Abstract
Coccidioidomycosis is a fungal disease caused by either Coccidioides immitis or Coccidioides posadasii. Anecdotal evidence suggests that camelids are particularly susceptible to this disease and that a relatively large percentage of pneumonias in these animals are caused by Coccidioides spp. In a search of 21 y (1992–2013) of records from the California Animal Health and Food Safety Laboratory, we found 79 cases of coccidioidomycosis diagnosed in camelids; 66 (84%) had pneumonia and 13 (16%) had lesions only in organs other than the lungs. The organs most frequently affected were lung (84%) and liver (78%). Coccidioides spp. were the cause of pneumonia in 66 of 362 (18%) camelid cases during the study period. The lesions in affected organs were multifocal-to-coalescing pyogranulomas, which in most cases were visible grossly. Ten of the 12 formalin-fixed, paraffin-embedded lung samples tested by a universal Coccidioides spp. PCR assay were positive (4 C. immitis, 2 C. posadasii); the species could not be determined in 4 of the 10 cases positive by PCR. Coccidioidomycosis is an important cause of pneumonia in camelids in California, and can be caused by either C. immitis or C. posadasii.
Introduction
Coccidioidomycosis, also called San Joaquin Valley fever, is a fungal disease that affects humans and other mammals, and is caused by Coccidioides immitis or Coccidioides posadasii. 14 Coccidioidomycosis is common in dogs, but cases have also been reported in other animal species, including llamas (Lama glama), alpacas (Vicugna pacos), non-human primates, cats, and horses. Hybrid strains of C. immitis and C. posadasii have also been described, indicating that exchange of genetic material occurs among them.10,15 C. immitis and C. posadasii cause almost identical disease, but they usually occur in different overlapping geographic regions. 2 Both species occur in dry, hot climates and sandy and silt soils. 4 However, C. immitis is found mostly in central and southern California and Mexico, whereas C. posadasii is found mainly in southern Arizona, western Texas, southern New Mexico, several Central and South American countries, and Australia.4,11 The reason for this difference in geographical distribution is not fully understood, although it has been suggested that soils in central and southern California and Mexico have high concentration of soluble salts, which favors the presence of C. immitis. 4
The spores of C. posadasii and C. immitis are commonly found in soil, and when rain falls, mycelia develop and release arthroconidia (the infectious form), which are disseminated in wind-blown dust. The most common mechanism of infection is by inhalation of arthroconidia, but direct inoculation of these structures through the skin occurs occasionally, causing cutaneous lesions that usually do not progress to systemic infection.1,13 Following inhalation and deposition within the lung, the arthroconidia transform into yeast forms that can be phagocytized by neutrophils and macrophages, within which they rapidly transition into immature spherules. 3 The latter are 10–20 µm diameter; when mature, spherules are typically 20–200 µm diameter and develop numerous 2–5 µm endospores. Mature spherules rupture, and the released endospores form either new spherules in tissue, or mycelia if they are released to a suitable environment.1,3
In mammals, lesions of coccidioidomycosis may be limited to the lungs, in which they vary from focal to multifocal. However, systemic lesions that result from hematogenous spread of the spherules from the lung to other organs may also occur. Rare cases have also been described in which systemic disease occured with no pulmonary lesions, and it has been postulated that these cases probably reflect resolution of the initial lung lesion rather than an extrapulmonary route of infection. Definitive evidence of this, however, is lacking.5,13
Scientific literature about coccidioidomycosis in South American camelids is scant.6,7,9 However, the few available references and anecdotal evidence suggest that these animals are particularly susceptible to coccidioidomycosis, and that a large percentage of pneumonias in camelids is caused by Coccidioides spp. 7 If this is the case, it would be relevant to take biosafety measures given that the culture of specimens containing Coccidioides spp. poses a risk for laboratory staff because of potential inhalation of arthroconidia. Herein, we further characterize the lesions of coccidioidomycosis in camelids, including the organ distribution of the lesions and the relative importance of both Coccidioides spp. as etiologic agents of pneumonia in these animal species.
Materials and methods
We searched the records of the California Animal Health and Food Safety (CAHFS) laboratory system for cases of South American camelids submitted for autopsy between 1992 and 2013 that had a diagnosis of pneumonia and/or coccidioidomycosis. In most cases, sections of multiple organs including liver, kidney, spleen, small and large intestine, pancreas, kidney, heart, lung, adrenal gland, trachea, brain, peripheral nerves, and skeletal muscle had been examined by the pathologist in charge of the case. Based on the information obtained from the autopsy reports, the cases of coccidioidomycosis were classified according to the number of organs affected. Cases in which more than one organ was affected were classified as disseminated coccidioidomycosis.
Archived histologic sections (hematoxylin and eosin, Giemsa, periodic acid–Schiff [PAS], and Gomori methenamine silver [GMS]) from 42 cases (21 alpacas, 21 llamas) were reviewed by one of the authors of this paper (JA Fernandez). Paraffin-embedded sections of lung from 7 cases in which pulmonary lesions were found (4 llamas, 3 alpacas), and sections of liver from 5 cases with hepatic lesions but without lung involvement (2 llamas, 3 alpacas), were initially tested by a universal Coccidioides spp. PCR capable of detecting both C. immitis and C. posadasii, but unable to differentiate between these 2 species. The specimens that were positive in this PCR were further processed to determine the Coccidioides species present, using a multi-sequence alignment strategy to target an area of homology within C. immitis and C. posadasii sequences (Vector NTI software, Thermo Fisher Scientific, Waltham, MA). Briefly, DNA was extracted from two 25-µm scrolls of formalin-fixed, paraffin-embedded (FFPE) tissue with xylene and ethanol. Lysis was achieved with proteinase K. DNA was eluted in diethyl pyrocarbonate–treated water. Nucleic acids were purified from the tissue lysate (DNeasy tissue kit, Qiagen, Valencia, CA) according to the manufacturer’s instructions. DNA was finally eluted in diethyl pyrocarbonate–treated water.
Quantitative real-time PCR (qPCR) was utilized for detection and speciation of Coccidioides spp. Three different qPCR assays (2 specific primers and 1 internal fluorescence-labeled probe) were designed (Primer Express software v.3.0.1, Applied Biosystems, Foster City, CA), as follows: 1) a universal assay that targets the ITS1 of the 18S gene (GenBank JX051631.1) at the 450–530 bp region; and 2) two species-specific assays for C. immitis and C. posadasii targeting the F-box gene (GenBank XM_001246343 and XM_003067910, respectively). The C. immitis– and C. posadasii–specific assays shared the same forward and reverse primers, (5’-TTTACGCCGTAGCCTTTGATG-3’) and (5’-CGT-GTGGCCTTGCAGTATAGC-3’), respectively, but a single nucleotide polymorphism on the minor groove binding probes allowed their differentiation. The probes were designed to hybridize to C. immitis (5’-6FAM-ATTGTCCAGTATGAG-GAT-MGB-3’) and C. posadasii (5’-6FAM-ATTGTC-CAGAATGAGGAT-MGB-3’) sequences. 12 Each 12-μL reaction contained 2 μL of DNA, 2× TaqMan universal master mix (Applied Biosystems), 2 nmol of each primer, and 400 pmol of the probe. Thermocycling conditions consisted of 50°C for 2 min, 95°C for 10 min, and 40 cycles at 95°C for 15 s, followed by 60°C for 1 min. Amplification, data acquisition, and data analysis were performed (7900HT Fast real-time PCR system, Applied Biosystems). Samples were considered positive if they had a quantification cycle (Cq) value of <40 and characteristic amplification plots. Three water negative controls and a sequence-confirmed positive DNA control were included in each run. The analytic sensitivity for each assay was a single copy of the targeted gene.
The amplification efficiency (E) of all assays was calculated from the slope of a standard curve generated on a 10-fold dilution in triplicate for every DNA sample, using the formula E = 10(-1/slope) – 1. In order to obtain accurate and reproducible results, all assays were assumed to have an efficiency of >95%. Based on the amplification efficiencies, detection limits were ~10 copies of DNA per reaction. The coefficient of variability of the qPCR determined for 10 replicates was ≤15%.
Results
A total of 2,101 camelids (1,018 llamas, 1,083 alpacas) were received for autopsy during the study period (1992–2013) in 3 of the CAHFS laboratories (Tulare, Davis, and San Bernardino). The ages of animals submitted ranged from neonate to 20 y, with an average of 7.4 y (SD ± 5.0). All of the cases originated from California. The average number of cases per year was 100, with a peak of 102 cases in 2006.
Of the 2,101 camelids studied, 362 animals (17%) had pneumonia as the cause of death, whereas 1,739 (83%) had died of causes other than pneumonia (data not shown). The most prevalent etiologic class was bacterial (36%), followed by aspiration (23%), fungal (20%), and viral (2%). The etiology of the pneumonia was not determined in 19% of the cases. In the viral pneumonia cases, the diagnosis was established based on gross and microscopic lesions, although a specific etiology could not be determined, thus the viral etiology remains presumptive (Table 1).
Agents isolated from 362 cases of pneumonia in alpacas and llamas received in the California Animal Health and Food Safety laboratory system, 1992–2013.

Numbers in parentheses are percentages.
Arcanobacterium spp., Corynebacterium spp., Salmonella spp., Pasteurella spp., Staphylococcus spp., Bacillus spp., and Listeria spp.
Aspergillus spp., Rhizopus spp., and Cryptococcus spp.
Presumptive.
Coccidioidomycosis was the cause of death in 79 (4%) of the 2,101 camelids received for autopsy during the study period (Table 2). Of the 79 animals with coccidioidomycosis, 66 (84%) had pneumonia, whereas in 13 (16%), pneumonia was not observed. Of the 66 cases in which pneumonia was observed, 11 (17%) had only pneumonia, whereas 55 (83%) had pneumonia and also lesions in at least one other organ (Table 2). The target organs affected in cases of disseminated coccidioidomycosis were, in decreasing order of prevalence, lung (84%), liver (78%), lymph nodes (54%), spleen and kidney (47% each), heart (34%), skin (9%), and skeletal muscle (5%). Of the 13 cases of coccidioidomycosis in which pneumonia was not observed (Table 2), 5 had lesions in only 1 organ, including liver (2), lymph node (1), and brain (2), and 8 of these animals had disseminated coccidioidomycosis. Of the 362 total pneumonia cases in camelids, 66 (18%) were diagnosed as coccidioidomycosis (42 llamas [25%] and 24 alpacas [13%]; Table 3).
Distribution of lesions and lung involvement in llamas and alpacas (n = 79) with coccidioidomycosis received in the California Animal Health and Food Safety laboratory system, 1992–2013.
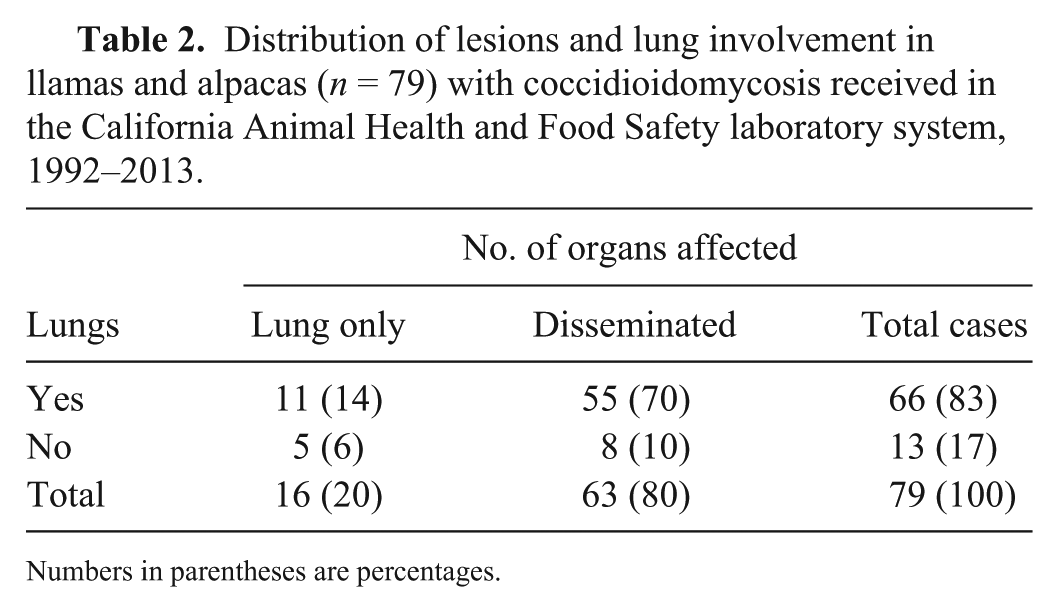
Numbers in parentheses are percentages.
Prevalence of coccidioidomycotic pneumonia in llamas and alpacas received in the California Animal Health and Food Safety laboratory system, 1992–2013.

Numbers in parentheses are percentages.
The gross anatomic lesions of coccidioidomycosis were similar in all animals and organs studied but varied in extent and severity, and consisted of multifocal-to-coalescing, yellow-white pyogranulomatous lesions randomly and roughly evenly distributed, and of variable sizes (0.2–10 cm diameter) with no predilection for any area of the organs affected (Figs. 1, 2). No predilection for right or left side was observed in lungs, kidneys, or liver.
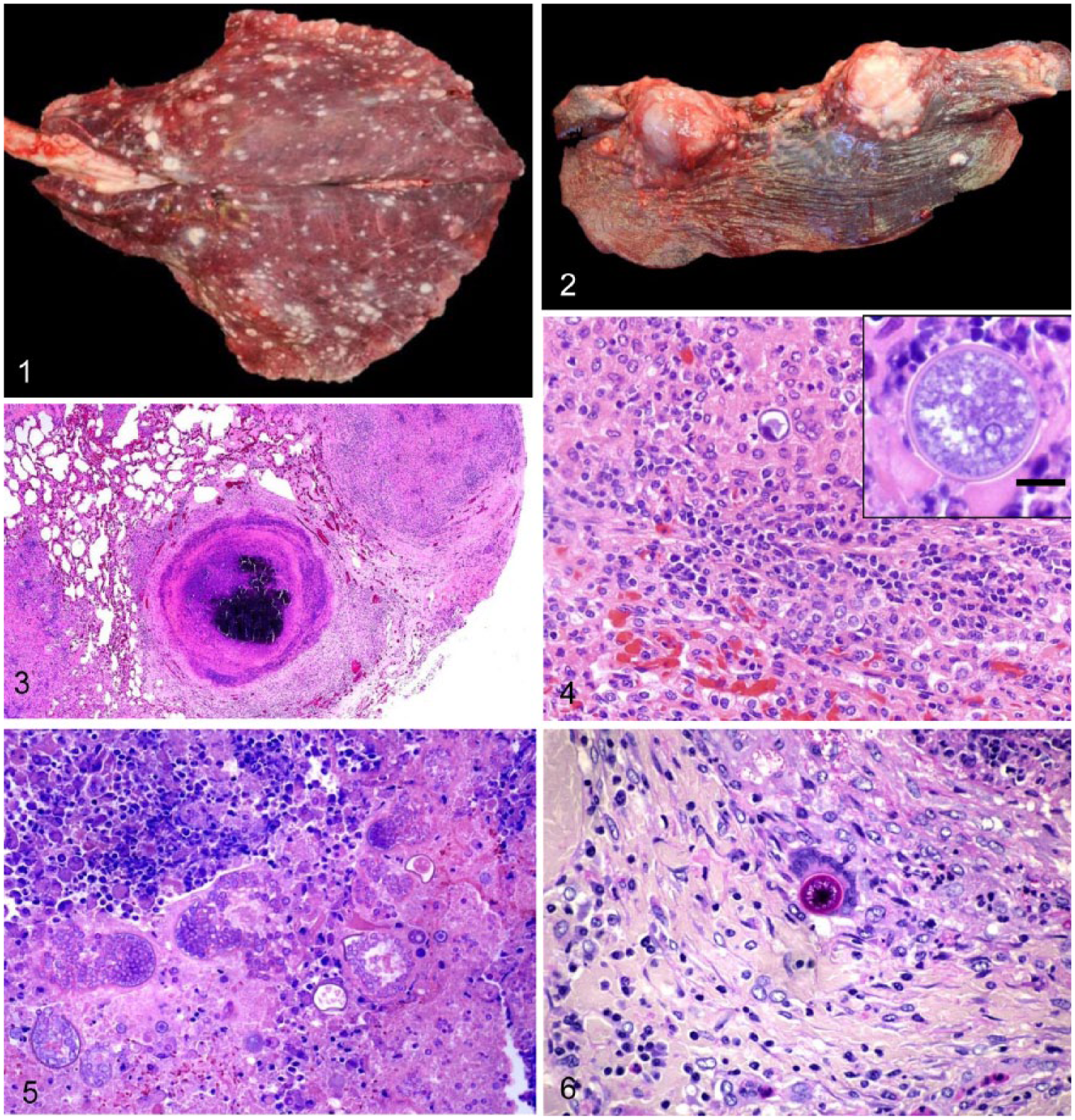
Coccidioidomycosis in South American camelids.
Microscopically, lesions of coccidioidomycosis were also similar, regardless of the organ and Coccidioides species detected, and consisted of multifocal- to-coalescing pyogranulomas, with central caseous necrosis and a mineralized core (Fig. 3), surrounded by a variable rim of tightly grouped neutrophils, lymphocytes, plasma cells, and macrophages.
Foreign-body and Langhans-type giant cells and epithelioid macrophages were observed in the periphery of most pyogranulomas (Fig. 4). Multifocally and randomly distributed within the pyogranulomas were variable numbers of round, 20–100 μm fungal spherules (sporangia) with a 4–5 μm thick refractile and hyaline double wall (Figs. 4–6). Sporangia contained flocculent basophilic material and, less frequently, numerous 5–7 µm endospores. Both sporangia and endospores stained positively with Giemsa, PAS, and GMS. Occasionally, no spherules were observed in some pyogranulomas. In those cases, the diagnosis was confirmed by finding spherules in other pyogranulomas of the same organ. All cases diagnosed as coccidioidomycosis had spherules in at least one granuloma. Most affected lungs had alveolar edema and congestion surrounding the areas of pyogranulomatous inflammation.
Ten of the 12 FFPE tissues tested were positive for Coccidioides in the universal Coccidioides spp. PCR assay. Of these 10 positive samples, 4 were positive for C. immitis and 2 were positive for C. posadasii; the signal was weak in the other 4, and the species could not be determined. C. immitis was detected in 2 samples of lung and in 2 samples of liver; C. posadasii was detected in 2 samples of liver, but not in lung of any of the animals tested.
Discussion
Almost one-fifth (18%) of all of the camelid pneumonias studied were caused by Coccidioides spp., which confirms that this microorganism is a significant cause of pneumonia in llamas and alpacas in California. Based on similar numbers of submissions for llamas and alpacas, coccidioidomycotic pneumonia was more common in llamas (42 cases, 25%) than in alpacas (24 cases, 13%). This may reflect differences in species susceptibility, although the reasons are unknown.7,6,9 The prevalence of coccidioidomycosis in other animal species (e.g., humans and mice) depends not only on environmental conditions, but also on other risk factors including genetic susceptibility, level of exposure, other diseases, and immunosuppression.8,13 It is possible that some of these factors affect the susceptibility of South American camelids to coccidioidomycosis.
Disseminated coccidioidomycosis was more prevalent than the purely pulmonary form (80% vs. 20%; Table 2), in agreement with previous reports of this disease in llamas. 7 Also, 84% of our coccidioidomycosis cases had pneumonia, whereas only 16% did not have pneumonia. The target organs in decreasing order of prevalence were lung, liver, lymph nodes, spleen and kidney, heart, skin, and skeletal muscle. Our results are consistent with previous finding in other species,1,13 confirming that the respiratory route is the main portal of entry for Coccidioides spp. in camelids, given that pneumonia was the lesion most frequently detected in cases of coccidioidomycosis. Given that this was a retrospective study based on autopsy reports prepared by several pathologists over 21 y, we cannot rule out that small pneumonic lesions were overlooked in some of the cases and not reported. A portal of entry for these infections other than the respiratory route is also possible. Maternofetal transmission has been demonstrated in a case of alpaca coccidioidomycotic abortion. 6 In other species, horizontal (animal-to-animal) and vertical (maternofetal) routes were also described, albeit infrequently. 4
It is usually assumed that most cases of coccidioidomycosis in California are produced by C. immitis (authors’ unpublished observation). However, in our study we also found a low percentage of animals infected by C. posadasii. These results, coupled with a previously reported case of abortion caused by C. posadasii in a llama in California 6 suggest that infection by C. posadasii is more prevalent in South American camelids of California than previously thought. Because no morphologic differences were observed in lesions produced by either of the Coccidioides species, gross or microscopic examination cannot define the species involved, and molecular methods are required. In our study, the only fungal structures detected were spherules. Hyphae have been described in human coccidioidomycosis cases. 13 Occasionally, no spherules were observed in some pyogranulomas. In those cases, the diagnosis was confirmed by finding spherules in other pyogranulomas of the same organ. In these cases, it was assumed that the lesions with no spherules were burnt out lesions from which the fungal elements were gone by the time of autopsy, as described previously. 5
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the California Animal Health and Food Safety Laboratory, School of Veterinary Medicine, University of California, Davis.
