Abstract
A 3-mo-old male llama was examined because of a 4-wk history of lethargy and ill thrift. Clinical examination revealed subcutaneous masses in the left prescapular and right inguinal regions, mild ataxia, a slight head tilt to the right, and right ear droop. The cria died before clinical workup was complete. At autopsy, there was generalized lymphadenomegaly, a hepatic nodule, a midbrain mass causing rostral compression of the cerebellum, and internal hydrocephalus. Microscopic findings included pyogranulomatous lymphadenitis, meningoencephalitis, hepatitis, and bronchopneumonia. Intralesional fungal spherules, most consistent with Coccidioides spp., were identified in the lymph nodes, lung, and brain. Fungal culture, single-nucleotide variation genotyping real-time PCR, and DNA sequencing confirmed Coccidioides posadasii. The dam of the cria was native to Arizona and had been moved to Missouri ~2.5 y previously. Agar gel immunodiffusion assay of the herd revealed that only the dam was positive for Coccidioides spp.; 6 herdmates were negative. Computed tomography of the dam revealed multiple nodules within the lungs and liver, which were presumed to be an active coccidioidomycosis infection. This case of systemic coccidioidomycosis in a llama native to Missouri was presumably acquired by vertical transmission from the dam.
Keywords
Coccidioidomycosis is a rare and highly infectious fungal disease that can affect many mammals, including humans. The etiologic agents, Coccidioides posadasii and C. immitis, have dimorphic, saprobic, and parasitic life cycles. The saprobic life cycle of Coccidioides spp. occurs in warm, arid to semi-arid environments, typically with alkaline soil, and the organism can remain viable for months to years. 6 In these environments, the mycelium asexually forms alternating arthroconidia, the infective agent. The arthroconidia are aerosolized following physical disturbance of the soil. 12
Infection in mammals most commonly occurs by inhalation of arthroconidia, but other routes of infection include cutaneous inoculation and, rarely, transplacental transmission. 7 The parasitic life cycle is initiated after inhalation of the arthroconidia followed by rapid maturation into spherules and eventually sporangia containing numerous endospores. Rupture of the sporangium leads to dissemination of the endospores throughout the body or return to the environment in which the endospores form mycelia, completing the life cycle. In some instances, infections can become latent and be reactivated during immunosuppressed situations. 3
Previous studies and anecdotal evidence have suggested that South American camelids are particularly susceptible to coccidioidomycosis. 9 In these species, affected organs typically develop multifocal-to-coalescing pyogranulomas. 9 The distribution of lesions in llamas and alpacas most commonly includes disseminated disease with and without pulmonary involvement. 9 There are a few reports of abortions in domestic animals or transplacental infections. 7 We describe here a case of systemic coccidioidomycosis in a Missouri-native llama cria with presumed vertical infection.
A 3-mo-old intact male llama was presented to the University of Missouri Veterinary Health Center (Columbia, MO) because of a 4-wk history of lethargy and ill thrift. The cria was small for his age and not gaining weight appropriately. The cria became dull and anorexic and developed clear bilateral ocular discharge a few days prior to presentation. During loading into the trailer, the cria fell and had difficulty rising. The 19-y-old dam of the cria was reported to be healthy. The dam originated from Arizona and had been transported to Missouri 2.5 y previously. The owner also owned the dam’s previous cria and reported that that animal was doing well. There was no history of travel or recent additions to the herd in the preceding months.
On physical examination, the cria was quiet, alert, and responsive. There was a slight head tilt to the right and the right pinna drooped. Bilateral serous ocular discharge was noted. Soft, freely moveable masses were palpated in the regions of the left prescapular lymph node and right inguinal lymph node; the remaining lymph nodes palpated within normal limits. The cria was mildly ataxic. Strength, proprioception, and reflexes were normal. The remainder of the physical exam was unremarkable.
A complete blood count, plasma biochemical profile, aural exam, ultrasound of the prescapular mass, and cytology of the prescapular mass were performed. The complete blood count revealed leukocytosis (59.5 × 109/L, reference interval [RI]: 8–21.4 × 109/L) characterized by neutrophilia (46.1 × 109/L, RI: 4.7–14.7 × 109/L) with band neutrophils (2.7 × 109/L, RI: < 0.2 × 109/L), lymphocytosis (5.1 × 109/L, RI: 0.7–4.8 × 109/L), and monocytosis (5.6 × 109/L, RI: < 1.0 × 109/L). 15 Anemia was also present (7.5 × 1012/L, RI: 10.1–17.3 × 1012/L) with decreased hematocrit (0.25 L/L, RI:0. 27–0.45)L/L. 15 The plasma biochemical profile revealed hyperproteinemia (93 g/L, RI: 53–73 g/L) characterized by normal albumin (32 g/L, RI: 30–42 g/L) and hyperglobulinemia (61 g/L, RI: 23–31 g/L). 15 Ultrasonography of the pre-scapular mass revealed a homogeneous soft tissue opacity determined to be likely lymphoid or fatty tissue. Cytology of a fine-needle aspirate revealed reactive fibroplasia with moderate inflammation. The inflammatory cells were primarily nondegenerate neutrophils and small-to-medium lymphocytes. No organisms were found.
The cria died before the clinical workup was complete and, thus, was submitted for autopsy. Many lymph nodes, including the left prescapular, perihilar, right inguinal, and iliac lymph nodes, were severely enlarged (up to 12 × 10 × 2 cm), firm, and on transections were diffusely tan-yellow with a peripheral, thin, circumferential band of yellow tissue (Fig. 1). Approximately 30% of the cranioventral lung lobes were diffusely dark red-purple and mildly rubbery on palpation. The liver was diffusely dark-red with a single, 1-cm diameter, yellow-white nodule in the left lobe. Within the brain, the lateral ventricles were markedly enlarged, contained clear, non-viscous fluid, and compressed the surrounding cerebral cortex. There was a 2 × 3 × 3 cm, soft, yellow-tan mass in the midbrain at the level of the fourth ventricle, compressing the rostral aspect of the cerebellum (Fig. 2).
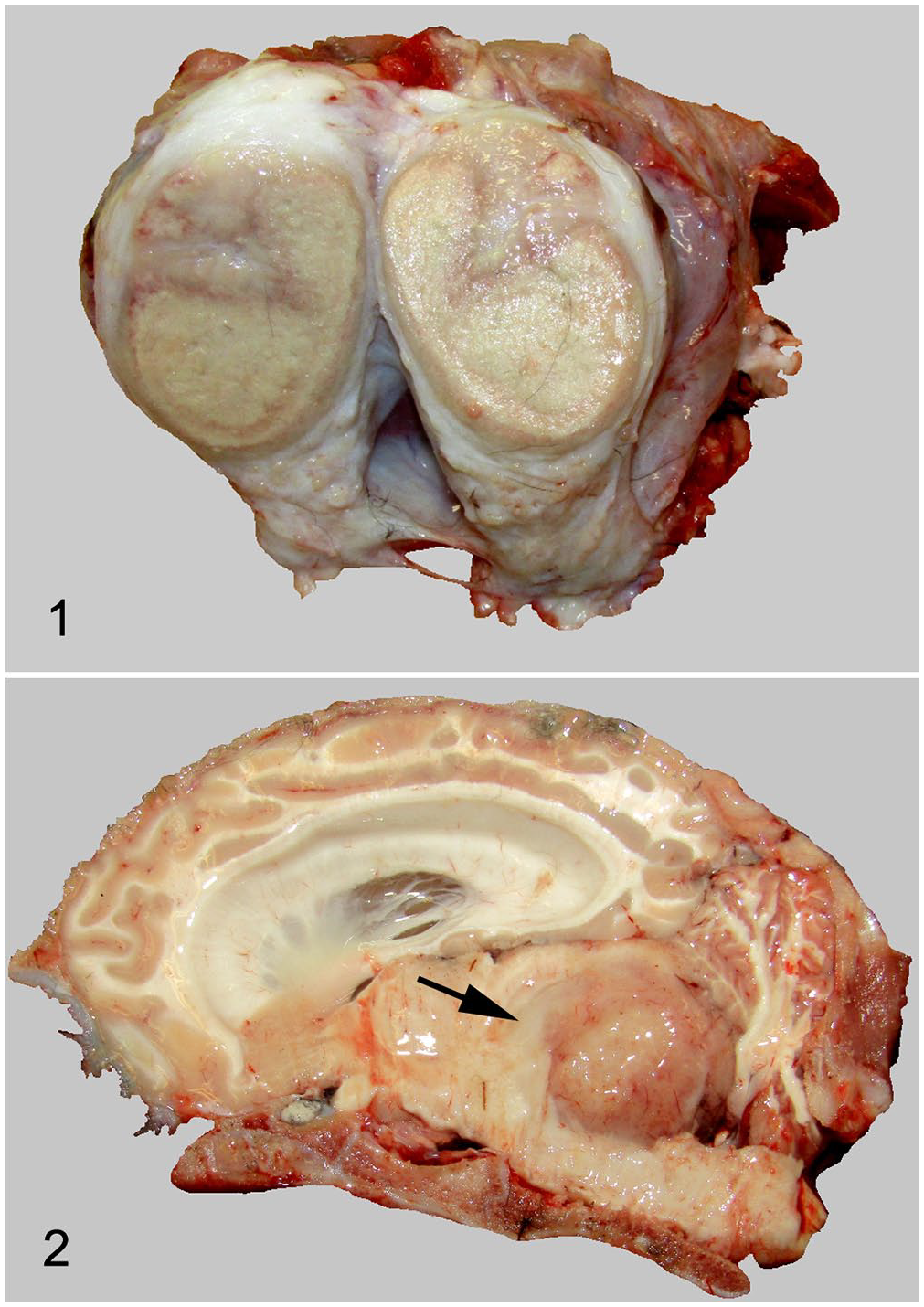
Systemic coccidioidomycosis in a llama.
Microscopically, the mass in the midbrain consisted of inflammatory cells, effacing and infiltrating the surrounding parenchyma. The core of the mass was multifocally necrotic, occasionally with dystrophic mineralization. The inflammatory cells were primarily degenerate neutrophils in the necrotic center; there were marked numbers of macrophages, neutrophils, epithelioid macrophages, multinucleate cells, and lymphocytes in the outer layer of fibrous connective tissue. Throughout the mass were large (20–40 µm diameter) uninucleate spherules (Fig. 3). Similar microscopic lesions were found in multiple lymph nodes (mediastinal, mesenteric, and inguinal), and in the lung. The liver nodule had granulomatous inflammation, but no organisms were found. The intestines had moderate numbers of coccidia in various developmental stages.
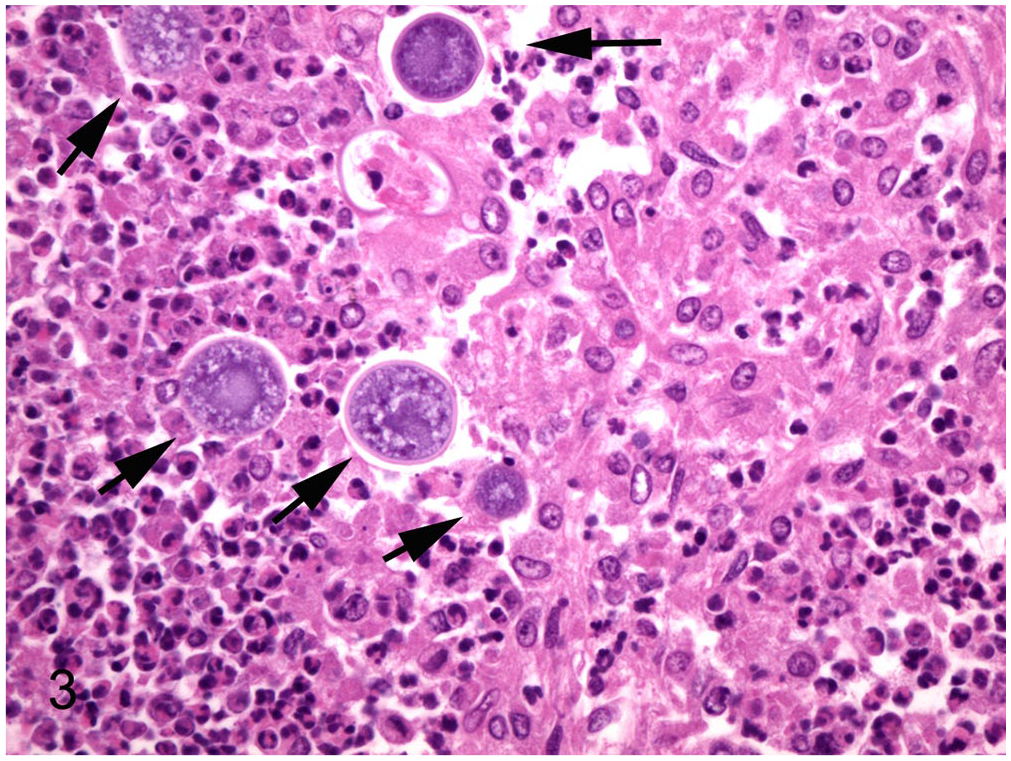
Left prescapular lymph node. Severe pyogranulomatous lymphadenitis with intralesional 20–40 µm fungal spherules (arrows). H&E. 400×.
The affected prescapular lymph node was ground and plated onto trypticase soy agar with 5% sheep blood, Sabouraud dextrose agar, and mycobiotic agar. Growth was noted on both blood agar (incubated at 35°C) and Sabouraud dextrose agar (incubated at 23°C). Both plates produced mycelia with alternating barrel-shaped arthroconidia that are characteristic of Coccidioides spp. For the fungal isolate, DNA sequencing of 16S rRNA was performed by following procedures reported previously. 14 Briefly, the primers were ITS1: 5′-TCCGTAGGTGAACCTGCGG-3′ and ITS4: 5′-TCCTCCGCTTATTGATATGC-3′. The PCR conditions were 95°C for 2 min; 30 cycles of 95°C for 30 s, 57°C for 30 s, and 72°C for 30 s; and final extension at 72°C for 6 min. The resulting amplicon was purified by using spin chromatography (Qiagen) and sequenced with amplification primers at the University of Missouri DNA Core Facility (Columbia, MO). The resulting 580-bp sequence was compared with sequences in GenBank by using BLAST (www.ncbi.nlm.nih.gov/Blast.cgi) and found to be 99.3% identical with C. posadasii and 98.9% identical with C. immitis.
A single-nucleotide variation (SNV) genotyping real-time PCR (rtPCR) was further employed to differentiate C. posadasii and C. immitis. 16 The forward (CF: 5′-TTTACGCCGTAGCCTTTGATG-3′) and reverse (CR: 5′-CGTGTGGCCTTGCAGTATAGC-3′) primers targeted a 113-bp fragment of the F-box gene of C. posadasii and C. immitis. Locked nucleic acid (LNA) probes Cp-P (5′-6-FAM-ATT +GT+C C+AG +tA+T G+AG GAT-3′-IABkFQ) for C. posadasii and Ci-P (5′-6-FAM-ATT +GT+C C+AG +aA+T G+AG GAT-3′-IABkFQ) for C. immitis have one nucleotide difference as shown in the lowercase letter in the probe sequences (the letters after each “+” are substituted with LNA). DNA was extracted from paraffin-embedded tissue (DNeasy blood and tissue kit; Qiagen). Amplification was performed (7500 fast real-time PCR system; Applied Biosystems) with the thermal cycling program of 50°C for 2 min, 95°C for 10 min, and 40 cycles of 95°C for 10 s, and 60°C for 1 min. The SNV genotyping rtPCR identified the sample as positive to the C. posadasii probe (Cp-P; cycle threshold [Ct] = 35.7) but negative to the C. immitis probe (Ci-P). To confirm the result of SNV genotyping rtPCR, the amplicon generated with the primers CF and CR was subjected to Sanger sequencing from both directions with CF and CR, respectively, at the University of Missouri DNA Core Facility. The resulting sequences were aligned with the reference sequences of XM_003067563 (C. posadasii) and XM_001245757 (C. immitis) in GenBank, which confirmed the isolate as C. posadasii.
Coccidioidomycosis in South American camelids is reported to appear grossly as yellow-white nodules of pyogranulomatous inflammation affecting the lungs, liver, lymph nodes, spleen, kidneys, heart, skin, skeletal muscles, and eye.5,9 Histologically, lesions are described as multifocal-to-coalescing pyogranulomas with necrotic centers, dystrophic mineralization, and variable numbers of coccidioidal organisms. The tissue forms of Coccidioides spp. include immature spherules, mature spherules (sporangia), and released endospores. Spherules and sporangia are 20–100 µm in diameter and have 4–5 µm thick refractile walls. Spherules can contain flocculent, basophilic material or, rarely, 5–7 µm diameter endospores. 9 Our case had disseminated disease with pyogranulomatous inflammation in the brain, lung, liver, and multiple lymph nodes. Coccidioidal meningoencephalitis is a rare manifestation but has been described in humans. 10
After diagnosing coccidioidomycosis in the cria, a serosurvey using an agar gel immunodiffusion assay was performed on 7 herdmates exposed to the cria. The dam of the cria was the only seropositive animal in the herd. At the time of the survey, the dam was asymptomatic. Of the 6 animals that were seronegative for Coccidioides spp., one was an offspring of the seropositive dam. Latency of infection in the dam was supported by the travel history of the dam, which was native to Arizona and had been transported to Missouri ~2.5 y prior to the presentation of the cria.
Physical examination of the seropositive dam revealed no overt abnormalities. To determine whether the dam had internal organ abnormalities consistent with coccidioidomycosis, computed tomography of the chest and abdomen was performed, which revealed multiple clusters of small, mineral attenuating nodules throughout the pulmonary parenchyma. The nodules had a predilection for the peribronchial regions. Similar mineral attenuating nodules were also present in the tracheobronchial lymph nodes, along the right lateral thoracic body wall, within the soft tissues at the level of the thoracic inlet, liver, and colon. These identified nodules were presumed to represent a latent infection with Coccidioides spp.
The dam died several months later at the farm, but no autopsy was performed because the authors were not informed of the death until a later date. Given that the environment in Missouri is not conducive to the Coccidioides spp. saprobic life cycle and given the serologic and computed tomographic evidence of disease in the dam, an intrauterine or neonatal infection is suspected to be the mode of transmission to the cria.
Multiple studies have demonstrated vertical transmission of Coccidioides spp. in domestic animals, which results in placentitis, disseminated disease in the fetus, and abortion.7,11,17 Abortion with disseminated infection in an alpaca fetus with C. posadasii has been reported, although it was thought to be an infrequent presentation. 7 In humans, pregnancy is considered a risk factor for coccidioidomycosis. 1 Transplacental transmission to the human fetus is rare because of the large size of spherules and granulomatous inflammation prohibiting transmission, unless there is significant vascular rupture or manipulation of the placenta during pregnancy.1,2 Many human cases of coccidioidal placentitis describe no evidence of fetal infection. 1 Neonatal infection may occur via aspiration of infected amniotic fluid or vaginal secretions during delivery.1–4
Without overt clinical signs, an antemortem diagnosis of coccidioidomycosis can be challenging. Fungal culture of infected fluids and tissues can provide a definitive diagnosis, but limitations to this method include potential false-negatives, longer turnaround times, and the risk of infection for laboratory personnel. In humans, antigen enzyme immunoassay and PCR assays on fluids and tissues are available to detect the antigen.8,13
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
