplastic large cell lymphoma. The IHC markers were positive for CD3, CD5, CD10, CD30, CD15, ALK1, CD117, CD23, PAX5, and Ki67, which further confirmed the diagnosis of classical HL, mixed cellularity subtype. Given the bone deposits in the baseline PET scan, HL was initially classified as Stage 4. The bone marrow aspiration test revealed marrow infiltration with HL, thus rendering it a high-risk scenario.
Treatment plan
The standard of care first-line treatment of HL: ABVD (Dosorubicin, bleomycin, vinblastine, and dacarbazine) protocol 7 was not offered to the patient considering the cholestatic jaundice, which was presumably attributed to the prior administration of ciprofloxacin. The metabolism of both adriamycin and vinblastine occurs in the liver. However, with a high bilirubin level at 17 mg/dL, Doxorubicin and vinblastine were contraindicated owing to the increased toxicity and severe myelosuppression.
Given the unique situation of HL in the setting of cholestasis, with all first-line chemotherapy regimen not being indicated, opinion was solicited from various National and International experts. Subsequently, nivolumab treatment was initiated which is the currently approved second-line therapy for the treatment of HL. This drug has a favorable safety profile even among patients with deranged liver function. On May 15, 2020 (11 months from presentation), the patient was started on nivolumab 200 mg (3 mg/kg IV) as a fixed dose, once in 2 weeks. However, after 40 days of nivolumab initiation (post administration of two doses), the patient’s total bilirubin increased from a baseline of 17–26 mg/dL with an increment in direct and indirect bilirubin components. Also, her hemoglobin (Hb) level significantly dropped to 5 from 12 mg/dL which led to the suspicion of autoimmune hemolytic anemia secondary to nivolumab therapy. The autoimmune hemolytic anemia was further confirmed from the following laboratory findings: high-serum LDH (lactate dehydrogenase), low-serum haptoglobin, and a positive direct and negative indirect Coomb’s test. 8 Given the CTCAE (common terminology criteria for adverse events) Grade IV hematological toxicity of nivolumab, the drug was discontinued. Despite the transfusion of multiple units of irradiated packed red blood cells (from June 18, 2020, to July 9, 2020) for compensating the low hemoglobin level, there was no improvement. On July 11, 2020, the direct Coomb’s test reported a negative result which was suggestive of the resolution of autoimmune hemolytic anemia. However, due to the persistently high bilirubin level (17 mg/dL), first-line chemotherapy for HL could not be administered.
Starting from July 11, 2020 (13 months from initial presentation), the treatment was changed to the GDP protocol, 9 (gemcitabine 1600 mg (1000 mg/m2 IV infusion) dexamethasone 40 mg (once daily orally), and carboplatin 600 mg infusion (target AUC of 4 to 6 mg/mL.min)) as a second-line treatment for HL, which includes a four-cycle Q3-week regimen. Given the peak of the COVID pandemic in July 2020 and the scarcity of inpatient beds, cisplatin infusion otherwise would require admission for 3 days. Hence, it was replaced by carboplatin. Unfortunately, on day 12 of the first cycle, the patient developed grade 3 febrile neutropenia (absolute neutrophil count (ANC): 600 cells/µL), which required the administration of higher antibiotics “Piperacillin-Tazobactam.” Her Hb level dropped to <5 g/dL, raising suspicion of a gemcitabine-induced hemolytic disease or hemolytic uremic syndrome. However, the investigation for hemolysis was negative (negative direct Coomb’s test and normal haptoglobin levels), which led to the suspicion of carboplatin-induced myelosuppression.
The patient’s treatment protocol was further altered with the administration of single-agent gemcitabine on day 1, and a combination of gemcitabine, carboplatin, and dexamethasone on day 8 of the cycle. As secondary prophylaxis, G-CSF support was provided for 5 days starting on day 8, and romiplostim supplement for the prevention of chemotherapy-induced neutropenia and thrombocytopenia, respectively. With the altered protocol, the patient tolerated chemotherapy without grade 3 neutropenia. Post three cycles of GDP protocol, interim PET-CT was done, and the results suggested good radiological, metabolic, and morphological response.
In September 2020 (15 months from initial presentation), the patient tested positive for SARS-2 corona virus infection with a CT severity score of 10 (out of 25) 10 and an ANC of 1200 cells/L. With the evolving guidelines for management of COVID and the patient’s deranged liver function undermining the administration of anti-viral drugs, she was given convalescent COVID plasma. Subsequently, the patient recovered from COVID. To avoid further complications, the GDP protocol was reinitiated following her recovery from COVID, with a 15% reduction in doses of gemcitabine and carboplatin. She completed six cycles of GDP protocol. A final FDG PET-CT scan was conducted during Jan 2021 (19 months from initial presentation), which suggested a complete metabolic and morphological response, with a score of 1 without residual mass on the five-point scale Lugano classification. 11 After completing 3 months of chemotherapy and achieving a complete response on the PET-CT scan, her bilirubin levels showed graded reduction. By March 2021, the total bilirubin was reduced to 6.2 mg/dL and by April 2021 it further reduced to 2.3 mg/dL.
This favorable change conduced the differential diagnosis of either VBDS as a paraneoplastic syndrome of HL, or the hepatic parenchymal involvement of HL which resolved post-chemotherapy. During May 2021, a retrospective review of the hepatic biopsy slides from 2019 did not show any malignant cells or atypical cellular IHC markers of HL. The initial scans (ultrasound, PET-CT) had not shown any mass lesions in the liver. However, the images of a liver biopsy done in August 2020 implied the rare diagnosis of VBDS.
Within 6 months (July 2021) of completion of chemotherapy and achieving complete remission, the patient’s liver function test showed a normalized total bilirubin value of 1 mg/dL. However, the patient continued to develop recurrent neutropenia for which she required split doses of pegylated G-CSF on a biweekly regimen. The split dosage is specific to this case as the patient developed severe body pain with a full dose (6 mg per chemotherapy cycle), which otherwise is the standard practice. 12 We gradually reduced the peg G-CSF dose to one-third dose (2 mg injection) given once every 3 weeks, which was continued for the following 6 months (June 2021–January 2022). Currently, as of June 2023, the patient is weaning off any medication with a normal CBC profile and liver function tests.
Figure 1(a) shows the baseline (April 2020) and interim (September 2020) PET-CT scans. In this figure, the first image is baseline scan, and the subsequent three images are interim scans. Figure 1(b) shows the complete remission image (January 2021).

PET-CT images. (a) Baseline and interim PET-CT image and (b) post-remission PET-CT image.
Both images are stained with hematoxylin & eosin (H&E). Figure 2(a) (40× magnification) shows Reed–Sternberg (R-S) cells (arrow) on a polymorphous background of lymphoid cells, which is pathognomonic of classical HL. Figure 2(b) (10× magnification) shows a granuloma which is consistent with the diagnosis of HL.

H&E-stained images of lymph node. (a) R-S cells and (b) Granuloma.
Both images are stained with H&E. Figure 3(a) and (b) (40× magnification) show portal tracts in the liver with total loss of biliary ductules. There is mild periportal lymphocytic infiltration and biliary stasis in the adjacent hepatocytes. There is no evidence of granuloma or plasma cells.

H&E-stained images of liver biopsy.
Figure 4(a) and (b) show the serial levels of liver function tests. Figure 4(a) plots the serial change in serum total bilirubin levels. Figure 4(b) plots the other liver enzymes. The liver function tests show normal values from July 2021 onward, which is 6 months after treatment completion in January 2021.
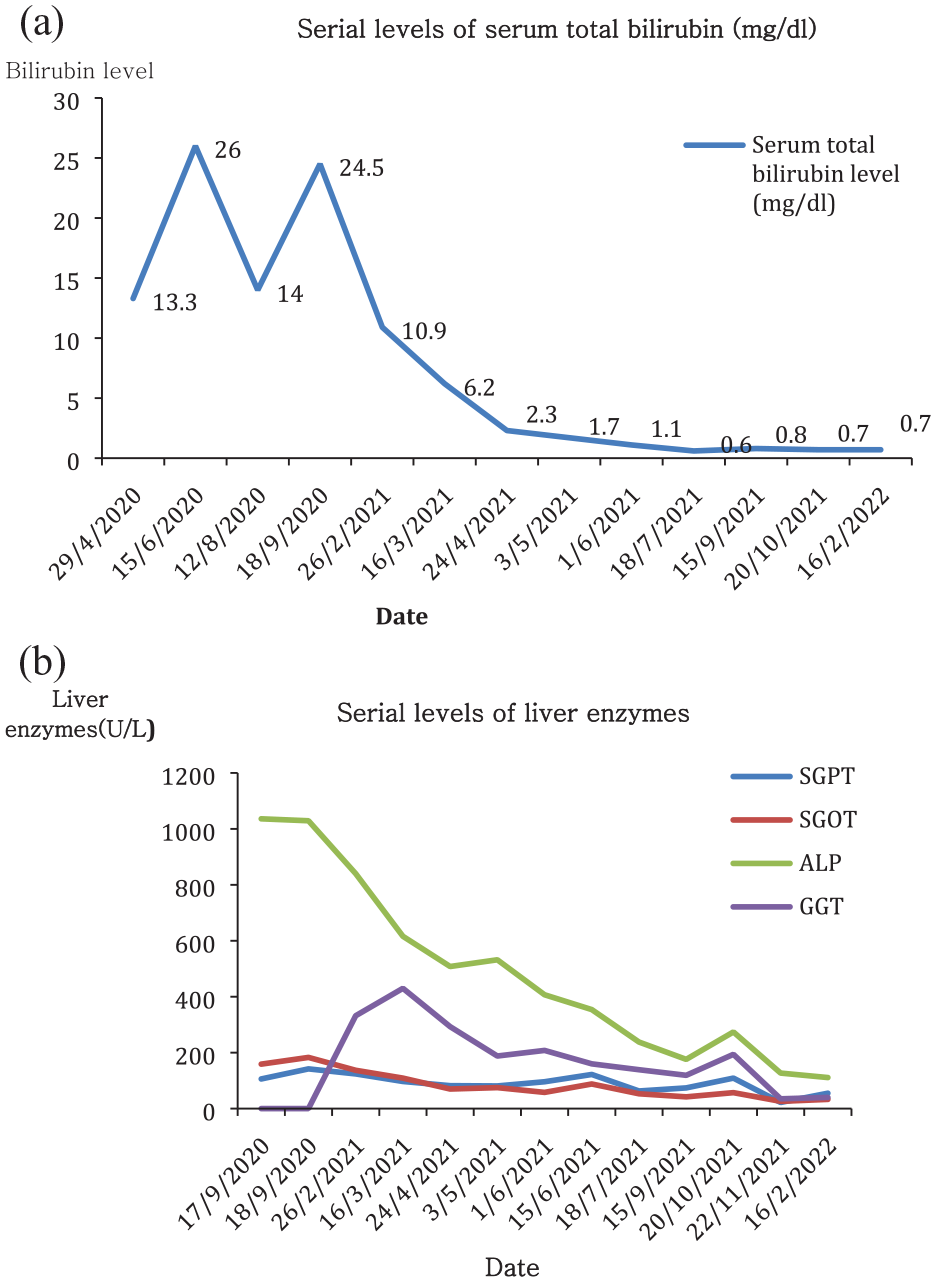
Plot of serial liver function tests. (a) Plot of serial bilirubin levels and (b) plot of serial levels of liver enzymes (other than bilirubin)*.
Table 1 shows the time course of events of the serum total bilirubin levels and the incidental treatment provided. The bilirubin levels were normalized following the completion of 6 months of chemotherapy and have thus been sustained.
Time course of events with regard to bilirubin levels and the treatment provided.

Discussion
In this report, we present the case of VBDS in the setting of HL. Given the rarity of primary hepatic HL 13 and its presentation among patients aged ⩾50 years, 6 we derived our diagnosis of VBDS by exclusion. VBDS is characterized by the disappearance of bile ducts, with the progressive damage resulting in cholestasis. 3 Prior evidence implies on the state of disease progression, which ultimately leads to hepatic failure. Earlier, patients with HL-related VBDS have been reported to have poor prognosis and overall survival.4,14 However, recent case reports suggest complete remission with adequate HL treatment and subsequent improvement in hepatic function.2,14
Wong et al. 5 discuss two possible mechanisms for explaining HL-related VBDS, which includes the direct damage to the bile ducts caused by the infiltrating lymphoma cells, and these cells releasing the paraneoplastic toxic cytokines. Evidence from cases of HL-related VBDS shows a minor load of lymphoma and severe cholestasis, which could possibly be due to the mediators released by inflammatory cells surrounding or infiltrating the tumor. 5 Scalabrini et al. 2 discuss the expression of major histocompatibility complex (MHC) antigens (both class I and II) and intercellular adhesion molecule 1 (ICAM-1) by the biliary epithelial cells, in response to the cytokines produced by HL. This contributes to the adhesion and cytotoxicity due to the T lymphocytes. Anugwom et al. 15 opine that in VBDS, apoptosis of biliary epithelial cells is favored with increased expression of B-cell lymphoma-2-associated X-protein and tumor necrosis factor-alpha.
In VBDS, small ductules of the biliary tree measuring <15 mm are usually affected, and hence hepatobiliary imaging may not provide the diagnosis. 15 The authors 15 opine that an adequate liver biopsy containing at least 10 portal tracts enables the diagnosis of VBDS. Ductopenia is diagnosed when >50% of the small portal tracts show loss of bile ducts. The diagnostic yield is increased by immunostaining for cytokeratin 7 and 19.
Patients diagnosed with HL-related VBDS are primarily in the <45 years age group. Ballonoff et al., 16 in their literature review of 37 cHL cases of VBDS (19 patients) or idiopathic cholestasis (16 patients), report the median age at diagnosis as 36.5 years. In a review by Wong et al., 5 the mean age at presentation was 28 years among 23 HL patients with VBDS. In our case, the patient was 25 years old at the time of presentation.
It is a big challenge to administer chemotherapy for patients with cholestasis. Evidence shows that although patients respond well to the initial MOPP-ABV (mechlorethamine, vincristine, procarbazine, prednisone/doxorubicin, bleomycin, vinblastine) regimen with complete remission, they soon tend to perish due to the irreversible liver failure. 17 Anugwom et al. 15 report a similar scenario wherein the patient was started on radiotherapy, a combination of high-dose steroids (dexamethasone), and chemotherapy. The chemotherapy regimen is comprised of rituximab, gemcitabine, and cisplatin. ABVD was not initiated given the significantly elevated levels of bilirubin and liver enzymes. The authors report an initial remission, thus enabling the later switch to ABVD regimen. Within 12 months of treatment initiation, the liver function test revealed normalization of bilirubin and transaminases.
Scalabrini et al. 2 report the therapeutic choice of using high-dose ursodeoxycholic acid (UDCA: 1350 mg/day) and prednisone (50 mg/day) in HL cases with VBDS, which resulted in the rapid improvement of patients. Although the patients were still icteric, a full-dose chemotherapy regimen (ABVD regimen) achieved a sustained complete remission, true to the well-known evidence of ABVD dose intensity affecting the outcome of HL. This is an important result in the scenario of a paraneoplastic disease. The authors 2 report that only 12% of patients receiving an upfront reduced chemotherapy dosage, overcame the VBDS when compared with 51% receiving the full dose. This study 2 reiterates the benefits of appropriate aggressive treatment (chemotherapy or radiotherapy) for a favorable outcome.
In the case scenario, nivolumab treatment was discontinued due to Grade IV hematological toxicity. Pastor ML’s 18 study reports that exogenous G-CSF enables preventing or reducing high-grade neutropenia among patients receiving cytotoxic drugs. In a study by Brockelmann et al., 19 the efficacy of nivolumab was compared to AVD (Doxorubicin, vinblastine, dacarbazine) among 109 early-stage unfavorable HL patients. As per the outcomes of the study, 19 nivolumab monotherapy was highly effective with 12-month progression-free survival and complete response after four cycles of drug treatment.
Given the carboplatin-induced myelosuppression following the GDP protocol, a modified GDP protocol regimen along with G-CSF and romiplostim was administered. A study by Al-Samkari et al. 20 on 42 patients with solid tumors undergoing chemotherapy evaluated the safety of romiplostim in treating chemo-induced thrombocytopenia. Based on the outcomes of the study, 20 romiplostim is a safe drug and increases the platelet count, thereby allowing subsequent use of chemotherapy at full dosage.
In the case scenario, we found a reduction in the bilirubin level following 3 months of completion of chemotherapy and achieving a complete response on the PET-CT scan (April 2021). Within the next 3 months (July 2021), the patient’s liver function tests were normalized with a total bilirubin level of 1 mg/dL. It has been reported that levels of bilirubin are reliable laboratory markers when compared with ALP, in the evaluation of response to treatment. 3
Ebadi et al.’s 21 study shows that, along with curative chemotherapy, tumor-directed radiotherapy, when used as a bridge in HL-associated VBDS (for lesions above the diaphragm), results in an improvement of liver function. However, the authors 21 could not measure the amount of tumor cytoreduction necessary to diminish the paraneoplastic liver damage and in turn permit the regeneration of the bile duct. Such interventions that improve liver function will subsequently enable standard curative-intent chemotherapy.
Wong et al. 5 demonstrated that among HL cases with VBDS receiving chemotherapy, additional autologous hematopoietic cell transplantation resulted in the survival of 44% of cases. Although autologous hematopoietic cell transplantation seems a feasible choice of treatment in this scenario, its long-term benefits need to be further elucidated.
Bakhit et al. 14 report that the immunological reaction which leads to VBDS can be treated with rituximab, a chimeric monoclonal antibody which targets CD20 frequently expressed in HL. Other strategies include using ursodeoxycholic acid and cholestyramine for improving cholestasis and rifampin, which impacts the bilirubin levels and symptoms of pruritis. The authors 16 also mention the beneficial effects of bilirubin apheresis treatment with an anion exchange adsorbent column, for reducing the bilirubin and bile acids. In case of persistent ductopenia and unresolving symptoms, liver transplant should be considered.
Conclusion
Our report describes the rare case of VBDS which heralded the diagnosis of HL and the efficacious treatment of HL which precedes the improvement of hepatic abnormality. Due to autoimmune hemolytic anemia induced by nivolumab, GDP chemotherapy regimen was initiated for the treatment of HL and its protocol was further modified for better tolerance. Given the delayed and prolonged myelosuppression following treatment of HL, split dose of G-CSF was administered. Another learning curve includes the use of split dose Peg-GCSF, which otherwise is administered as full dose in standard practice.
Footnotes
Acknowledgements
Anisha Raju: Senior Medical writer, Technology healthcare big data analytics, Gurugram, India. Dr. Radheshyam Naik, Group Medical Advisor, Healthcare Global, Bangalore, India. Dr. Veena R, Consultant Oncopathologist, Triesta Sciences—A Unit of HCG, KR Road, Bangalore, India. Dr. Prakash Vishnu, Consultant: Haematology, Medical Oncology, Valley Medical Center Specialty care, University of Washington—Medicine, Renton, Washington, USA.
Availability of data and material
Presently in the Institutional data repository. Available on request from the Corresponding Author.
Authors’ contribution
B.P. managed the treatment and conceptualized the case report; V.K.R. drafted and submitted the article; P.N.R. and M.S. were part of the team who managed the treatment; S.P. reviewed the article and D.N.R. monitored the treatment management.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: However, the publication charges were partly funded by Ms.Kamala Rao, UK, Mr.Annaiah and Mr.Kotreshappa R, India.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
