Abstract
Objectives
The goal of this study was to describe the clinical presentation, diagnosis and treatment of coccidioidomycosis in cats residing in a region endemic for Coccidioides species.
Methods
A retrospective review of records was performed at both primary and tertiary care veterinary practices in Tucson and Phoenix, Arizona, USA. Data collected included signalment, clinical signs, physical examination findings, diagnostic test results, treatment and outcome.
Results
Fifty-one feline cases were identified from six veterinary hospitals. Cats presented with clinical signs and laboratory abnormalities similar to what has been seen in dogs, including respiratory illness (n = 20/51), neutrophilia (n = 24/31), monocytosis (n = 17/31) and hyperglobulinemia (n = 16/30). However, cats at diagnosis were typically significantly ill, with 31/51 having disseminated infection, most commonly to the skin (n = 22). Additionally, 43/44 cats that had serum antibody tests performed were positive, and median titer at diagnosis was 1:32 (range 1:4 to ⩾1:256). Serum antibody titers were significantly reduced (P ⩽0.001) in cats that responded to treatment compared with cats that did not clinically improve. Forty of 46 cats that were treated with oral fluconazole responded and did not require additional therapy. Fourteen cats developed recurrent disease and all but one had antifungal therapy successfully reinstituted.
Conclusions and relevance
Coccidioidomycosis is a disease of concern for cats residing in the regions endemic for Coccidioides species. Disease is most often disseminated at the time of diagnosis, possibly due to delays in presentation for care and recognition of the infection. Suspicion of disease, serum chemistries, blood cell counts, presence of antibody and imaging aid in the diagnosis of coccidioidomycosis in cats. Serum antibody reduction during treatment frequently correlated with an adequate response to medication. Consideration of coccidioidomycosis as a cause of illness will lead to earlier diagnosis and potentially better treatment outcomes in cats.
Introduction
Coccidioidomycosis, also known as Valley Fever, is a fungal disease prevalent in semi-arid regions of the western hemisphere. In the USA, endemic regions are found in Arizona, California and parts of New Mexico, Texas, Nevada and Utah.1,2 Recently, several locally acquired human cases were identified in eastern Washington state.3,4 The causative agents are the dimorphic fungi Coccidioides immitis and Coccidioides posadasii, with no noteworthy variances in the disease course between the two species. 5
Infection is acquired by inhalation of the 3–5 µm arthroconidia (spores) which grow from mycelia in the soil and are easily aerosolized by natural or human-caused disturbance. 1 In the lungs, the tiny spores easily reach the distal airways and rapidly transform into the parasitic spherule phase. Rapid expansion of the fungus by endosporulating spherules results in a respiratory infection ranging from subclinical to severe. In hosts that are unable to control the infection early, Coccidioides species may disseminate via the bloodstream to virtually any extrapulmonary tissue.1,6
Published literature on dogs with coccidioidomycosis cites a wide variety of clinical presentations, dependent on the organ systems involved, but coughing, lethargy and anorexia are the most common features, followed by lameness from osteomyelitis.6–8 Typical clinicopathological findings in dogs include hyperglobulinemia, monocytosis and hypoalbuminemia.9,10 Less information is available about the disease in cats. At one time, cats were believed to be resistant to infection by Coccidioides species; 11 however, published case reports have proven this to be incorrect.5,11–13 As the historic incidence of coccidioidomycosis is not known in cats, it cannot be ascertained whether infections are increasing or whether veterinarians are more aware of the disease in this species.
The purpose of this retrospective study was to learn more about how coccidioidomycosis presents, progresses and is treated in the feline species.
Materials and methods
Study design and case selection
A retrospective record review of feline coccidioidomycosis cases was performed at six veterinary practices in Arizona (three feline only, two referral and one small-animal). Records between 2004 and 2018 (where available) were searched for potential cases. All database searches were performed by staff at each clinic, except for one specialty hospital, where investigators searched the database for ‘coccidioidomycosis’ and ‘Valley Fever’. Cases were included in this study if they were definitively diagnosed via cytology, histology or culture, or if they had at least two of the following: clinical signs that can be related to coccidioidomycosis in other species, positive anti-coccidioidal antibody serology, clinicopathological changes similar to dogs and radiographs supportive of the diagnosis. Cases were excluded if inadequate information was available and if the diagnosis was inconclusive.
Data collection
Medical records were reviewed by one of the authors. The data collected included signalment, lifestyle, clinical signs, complete blood cell count (CBC), serum chemistries, radiographs or other imaging, coccidioidal serology, treatment and outcome. As multiple diagnostic laboratories were used, clinicopathological data was collected as the laboratory interpretation rather than the raw data. Coccidioidal serology was performed by commercial laboratories, which used agar gel immunodiffusion to identify antibodies (IgG and IgM) and quantitate IgG using serial dilution of positive serum.
Statistical analysis
Descriptive analysis of collected data was performed in Microsoft Excel (2016). Data were migrated to GraphPad Prism version 7.00 for Windows (GraphPad Software) for statistical analysis. The non-parametric Mann–Whitney U-test was performed to compare titers. Significance was set at P ⩽0.05.
Results
Case inclusion
Fifty-one feline cases of coccidioidomycosis were included in the analysis. While demographic data of the hospital populations were largely unavailable, we were able to obtain estimates of feline patient visits for two hospitals. A feline-only clinic had a rate of 2.3 coccidioidomycosis cases/1000 visits, while a specialty clinic had a rate of 1.5 feline coccidioidomycosis/1000 unique cat visits.
Signalment
Most of the cats were domestic crossbreeds (n = 46; 90%). There was one of each of the following breeds: Abyssinian, Himalayan, Maine Coon, Persian and Siamese. Age at diagnosis ranged from 1 to 15.5 years (mean ± SD 6.8 ± 4). There were 30 neutered males (59%) and 21 females (41%), of which all but one were spayed. Most cats were reported as indoor only (n = 30), with 15 indoor/outdoor and one outdoor only. Lifestyle information was unavailable for five cats. Body weights at time of diagnosis ranged from 1.86 to 7.82 kg (mean ± SD 4.58 ± 1.27 kg). Eighteen cats had lost weight from a previous visit, while 12 cats experienced no weight loss. Information about previous weight was unavailable for 21/51 of the cats. Five cats had comorbidities that could have affected immune function (feline immunodeficiency virus [n = 3], feline leukemia virus [n = 1] and diabetes mellitus [n = 1]).
Clinical signs
Clinical signs at presentation are summarized in Table 1. Dermal lesions were the most commonly reported complaint (n = 22; 43%), with most of those lesions described as nodular (n = 13/22). Additional descriptions of dermal lesions included non-healing wounds (n = 7/22) and crust or plaque-like lesions (n = 2/22). Respiratory signs were present in 20 (39%) cats. These cats were described as having some form of respiratory distress (n = 14/20), including tachypnea, wheezing or labored breathing. Six of the 20 cats presented with a cough only, while 12/20 cats had both a cough and respiratory distress described. Signs associated with systemic illness, such as decreased appetite or anorexia (n = 14) or fever (⩾103°F/39.4°C; n = 4) were less frequently reported. Lameness, paresis or paraparesis was seen in 11 (22%) cases.
Presenting clinical signs

These reflect the percentage of animals that presented with that category of clinical signs
One cat with dermatologic lesions did not have a description of the lesions provided in the record
Radiography and imaging
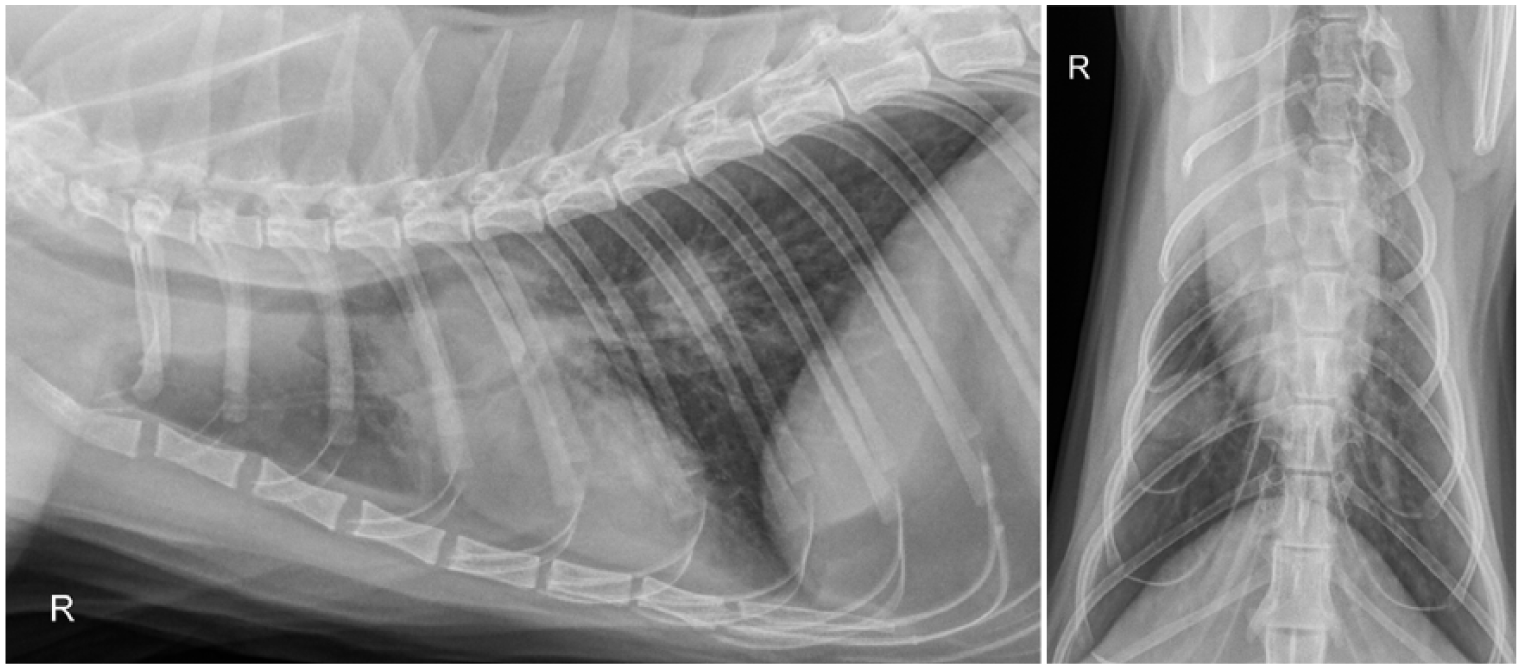
Radiographs were acquired in 22 cats at the time of diagnosis (19 thoracic, two thoracic and skeletal, and one skeletal only). One cat had a thoracic ultrasound performed and four cats had both thoracic ultrasound and radiographs performed. Results are summarized in Table 2. Half of the cats had multiple radiographic abnormalities noted. Overall, a bronchial, interstitial and/or alveolar pattern or consolidation of one or more lung lobes were noted most frequently (n = 9 [41%] each). Hilar lymphadenopathy was observed in six (27%) of the cases. Four cats (18%) had solitary lung masses/nodules and five (24%) had pleural effusion (Figures 1–3). All three cats with skeletal radiographs performed were found to have an osteomyelitis characteristic of fungal infection in a forelimb. Figure 4 shows a typical lytic/proliferative bone lesion, though the radiograph is not from one of the cats reviewed. Thoracic ultrasound showed pericardial effusion in two cats. CT documented an intramedullary mass in one cat with progressive paraparesis (Figure 5).
Radiographic abnormalities noted

Twenty-two cats had radiographs (19 thoracic only, one skeletal only, and two thoracic and skeletal)
Lateral and ventrodorsal radiographs of a 3-year-old male castrated domestic shorthair cat, presented for a 2 week history of intermittent coughing, show a multilobular alveolar lung pattern, right caudal lung lobe solitary mass and hilar lymphadenopathy with dorsal displacement of the trachea. Coccidioidomycosis was diagnosed based on radiographic changes and positive serology, and the cat responded to treatment with fluconazole

Pleural effusion and increased opacity in the left cranial lung lobe in an approximately 1-year-old male castrated domestic shorthair found stray 1 month prior to presentation for an intermittent cough. The initial anti-coccidioidal IgG titer was 1:64

Tracheal collapse and a diffuse bronchial pattern in a 10-year-old female spayed domestic shorthair presented for nasal congestion and wheezing of 4 months’ duration. A presumptive diagnosis of asthma was made from radiographs with a bronchial pattern only obtained by another veterinarian 1 month earlier, but the cat did not respond to treatment with prednisolone and amoxicillin/clavulanic acid. The radiologist who reviewed the radiographs commented asthma was the most likely diagnosis and did not include fungal disease in their differential. The initial coccidioidomycosis IgG titer was ⩾1:256
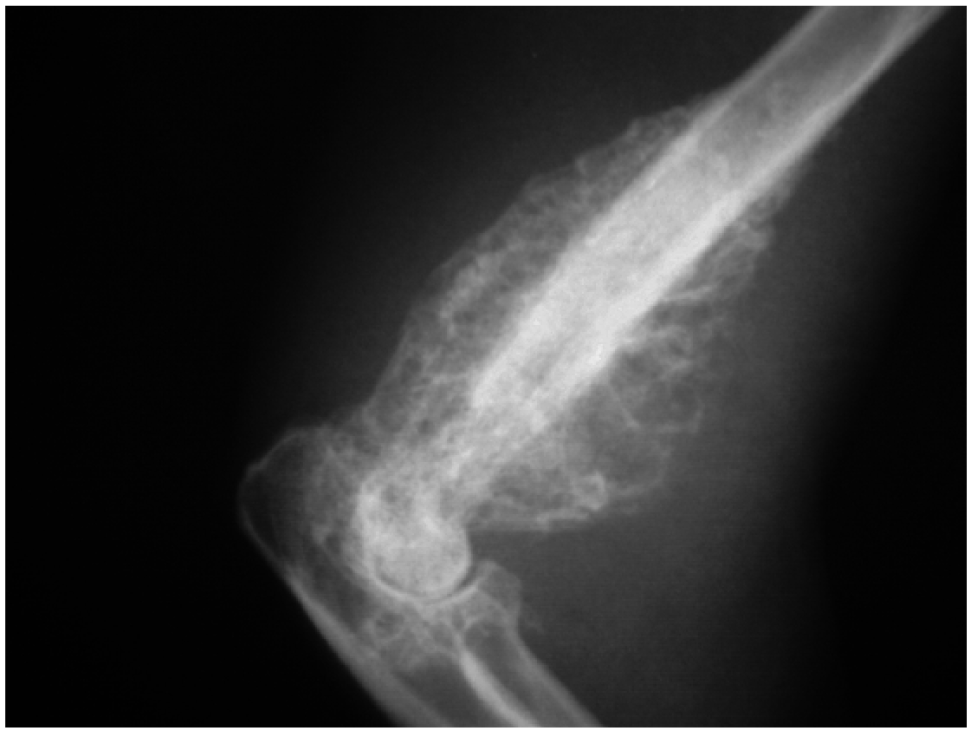
Lytic/proliferative osteomyelitis with soft tissue swelling was identified in a male castrated cat with several months’ history of lameness. The coccidioidomycosis titer was 1:256 at the time of radiography. The lytic/proliferative lesion is characteristic of coccidioidal osteomyelitis, though this cat was not one of the cases reviewed herein

Coronal section of (a) thorax and (b) sagittal section of thoracic spine of a female spayed domestic shorthair with a 3 week history of progressive hindlimb paresis and ataxia with weight loss. (a) The thoracic section shows an enlarged mediastinal lymph node (LN) and pulmonary consolidation, believed to be a granuloma (G), in the right cranial lung lobe. (b) There is a contrast-enhancing, intramedullary lesion of the spinal cord at the level of T8. Coccidioides species spherules were identified by biopsy of a sublingual mass and the cat responded to oral fluconazole
Hematology and serum chemistries
Thirty cats had serum chemistries and a CBC performed at the time of diagnosis and one additional cat had only a CBC performed. Six cats (20%) had no abnormalities noted on either the serum chemistries or the CBC. Ten cats (33%) had a neutrophilia ± monocytosis with normal serum chemistries. Hyperglobulinemia was present in 13 cats (43%); all had a normal CBC. The remaining 10 cats had abnormalities in both the CBC and serum chemistries (Table 3).
Serum chemistry and complete blood count abnormalities

Serology
Forty-four cats had coccidioidal serology performed at the time of diagnosis and 43 were seropositive. Titers ranged from 1:4 to ⩾1:256 (median 1:32). The seven cases in which serology was not performed were definitively diagnosed through cytology, histopathology or culture, as was the seronegative cat. Post-diagnosis serology results were available for 40 cats. The latest available serology result for each of these 40 cats was compared to the serology results at diagnosis, with the available follow-up time ranging from 2 months to 12 years (mean ± SD 2.67 years). The post-diagnosis serology results ranged from negative to ⩾1:256 (median ⩽1:4).
The 40 cats with follow-up serology were divided into two groups based upon whether there was clinical improvement. Thirty-three cats showed improvement, while seven cats were clinically worse. On average, the cats with clinical improvement had the titer decrease by three dilutions, while cats that were not clinically better had an average increase of the titer by one dilution. There was a statistically significant difference in the change in quantitative dilutions between these groups (P ⩽0.001).
Clinical pathology
Cytology, histology and/or culture were performed and confirmed the diagnosis of coccidioidomycosis in 18 (35%) of the cats in the population; all but one of these cats had disseminated disease. The most common confirmed site of dissemination was to the cutaneous or subcutaneous tissue (n = 6), followed by ocular tissue (n = 4). One cat each had dissemination to the pleural space, a cranial thoracic mass, the liver and an extradural mass. Four cats had positive culture and/or cytology from multiple sites.
Disseminated disease
In addition to the 17 cats that received a confirmation of extrapulmonary dissemination through histology, cytology or culture, 14 (27%) had a combination of evidence supporting a diagnosis of disseminated disease by the treating veterinarian, although etiologic testing was not pursued. Of these, nine cats had dermal lesions consistent with coccidioidomycosis. Two cats had skeletal radiographic changes characteristic for fungal osteomyelitis. Two cats had both dermal and ocular disease and one cat was believed to have dissemination to the skin, eye and tongue based on records (Table 4).
Sites of dissemination

Four cats within this group had confirmed dissemination to multiple sites
Three cats within this group had suspected dissemination to multiple sites
Treatment
Treatment was instituted in 50/51 cats, while the remaining cat was euthanized at diagnosis owing to the severity of the disseminated disease. The most commonly prescribed antifungal was fluconazole as sole agent and the typical dosage of fluconazole was 50 mg/cat PO q12h.
Forty-six (92%) cats were treated with fluconazole initially, and 40 of these never received another antifungal drug. Six cats received additional antifungal drugs, either concurrently or sequentially, owing to failure to respond to fluconazole alone. Additional antifungal drugs included itraconazole, amphotericin B, posaconazole and terbinafine. Twenty-five cats were still receiving oral antifungal medication at the time of the review (22 on fluconazole, two on itraconazole and one on posaconazole). For this group of cats, the time on antifungal medication ranged from 6 months to 7.5 years.
Itraconazole was instituted as the initial treatment in the other four (8%) cats. One of these cats was maintained on itraconazole only. One cat, which had severe skeletal disease requiring amputation of the affected limb, was changed to fluconazole post-amputation. The remaining two cats received multiple therapies (Table 5).
Antifungal treatments

One cat in this group had amphotericin B discontinued after one dose due to rising renal values; the cat was then maintained on fluconazole alone
This cat was euthanized approximately 1 year after diagnosis due to worsening disseminated disease
A recurrence of coccidioidomycosis was described in 14 cats. All of these cats received additional antifungal therapy, which was successful in 13/14. Fourteen cats were deceased at the time of the review. As previously described, one cat was euthanized at diagnosis owing to the severity of disease and one was euthanized after failing to respond to antifungal therapy after a recurrence of disease. Five cats were euthanized for unrelated medical conditions, and the circumstances of the death or euthanasia could not be determined for the remaining seven cats.
Discussion
Reports of cats with coccidioidomycosis have appeared in the literature only within the past five decades and constitute a few case reports and retrospective reviews.5,11,12,14,15 These cases represent a small fraction of the cats residing within the endemic region for Coccidioides species. Coccidioidomycosis is not currently reportable in veterinary species, allowing only for estimates of the impact of disease in our animal populations. Previous work has shown that the rates of infection and disease in dogs are similar to, or higher than, those seen in people; 16 however, the frequency of disease and subclinical infection in cats is unknown.
In this study population, the average age at diagnosis was 6.8 years, which is similar to cats in a previous retrospective study. 11 Two-thirds of the cats diagnosed with coccidioidomycosis lived exclusively indoors at the time of diagnosis, suggesting that being outdoors exposed to soil is not requisite for acquiring infection. This has also been noted in cats with blastomycosis,17,18 cryptococcus 19 and histoplasmosis. 20 Spores may be introduced to indoor cats through air conditioners, open doors and windows or on fomites that enter the residence.
Respiratory disease has been previously reported to be present in only 25% of cats with coccidiodomycosis. 11 In contrast, 40% of the cats in this population exhibited significant respiratory signs. Hilar lymphadenopathy was not frequently identified on the thoracic radiographs of this cohort, despite being a common finding in dogs with coccidioidomycosis.10,21,22 What is most notable about cats is that >60% of this population had disseminated disease at the time of diagnosis, and nearly half of all the cats in this study had dermal lesions as the primary feature leading to diagnosis of coccidioidomycosis. For all cats residing in the endemic region, even those exclusively indoors, coccidioidomycosis should be considered as a differential diagnosis for antibiotic-unresponsive dermal lesions, respiratory distress or cough, pleural effusion, ocular disease, chronic skeletal pain/lameness or paresis/paralysis. Coccidioidomycosis should be strongly considered in cats that present with chronic dermal lesions that do not respond to empirical treatment.
Clinicopathologic abnormalities were more common in this feline population compared to previous reports. 11 The most common abnormalities, hyperglobulinemia, neutrophilia and monocytosis, are supportive of a diagnosis of coccidioidomycosis based on canine disease.9,10 While the CBC and serum chemistries can aid in the diagnosis of coccidioidomycosis, the changes are not pathognomonic for coccidioidomycosis and the lack of inflammatory parameters cannot rule it out in cats.
Agar gel immunodiffusion uses doubling serial dilutions of serum to quantitate anti-coccidioidal antibody levels. The assay has been demonstrated to have high specificity for anti-coccidioidal antibodies, although there are no published validity studies of its use in cats. 23 In dogs, overlap in titers has been shown with clinical and subclinical disease and the magnitude of the titer does not consistently correlate with the presence or severity of illness. 16 Additionally, negative serology does not rule out coccidioidomycosis in humans or dogs.9,21,24 The overwhelming majority of the cats in this study that were tested did have anti-coccidioidal antibodies, which is similar to the only other review of feline coccidioidomycosis. 11 At this time, the rates of healthy seropositive cats, as well as seronegative cats with clinical coccidioidomycosis is unknown. A serosurvey of both healthy and sick cats, as has been undertaken in dogs, 16 would be needed to determine this information.
Confirmation of the presence of the fungus through histopathology, cytology and/or fungal culture is considered the gold standard for diagnosis; however, dogs are typically diagnosed through a combination of clinicopathology, diagnostic imaging and relevant clinical signs. 6 The large number of cats in this study with confirmed coccidioidomycosis is likely due to the frequency and location of dissemination. Extrapulmonary dissemination is rare in humans.2,25 It appears to be more common in dogs, with previous studies showing dissemination rates from 20% to 42% of cases.10,26 The most common site of dissemination in dogs is to the skeletal system and biopsy is infrequently pursued due to cost and invasiveness of the testing, as well as a strong radiographic indication of disease. However, the dermatologic dissemination seen in cats5,11,12,15 allows for easily obtained aspirates and biopsies. Also, the specimens for definitive diagnosis are usually obtained after lesions have failed to respond to antimicrobial or other empiric treatments.
Most cats in this study were treated with the oral antifungal fluconazole, as is typical for dogs in Arizona. 6 Fluconazole has the advantage of a high bioavailability and excellent tissue penetration.27,28 The tissue penetration makes fluconazole a particularly good choice due to the level of disseminated disease in multiple tissues seen in these cats. It is likely that fluconazole was chosen due to convenience, availability and familiarity, but it appears that most cats were treated successfully with it. Most of the cats in this study that were successfully treated required medication for over 1 year, and often several years. Recurrence of disease was evident in over a quarter of the cats in this cohort, although all but one of the cats was treated to clinical remission. While coccidioidomycosis in cats may require lengthy treatment with antifungal therapy and monitoring for recurrence, overall most cats will respond to treatment.
A limitation of this study is the retrospective nature of data collection. In order to identify the greatest number of cases, we purposefully approached cat only and specialty hospitals, which may have created a bias towards cats that were more ill at the time of diagnosis. However, the cases from the small animal practice did not have notable differences in the severity of illness. Owing to privacy concerns, we relied upon most of the clinics to perform their own database searches and we suspect that the true number of feline coccidioidomycosis cases is underrepresented as we could not control for search terms. We were also unable to control for radiographic interpretation, diagnostic laboratory and treatments. There is currently no information regarding the number, sex, lifestyle and breed of cats living in Tucson and Phoenix, making it impossible for us to identify risk factors for disease. A longitudinal study of coccidioidomycosis in cats would provide stronger evidence about the presentation of disease and outcome for cats residing in the endemic region.
Our cases indicate that cats experience a range of clinical disease as a result of infection with Coccidioides species. Extrapulmonary dissemination was evident in over half of the cats, often with spread to multiple organ systems. It is possible that the independent nature of cats and their ability to hide subtle indications of illness leads them to be diagnosed later in the course of disease. However, most of the cats in this study were reported to live exclusively indoors, suggesting that this population of cats did have owners who could monitor their cats daily for behavioral changes indicating illness. The extent of disease associated with coccidioidomycosis reported here and in previous work 11 may indicate a difference in host control of disease compared with dogs or humans.
Conclusions
Coccidioidomycosis presents a diagnostic challenge due to the overlap of clinical signs, clinicopathologic changes and radiographic abnormalities with other diseases. The results of our study indicate that clinical suspicion of coccidioidomycosis should be high in cats with dermatologic lesions or severe respiratory illness. Positive serology appears to be correlated with clinical illness, although biopsy and culture samples can provide a definitive diagnosis. Orally administered fluconazole was the treatment of choice in this population of cats and proved to be effective in most animals treated.
Footnotes
Acknowledgements
We are grateful to the following veterinary clinics for sharing their information with us: Desert Veterinary Medical Specialists (Gilbert, AZ), Feline, Ltd (Tucson, AZ), Orange Grove Animal Hospital (Tucson, AZ), Scottsdale Cat Hospital (Scottsdale, AZ) and the Veterinary Specialty Center of Tucson (Tucson, AZ).
Conflict of interest
One author (MK) is the owner of a veterinary hospital in Tucson that participated in this retrospective review. There are no other conflicts of interest to report.
Funding
This work was funded by the Valley Fever Center for Excellence Companion Care Fund, The University of Arizona, Tucson, AZ.
