Abstract
Polyethylene glycols (PEGs) are amphiphilic polymers that are used extensively in consumer products and PEGylated biotherapeutics. Although PEGs are considered biologically inert with a low toxicity, anti-PEG antibodies have been detected in patients receiving treatment with PEGylated biotherapeutics as well as in healthy individuals. Despite continual exposure in daily life, the prevalence of PEGs within the general population remains unclear. This study aimed to evaluate a healthy population for the presence of PEGs. A validated enzyme-linked immunosorbent assay (ELISA) platform was used to analyze 200 plasma samples from healthy adults for the presence of PEGs. High-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) was also used to analyze the samples for the presence of four PEG analytes: PEG3350; PEG4000; PEG10000; and PEG2ru20K COOH, a 20 kDa PEG used to modify therapeutic proteins. The detection limit for PEG3350 calculated by extrapolation with linear regression was assumed to be 3.60 ng/mL. PEG levels above the lower limit of quantification (LLOQ) (21.6 ng/mL) were detected in 41 plasma samples, range 22.1-43.9 ng/mL, using the ELISA platform. None of the samples were found to contain detectable levels of PEG4000, PEG10000, or PEG2ru20K COOH. Following extrapolation, PEG3350 levels above the LLOQ (3.60 ng/mL) were detected in 154 plasma samples, range 3.69-46.0 ng/mL. Using an ELISA platform and HPLC-MS/MS analysis, PEGs were detected in the plasma of 77.4% of healthy adults. The findings are suggestive that expanding worldwide exposure to PEGs may have resulted in a much higher incidence of detectable plasma burdens of PEG than was previously realized.
Introduction
Polyethylene glycols (PEGs), also known as macrogols, are amphiphilic polymers with a repetitive chemical structure, general formula H–(O–CH2–CH2)n–OH, that can be produced in various chain lengths and molecular weights, are chemically inert, and have a low toxicity.1-4 PEGs can also exist in different conformations and carry different functional end-groups to generate PEG derivatives, such as PEG ethers, PEG fatty acid esters, and PEG soy sterols. Polyoxyethylene sorbitan esters (also known as polysorbates), such as Tween 80, are also PEG derivatives and are frequently used as drug excipients.5,6
PEGs and PEG derivatives are used extensively in cosmetics as surfactants, skin conditioning agents, emulsion stabilizers, and humectants.5,7-11 Products containing these polymers include toothpaste, body lotions, deodorants, and makeup as well as bath and hair-care products. 5 PEGs and PEG derivatives also have numerous applications in the pharmaceutical industry as the basis for ointments, as tablet/pill binders, and in capsules, laxatives, and suppositories.1,5 PEGs of different molecular weights are used for different applications; for example, PEGs 600-3350 are typically used in toothpaste, while PEGs 3350-4000 are used in laxatives.1,2 PEGs can also be conjugated to biotherapeutic agents such as proteins or aptamers, through a process known as PEGylation, to enhance the agent’s pharmacological and pharmaceutical properties by increasing half-life, tolerability, and stability. 12 Both linear and branched-chain PEG reagents, typically ranging from 5 to 60 kDa in size, and PEG derivatives with different linker chemistries have been used to chemically modify biotherapeutic agents.12,13
Although PEGs are considered biologically inert, the development of anti-PEG antibodies has been observed in animal models and in patients receiving treatment with PEGylated biotherapeutics.14-19 Anti-PEG antibodies are directed against either the repeated ethane-1,2-diol backbone of the polymer or its terminal end-groups. 13 The presence of anti-PEG antibodies has been correlated with a loss of therapeutic efficacy and an increased risk of adverse immune reactions in some patients receiving treatment with PEGylated biotherapeutics.14,15,17
Anti-PEG antibodies have also been detected in healthy individuals as well as in patients who have not previously received treatment with PEGylated biotherapeutics.15,16,20,21 The development of anti-PEG antibodies within these populations is possibly caused by exposure to PEGs and PEG derivatives within consumer products.15,20 Although PEGs have been detected in the placentas of some pregnant women, 22 the prevalence of PEGs within the general population remains unclear. The aim of this study was to evaluate the presence of different molecular weight PEGs in a healthy population.
Methods
Human Plasma Samples
Plasma samples from 200 healthy adult donors were utilized in the study, of which 120 were pre-selected by the study sponsor and 80 were commercially purchased from BioIVT, NY, USA. Of the 120 samples that were pre-selected, 20 plasma donors were from Austria, and 20 plasma donors each were from Ammon (ID), Elkhart (IN), Fargo (ND), Lakeland (FL), and Laredo (TX) in the USA. No plasma samples from pediatric donors were included in the study. Written informed consent was obtained from all individuals who provided plasma samples.
Detection of PEGs
ELISA-Based Approach
Plasma samples were analyzed using a validated PEGylated protein enzyme-linked immunosorbent assay (ELISA) kit purchased from Enzo Life Sciences, Lausen, Switzerland. The assay was a competitive ELISA that used a monoclonal antibody specific to the repeated ethane-1,2-diol units that form the backbone of PEGs. No polyethylene derivatives, such as Tween, were used in the assay. Details of the sensitivity of the ELISA are provided in the Supplemental Materials. The assay was validated for the detection of PEGs in human plasma in terms of precision (inter- and intra-assay variability), selectivity, linearity, and robustness. Details of the validation procedures are given in the Supplemental Materials.
Plasma samples were tested in duplicate at a minimal required dilution of 1:5. A 20 kDa branched PEG (Nektar Therapeutics, CA, USA) containing a carboxyl group (COOH) was spiked at different concentrations into PEG-negative human plasma and used as a reference standard. Negative controls were pools of human plasma that had tested negative for PEG antigens.
Data were analyzed using Gen5 software, version 3.04 (BioTek Instruments, Inc., VT, USA). Results were considered valid if the deviation (inter-dilution variance) to the mean of the duplicate measurement was ≤15%. The lower limit of quantification (LLOQ) was 21.6 ng/mL, and the upper limit of quantification was 300 ng/mL.
HPLC-MS/MS-Based Approach
High-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) was used to investigate the presence of four PEG analytes in the plasma samples: PEG3350 (average molar mass: 3350 g/mol); PEG4000 (molar mass: 3500-4500 g/mol); PEG10000 (average molar mass: 10676 g/mol); and PEG2ru20K COOH (molar mass: 20000 g/mol). The presence of all four analytes was evaluated in samples that had not undergone acidic cleavage. The presence of PEG2ru20K COOH was also evaluated in samples that had undergone acidic cleavage.
Samples were injected into a HPLC-MS/MS system (QTRAP® 6500, AB SCIEX, MA, USA/Canada), sample volume 50 μL. An Onyx™ monolithic C18 column (100 × 4.6 mm; Phenomenex, CA, USA) was used for separation. Chromatographic separations were achieved at a column temperature of 40°C and a flow rate of 1 mL/min. The column was eluted with a linear gradient of 1% formic acid in 55% methanol to 1% formic acid in 85% methanol in 9.5 min. Retention times were 4.9, 5.4, 7.6, and 8.5 min for PEG3350, PEG4000, PEG10000, and PEG2ru20K COOH, respectively.
The analysis was run in multiple reaction monitoring mode with positive polarity: m/z 675 → 124 for PEG3350; m/z 675 → 124 for PEG4000; a sum of three transitions m/z 900/950/1000 → 124 for PEG10000; and a sum of three transitions m/z 950/1000/1050 → 124 for PEG2ru20K COOH. A sum of three transitions m/z 750/800/850 → 49 was used for D-PEG-22K, the internal standard.
Analytical reference standards were PEG3350, PEG4000, PEG10000, and PEG2ru20K COOH. D-PEG-22K was used as a reference standard for the internal standard. Calibration standards were spiked into commercially available pooled human plasma (Trina Bioreactives, Switzerland; BioIVT, NY, USA) at eight different concentrations: 20, 40, 70, 100, 250, 500, 750, and 1000 ng/mL. Quality control samples were spiked into commercially available pooled human plasma at four different concentrations: 20, 60, 500, and 800 ng/mL. Calibration standards and quality control samples were analyzed without acidic cleavage, except for the quality control samples that were run to determine cleavage efficiency. The calibration range was 20-1000 ng/mL and the LLOQ was 20.0 ng/mL. The precision and accuracy of the calibration standards are given in Supplemental Table 1.
Data were analyzed using Analyst® software, version 1.7.1 (AB SCIEX, MA, USA/Canada). A linear regression model was used to extrapolate concentration values for PEG3350 in which the calibration standard 0 was 5.12 ng/mL (calculated from area ratios from calibration standards 0 and 1), calibration standard 1 was 20.0 ng/mL, calibration standard 2 was 40.0 ng/mL, and calibration standard 3 was 70.0 ng/mL. The detection limit was assumed to be 3.60 ng/mL. All concentrations <3.60 ng/mL were reported as below the LLOQ.
Results
ELISA-Based Approach
All 200 plasma samples were analyzed using the ELISA platform. PEG levels above the LLOQ (21.6 ng/mL) were detected in 41 (20.5%) plasma samples, range 22.1-43.9 ng/mL (Supplemental Table 2). PEG levels ranging from ≥25.0 to <35.0 ng/mL were detected in 29 of the 41 plasma samples (Figure 1). PEG levels above the LLOQa in 41 plasma samples from healthy adult donors analyzed by an ELISA platform. aLLOQ, 21.6 ng/mL. Abbreviations: ELISA, enzyme-linked immunosorbent assay; LLOQ, lower limit of quantification; PEG, polyethylene glycol.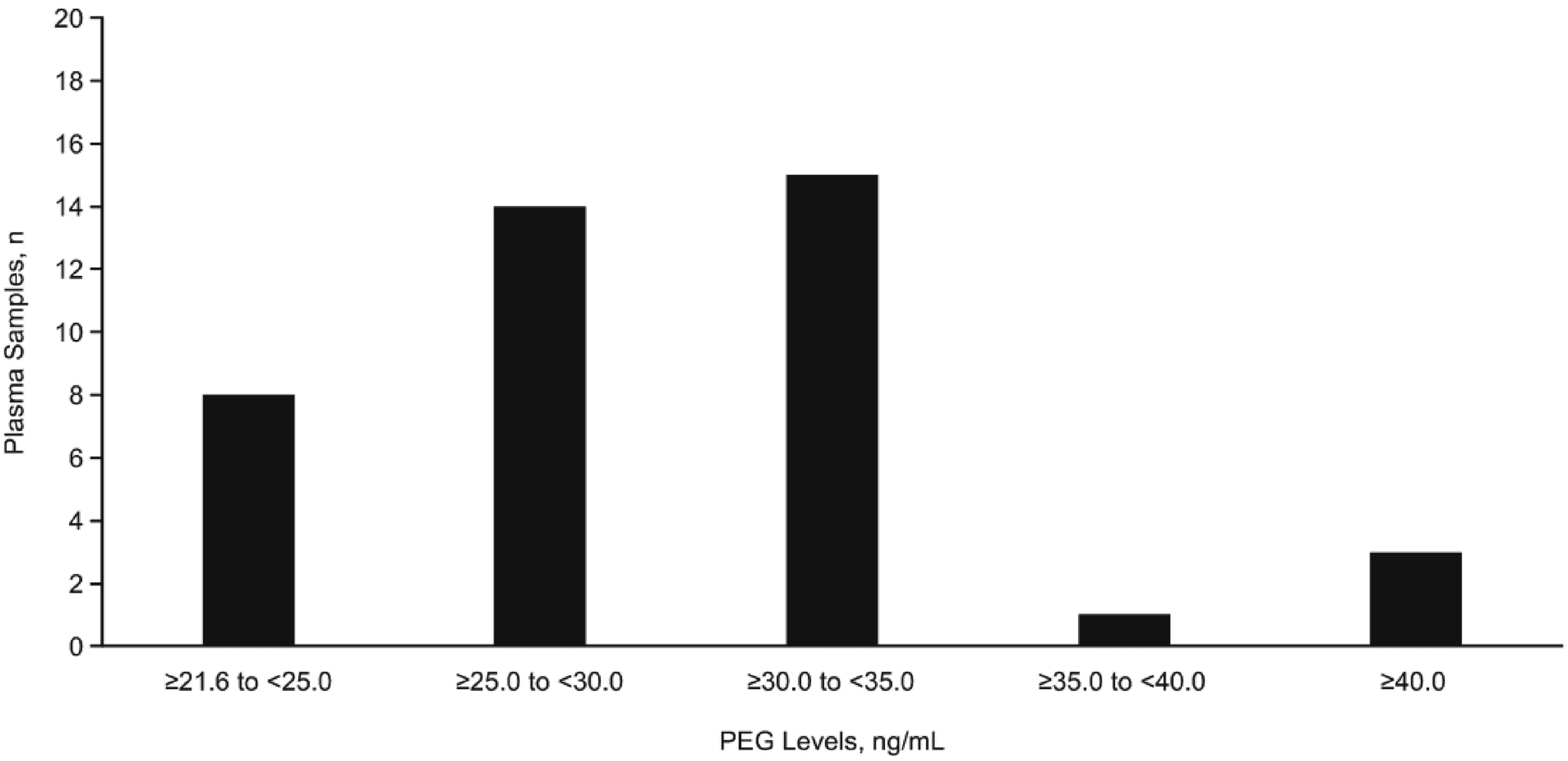
HPLC-MS/MS-Based Approach
All 200 plasma samples were analyzed using HPLC-MS/MS. One sample had to be reanalyzed owing to improper sample injection. Five plasma samples were analyzed at a smaller volume (20 and 25 μL) owing to an insufficient sample size. Higher LLOQs were used for these five samples: LLOQ2 (40.0 ng/mL) and LLOQ3 (50.0 ng/mL).
None of the samples were found to contain detectable levels of PEG4000, PEG10000, or PEG2ru20K COOH analytes, and none were found to contain detectable levels of PEG2ru20K COOH analytes following acidic cleavage. Two plasma samples were found to contain PEG3350 levels above the LLOQ (20.0 ng/mL): 46.4 ng/mL and 20.3 ng/mL, respectively.
A linear regression model was used to extrapolate concentration values for PEG3350 in 199 plasma samples. Data from the sample that had to be reanalyzed owing to improper sample injection were not extrapolated. PEG3350 levels above the LLOQ (3.60 ng/mL) were detected in 154 (77.4%) plasma samples (n = 199), range 3.69-46.0 ng/mL, following extrapolation (Figure 2 and Supplemental Table 3). PEG3350 levels above the LLOQa in 154 plasma samples from healthy adult donors detected following the extrapolation of concentrations identified through HPLC-MS/MS. aLLOQ, 3.60 ng/mL. Abbreviations: HPLC-MS/MS, high-performance liquid chromatography–tandem mass spectrometry; LLOQ, lower limit of quantification; PEG, polyethylene glycol.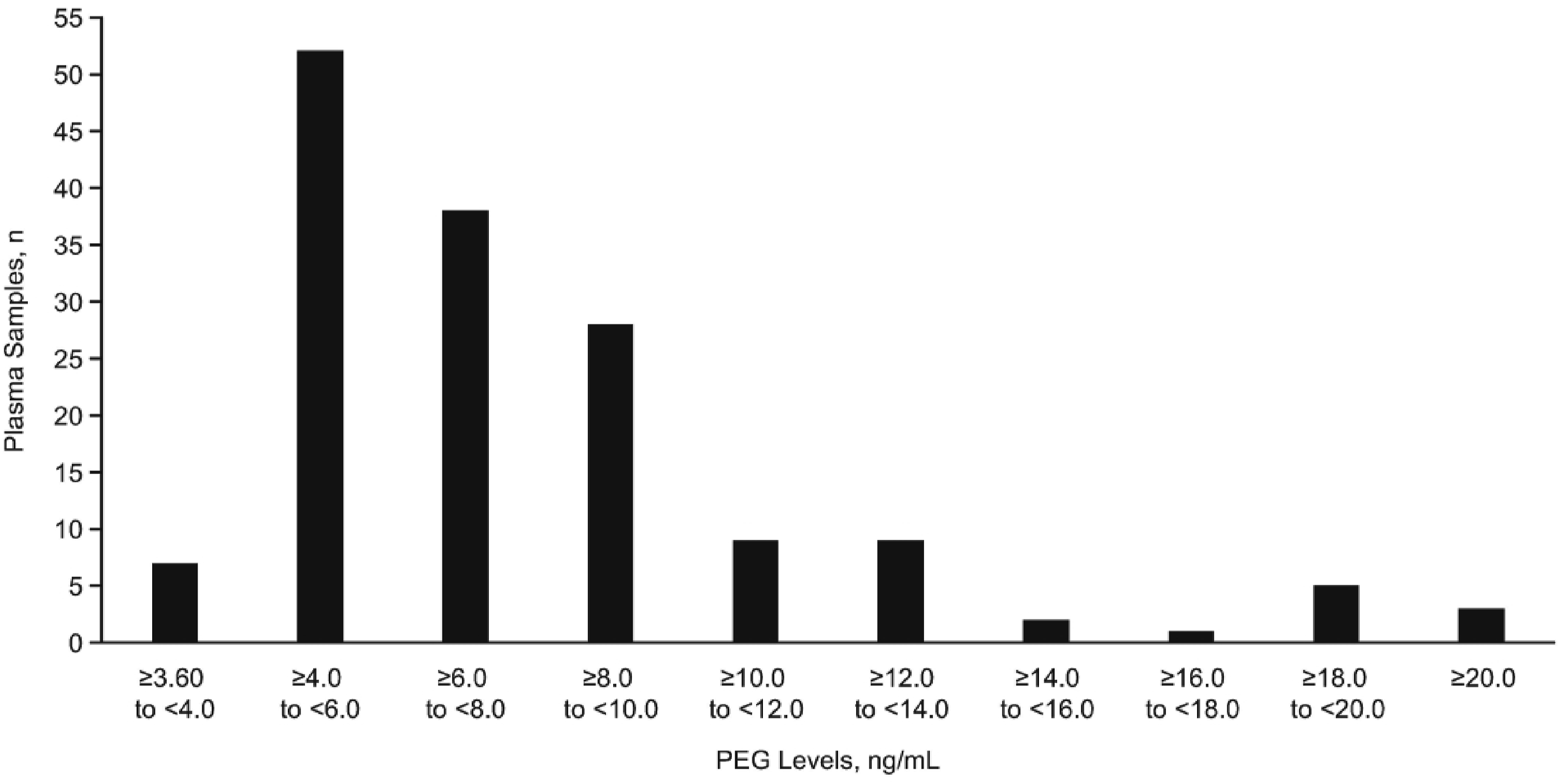
Discussion
In this study, 200 plasma samples from healthy adult donors were evaluated for the presence of PEGs using both an ELISA platform and HPLC-MS/MS. Approximately one-fifth of the plasma samples were found to contain detectable levels of PEGs using the ELISA platform. None of the samples were found to contain detectable levels of PEG4000, PEG10000, or PEG2ru20K COOH analytes using HPLC-MS/MS; however, more than three-quarters of the samples tested were found to contain detectable levels of PEGs approximately 3350 Da in size, also known as PEG-75, 1 following extrapolation using a linear regression model.
The study used two different approaches for the detection of PEGs. Although the ELISA platform was an effective way of detecting PEGs in plasma samples, PEGs of different sizes could not be differentiated using this approach given that the assay used a monoclonal antibody specific to the repeated ethane-1,2-diol units that form the backbone of all PEGs. HPLC-MS/MS enabled the detection of PEGs of specific sizes and the four analytes investigated in this study were chosen to encompass a large range of molecular weights. PEG3350 was the smallest PEG analyte investigated in this study. Smaller analytes such as PEG400 were not investigated, as HPLC-MS/MS is not optimally suited for their detection. Owing to the ubiquity of small PEGs, it would also be very difficult to obtain a suitable blank matrix to use as a calibration standard. Not surprising, given the different assay principles and the different validation criteria, the two different methods for the detection of PEGs, ELISA and HPLC-MS/MS, resulted in different levels of PEG found in the study population. PEG levels above the LLOQ were detected in 20.5% of plasma samples by the ELISA platform, while with HPLC-MS/MS, which can discriminate PEGs of different molecular sizes, PEG3350 was found in 77.4% of healthy adults. However, it required a linear regression model to extrapolate concentration values of PEG3350 because non-quantifiable signals of PEG were detected below the validated LLOQ of 20.0 ng/mL. Although already highly sensitive, further lowering the validated detection limit of the HPLC-MS/MS method would be required to substantiate the numbers found in the study.
PEGs and PEG derivatives are used extensively in consumer products, with 10 mg/kg considered the acceptable daily maximum intake by the World Health Organization.1,5,23 The PEG molecules used in cosmetics are generally considered to be safe, with a high median lethal dose (LD50).5,23 In animals, the oral ingestion of PEG has a LD50 of 2000 mg/kg bw, while PEG combined with fatty acids, sorbitan esters, or castor oils has a LD50 between 1000 and 2000 mg/kg bw.5,23 The LD50 for dermal exposure in animals is generally >10000 mg/kg bw.5,23 In humans, systemic toxicity effects possibly associated with PEG have been reported in the cases of three burn victims who received treatment with topical antimicrobial ointments containing PEG.5,23
Cytoplasmic vacuolation affecting different cell types, including renal tubule cells, macrophages, and epithelial cells of the choroid plexus, has been observed in a number of in vivo animal model toxicology studies following the repeated administration of high doses of PEGylated biotherapeutics.24,25 The potential for PEGylated biotherapeutics to cause cytoplasmic vacuolation in humans is unclear. A recent in vitro study in which monocyte-derived macrophages were exposed to 20-kDa PEG showed that vacuolation only occurred following exposure to concentrations that far exceeded clinically relevant doses for PEGylated drugs. 26 The study also showed that vacuolation did not affect the viability or functionality of the cells, which might suggest that PEG-mediated vacuolation is an adaptive cellular response rather than a toxic effect. 26
Although considered biologically inert, PEGylated biotherapeutics have been shown to elicit an immune response in some patients through the development of anti-PEG antibodies.14-17,27 These antibodies have been shown to correlate with a loss of therapeutic efficacy in some patients.14,15,17,27 In a study characterizing the antibodies that developed in 2 out of 46 patients with mild hemophilia A receiving treatment with a factor VIII therapeutic PEGylated with a 40-kDa PEG, it was shown that both patients developed neutralizing antibodies against the PEG moiety and that these antibodies abolished the efficacy of the drug. 27
Notably, anti-PEG antibodies have also been detected in healthy individuals and in patients who have not previously received treatment with PEGylated biotherapeutics.15,16,20,21 It has been suggested that the development of these antibodies is due to exposure to PEGs and PEG derivatives within consumer products.15,20 The findings from this study would support this suggestion given that the PEGs detected in this study are similar in size to some of the PEGs that are used in consumer products.1,2
Anti-PEG antibodies have also been shown to be associated with adverse safety outcomes, such as hematologic complications and hepatic toxicity, as well as hypersensitivity reactions which can range from local inflammation to complement activation-related pseudoallergy syndrome.28-30 In a study evaluating the prevalence of anti-PEG antibodies in patients with acute lymphoblastic leukemia prior to and/or immediately after receiving their first dose of PEGylated Escherichia coli asparaginase, the activity levels of the drug were significantly lower among patients with elevated anti-PEG IgG or IgM antibodies prior to their first dose. 29 Pre-existing anti-PEG IgG antibodies were also significantly associated with first-exposure hypersensitivity reactions in some patients. 29 It should be acknowledged that the presence of anti-PEG antibodies is not necessarily predictive of antibody-mediated toxicity as demonstrated by an in vitro study in which the presence of anti-PEG antibodies in human plasma did not correlate with complement activation in response to PEGylated liposomes. 31 The impact of anti-PEG antibodies on the safety of PEGylated biotherapeutics is currently a subject of scientific investigation and debate.32,33 While the safety considerations regarding PEGylated biotherapeutics are valid and warrant ongoing vigilance, some research, such as the low incidence of inhibitory antibodies observed in previously untreated patients with severe hemophilia A who were receiving treatment with a 20 kDa PEGylated recombinant factor VIII, might suggest that the benefits of PEGylated biotherapeutics may outweigh the risks associated with PEGylation.34,35
The worldwide exposure to PEGs is likely to have increased since the COVID-19 pandemic owing to the development and use of PEGylated lipid nanoparticle (LNP) COVID-19 vaccines. 36 PEGylated LNPs are synthetic non-viral vectors that are used as a delivery system for nucleic acids such as mRNA. 36 Before the COVID-19 pandemic, a few PEGylated nanocarriers had been approved for the treatment of conditions including ovarian cancer and breast cancer. 36 During the pandemic, two mRNA-based PEGylated LNP vaccines against COVID-19 were developed. 36 Similar to other PEGylated biotherapeutics, anti-PEG antibodies have been detected in some individuals who received these vaccines.37,38 All the samples used in this analysis pre-date the pandemic, thereby providing an insight into PEG exposure prior to the advent of mRNA COVID-19 vaccines. Research evaluating the presence of PEGs in the plasma of healthy individuals collected after the COVID-19 pandemic might be of interest to investigate any possible effects that mRNA COVID-19 vaccines might have had on PEG exposure.
The therapeutic efficacy and safety profile of PEGylated biotherapeutics is challenged by the presence of anti-PEG antibodies and research is ongoing into how these challenges might be overcome, with possible strategies including modifications to the surface of PEGylated liposomes or alterations to PEG-lipid linkages. 36 There are also several possible alternatives to PEG, such as zwitterionic polymers or polyoxazoline, that are under investigation.36,39 It is important to acknowledge, however, that PEG alternatives are not immunologically inert and that much remains unclear around the mechanisms that underpin the immunological effects of PEGylated biotherapeutics. 40 Continued research into PEGylated biotherapeutics, therefore, remains essential to addressing these knowledge gaps.40,41
Conclusion
PEGs were detected in the plasma of a proportion of up to 77% of healthy adults. The presence of these PEGs is possibly the result of exposure to PEGs and PEG derivatives in consumer products. A limitation of the study is the small sample number. More extensive studies to better characterize the global incidence of plasma burdens of PEG in a larger population would be required. Such studies should also consider measuring samples from healthy individuals of different age groups, from different social environments, and from countries with different development status defined using a combination of economic, social, and political indicators such as the Human Development Index established by the United Nations Development Programme. Additionally, further research into whether anti-PEG antibodies correlate with PEG levels detected in healthy individuals and whether PEG levels in healthy individuals fluctuate over time or have any adverse effects would be of interest.
Supplemental Material
Supplemental Material - Evaluation of Plasma Polyethylene Glycol Levels in a Healthy Adult Population
Supplemental Material for Evaluation of Plasma Polyethylene Glycol Levels in a Healthy Adult Population by Klaus Kubesch, Christian Lubich, Daniel Mascher, Srilatha Tangada, and Peter L. Turecek in International Journal of Toxicology
Footnotes
Acknowledgements
Under the direction of the authors, medical writing support was provided by Sarah Morgan, PhD, employee of Envision Catalyst, an Envision Medical Communications agency, a part of Envision Pharma Group (Fairfield, CT, USA), and was funded by Takeda Development Center Americas, Inc., Cambridge, MA, USA.
Author Contributions
Study conception and design: S.T. and P.L.T. Data acquisition: K.K., C.L., and D.M. Data analysis and interpretation: C.L., S.T., and P.L.T. All authors were involved in drafting the article and/or revising it critically for important intellectual content. All authors approved the final version to be published, and all authors agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.K. is an employee of pharm-analyt Labor GmbH. C.L. is an employee of Institute Krems Bioanalytics, which received funding from Baxalta Innovations GmbH, a Takeda company, for this study. D.M. is an employee of pharm-analyt Labor GmbH. S.T. is an employee of Takeda Development Center Americas, Inc., Cambridge, MA, USA, and a Takeda stock owner. At the time of the study P.L.T. was an employee of Baxalta Innovations GmbH, a Takeda company, and a Takeda stock owner.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Baxalta Innovations GmbH, a Takeda company, Vienna, Austria.
Ethical Statement
Written informed consent was obtained from all individuals who provided plasma samples. Local ethical committees approved the study.
Data Availability Statement
All relevant data are within the paper and its supporting information.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
