Abstract
BAY 94-9027 (Jivi) is a site-specifically PEGylated human B-domain-deleted (BDD) recombinant factor VIII (FVIII), with a 60 kDa branched PEG molecule attached. The nonclinical safety of BAY 94-9027 was evaluated in a toxicology program that included 2 weeks intravenous (IV) toxicity studies in rats and rabbits, a juvenile toxicity study in rats as well as a 26-week chronic study in rats. Doses of 75, 750, or 2250 IU/kg given every other day for 2 weeks did not elicit any findings related to BAY 94-9027. Specifically, no thrombus formation or histological changes such as cellular vacuolation were seen. In the chronic toxicity study, 40, 400, and 1200 IU/kg of BAY 94-9027 given twice weekly did not induce adverse effects related to BAY 94-9027, and no tissue vacuolation was observed. There was no PEG detected in choroid plexus or other areas of the brain, cerebrospinal fluid or in spleen or kidneys. These results were supported by toxicity studies in rats and rabbits treated with PEG 60 kDa attached to the maleimide linker (PEG-60-Mal-Cys). No findings related to PEG-60-Mal-Cys were seen. These results demonstrate the safety of BAY 94-9027 for long-term use.
Keywords
Introduction
People with the coagulation disorder hemophilia A are treated with factor VIII (FVIII) as replacement for their missing or inactive intrinsic FVIII protein, either on demand (treatment when a bleed occurs) or prophylactically (to prevent bleeding). While prophylactic use can provide continuous protection from bleeding, intravenous (IV) infusions have to be given several times per week due to the short half-life of FVIII. Extending protection with fewer infusions has been a goal in the development of new coagulation factors and can be achieved, for example, by linking the recombinant human FVIII (rFVIII) protein to other, inert large molecules such as the Fc part of an antibody (rFVIIIFc, Elocta/Eloctate) or to polyethylene glycol (PEG) molecules as seen in BAY 94-9027 and Adynovate. These techniques have enabled less frequent IV infusions while maintaining pharmacological activity. 1 –7
Bay 94-9027 (Jivi) was recently approved for the treatment of hemophilia A. In humans, 2 pharmacokinetic studies have shown that BAY 94-9027 has improved pharmacokinetic parameters compared to recombinant FVIII (rFVIII; Kogenate FS) and extended half-life rFVIIIFc (Elocta/Eloctate) products. 2,8 In the PROTECT VIII study and its extension, BAY 94-9027 was efficacious in the prevention of bleeds in previously treated adults and adolescents. 9,10 BAY 94-9027 was generally well tolerated, and no patient developed FVIII inhibitory antibodies. 11,12
BAY 94-9027 is a site-specifically PEGylated B-domain-deleted (BDD) rFVIII containing a single branched 60 kDa PEG molecule. 1 It contains the largest PEG molecule conjugated to a drug protein to date. BAY 94-9027 itself is inactive and requires activation to FVIIIa to be effective in the intrinsic coagulation pathway. The molecule was selected out of several drug candidates with different, smaller PEG sizes and different PEG attachment sites based on its longer half-life in hemophilia A mice and dogs and full coagulation activity. PEGylation was used for BAY 94-9027 since it has been a successful approach in the modification of biopharmaceuticals to extend their circulation half-life. 13 –16 A single clinical dose of 60 IU/kg body weight of BAY 94-9027 contains a very small amount of PEG, approximately 0.002 mg. Therefore, the overall amount of PEG, also with long-term treatment, is very small in humans.
More than 12 PEGylated biotherapeutics have been approved for IV or subcutaneous (SC) use over the last 2 decades, 13,16 and no safety signals related to PEG have been identified in humans. 13 –16 Polyethylene glycol molecules ≥2 kDa in size are inert, highly water soluble, and have no specific receptor or other target in organs or tissues in the body. 13 The toxicity of PEGylated drugs usually reflects the toxicity of the parent (unconjugated) drug molecule. 17 The only effect related to PEG observed in nonclinical toxicology studies was described as cellular vacuolation visible in histological evaluations by light microscopy at higher PEG doses, 18,19 above the amount of PEG in a clinical dose of BAY 94-9027. In publicly available toxicological information, vacuolation has not been linked with changes in organ function in the absence of changes in cell morphology or changes in the surrounding tissue and therefore cellular vacuolation alone is not considered adverse. Nor have there been any reports of adverse events due to PEG with PEGylated drugs in humans. 13,16 Cellular vacuolation has been seen histologically with PEG sizes starting already at 5 kDa after repeated high doses in toxicology studies 14 and has also been reported after a single dose for a different product. 20 The light microscopically visible cellular vacuoles were round and discrete clear cytoplasmic vacuoles in hematoxylin and eosin (H&E) staining. 18,19 Polyethylene glycol does not stain with standard histological H&E dyes, and specialized immunohistochemistry (IHC) methods have to be used to detect PEG in cellular vacuoles and recently also in the cytoplasm of cells. 18,19 These vacuoles likely represent lysosomal/endosomal uptake of PEG or PEGylated drugs. Polyethylene glycol–related vacuolation is seen most frequently in macrophages and other cells of the reticulo-endothelial (RES) system and is likely an adaptive response to remove molecules such as PEG. 18,21 Vacuoles have also been observed in nonphagocytic cell types including renal tubular epithelium and choroid plexus epithelium. Distribution patterns of PEGylated biotherapeuticals may depend on drug receptor/target-mediated uptake and/or ligand-specific transport mechanisms, and therefore organs or tissues with vacuolation and the time and dose when this occurs can vary for different PEGylated biopharmaceuticals. This has been shown by Ivens et al. 13 in an industry survey of 17 PEGylated biopharmaceutics under development, which, for example, found that vacuolation of the choroid plexus was seen with 20-, 30-, and 40-kDa site-specific-PEGylated proteins. FVIII and BAY 94-9027 have no cellular receptors, it is unlikely that there is specific uptake of BAY 94-9027 in any organ or tissue.
Basophilic storage granules (cytoplasmic of basophilic granules) similar in type to vacuolation seen with PEG have been described for antisense oligonucleotides (ASOs; stained with H&E), and vacuolation has been detected following administration of some sugars and also methylcellulose. 22
The nonclinical toxicity program for BAY 94-9027 included systemic, juvenile, and chronic (long-term) toxicity studies in rats and rabbits to address overall safety and specifically the potential for PEG-related cellular vacuolation. The chronic toxicity study with 26 weeks of twice-weekly dosing is the main focus of this publication. It was conducted in immunodeficient (IDF) athymic male rats (Crl: NIH-Foxn1 rnu ), which do not mount a humoral immune response against species foreign proteins. The study provides a comprehensive toxicology investigation of BAY 94-9027 utilizing IHC in combination with standard histopathology, and PEG analysis in cerebrospinal fluid (CSF) plus plasma to address recent concerns about PEG accumulation in tissues after administration of PEGylated biopharmaceuticals. The toxicology program BAY 94-9027 was supplemented by studies with the PEG 60 kDa linker moiety alone (unconjugated PEG-60-Mal-Cys) at high doses to assess the potential for formation of cellular vacuolation and the genotoxicity of the maleimide linker.
Materials and Methods
Toxicology Program
To assess the safety of BAY 94-9027, the nonclinical program was tailored specially for the patient population with hemophilia A. The toxicology program for BAY 94-9027 was conducted in male animals only to reduce the overall animal number since the patient population is male. Factor VIII and BAY 94-9027 are active in the rat and rabbit coagulation cascades (data not shown); therefore, rats and rabbits are relevant species.
The program included 2-week IV systemic and juvenile (requested by one regulatory agency and not triggered by any findings in other toxicity studies) studies in rats and rabbits and a chronic 26-week IV toxicity studies in IDF rats. The chronic rat study is the main focus of this publication and is reported in detail, while the other toxicology studies are summarized more briefly.
In the systemic studies, 2250 IU/kg BAY 94-9027 was given IV as highest dose every other day for 2 weeks. This is approximately 30 times the highest human dose of 60 IU/kg. The following endpoints were evaluated: body weight, mortality, clinical signs and symptoms, ophthalmology, hematology and hemostasis (at end of study), clinical chemistry and urinalysis (at end of study and recovery), toxicokinetic (TK), antidrug antibodies (binding and neutralizing), gross pathology and organ weights, histopathology of a set of >40 organs, and tissues per animal according to the Guideline on Repeat Dose Toxicity. 23 Furthermore, a 2-week study in juvenile rats was conducted with twice-weekly IV dosing of 200 and 1000 IU/kg starting on postnatal day (PND) 17 employing similar end points as the systemic studies.
Due to recent concerns about PEG safety, 21 and since the toxicology studies with BAY 94-9027 were limited in duration and doses, the PEG 60 kDa moiety connected to the maleimide linker (PEG-60-Mal-Cys) was evaluated in further studies up to 4 weeks duration in rats and rabbits. In addition, a set of genotoxicity studies was conducted, since PEG-60-Mal-Cys including the maleimide linker had not been tested before. PEG-60-Mal-Cys was produced by mixing PEG 60 kDa maleimide used in production of BAY 94-9027 with cysteine to quench the reactive part of maleimide by binding it to this amino acid.
Doses of PEG-60-Mal-Cys were selected as very high multiples of the weekly PEG dose present in BAY 94-9027 assuming that a high dose given for 4 weeks will reveal the potential for formation of cellular vacuolation. In the 4-week rat study, animals were dosed with 11 mg/kg given every second day, which is more than 5000-fold higher than the amount of PEG 60 kDa in a single human dose of 60 IU/kg. The same end points summarized earlier for the 2-week studies with BAY 94-9027 were evaluated at the end of these supplementary in vivo studies. As previously reported, the pharmacokinetics, distribution, excretion, and metabolism of the PEG-60-Mal-Cys (= BAY 1025662) at 11 mg/kg was previously investigated in the rat at a single high dose. 24
The chronic toxicity study was conducted in IDF athymic male rats (Crl: NIH-Foxn1 rnu IDF nude rat) since immune-competent animals mount an antibody response to human FVIII within 2 weeks due to the differences in the protein structure between humans and animals. Binding antibodies (ADA) and drug-neutralizing antibodies (nADA) are generated, which abolish exposure to active FVIII.
The study design of the chronic toxicity study in IDF rats is described in more detail below, and Table 1 summarizes the toxicology studies conducted with BAY 94-9027, while Table 2 lists supplemental toxicology studies with PEG-60-Mal-Cys including group size and doses.
List of Repeated Administration Toxicity Studies With BAY 94-9027.

Abbreviations: IV, intravenous; PND, postnatal day.
List of Repeated Administration Toxicity Studies With PEG 60 kDa Bound to Maleimide (PEG-60-Mal-Cys).

Abbreviations: IV, intravenous; PEG, polyethylene glycol.
Good laboratory practice status and regulations applied
The chronic study in IDF rats complied with the US Animal Welfare Act. 25 The models in immune-competent rats and rabbits for the initial characterization of BAY 94-9027 were chosen in line with the international guidelines for assessment of the toxicological profile on drugs, especially biologics. 23,26,27 In line with German regulations, animal studies requested by regulations for risk assessment of human use were authorized by the local authority responsible for animal protection by granting a “license for the use of animals for scientific purposes” (Anzeige der Verwendung von Tieren für wissenschaftliche Zwecke) for the specific type of experiment. This includes the animal species used, the procedures performed, and the general size of the study based on the German Animal Welfare Law. 28 The approval process included a thorough check whether the principle design of the animal studies was adequate for the intended purpose and the potential suffering of the animals ethically justified. All animal studies performed in Germany were performed with valid licenses for such experiments.
All in vivo studies were good laboratory practice (GLP) compliant. The peer reviews and all assays and assessments except the assay for PEG concentration in rat plasma in the chronic rat study were conducted under GLP.
Chronic Toxicity Study With BAY 94-9027 in IDF Rats (Crl: NIH-Foxn1rnu)
Animals and study design
Male Crl: NIH-Foxn1 rnu IDF rats were approximately 8 weeks old at the start of treatment, assigned to study groups using computer-generated randomization procedures, housed two per box when possible in isolator equivalent animal rooms, and handled according to procedures for immunocompromised rodents. Rats were housed in filter-top, autoclaved, polycarbonate nesting boxes containing environmental enrichment with autoclaved bedding and nesting material. Rats were provided with Irradiated Rodent Diet available ad libitum, and sterilized water in sipper tube bottles was available ad libitum. Veterinary care was available throughout the course of the study. All cage sizes and housing conditions are in compliance with the US Guide for the Care and Use of Laboratory Animals.
The highest dose of 1200 IU/kg twice weekly was selected to be 30-fold above the highest weekly dose in humans. BAY 94-9027 and the control item were administered to the animals via IV injection into the tail vein (alternating between the left and right caudal veins, when possible) by straight needle bolus administration. The lots used were tested for potency, stability, and other attributes to assure the accuracy of dosing in the study.
Animals received volumes ranging between 0.1 and 3.0 mL/kg body weight. Groups of rats received 0, 40, 400, and 1200 IU/kg of BAY 94-9027 twice weekly IV for 13 or 26 weeks at volumes between 0.1 and 3.0 mL/kg. Additional animals received the test item for 26 weeks twice weekly followed by 26 treatment-free weeks (see Table 3 for the study design). The following parameters and end points were evaluated in this study: clinical signs, body weights, body weight gains, food consumption, ophthalmology, clinical pathology parameters (hematology, coagulation, clinical chemistry, and urinalysis), TK parameters for BAY 94-9027 to show exposure, PEG concentrations in plasma, PEG in CSF, ADAs in serum, gross necropsy findings, organ weights, IHC evaluation for PEG in tissues, and histopathologic examinations.
Study Design of 26 Weeks Chronic Toxicity Rat Study Followed by 26 Weeks of Recovery With BAY 94-9027 Dosed Twice Weekly.
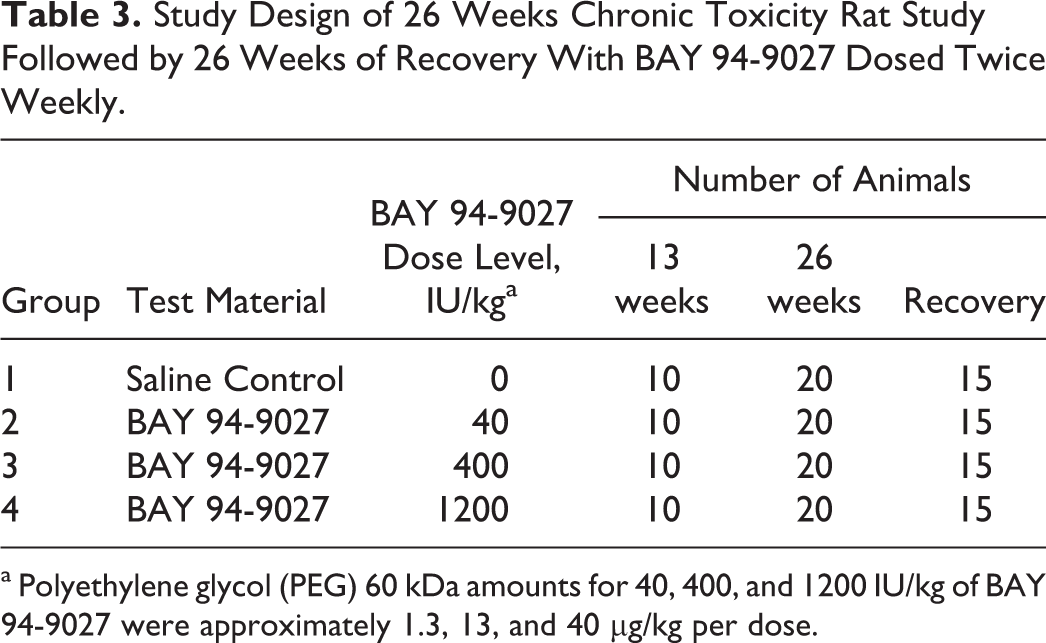
a Polyethylene glycol (PEG) 60 kDa amounts for 40, 400, and 1200 IU/kg of BAY 94-9027 were approximately 1.3, 13, and 40 µg/kg per dose.
Laboratory evaluations for clinical chemistry, hematology, coagulation and urinalysis
Blood was collected from jugular vein (in-life) or via the vena cava while under isoflurane/oxygen anesthesia (terminal) from individual animals. After collection, samples were transferred to the appropriate laboratories for processing. Table 4 shows the sample schedule for clinical pathology. Clinical pathology parameters were assessed after 13 and 26 weeks and after recovery.
Scheduled Clinical Pathology Time Points for Chronic Rat Study.
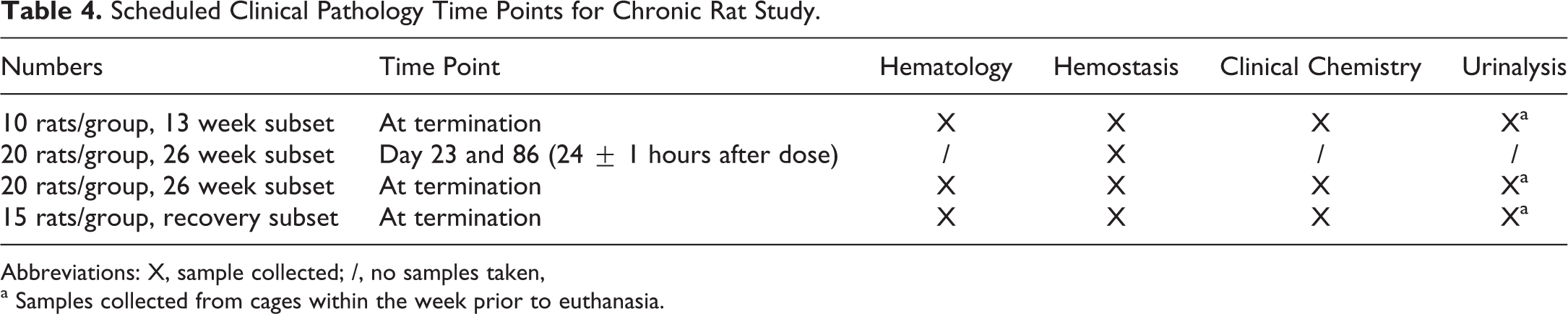
Abbreviations: X, sample collected; /, no samples taken,
a Samples collected from cages within the week prior to euthanasia.
Hematology parameters included red blood cell count, hemoglobin concentration, hematocrit, mean corpuscular volume, red blood cell distribution width, mean corpuscular hemoglobin concentration, mean corpuscular hemoglobin, reticulocyte count (absolute), platelet count, white blood cell count, neutrophil count (absolute), lymphocyte count (absolute), monocyte count (absolute), eosinophil count (absolute), basophil count (absolute), large unstained cells, and other cells (as appropriate).
Clinical chemistry parameters included alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, gamma-glutamyltransferase, creatine kinase, total bilirubin, urea nitrogen, creatinine, calcium, phosphorus, total protein, albumine, globulin (calculated), albumine/globulin ratio, glucose, cholesterol, triglycerides, sodium, potassium, and chloride.
Coagulation parameters included activated partial thromboplastin time by assay that did not react to PEG, fibrinogen, and prothrombin time.
Urinary parameters included color, appearance/clarity, specific gravity, pH, protein, glucose, bilirubin, ketones, blood. Urine were collected from individually housed animals overnight. Animals were fasted overnight prior to collection.
Toxicokinetics and antidrug antibody analysis
Plasma concentrations of BAY 94-9027 were determined by a validated method from individual rats. Starting on dosing day 78 (week 11) and dosing day 169 (week 24), blood samples were collected at 0, 0.25, 8, 24, 48, and 72 hours after dosing. Due to the spacing of the time points, each animal was sampled at least twice, allowing assessment if exposure was impacted/low due to antidrug antibodies. BAY 94-9027 activity was determined in diluted plasma by an enzymatic assay with absorbance readout using an antihuman FVIII monoclonal antibody as capture and a commercially available kit (Biophen FVIII: C; Hyphen BioMed, Neuville-sur-Oise, France) for measuring activity. The ADAs were evaluated in serum samples for all animals at termination using a validated enzyme-linked immunosorbent assay (ELISA). Samples were heat treated at 56°C for 30 minutes before analysis at a minimum required dilution of 1:20. BAY 94-9027 was coated onto a microtiter plate and used to capture antidrug antibodies present in samples. Captured antibodies were detected using a goat anti-rat IgG antibody conjugated to horseradish peroxidase (HRP) and tetramethylbenzidine as a substrate. Samples were analyzed initially in a screening assay followed for samples screened positive by a confirmatory assay.
Polyethylene glycol analysis in plasma
Plasma samples for PEG analysis were collected at necropsy from each animal. Samples were analyzed under non-GLP conditions for PEG by liquid chromatography–mass spectrometry after protein precipitation with methanol. PEG-60-Mal-Cys was used as reference standard and a stable isotopically labeled 60 kDa PEG as internal standard. The method was validated for a lower limit of quantitation (LLOQ) of 20 ng/mL.
Cerebrospinal fluid collection and analysis
Cerebrospinal fluid was collected from all animals (when possible) at termination under anesthesia before necropsy for analysis of PEG. The determination of PEG in CSF was conducted using a validated direct competitive ELISA with an LLOQ of 20 ng/mL: Diluted samples were added on a 96-well plate coated with bovine serum albumin conjugated to methoxy-PEG. An anti-PEG antibody specific for the polyoxyethylene backbone and conjugated to HRP was used as detection reagent. PEG-60-Mal-Cys was used as the reference standard. Some samples were excluded from the analysis since they had a blood contamination level higher than 0.5%, which was shown to generate false-positive results during validation.
Necropsy, histology, and immunohistochemistry
A gross necropsy was performed for all animals, and organ weights were taken. Tissues were collected for histology and IHC in 10% neutral-buffered formalin. The number of organs and tissues weighed and the list of 46 organs and tissues per animal fixed and evaluated histologically closely followed the Guideline on Repeated Dose Toxicity. 23 Histological evaluation was performed by a pathologist and peer-reviewed according to internal Standard Operating Procedures (SOPs).
Sections for IHC of brain with choroid plexus, kidneys, and spleen (weeks 13, 26, and 52) were prepared after fixation in 10% neutral-buffered formalin (2-7 days; approximately 5µm thick), dehydration, and embedding in paraffin wax. From each animal, 4 unstained sections from each of the 3 organs were mounted on Super frost slides and air dried for at least 24 hours at room temperature.
Positive controls for IHC were sectioned from formalin-fixed, paraffin-embedded rat skeletal muscle injected postmortem with cell culture suspension containing the human acute monocytic leucemia cell line THP-1 differentiated into human macrophage phenotype and preincubated with BAY 94-9027 for >20 hours and/or test item BAY 94-9027. The true negative controls included tissue sections from group 1 control animals incubated with the primary antibody, and sections from formalin-fixed, paraffin-embedded rat skeletal muscle injected postmortem with cell culture suspension containing untreated THP-1 human macrophages. The design of the positive controls was similar to the one described by Rasmussen et al. 29 Tissue sections were deparaffinized and rehydrated, and for each IHC procedure antigen-blocking was performed to eliminate nonspecific binding by endogenous avidin/biotin and 10% normal goat serum for proteins. The primary antibody (Ab) used was a rabbit monoclonal anti-PEG Ab (PEG-B-47; Epitomic, CA, USA). The company indicates binding to the terminal methoxy group of conjugated PEG molecules, and Rudmann et al 18 have shown that this detection antibody also binds to unconjugated PEG. The optimal antibody concentration was determined to be 5 µg/mL. In order to confirm negative staining results at 5 µg/mL, the 2-fold concentration of 10 µg/mL antibody was applied on brain, spleen, and kidney sections from 5 of 20 controls and 5 of 17 high-dose animals from the 26-week test period. The secondary antibody was a link anti-rabbit antibody (HK326-UR; Biogenex, 49026 Milmont Drive, Fremont, CA 94538, USA) used per manufacturer recommendation, and labeling was performed with a streptavidin-alkaline phosphatase followed by development with the chromogen permanent AP-red solution and counterstaining with hematoxylin. Isotype-matched immunoglobulin from nonimmunized rabbit serum (rabbit IgG, IS600, Dako, 5301 Stevens Creek Blvd, Santa Clara, CA 95051. USA) matched by concentration was used in place of the primary antibody for negative controls. All steps were performed at room temperature, and incubation was performed in a humid chamber at Bayer AG, Germany. Peer reviews of the immunohistochemical data were conducted according to internal SOPs.
Statistical Analysis
Proportions and/or incidences were calculated to summarize categorical data as appropriate. Means and standard deviations were calculated, where appropriate. For clinical pathology data, analysis of variance was employed, comparing the groups treated with BAY 94-9027 to saline control (group 1).
Results
Two-Week Toxicity Studies With BAY 94-9027
The following briefly summarizes the results of the 2-week studies. The analysis of anti-FVIII antibodies confirmed that approximately 50% of rats and rabbits had developed nADAs after 2 weeks, while animals treated with full-length FVIII in concurrent dose groups had a higher frequency of close to 100%. This confirmed that a longer duration of the studies would not be meaningful since the nADAs abolish exposure to active drug.
Rats and rabbits dosed IV with BAY 94-9027 every other day at 75, 750, and 2250 IU/kg for 2 weeks tolerated BAY 94-9027 without any toxicity. No treatment-related mortality, effects on general behavior, or relevant changes in organ weights or histopathologic changes were observed. Specifically, no PEG-related cellular vacuolation in any of the >40 organs and tissues evaluated per animal was seen. In rabbits, a prolongation of the activated partial thromboplastin time (aPTT) was recorded after 2 weeks of dosing in the 750 (153% of controls) and 2250 IU/kg (166%) dose groups. This was also seen at the end of the recovery period. Most likely, the prolongation is due to the development of inhibitory antibodies (nADAs) that cross-reacted with rabbit FVIII, causing the prolongation of aPTT that persisted throughout the recovery period. No anti-PEG antibodies were detected in rats or rabbits. The no-adverse effect level (NOAEL) is the highest dose of 2250 IU/kg given every other day. No test article–related adverse effects were seen in juvenile rats (first dose at PND 17) up to the highest dose of 1000 IU/kg given twice weekly for 2 weeks.
Four-Week Toxicity Studies With PEG 60 kDa
PEG-60-Mal-Cys was well tolerated after single and repeat administration up to very high doses (Table 2). In none of the studies conducted, treatment-related mortality or effects on general behavior or organ weights were observed. Up to the highest doses tested, no evidence of any organ-specific or other toxicities were seen. Specifically, no tissue vacuolation in histopathological evaluations was seen after 4 weeks of high doses of PEG-60-Mal-Cys in rats and rabbits.
A juvenile animal study was conducted with PEG-60-Mal-Cys with the first IV dose on PND 17, dosed twice weekly for 4 weeks. No toxicologically relevant effects were observed for PEG-60-Mal-Cys up to 2 mg/kg, the highest doses tested in these young animals.
Chronic Toxicity Study With BAY 94-9027
The IDF rat model has been used previously to evaluate other coagulation factors such as FVIII and FIX. 29,30 This rat strain has severely impaired thymus-related immunoreactions and T-cell development, 31 –33 preventing ADA and nADA formation as shown for FVIII or FIX. 29,30 The number of granulocytes, monocytes/macrophages, erythrocytes, B cells, and natural killer (NK) cells in the blood of IDF rats were usually within the normal range; the innate immune system in IDF rats is well developed. 32 There is indication that the IDF rat strain has a reduced survival time likely due to pathogen-free housing requirements with a reported life span of 1.5 years. 31 –33 Also, detailed mortality data and histological background lesion information are not available since this model is usually not used for toxicology studies.
Results of In-life Assessment
The following summarizes the study results. No BAY 94-9027-related observations were recorded during in-life or recovery phase up to the highest dose of 1200 IU/kg twice weekly. There were no treatment-related changes in body weights, body weight gains, or food consumption (data not shown). In the highest dose group, 4 of 45 animals per group died or were killed moribund within 52 weeks (end of recovery phase), while in the other groups 1 or 2 deaths were recorded (Table 5). For 2 of the 8 animals that died, no clear cause of death could be determined (animal number 78 and 136). Findings such as paraphimosis (1 rat in group 4), kidney pathology (1 rat in group 4), and lymphoma (2 rats in group 4) are known to also occur in other rat strains as background changes, especially in older rats. It is likely that the IDF rat strain can develop these pathologies and that the mortality seen at the high dose is likely unrelated to treatment with BAY 94-9027.
Mortality Up to Week 13, 26, and at End of the Recovery Period (After 52 Weeks) in the Chronic Rat Toxicity Study With BAY 94-9027 Dosed Twice Weekly.
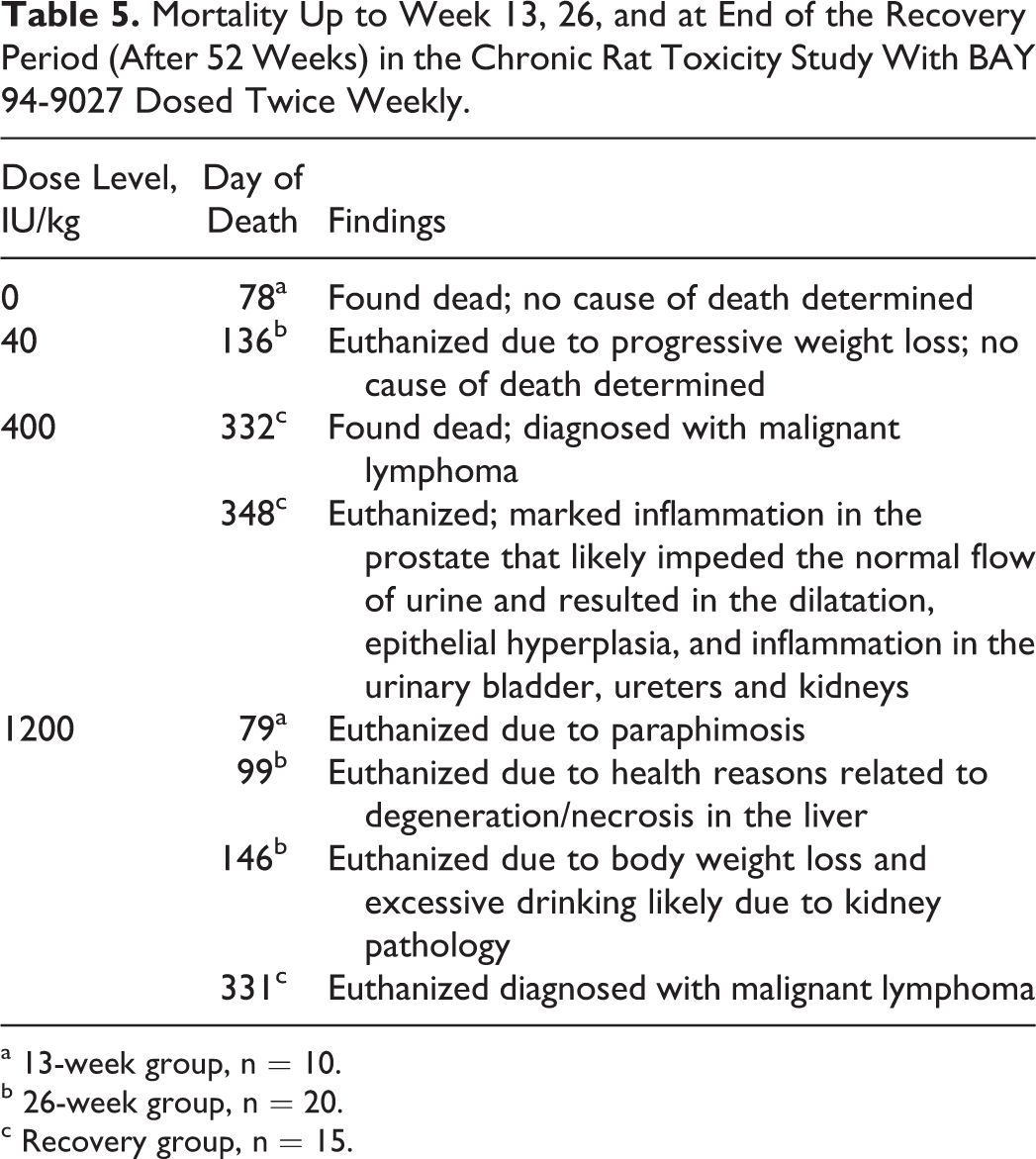
a 13-week group, n = 10.
b 26-week group, n = 20.
c Recovery group, n = 15.
Clinical Chemistry, Hematology, Coagulation, and Urinalysis
No changes were seen in any of the hematology, clinical chemistry, or urinary parameters. The only effect measured after administration of BAY 94-9027 was limited to a transient minimal decrease in the aPTT, mainly at the highest dose on days 23 and 86. This was not noted at subsequent time points. A reduction in aPTT is an expected pharmacological effect of BAY 94-9027. There were no BAY 94-9027-related alterations in clinical chemistry parameters. There were no BAY 94-9027-related alterations in urinalysis parameters.
Toxicokinetics and Antidrug Antibody Evaluation
The goal of the TK assessment of BAY 94-9027 was to demonstrate exposure and identify animals that may have had low/no exposure due to antibodies to rFVIII. The data were supported by the ADA analysis. The nADAs were not measured since ADAs together with TK already indicates neutralization of exposure. After 13 and 26 weeks, the lowest dose of 40 IU/kg was mostly below limit of quantitation of the assay. At the higher dose groups, all animals showed exposure except one rat in the mid-dose group. This animal also was the only animal with ADAs. Group means are listed in Table 6.
Toxicokinetics: Summary of Mean Plasma Concentrations (Geometric Mean and SD) of BAY 94-9027 After 11 and 24 Weeks of Dosing (Each Value is Comprised of 3 [Week 11] or 5 [Week 24] Rats Per Time Point).
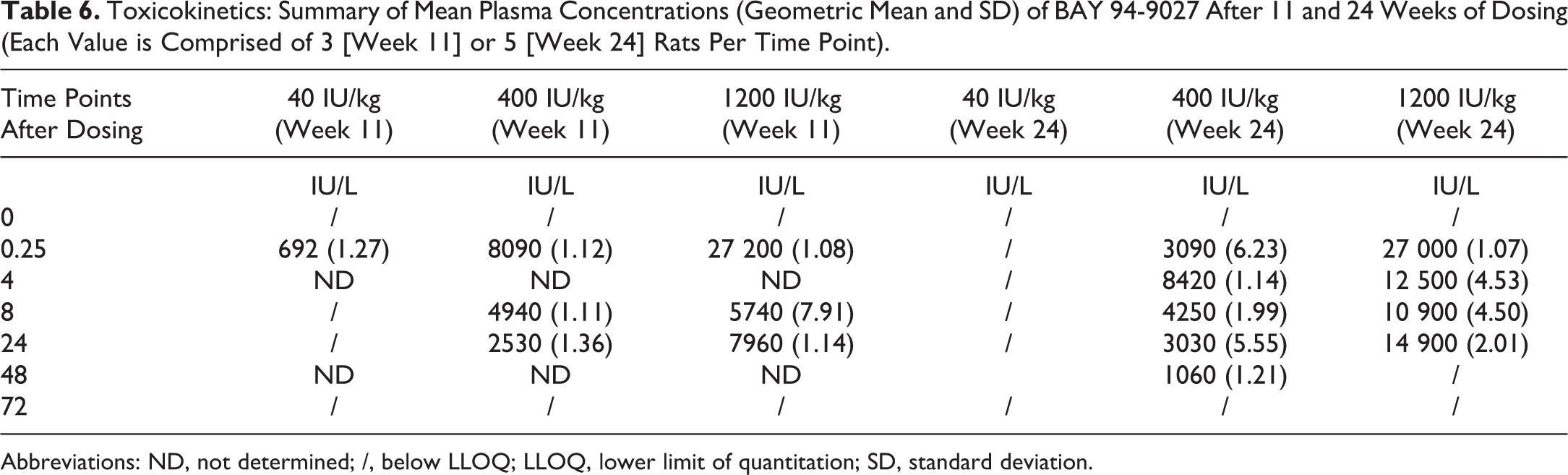
Abbreviations: ND, not determined; /, below LLOQ; LLOQ, lower limit of quantitation; SD, standard deviation.
Analysis of PEG in Plasma
The analysis of PEG in plasma showed that only 1 sample from each of the week 13 and week 26 time points had measurable PEG levels in the group 2 (40 IU/kg) animals (21.1 and 24.5 ng/mL), and all other group 2 samples were below the LLOQ (20 ng/mL). Mean concentrations of PEG in plasma increased with dose in the group 3 (400 IU/kg) and group 4 (1200 IU/kg) animals as presented in Table 7. No PEG was measurable in plasma after 26 treatment-free weeks.
Plasma Levels of Free PEG After 13 and 26 Weeks.
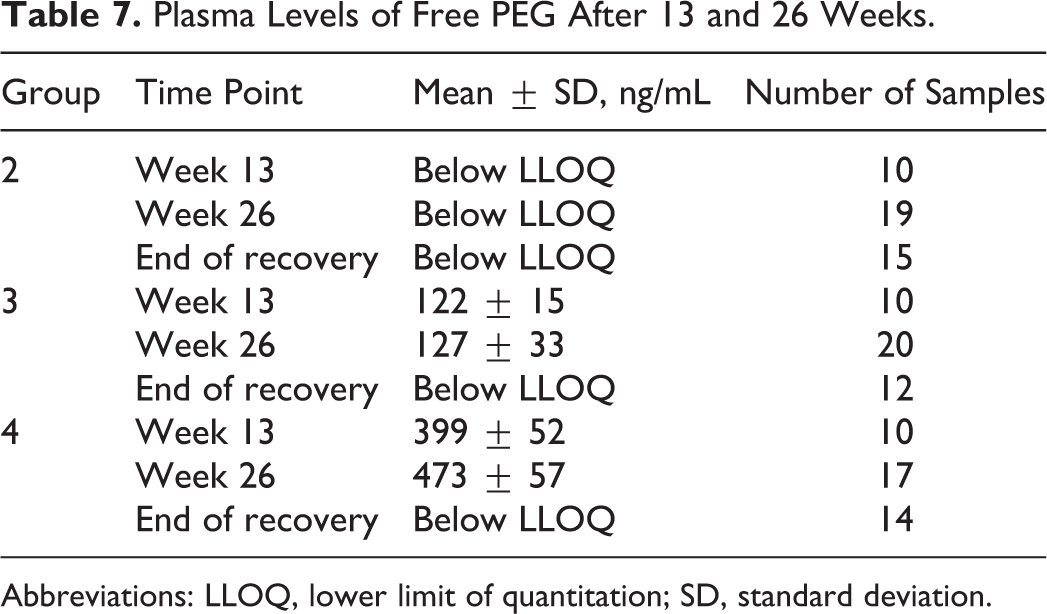
Abbreviations: LLOQ, lower limit of quantitation; SD, standard deviation.
Polyethylene Glycol in CSF
All CSF samples analyzed for PEG had values below the method LLOQ of 20 ng/mL.
Macroscopic and Microscopic Pathology
No BAY 94-9027-related macroscopic or microscopic findings were noted after 13 and 26 weeks or at the end of the recovery phase. The macroscopic findings observed were considered incidental, of the nature commonly observed in this strain and age of rat, and/or were of similar incidence in control and treated animals and, therefore, were considered unrelated to administration of BAY 94-9027. No test item–related organ weight changes were seen in any of the organs evaluated at the 3 necropsy time points.
No BAY 94-9027-related microscopic findings were noted (Figure 1). Only high-dose and control animals were evaluated by histology since no BAY 94-9027-related findings were seen that would have triggered an evaluation of the other dose groups.
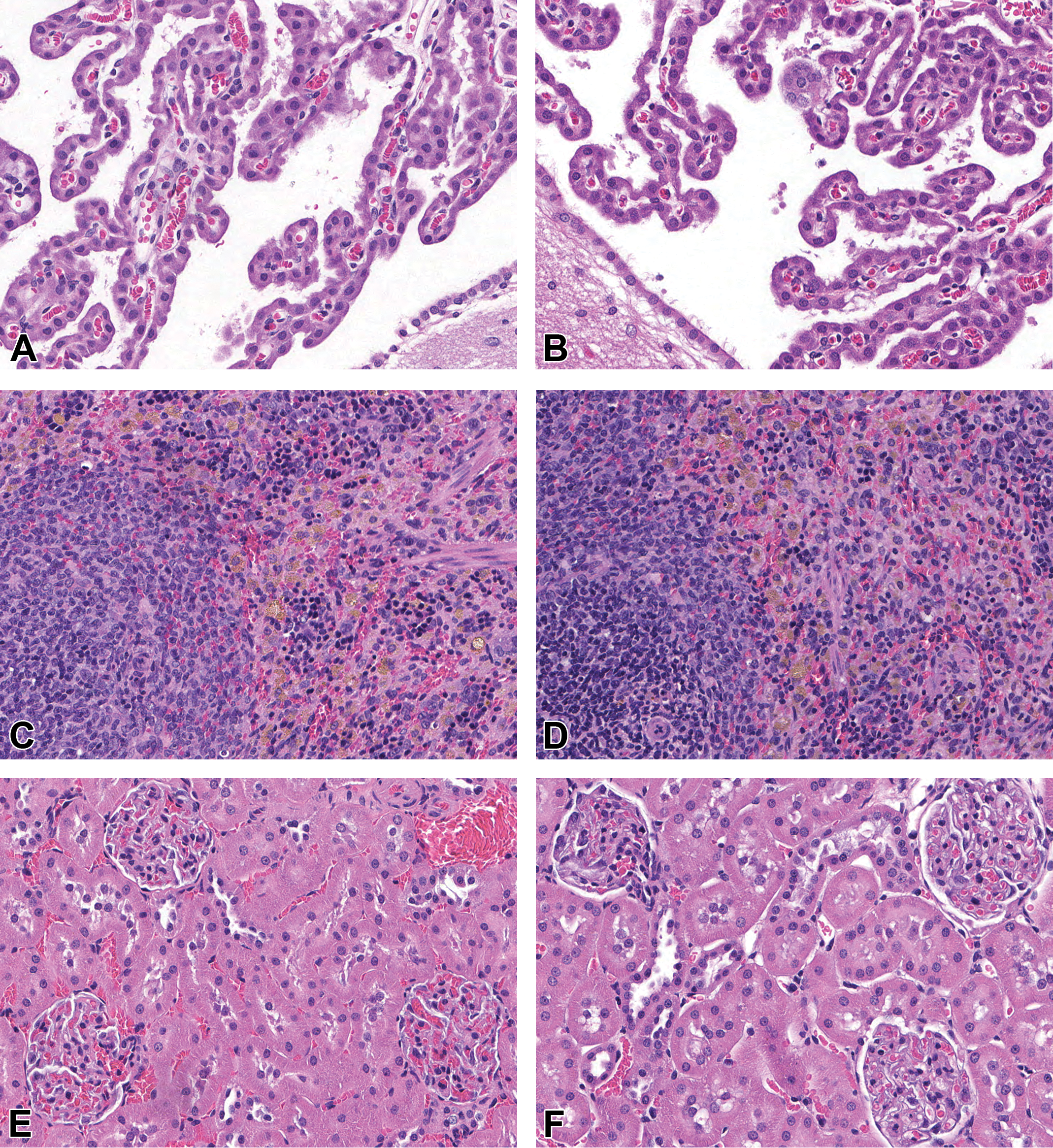
Histopathological evaluation after 13 and 26 weeks of treatment or at the end of the 26 weeks recovery period noted no BAY 94-9027-related microscopic findings up to the highest dose of 1200 IU/kg in any of the more than 40 organs or tissues evaluated per animal. Brain with the choroid plexus epithelia (A and B), spleen (C and D), and kidneys (E and F) from rats administered BAY 94-9027 at 1200 IU/kg twice weekly (B, D, and F) showed no indication of test item–related cellular vacuolation when compared to the controls given the vehicle (A, C, and E). Note the golden brown pigment in macrophages unrelated to BAY 94-9027 present in both controls and animals given BAY 94-9027 (C and D). Hematoxylin and eosin (H&E), original scan ×20.
Cellular vacuolation seen in some animals of all groups including those in the control group in cortical epithelial cells in the adrenal glands, tubules of the kidneys, and hepatocytes was considered normal morphologic variability for these tissues and not attributed to PEG.
In the brains of some animals including controls, free erythrocytes, pigmented macrophages (golden-brown/brown), and/or erythrophagocytosis were seen primarily in the third ventricular space and meninges and were considered related to the terminal cerebral spinal fluid collection at necropsy. At the end of the recovery period, no BAY 94-9027-related microscopic findings were noted.
Immunohistochemistry for PEG Detection
No positive immunoreaction for PEG was present in the following tissues examined: brain (choroid plexus [lateral, third, and fourth ventricle], cerebral cortex, corpus callosum, globus pallidus, hippocampus, thalamus, hypothalamus, pyramids, cerebellum, trigeminal nuclei and tracts, lateral [dentate] cerebellar nuclei, medulla oblongata, circumventricular organs and pineal gland), spleen, and kidney (Figure 2). In positive controls, a strong immunopositive reaction for PEG was observed in the expected structures (Figure 3). Negative controls were negative (data not shown).
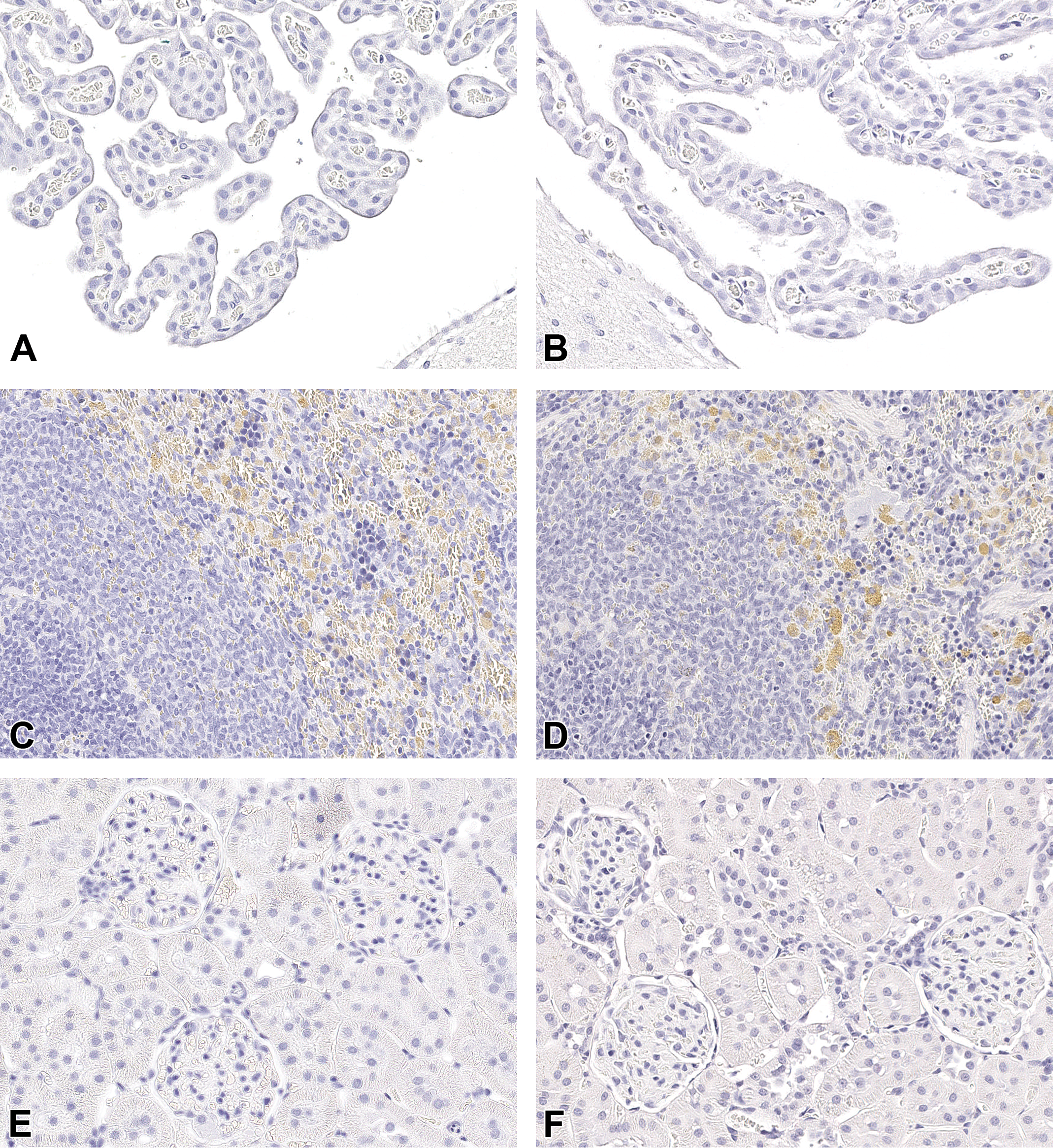
In the immunohistochemical evaluation, no PEG immunoreactivity could be detected after 13 weeks, 26 weeks, or at the end of the 26 weeks treatment-free period in the brain and in the choroid plexus epithelial cells (A and B), spleen (C and D), and kidneys (E and F) from rats administered vehicle (A, C and E) or BAY 94-9027 (B, D, and F). Note the brown background staining in macrophages unrelated to BAY 94-9027 present in both controls and animals given BAY 94-9027 (C and D). Immunohistochemistry (IHC), original scan 20×.
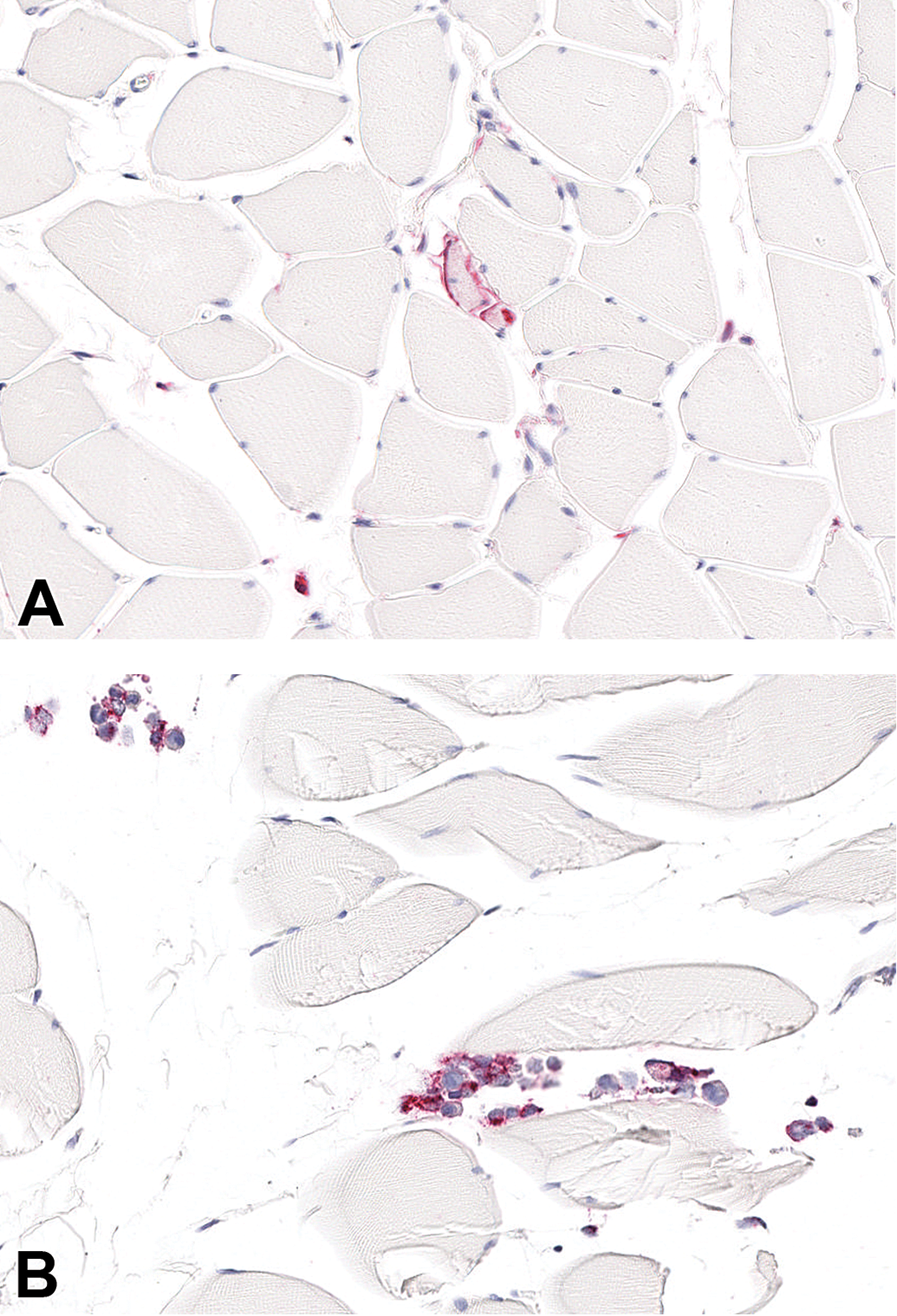
For the immunohistochemical assessment, positive and negative controls were evaluated and polyethylene glycol (PEG) staining (red) could be detected. Positive (A and B) and negative (pictures not shown) controls were included in each immunohistochemistry (IHC) run. Rat skeletal muscle injected postmortem with BAY 94-9027 directly (A) or cell culture suspensions containing THP-1 human macrophages preincubated with BAY 94-9027 for 21 hours (B) served as positive controls. A strong immunoreaction, visible as strong red staining, was present in the musculature injected with the test item (A) and in preincubated THP-1 cells (B). The negative control, native THP-1 macrophages, and the isotype control applied to skeletal muscle injected with the test item, preincubated THP-1 macrophages, and native macrophages were negative. Immunohistochemistry (IHC), original scan 20×.
Discussion
Safety of Large PEG Molecules Used in PEGylated Biopharmaceuticals
Concerns have been raised about the detection of PEG-related cellular vacuolation seen in toxicology studies with some PEGylated biopharmaceuticals. 13,21 Vacuoles were observed by histopathology in cells of the RES but also in some studies in pivotal organs such as kidneys and the choroid plexus. 13 –16,18,21 Recently published nonclinical studies and reviews with molecules of different PEG sizes either unbound 18 or conjugated to drug protein 13,14,19 have shown that as long as vacuolation is not associated with pathologic changes like tissue degeneration, inflammation, necrosis, and cellular distortion related to vacuolation, or changes in study end points including hematology, clinical chemistry, urinalysis, or organ weight vacuolation, cellular vacuolation is not considered to be adverse. It was shown that cellular vacuolation was dose and time dependent when testing either unconjugated or conjugated PEG molecule 13,18 and seen at higher doses than present in BAY 94-9027, which is in the microgram/kg range, 14 although comparing data between different PEGylated biotherapeuticals is difficult.
Rudmann et al 18 evaluated very high IV doses of linear unconjugated PEG 10, 20, or 40 kDa. Normalized to a weekly dose the PEG doses were 10 kDa—700 mg/kg/week; 20 kDa—350 mg/kg/week; and 40 kDa—200 mg/kg/week. The 3-month study in rats showed that the molecular weight of PEG influenced both the tissue distribution and the vacuolation. For 10 and 20 kDa PEG, PEG immunoreactivity was most prominent in the renal tubule epithelium, in alveolar macrophages, and hepatic Kupffer cells, while cellular vacuolation was absent. Rats given 40 kDa PEG had strong PEG immunoreactivity in splenic subcapsular red pulp macrophages, renal interstitial macrophages, and choroid plexus epithelial cells frequently associated with cellular vacuolation. No PEG IHC staining or vacuolation was detected in the brain parenchyma, suggesting that even at very high PEG doses the choroid plexus acts as a barrier between the blood and the ventricular CSF for PEGs. It is not known how far data from unconjugated PEG can be accurately extrapolated to PEGylated biopharmaceutics since vacuolation, distribution and excretion patterns of PEGylated biotherapeuticals may depend more on drug receptor/target-mediated cellular uptake or ligand-specific transport mechanisms, and therefore organs or tissues with vacuolation and the time and dose when this occurs can vary.
In an industry survey summarizing data from 17 PEGylated biopharmaceutics under development in 2013 found that vacuolation of the choroid plexus was seen also with 20, 30, and 40 kDa site-specific-PEGylated proteins. 13 Again, no PEG was detected in the brain parenchyma except in one case where the PEGylated molecule was designed to cross the blood–brain barrier (development of this molecule was stopped). 13
Long-term Safety of PEGylated Coagulation Factors
PEGylated rFVIII (turoctocog alfa pegol, N8-GP) with a 40 kDa branched PEG and PEGylated FIX (Refixia, nonacog beta pegol, N9-GP) using the same 40 kDa PEG were tested in the IDF chronic rat model. 29,30 With turoctocog alfa pegol, no cellular vacuolation was detected after 26 or 52 weeks of every fourth day dosing up to 1200 IU/kg (containing up to 0.024 mg PEG/kg/dose). In the chronic study with Refixia with 6 and 26 weeks of treatment, no light microscopic vacuolation was reported by the authors, but PEG was detectable in the choroid plexus and spleen macrophages by IHC.
The Refixia doses given every fifth day were 40 IU/kg (clinical dose; 0.2 mg PEG/kg/dose), 150 IU/kg (0.8 mg PEG/kg/dose), 600 IU/kg (2.7 mg PEG/kg/dose), and 1200 IU/kg (6.4 mg PEG/kg/dose). At the low dose of 40 IU/kg, IHC was positive for PEG in choroid plexus of about 50% of examined animals after 26 weeks, while the high dose of 1200 IU/kg showed positive IHC after 6 weeks of treatment.
N9-GP, turoctocog alfa pegol (N8-GP), and BAY 94-9027’s distribution to the blood stream as blood coagulation proteins may differ to many of the other PEGylated biopharmaceutics, which usually have a drug target receptor or bind in organs/issues. Factor VIII and also BAY 94-9027 are usually bound to van Willebrand Factor (vWF) in circulation. The only receptors known for FVIII are clearance receptors in the liver although the pathways that regulate the clearance of vWF-bound and vWF-free FVIII are not well characterized. 34,35 It is likely that BAY 94-9027 including its PEG-60 will be removed by the liver, the protein part is degraded intracellularly, while PEG 60 kDa remains and is excreted or recirculated. 24 It was shown that unconjugated PEG-60-Mal-Cys is excreted mainly via the kidney. 24
It can be assumed that a small amount of PEG 60 kDa from BAY 94-9027 may be taken up also in the liver as was shown with PEG 40 kDa 36 for N8-GP and may be excreted in bile although this has not been confirmed. The small difference in organ distribution seen between conjugated and unconjugated PEG 40 kDa did not have any impact on the liver function or morphology as seen from the nonclinical studies since no vacuolation in any of the liver cell types including Kupffer cells occurred with BAY 94-9027, turoctocog alfa pegol, and Refixia in the chronic IDF rat model.
The PEG doses in Refixia in the chronic rat study were distinctly higher than those tested here for BAY 94-9027. They were a multiple of 100 to 3200 over a clinical dose of 0.002 mg/kg PEG in BAY 94-9027. The findings with turoctocog alfa pegol show that PEG doses in the range of 0.024 mg PEG/kg/dose giving as multiples of approximately 12 over the clinical dose of BAY 94-9027 are safe when given over 52 weeks. In the chronic study with BAY 94-9027, a dose of 0.04 mg PEG/kg/dose or up to 20 times the clinical dose of BAY 94-9027 was shown safe for chronic treatment.
Safety of BAY 94-9027
The toxicology program for BAY 94-9027 evaluated the systemic, juvenile, long-term, and genotoxic properties of BAY 94-9027 or its PEG 60 kDa moiety. There was no thrombus formation in rats or rabbits up to the highest dose of 2250 IU/kg (>30 times the maximal human dose) in the 2-week studies. Thrombus formation or other reactions in the coagulation system could be a potential effect at high doses when FVIII is activated to FVIIIa. In the systemic and juvenile toxicity studies with BAY 94-9027 or PEG-60-Mal-Cys, there was no cellular vacuolation and no toxicity seen.
The chronic toxicity of BAY 94-9027 was assessed in the IDF rat. These animals mount a no humoral immune response to species-foreign proteins such as FVIII. They can therefore be treated longer than immune-competent animals although due to the special housing requirements (pathogen free) and the sensitivity to infections, this model can only be utilized on a case-by-case basis.
No signs of toxicity were seen with BAY 94-9027 in the chronically treated IDF rats with twice-weekly dosing of up to 1200 IU/kg for 26 weeks. The number of animals that died in the highest dose group of 1200 IU/kg (4 out of 45 over the course of 52 weeks on study) was slightly higher than in the other dose groups or control (1 or 2 animals out of 45). These deaths are not attributed to treatment with BAY 94-9027. All animals that died in the highest dose group showed pathologies that can be attributed to background changes. It is concluded that there was no BAY 94-9027-related mortality, adverse clinical observations, body weight or food consumption changes, or ophthalmology observations. No toxicologically important differences were noted in hematology, clinical chemistry, or urinalysis parameters. There were no BAY 94-9027-related macroscopic or microscopic findings or organ weight changes at weeks 13 and 26 and through the recovery portion of the study. No PEG-related cellular vacuolation was seen in any of the > 40 organs and tissues (including macrophages) evaluated histologically. Toxicokinetic assessment revealed good exposure in all animals except 1 animal dosed with 400 IU/kg that had also a positive ADA titer.
There was no accumulation of PEG in pivotal organs (brain including choroid plexus, kidney, or spleen) as demonstrated by the lack of a positive immunoreaction to anti-PEG monoclonal antibody by IHC. Cerebrospinal fluid samples analyzed from all time points of all dose groups with a sensitive assay were negative for PEG supporting the IHC results. The concentrations of PEG in plasma were not measurable in the low-dose group (40 IU/kg) and increased with dose in the group 3 (400 IU/kg) and group 4 (1200 IU/kg) rats. In the highest dose group, the mean PEG concentration measured in plasma 72 hours after last dose on week 26 was 473 ± 57 ng/mL in the rats. No PEG was measured in plasma after the 26-week treatment-free period.
Toxicokinetics of BAY 94-9027 and PEG in plasma in the IFD rats were comparable to the results from the experiments with euthymic rats (data not shown), indicating no relevant differences in exposure to BAY 94-9027 between the different strains. It is reported 32 that the number of granulocytes, monocytes/macrophages, erythrocytes, B cells, and natural killer (NK) cells in the blood of IDF rats were usually within the normal range; the innate immune system in IDF rats is well developed. It can be concluded that macrophage function and the role of the RES in PEG clearance are likely comparable between IDF rats and immunocompetent animals and that the chronic study with BAY 94-9027 is an adequate model for long-term treatment and risk assessment for humans.
In combining sensitive methods evaluating potential PEG presence in organs and tissues (IHC), and evaluating the plasma and CSF concentrations in combination with standard H&E histopathology technique, the chronic study showed that BAY 94-9027 did not induce PEG-related changes at multiples of the human dose after chronic treatment.
Conclusions
The nonclinical toxicology program supports that BAY 94-9027 is safe when given on demand and prophylactically as chronic treatment. There were no toxicities seen related to the rFVIII protein or the PEG moiety in any of the studies. Specifically, no cellular vacuolation or PEG accumulation was detectable up to high multiples of the clinical dose and after chronic dosing. The NOAEL in the chronic rat toxicity study was the highest dose of 1200 IU/kg or 0.04 mg PEG/kg given twice per week.
Footnotes
Acknowledgments
We are grateful to Dr. Claudia Stark for her toxicology expertise, critical review and helpful comments to the manuscript. Further, we thank the many people who contributing to the experimental data.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
