Abstract
Autoimmune diseases increase the risk of thrombosis. Neutrophil extracellular traps (NETs) are webs of DNA and protein that may mediate thrombosis in autoimmune diseases. Human and murine studies show NET-releasing neutrophils within a thrombus promote its growth, but it is unclear to what extent NET fragments released into circulation during inflammation are prothrombotic. This study hypothesized that canine NETs promote clot formation and impair lysis even in the absence of neutrophils. NETs were prepared from PMA-stimulated neutrophils and added to fibrinogen and thrombin or to recalcified pooled canine platelet-poor plasma, tissue factor, and tissue plasminogen activator. Clot formation and lysis were measured spectrophotometrically. NETs did not alter fibrin clot formation, but NETs increased maximum clot formation velocity (P = .001) and delayed lysis (P = .009) of plasma clots compared with supernatants from nonstimulated neutrophils. DNase digestion of NETs reduced their effect on clot lysis but not maximum clot formation velocity. This suggested impaired lysis was principally mediated by DNA within NETs but that NET proteins were principally responsible for increased speed of clot formation. Previous reports suggested elastase or histones might be responsible for the effect of NETs on clot formation. Elastase activity was greatly reduced by plasma, and addition of histones to plasma did not increase formation velocity, suggesting these proteins were not responsible for increasing maximum formation velocity. This study showed that NETs enhanced clot formation and impaired clot lysis in canine platelet-poor plasma. These in vitro findings suggest both NET proteins and DNA may contribute to thrombosis in inflammatory disease.
Human autoimmune diseases are associated with an increased risk of thrombosis. 31 Similarly, thrombosis is common in canine autoimmune diseases, 15,21 particularly immune-mediated hemolytic anemia (IMHA), in which thrombi are present in about 80% of dogs that die or are euthanized. 5
Links between autoimmunity and thrombosis are multifactorial, 11,19,23 but neutrophil extracellular traps (NETs) are one likely contributor. NETs are webs of DNA associated with nuclear and non-(i.e. non-nuclear)nuclear proteins that neutrophils release in response to a wide range of stimuli, including substances increased in autoimmune diseases such as immune complexes. 3,4 In addition to increased NET release, reduced degradation of NETs occurs in a subset of human patients with autoimmune diseases. 16,17
Although interactions between NETs and the coagulation system have not been previously investigated in dogs, murine studies show NETs promote thrombosis when released at sites of vascular injury. 27 NETs act on multiple components of the coagulation system to promote clot formation and impair lysis (summarized in Fig. 1). These prothrombotic effects of NETs have created considerable interest in circulating NETs as therapeutic targets in human and canine autoimmune diseases. Elevated circulating markers specific for, or correlated with, NETs have been detected in dogs with IMHA 13 and humans with autoimmune diseases including systemic lupus, 32 rheumatoid arthritis, 24 and thrombotic microangiopathies. 8 However, it is not clear whether NETs are prothrombotic when present free in circulation rather than within a developing thrombus.
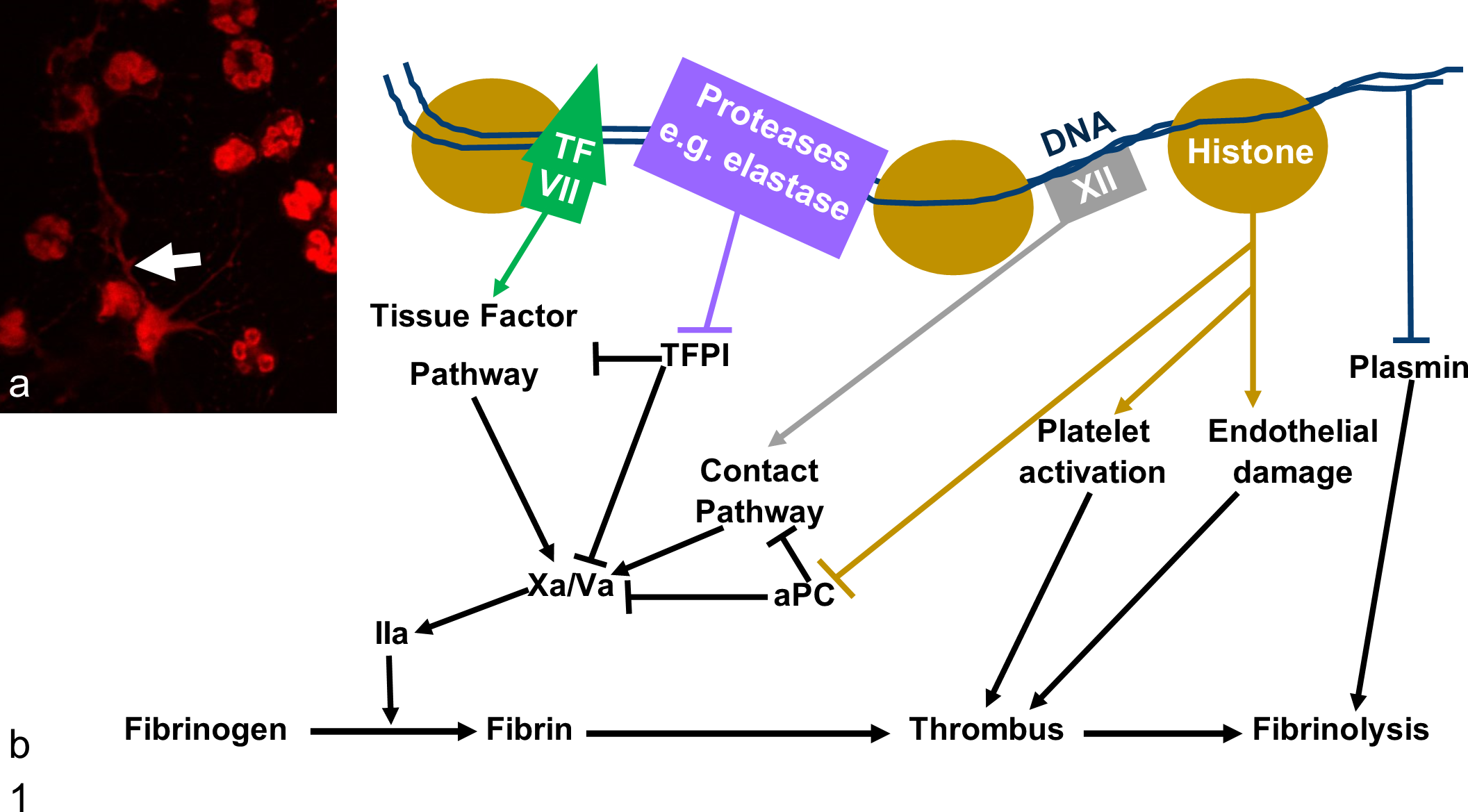
Neutrophils release neutrophil extracellular traps (NETs), which promote thrombus growth and impair lysis through multiple mechanisms. (a) NETs are webs of DNA and protein released in response to a wide range of stimuli. In this image, canine neutrophils were stimulated with platelet activating factor for 4 hours and stained with DAPI (red). A NET is indicated by the white arrow. (b) Human and murine studies have identified multiple prothrombotic effects of NETs. NETs may contain tissue factor (TF) and so activate the tissue factor pathway of the coagulation cascade. 15 Negatively charged DNA activates the contact pathway. 11 NET proteins such as elastase and histones inactivate endogenous anticoagulants including tissue factor pathway inhibitor (TFPI) 20 and impair activation of the inhibitory protein C (aPC). 1 Histones also activate platelets 11 and damage endothelial cells. 23 DNA impairs fibrinolysis through multiple mechanisms, including forming inhibitory complexes with plasmin and fibrin. 12 Histones may contribute to impaired fibrinolysis. 26
Investigation of interactions between NETs and plasma clotting have largely focused on the effects of neutrophils undergoing NETosis, 10,18,26,27 with few authors investigating the effect of NETs in the absence of intact neutrophils. 12,28 Previous studies, therefore, more closely reflect NET release within a leukocyte-rich thrombus rather than circulating NETs. The ability of NETs to trigger thrombosis in circulation is potentially clinically relevant as it could dictate the extent to which anti-NET therapies, such as DNase or anti-histone antibodies, might be useful for thrombus prevention in autoimmunity rather than only as agents to reduce damage associated with thrombosis. This study investigated the ability of canine NETs to promote clot formation and impair clot lysis in vitro in the absence of neutrophils and sought to determine if the effects of NETs were largely mediated by DNA or protein.
Methods
A schematic overview of methods and results is provided in Supplemental Figure S1.
Generating Cell-free NETs
This study was performed with ethical approval from Iowa State Institutional Animal Care and Use Committee (protocol 12-13-7687-K) and with informed owner consent. Blood was collected into EDTA from healthy pet dogs via cephalic or jugular venipuncture.
Neutrophils were isolated and NET formation induced as previously described. 13 After washing in phosphate-buffered saline (PBS), neutrophils were seeded in Roswell Park Memorial Institute medium (RPMI; 0.5% fetal calf serum, Gibco, ThermoFisher, Waltham, MA) in a 12-well polystyrene plate (Corning Inc., Corning, NY) and incubated for 1 hour at 39°C to allow attachment before a 2-hour incubation with the NET agonist phorbol 12-myristate 13-acetate (PMA; Sigma Aldrich, St Louis, MO; final concentration 7 μM). Supernatants containing NETs were obtained using a published method for generating NET fragments of sufficient size to retain functional properties. 2 Briefly, culture medium was drawn off and replaced with 400 μl of the restriction enzyme Alu1 (ThermoFisher, 4 U/ml). After 30 minutes, supernatants were collected and centrifuged to remove intact cells.
Controls from nonstimulated neutrophils were prepared identically, except PMA was replaced by an equal volume of RPMI. To control for residual PMA or RPMI contaminating the supernatants from PMA-stimulated neutrophils, PMA controls were prepared identically to the NETosis samples, except neutrophils were not present. To investigate the effects of intact NETs versus their protein components, a subset of Alu1-digested NETs were incubated with 100 U/ml (final concentration) DNase 1 (Pulmozyme, Genentech, San Francisco, CA) at 37°C for 30 minutes. To control for direct effects of DNase, DNase was added to Alu1 in the absence of NETs.
DNA was measured in supernatants using the double-stranded DNA dye PicoGreen (ThermoFisher) and protein concentration by a bicinchoninic acid assay (Pierce BCA Protein Assay Kit, ThermoFisher) according to the manufacturer’s instructions.
To confirm Alu1 generated fragments large enough to retain functional properties and DNase effectively degraded canine DNA, canine leukocytes were lysed by overnight incubation at 37°C in lysis buffer (10 mM TrisHCl, pH 8.5; 5 mM EDTA; 0.2% SDS; 200 mM NaCl in DNase free water). DNA was purified from lysed cells and supernatants collected from PMA-stimulated, Alu1-treated neutrophils using silica membrane microcentrifuge tubes (QIAamp DNA Blood Mini Kit, Qiagen, Valencia, CA) according to the manufacturer’s instructions and concentrated by elution into half the volume of the original sample. Genomic DNA was subjected to Alu1 or DNase digestion at the same concentrations as in the NET experiments. Genomic DNA and supernatants from PMA-stimulated, Alu1-treated cells were run on a 0.5% agarose gel at 100 V and imaged by ethidium bromide (ThermoFisher) fluorescence.
Effect of ALU1- and DNase-Digested NETs on Fibrin or Plasma Clot Formation and Lysis
For plasma clot studies, pooled platelet-poor plasma from healthy dogs supplemented with bovine plasminogen-depleted fibrinogen (Enzyme Research Laboratories, South Bend, IN; final concentration 4.2 mg/ml) was incubated with an equal volume of supernatants from PMA-stimulated, Alu1-treated cells +/− DNase-digestion or supernatants from nonstimulated cells for 15 minutes at room temperature in the wells of a polypropylene 96-well plate (Corning Inc.). Clot formation and lysis were initiated by recombinant tissue factor (Dade Innovin, Siemens, Malvern, PA; final dilution 1:10 000), calcium chloride (final concentration 5 mM), and recombinant tissue plasminogen activator (Activase, Genentech, San Francisco, CA; 100 μg/ml).
For fibrin clots, bovine plasminogen-depleted fibrinogen (final concentration 4.5 mg/ml) was incubated with an equal volume of supernatants from PMA-stimulated, Alu1-treated cells +/− DNase-digestion or supernatants from nonstimulated cells. Clotting was induced by bovine thrombin (Sigma Aldrich; final concentration 2 U/ml) in HEPES saline (10 mM HEPES, 150 mM NaCl, pH 7.5) and calcium chloride in HEPES saline (Sigma Aldrich; final concentration 5 mM).
Immediately after adding agonists, light absorbance (405 nm) was measured every 10 seconds for 25 minutes using a microplate reader (BioTek Synergy, BioTek, Winooski, VT). Fibrin clots were blanked by subtracting the absorbance of wells in which thrombin and calcium were replaced by HEPES saline. Plasma clots were blanked by subtracting the lowest recorded optical density (OD) for each well. Maximum formation velocity was calculated from a line of best fit based on 15 to 20 points; peak was the maximum absorbance achieved and area under the curve (AUC) calculated using the polyhedral method. 33 For plasma clots, 50% lysis time was the time from peak OD to a 50% reduction in OD. Clot lysis was not assessed for fibrin clots, as they were formed in the absence of plasmin(ogen).
Five to six technical replicates were prepared using supernatants from PMA-stimulated, Alu1-treated neutrophils +/− DNase digestion or supernatants from nonstimulated cells and the average value of the technical replicates for each dog (n = 4–5) used in statistical analysis. To test for a significant effect of DNase or residual PMA/RPMI on fibrin or plasma clotting kinetics, five to six technical replicates of each control and Alu1 alone were performed.
Determination of Fibril Thickness by Turbidity
Fibrin fiber radius was estimated from turbidity using the method described by Yeromonahos et al. (2010). 29 Fibrin clots were prepared as described and allowed to mature for 2 hours at room temperature. Light absorbance was measured at 400 to 800 nm. After subtraction of blanks prepared identically except thrombin and calcium were replaced by HEPES saline, turbidity (τ) was calculated by τ = OD.ln(10). τλ5 was plotted against λ2, and the fiber radius (r, cm) was calculated from the line of best fit using the equation τλ5 = Aμ(λ2-Br2) rearranged to produce r = √(intercept/slope × B), where λ is the wavelength of light (cm) and B is equal to (184/154)π2ns2, where ns = 1.33. For each dog (n = 4), the average radius of fibrils in clots formed in the presence of supernatants from PMA-stimulated, Alu1-treated neutrophils +/− DNase digestion or supernatants from nonstimulated neutrophils was calculated based on 5–6 technical replicates. Five technical replicates were prepared for each reagent control.
Effect of Histones Alone on Clot Formation and Lysis
Purified histones from calf thymus (H1, 2, 3, 4; Sigma Aldrich) or PBS were added in a 1:1 ratio to pooled, fibrinogen-supplemented normal dog plasma (end histone concentration 150 ng/ml, approximately the concentration present in supernatants from PMA-stimulated, Alu1-treated neutrophils estimated from DNA concentration). Clotting was initiated with tissue factor and calcium chloride as above and light absorbance measured every 10 seconds for 25 minutes. Maximum formation velocity was calculated using a line of best fit based on 15 points. Fourteen technical replicates were prepared for each condition.
Elastase Activity in Dog Plasma
Human neutrophil elastase (Sigma Aldrich; 1–0.125 U/ml) was incubated in a polypropylene 96-well plate in a 1:1 ratio with either PBS or pooled, normal dog plasma for 30 minutes at 37°C before addition of the elastase colorimetric substrate, N-Succinyl-Ala-Ala-Ala-p-nitroanilide (Sigma Aldrich; final concentration 1.3 mg/ml in 100 mM Tris HCl, pH 8). Two technical replicates were prepared for each elastase concentration and condition. Light absorbance at 410 nm was measured every 25 seconds for 10 minutes, and elastase activity measured by the increase in light absorbance between the first and last reading. To determine if NETs prevent inhibition of elastase, plasma was also incubated with PBS or supernatants from PMA-stimulated, Alu1-treated neutrophils (1:2 ratio; four technical replicates) for 30 minutes before addition of 1 U/ml elastase.
Statistical Analysis
After confirming data were normally distributed by Shapiro-Wilk normality testing, a one-way repeated-measures analysis of variance (ANOVA; i.e., block design) was used to compare the average of the technical replicates for each dog for supernatant DNA concentrations and the parameters of the fibrin and plasma clot studies. One-way ANOVA was used to test for effects of DNase or residual RPMI/PMA on clotting studies. Multiple comparisons with Tukey’s correction were performed if pANOVA < .05. Unpaired t-test was used to assess the effect of histones on maximum formation velocity and the effect of NETs on the plasma’s inhibition of elastase. Analytical coefficients of variation were calculated from 10 replicates to which PBS (rather than NETs or controls) were added. Analysis was performed using GraphPad Prism 6 for Windows (GraphPad Software, La Jolla, CA).
Results
PMA Increases DNA and Protein Concentrations of Supernatants
The mean protein concentration in supernatants of PMA-stimulated, Alu1-treated neutrophils was 55 ± 21 μg/ml (mean ± standard deviation) and was below the linear range of the assay (20 μg/ml) for supernatants from nonstimulated cells (n = 5 dogs). The mean DNA concentration was higher in supernatants from PMA-stimulated, Alu1-treated neutrophils (401 ± 94 ng/ml) than supernatants from ALU1-treated nonstimulated cells (mean 106 ± 49 ng/ml; P = .0004; Fig. 2). DNase digestion of supernatants from PMA-stimulated, Alu1-treated neutrophils reduced measurable DNA concentration (mean 113 ± 18 ng/ml; P = .0005) to levels not significantly different from supernatants from nonstimulated cells (P = .9873). By agarose electrophoresis, DNA in supernatants from PMA-stimulated, Alu1-treated neutrophils was predominantly between 600 and 1000 base pairs, equivalent to approximately 3 to 5 nucleosome units in length and in the range known to bind human fibrin and plasmin (Fig. 2). 9
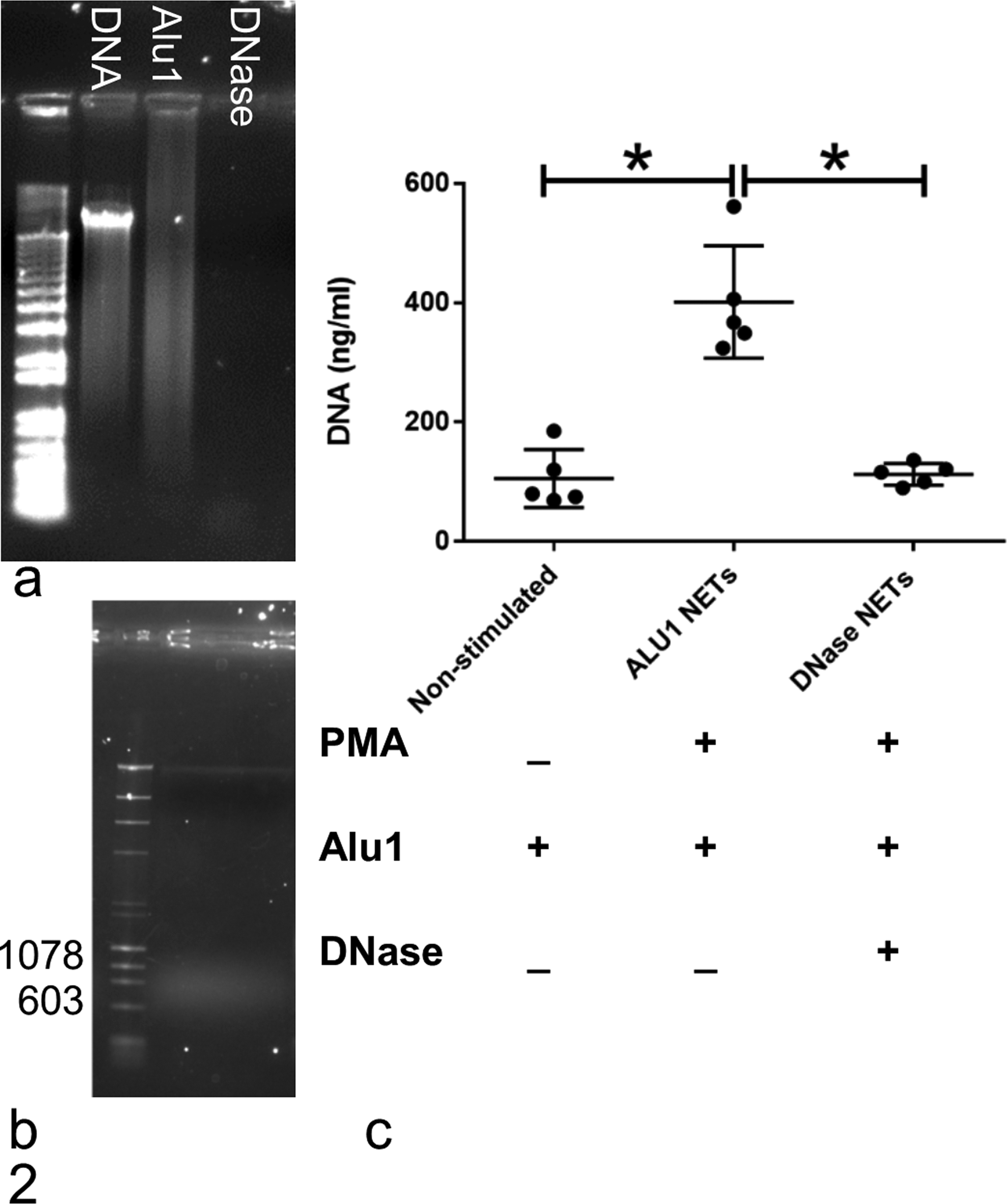
Restriction enzyme digestion of canine neutrophil extracellular traps (NETs) results in NET fragments of the desired size for experiments while DNase effectively digests DNA. (a) Genomic DNA before and after digestion, imaged by ethidium bromide fluorescence. Undigested canine genomic DNA produces a high-molecular-weight band. Digestion with Alu1 (4 U/ml) produces fragments of variable size, resulting in a smeared band. DNase almost completely digests genomic DNA. (b) Supernatants from neutrophils treated with phorbol 12-myristate 13-acetate (PMA; 7 μM) and Alu1 (4 U/ml) contain DNA that is predominantly between 600 and 1000 base pairs. (c) DNA concentration, quantified using the nucleic acid dye Picogreen, was higher in supernatants from cells treated with PMA and Alu1 than from cells treated with Alu1 alone. DNase digestion reduced DNA concentration in supernatants from PMA-stimulated, Alu-1 treated cells. *P < .001, n = 5 dogs. Error bars: mean and SD.
As these data are consistent with successful induction of NETosis by PMA and release of NET fragments by Alu1-digestion, supernatants from PMA-stimulated, Alu1-treated cells will be referred to as NETs for the remainder of the article.
NETs Increase the Speed of Plasma Clot Formation and Slow Lysis
NETs increased maximum formation velocity, and clots formed in the presence of NETs took longer to lyse, as measured by the time from peak OD to a 50% reduction in OD (Fig. 3; Supplemental Table S1). The effect of NETs on maximum formation velocity but not time to 50% clot lysis time remained significant after DNase digestion, consistent with the delay in clot lysis being principally mediated by DNA and NET proteins being principally responsible for increasing maximum formation velocity.
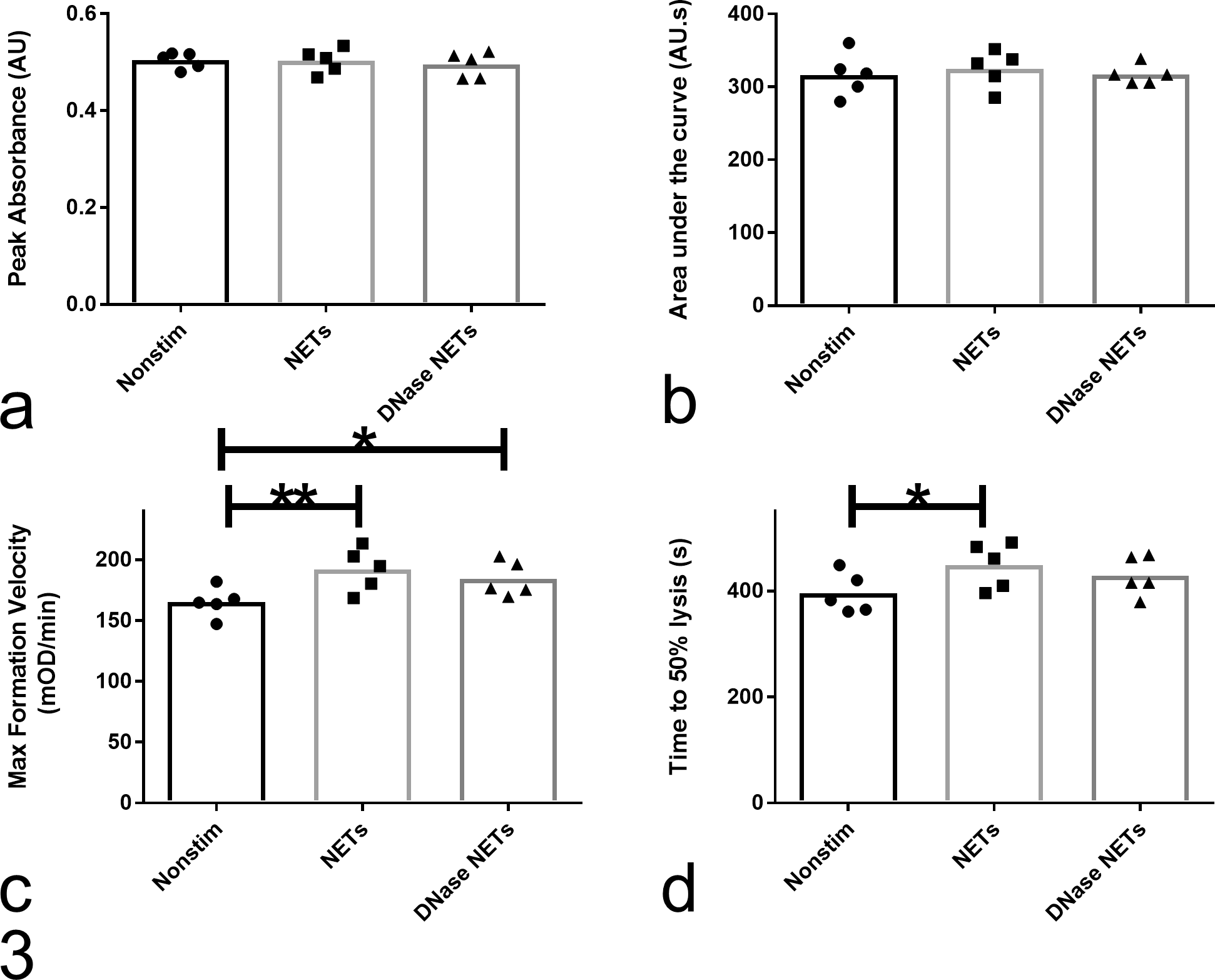
Effect of neutrophil extracellular traps (NETs) on tissue factor–induced plasma clot formation and tissue plasminogen activator–induced clot lysis. In canine plasma, NETs significantly increased maximum clot formation velocity (c) and time to 50% clot lysis (d) (pANOVA < .05) but had no effect on peak absorbance (a) and area under the curve (b) (pANOVA > .05). The effect of NETs on clot lysis (d), but not clot formation (c), was abrogated by the addition of DNase, indicating that the former is largely mediated by NET DNA and the latter by NET proteins. Five to six technical replicates were performed for each condition, and the average value for each dog (n = 5) was compared by a repeated-measures 1-way analysis of variance (ANOVA; i.e., block design). Conditions were compared by Tukey’s multiple comparisons test and multiplicity adjusted P values calculated (**P = .001; *P = .009). DNase/Alu1- and RPMI/PMA/Alu1-containing controls were not significantly different from Alu1 alone (one-way ANOVA, 5–6 technical replicates; pANOVA ≤ .717; data not shown).
For reagent controls, there were no significant differences (pANOVA ≥ .717) between clots formed in the presence of Alu1, residual PMA, or DNase alone (5–6 technical replicates). To aid in interpretation of the magnitude of the effect of NETs, 10 technical replicates were performed and coefficients of variation calculated as 5% (maximum formation velocity), 3% (peak OD), 6% (AUC), and 4% (50% lysis time).
Interactions Between NET Proteins and Plasma
After 30 minutes of incubation with canine plasma, elastase activity was undetectable for all elastase concentrations tested (Fig. 4A). Preincubation of plasma with NETs did not reverse inhibition of elastase activity (Fig. 4B).
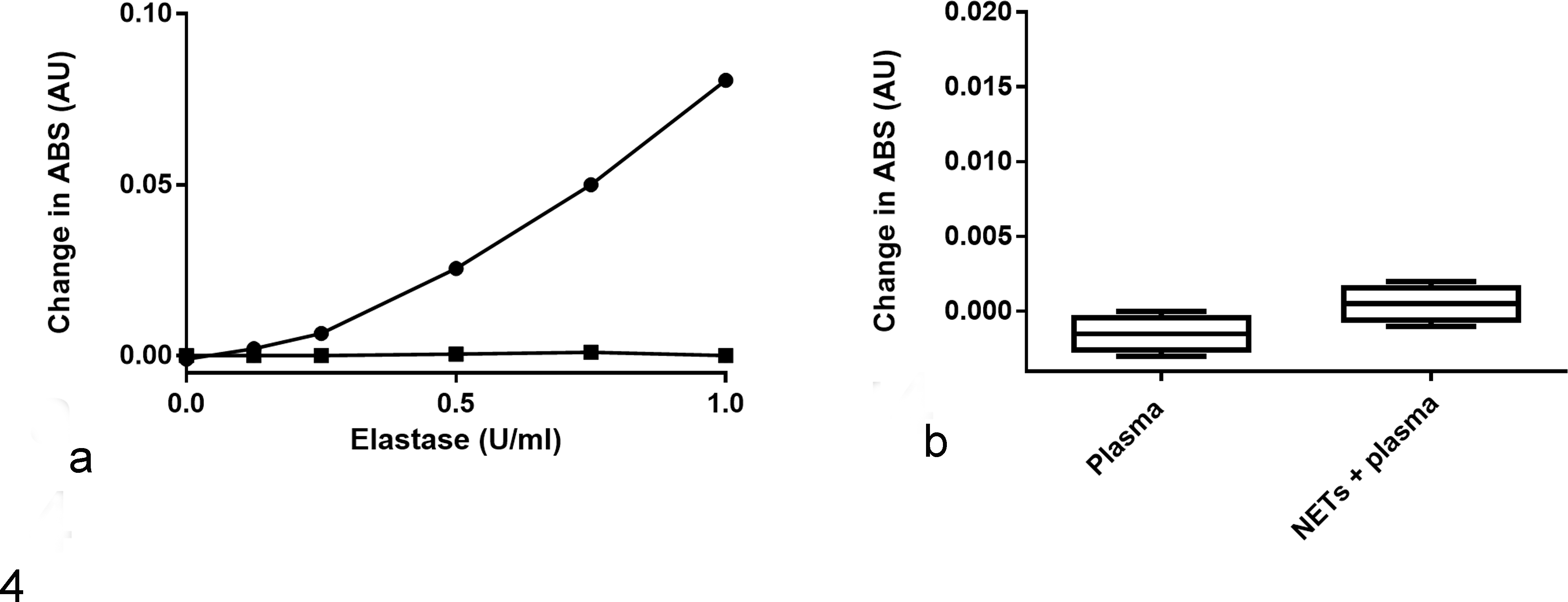
Elastase is inactivated by preincubation with canine plasma, an effect that is not rescued by neutrophil extracellular traps (NETs). (a) Human elastase was incubated with phosphate-buffered saline (circles) or pooled normal dog plasma (squares) before addition of the colorimetric elastase substrate, N-Succinyl-Ala-Ala-Ala-p-nitroanilide. Elastase activity, as measured by change in absorbance at 410 nm over 10 minutes, was almost completely abolished by preincubation with plasma. (b) Addition of NETs to plasma prior to incubation with elastase (1 U/ml) did not significantly increase change in absorbance (P = .071; n = 4 technical replicates).
Influence of elastase on maximum formation velocity was not investigated further. Mean maximum formation velocity was not significantly increased by addition of 150 ng/ml purified histones (mean maximum formation velocity histones: 101 ± 12.03 mAU/min; PBS: 98.86 ± 14.16 mAU/min, P = .66, 14 technical replicates).
NETs Do Not Alter the Kinetics of Fibrin Clot Formation or Fibrin Fiber Radius
There were no differences (pANOVA > .05) between the kinetics of fibrin clot formation in the presence of NETs or DNase-digested NETs and supernatants from nonstimulated cells for AUC, peak or maximum formation velocity (Fig. 5; Supplemental Table S2; n = 4 dogs). There were no differences (pANOVA > .09) between clots formed in the presence of Alu1, residual PMA, or DNase (5 technical replicates). The coefficient of variation of the method was ≤3% for all parameters based on 10 technical replicates.
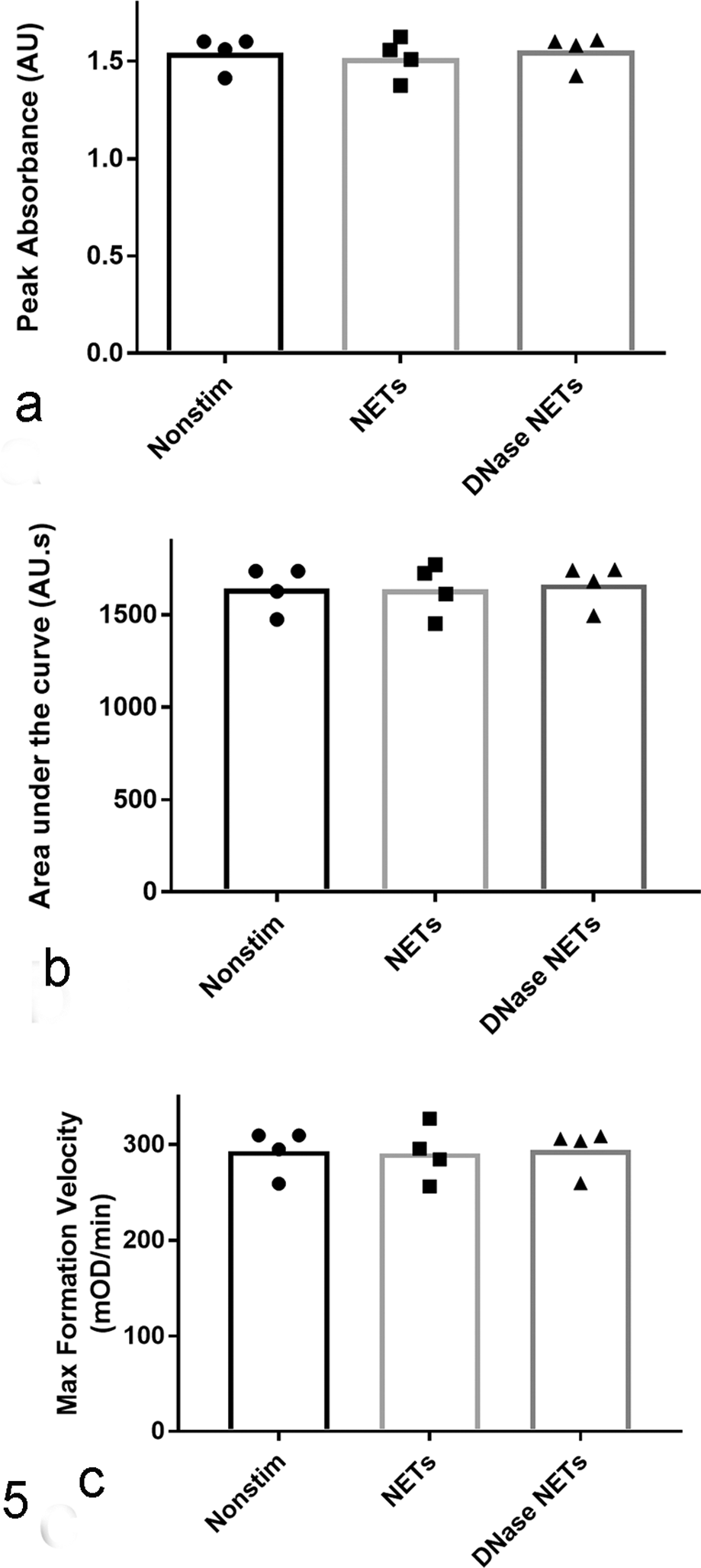
Effect of neutrophil extracellular traps (NETs) on thrombin-induced clotting of purified fibrinogen. NETs added to purified fibrinogen did not have any significant effect on thrombin-induced clot formation as assessed by peak absorbance (a), area under the curve (b), and maximum clot formation velocity (c), pANOVA > .05. Five to six technical replicates were performed for each condition, and the average value for each dog (n = 4) was compared by a repeated-measures one-way analysis of variance (ANOVA). DNase/Alu1- and RPMI/PMA/Alu1-containing controls were not significantly different from Alu1 alone (one-way ANOVA, 5 technical replicates, pANOVA > .09), data not shown.
There was no detectable difference (pANOVA = .2117) between the mean fiber radius of clots prepared in the presence of NETs (73 ± 3.9 nm) or DNase-digested NETs (73 ± 4.5 nm) and supernatants from nonstimulated cells (75 ± 3.0 nm; n = 4 dogs). There was no evidence of an effect of Alu1, PMA/RPMI, or DNase on fibrin clot radius (pANOVA = .5153; n = 5 technical replicates). The coefficient of variation for the method was 1.7% based on 10 clots formed with PBS. The R2 for the line of best fit from which the radius was calculated was >0.990 in all cases, confirming that the model of Yeromonahos et al 29 adequately fitted the data.
Discussion
Canine NET fragments increased maximum clot formation velocity and slowed clot lysis in platelet-poor plasma. Increased maximum clot formation velocity but not slowed lysis remained significant after DNase digestion of NETs, consistent with the former effect being largely mediated by NET proteins and the latter by DNA.
The ability of canine NET fragments to slow clot lysis is similar to the effect of NETing neutrophils on human plasma. 26 The ability of DNA to impair fibrinolysis appears multifactorial, with evidence that DNA intercalates with fibrin providing a scaffold stabilizing clot structure, 7 reduces plasminogen activation, 26 and forms inhibitory tertiary complexes with plasmin and fibrin. 9
The origin of the protein-mediated effect of NETs on maximum clot formation velocity is less clear. The effect was absent when NETs were added to purified fibrinogen and thrombin, and there was no turbidimetrically detectable effect of NETs on fibrin clot structure. This suggests that in this in vitro model, NET proteins did not increase clot formation velocity by direct interactions with thrombin or fibrinogen. However, bovine fibrinogen and thrombin were used for fibrin clot studies, so interactions between NETs, canine fibrinogen, and thrombin cannot be completely ruled out.
Although the effect of NETs on plasma but not fibrin clots suggests that NET proteins interact with earlier components of the coagulation cascade, identifying the specific protein or proteins responsible for increasing maximum clot formation velocity is challenging. Previously proposed procoagulant NET proteins do not act via the tissue factor or contact pathway alone but affect multiple pathways. For example, histones impair protein C activation, which affects both the common and contact pathway. 1 This limits the extent to which the protein(s) responsible for enhancing clot formation can be identified using inhibitors or factor-deficient plasma.
We therefore investigated specifically the effects of two previously proposed procoagulant NET proteins, histones 10 and elastase. 18 While histones alone were not responsible for the enhanced maximum clot formation velocity, it should not be assumed that canine histones have no procoagulant effects. Their impact on platelets 10 and endothelial cells 22 is well established in human and murine studies, but these effects would have been absent in the current platelet-poor plasma model. Furthermore, the concentration of histones was selected based on the amount of DNA present in the current study. The 200 ng/ml DNA concentration is similar to that reported in human lupus patients 32 but is lower than in other potentially immune-mediated diseases such as thrombotic microangiopathies in which DNA and histone concentrations more closely approximate microgram concentrations used in previous in vitro studies. 9,26
The greatly reduced activity of elastase after incubation with canine plasma is not unexpected, as it is known that alpha-1 antitrypsin rapidly inhibits elastase in human plasma. 20,30 However, it does suggest that elastase in NET fragments was unlikely to be responsible for increased maximal clot formation in this system.
It is intriguing that circulating NETs could contribute to a hypercoagulable state in autoimmunity and other inflammatory states. The effects of NET fragments we observed were subtle, as evidenced by the lack of significant changes in the marker of overall clot size, AUC. However, the magnitude of effects reported here are similar to the differences between healthy humans and patients with various hemostatic disorders. For example, the mean difference between the effect of NETs compared with supernatants from nonstimulated cells was 17% for maximum clot formation velocity and 12% for time to 50% lysis. Women with heavy menstrual bleeding have a mean 17% increase 25 and patients with antiphospholipid antibody syndrome a 10% decrease in clot lysis time, 6 another spectrophotometric measure of clot lysis.
Our results therefore suggest canine NETs have promise as markers of thrombotic risk, and drugs targeting NETs could have value in thromboprophylaxis. It is, however, important to recognize that the current study offers only in vitro data and is limited to examination of the effects of NETs on platelet-poor plasma. This simple model is useful for detecting subtle interactions between NETs and the enzymatic coagulation system but does not include interactions between NETs and platelets, endothelial cells, or leukocytes nor the effects of shear stress. The complexity of these interactions is becoming clear, with, for example, the recent discovery that von Willebrand factor can provide a bridge between NETs and the vasculature. 14 Further studies addressing the role of NETs in vivo and the relative importance of targeting DNA versus NET proteins are needed before anti-NET therapies are trialed in clinical patients.
In conclusion, NETs impaired fibrinolysis and enhanced maximum clot formation velocity in canine platelet-poor plasma in the absence of neutrophils. Although firm conclusions about the physiologic relevance of these changes cannot be drawn from this in vitro system, the current study provides the first evidence that NETs have procoagulant effects in dog plasma. This suggests further investigation of NETs could improve our understanding and treatment of hypercoagulability in canine autoimmunity and other inflammatory states.
Footnotes
Author Note
This work has been presented as a poster abstract at the American Society of Hematology’s 2016 annual meeting and forms part of U. Jeffery’s PhD thesis.
Acknowledgements
We would like to thank the volunteer dogs and their owners for participating in the study, Dr James Roth for his advice and support, and Amy Hodnefield and Carrie Schwake for assistance with sample collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
