Abstract
Colorless, intracytoplasmic vacuoles occur in multiple tissues in animals following repeated administration of polyethylene glycol (PEG)-conjugated molecules. The extent of vacuolation depends on physical characteristics and molecular backbone of the PEG and the dose, product, drug target/pharmacology, and duration of exposure. The collective experience gathered from multiple nonclinical toxicology studies of PEGylated biopharmaceuticals indicates that in general, PEG-related vacuolation is not associated with demonstrable cell and tissue damage or dysfunction and is reversible with sufficient duration of drug-free periods. Existing data are insufficient to predict whether nonclinical animal species differ in their sensitivity to develop PEG-associated vacuoles; however, recent data suggest that there may be species differences. Recent comprehensive reviews have addressed the basic challenges in developing PEGylated pharmaceutical products, including general reference to and description of PEG-associated tissue findings. These manuscripts have identified gaps in our current understanding of PEG-associated vacuolation, including the lack of a widely accepted standardized histological terminology and criteria to record and grade the severity of vacuolation as well as insufficient knowledge regarding the nature of the contents of these vacuoles. The goal of this article is to help address some of the gaps identified above by providing points to consider, including a pictorial review of PEG-associated microscopic findings, when evaluating and reporting the extent, severity, and significance (adversity or lack of adversity) of PEG-associated cytoplasmic vacuolation in safety assessment studies.
Note: This Points to Consider article is a product of a Society of Toxicologic Pathology (STP) Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP). It has been reviewed and approved by the SRPC and Executive Committee of the STP, but it does not represent a formal best practice recommendation of the Society; rather, it is intended to provide key “points to consider” in designing studies or interpreting data from toxicity and safety studies intended to support regulatory submissions. The opinions expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the editor.
Approved polyethylene glycol (PEG)-containing therapeutic products are of three general types. The first employs unconjugated PEG itself as the therapeutic product, such as the oral laxative MiraLAX®. The second type employs unconjugated PEG as an excipient for a purified biopharmaceutical, such as the immunoglobulin replacement therapy Gammagard®. The third type is produced by covalent attachment of PEG, in various conformations and sizes, to a biopharmaceutical (commonly proteins, peptides, or oligonucleotides) to extend the circulating half-life or alter its immunogenicity (Harris and Chess 2003; Turecek et al. 2016). The latter is referred to as PEGylated biopharmaceuticals herein. Examples of PEGylated biopharmaceuticals include Adagen®, Cimzia®, Krystexxa®, Neulasta®, Pegasys®, and Rebinyn®. The PEG moieties used for conjugation are typically of molecular weight (MW) ≥ 5 kDa and are unlikely to be metabolized to any significant degree in mammals. Instead, clearance is largely through glomerular filtration and excretion in the urine and/or uptake by phagocytes or other cells, depending on the size of the combined PEG-biopharmaceutical construct and/or the geometric complexity of the PEG side chain (Webster et al. 2007; Xu et al. 2013).
The principal light microscopic changes in paraffin-embedded, hematoxylin and eosin (H&E)-stained tissue sections attributed to the repeated administration of PEG alone (Fruijtier-Pollöth 2005) or PEG conjugated to another molecule (Bendele et al. 1998; Veronese and Mero 2008; Jain and Jain 2008; Ivens et al. 2013, 2015; Kaufman, Conover, and Buchbinder 2014; Turecek et al. 2016) have been characterized as cytoplasmic vacuolation of various tissues and cells. Vacuole formation is dose dependent (Kaufman, Conover, and Buchbinder 2014; Rasmussen et al. 2016; Forest et al. 2017) and product dependent (Stidl et al. 2016; Byrd et al. 2017), since vacuolation is not observed for all PEG-conjugated molecules. The observed vacuoles are single or multiple, clear, colorless, variably sized, and generally round in shape and are reported to arise mainly from endocytic sequestration of PEG-linked molecules within lysosomes (Bendele et al. 1998), although binding of PEG to the polar heads of membrane phospholipids has also been posited based on in vitro experiments using endothelial cells (Beckman et al. 1988). The exact contents of these vacuoles, in vivo or ex vivo, have not been comprehensively elucidated. However, these may contain variable amounts of PEG as has been demonstrated using anti-PEG immunohistochemical (IHC) procedures (Rudmann et al. 2013; Kronenberg et al. 2013; Ivens et al. 2015). Some authors have indicated that over time, vacuoles may increase in number and/or size (Bendele et al. 1998; Bolon et al. 2001); it is unknown whether this can be generalized to all PEGylated biopharmaceuticals and across all cell types. Phagocytic cells, such as macrophages, are a frequently affected cell type; however, other cells and tissues may also exhibit vacuolation. Recent publications have highlighted that the distribution and occurrence of PEG-associated vacuolation may also depend on the species of animal to which the PEGylated biopharmaceutical was administered (Ivens et al. 2015; Forest et al. 2017).
The degree of vacuolation produced by PEG-conjugated biopharmaceuticals is in large part associated with the physical properties of the PEG moiety as revealed by investigative studies (Bendele et al. 1998; Bolon et al. 2001; Rudmann et al. 2013; Xu et al. 2013). For example, the number and size of PEG-associated vacuoles within renal tubular epithelial cells was highest for proteins conjugated to a single linear, low–molecular weight PEG (Bolon et al. 2001). The size and number of renal cytoplasmic vacuoles may be reduced by any of several modifications. Examples include boosting the PEG size (Bendele et al. 1998; Bolon et al. 2001), increasing the PEG complexity (by using branched rather than linear PEG, Bolon et al. 2001; Xu et al. 2013), altering the PEG conjugation site (from the molecule terminus to elsewhere, thereby increasing the molecular diameter of the final product, Bolon et al. 2001), and/or attaching two PEGs to the same biopharmaceutical (Bolon et al. 2001).
In general, PEG-associated cytoplasmic vacuolation has been considered an adaptive, nonadverse finding. This interpretation is supported by the lack of microscopic findings and/or changes in circulating biomarkers indicative of degeneration or dysfunction in affected cells and tissues from many nonclinical toxicity studies of PEGylated biopharmaceuticals (Rudmann et al. 2013; Ivens et al. 2015; Turecek et al. 2016). Nonetheless, the increasing number and complexity of PEGylated materials currently in development or being marketed, and particularly the potential need for lifelong treatment with such agents, has led drug regulatory agencies (DRAs) to pay close attention to the biodistribution, disposition, and long-term impact of PEGylated products (European Medicines Agency [EMA] 2012a; Peyvandi 2015; Ivens et al. 2015). Recent reviews have addressed the basic challenges in developing PEGylated pharmaceutical products, including general reference to and description of the aforementioned PEG-associated tissue findings (Ivens et al. 2015; Turecek et al. 2016). Gaps remain in our current understanding of PEG-associated tissue vacuolation, including the lack of widely accepted standardized histological criteria to record and grade the severity of vacuolation as well as insufficient knowledge regarding the exact mechanism(s) of vacuole formation and the nature of their contents (Ivens et al. 2015). The Scientific and Regulatory Policy Committee of the Society of Toxicologic Pathology (STP) commissioned a Working Group with the remit of surveying and further characterizing features of PEG-associated cellular vacuolation, including advice regarding possible schemes for microscopic severity grading. The goal of this article is to help address some of the previously identified gaps with an expanded pictorial review of PEG-associated microscopic findings and providing points to consider when evaluating and reporting the extent, severity, and significance/adversity of cytoplasmic vacuolation consequent to the administration of PEGylated biopharmaceuticals in safety assessment studies.
Points to Consider
Pictorial Review of PEG-associated Tissue Vacuolation
In nonclinical toxicity studies, PEGylated biopharmaceutical products have been associated with vacuolation in various cell types and in numerous tissues (Table 1). Macrophages are predominantly affected. However, other cell types that have been identified frequently as affected are various epithelial cells, particularly of the renal proximal tubules and the choroid plexus of the brain.
Types of Tissues and/or Cells Known to Be Susceptible to PEG-associated Vacuolation.

Note: PEG = polyethylene glycol.
Important documentary sources of information regarding PEG-associated microscopic vacuolation include publicly available reviews by global DRAs, including the U.S. Food and Drug Administration (FDA) and European Medicines Agency. A limitation of these sources is the lack of images that demonstrate a representative spectrum of PEG-associated changes in tissues. Peer-reviewed manuscripts will sometimes provide images of PEG-associated findings, but these illustrations are usually limited in number and scope. To augment the availability of representative images in the scientific literature and thus facilitate the discussion of appropriate terminology/descriptors and severity grades, we herein provide a compendium of images demonstrating the characteristic appearance, distribution, and extent of PEG-associated vacuolation in selected tissues of nonhuman primates, rodents, and dogs from nonclinical toxicity studies conducted during the development of PEGylated biopharmaceuticals (Figures 1 –10).
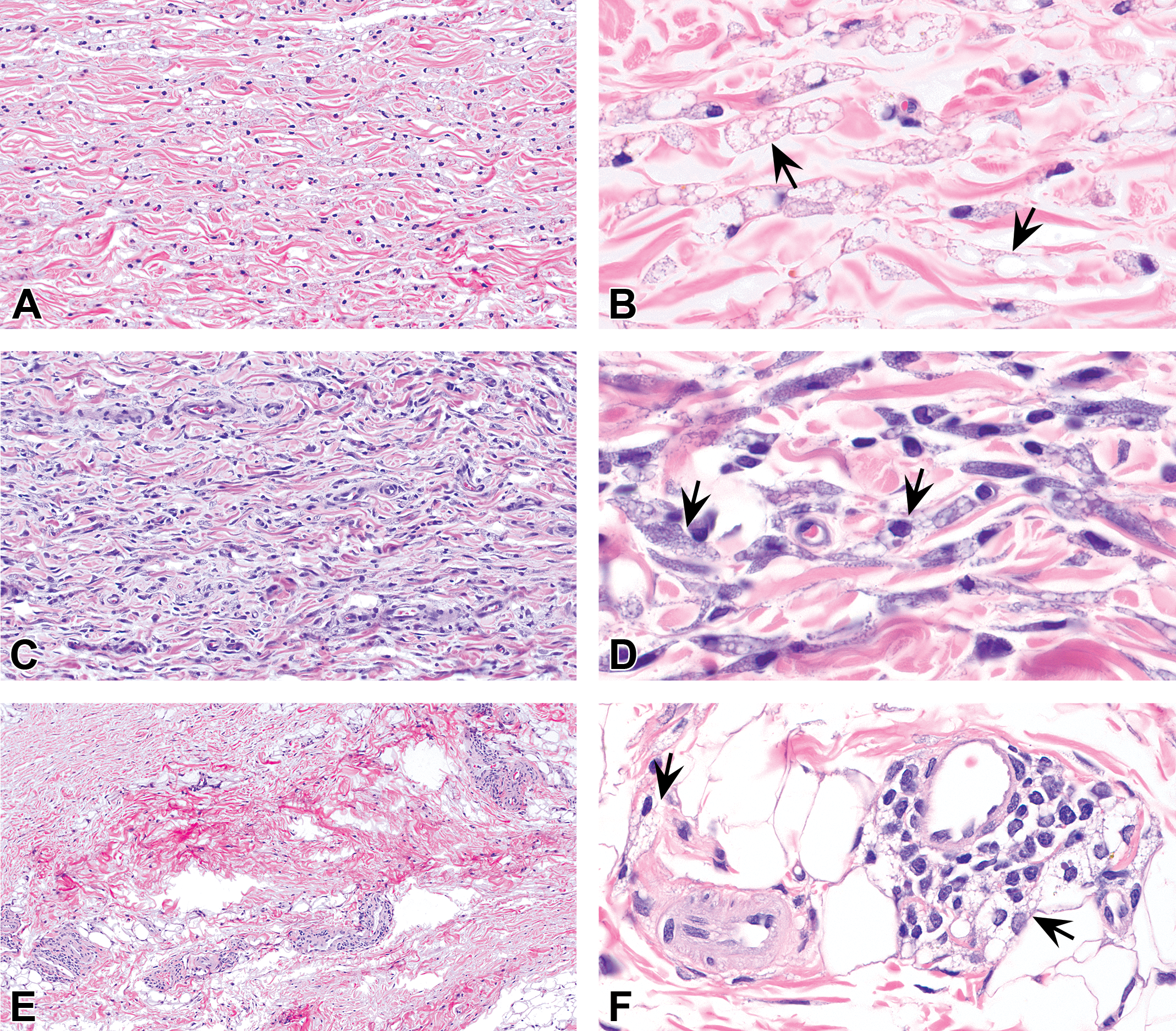
Subcutaneous injection sites from rats (A–D) and cynomolgus monkeys (E–F) administered PEGylated biopharmaceuticals. Vacuolation may manifest as infiltrates of vacuolated macrophages in the subcutaneous tissue (arrows). The macrophages may have variably sized, clear, colorless cytoplasmic vacuoles and basophilic-staining cytoplasm. Panels E and F show infiltrates of macrophages located next to small blood vessels. Panels B, D, and F are higher magnifications of A, C, and E, respectively. Hematoxylin and eosin stain. PEG = polyethylene glycol.
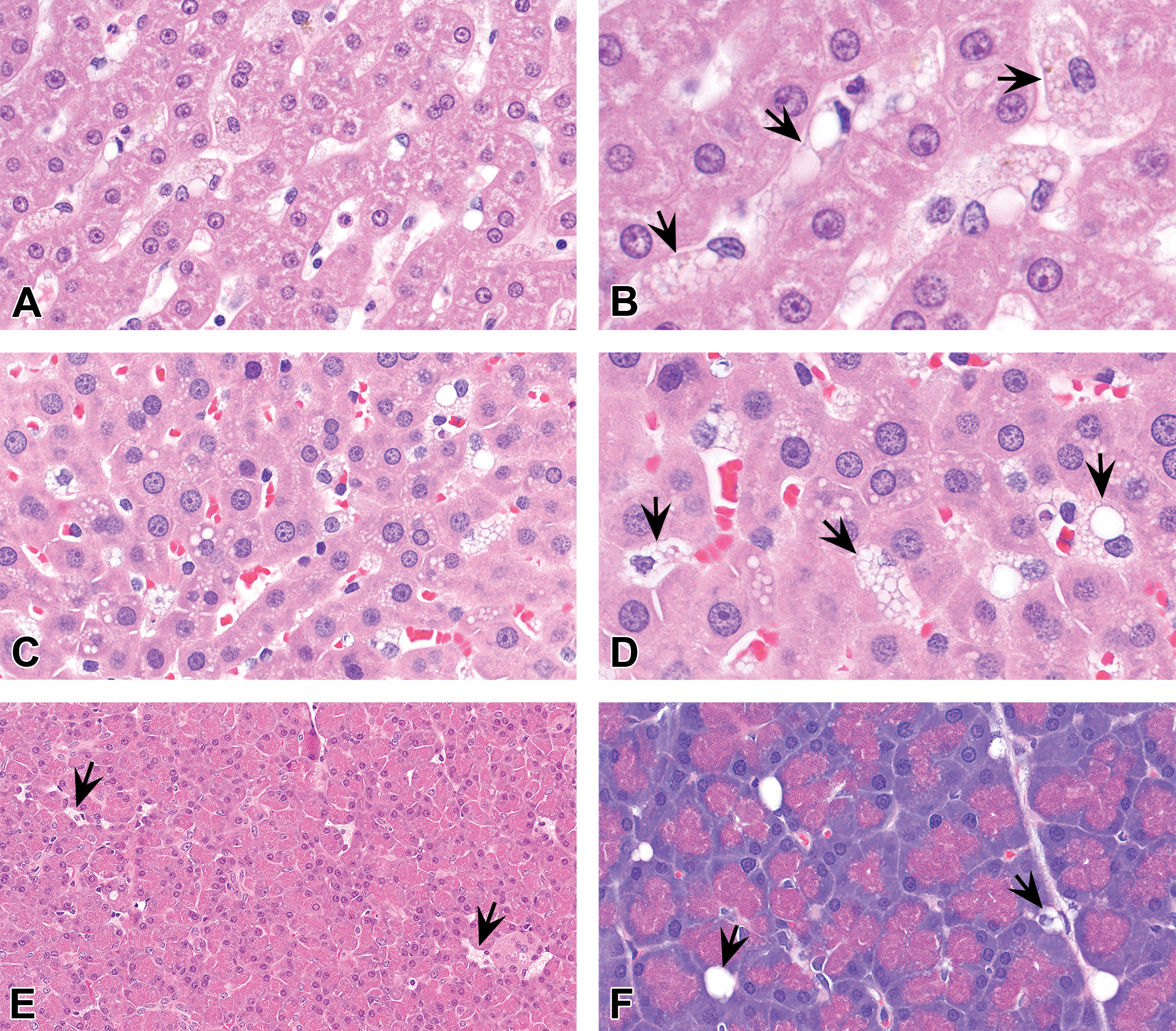
Liver (A–D) and pancreas (E–F) from cynomolgus monkeys (A–B, E) and rats (C–D, F) administered PEGylated biopharmaceuticals. Vacuolation may manifest as vacuolated sinusoidal macrophages (Kupffer) cells and/or infiltrates of macrophages (arrows) in these organs. Cytoplasmic vacuolation in hepatocytes was comparable to that present in control animals receiving vehicle and so was not deemed to be due to exposure to PEG. B and D are higher magnifications of A and C, respectively. Hematoxylin and eosin stain. PEG = polyethylene glycol.
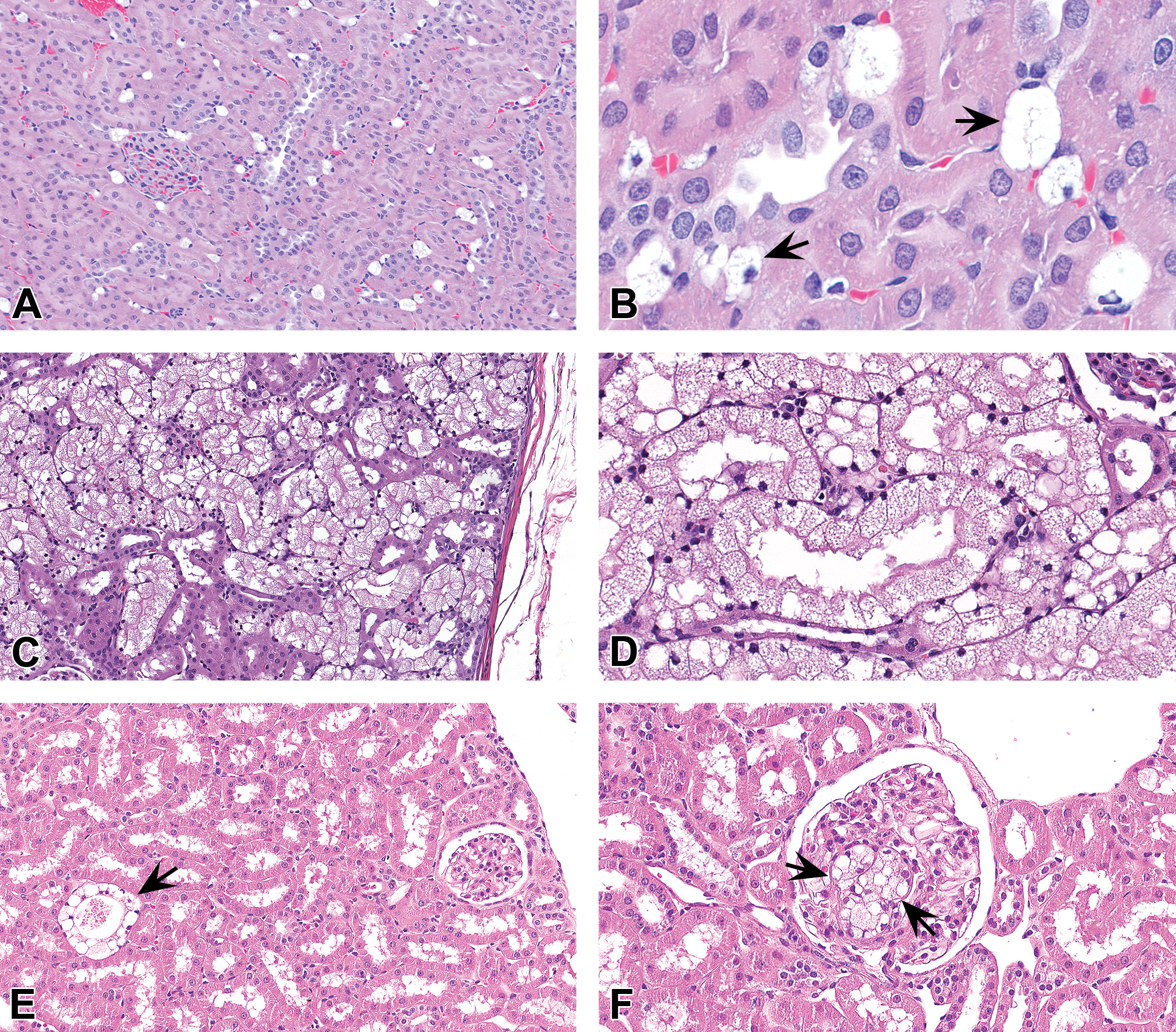
Kidney from rats (A-B) or cynomolgus monkeys (C-F) administered PEGylated biopharmaceuticals. Vacuolation may manifest as vacuolated macrophages in the interstitium (A–B) or glomeruli (F), or as vacuolated tubular epithelium (C–E) (arrows). B and D are higher magnifications of A and C respectively. Hematoxylin and eosin stain. PEG = polyethylene glycol.
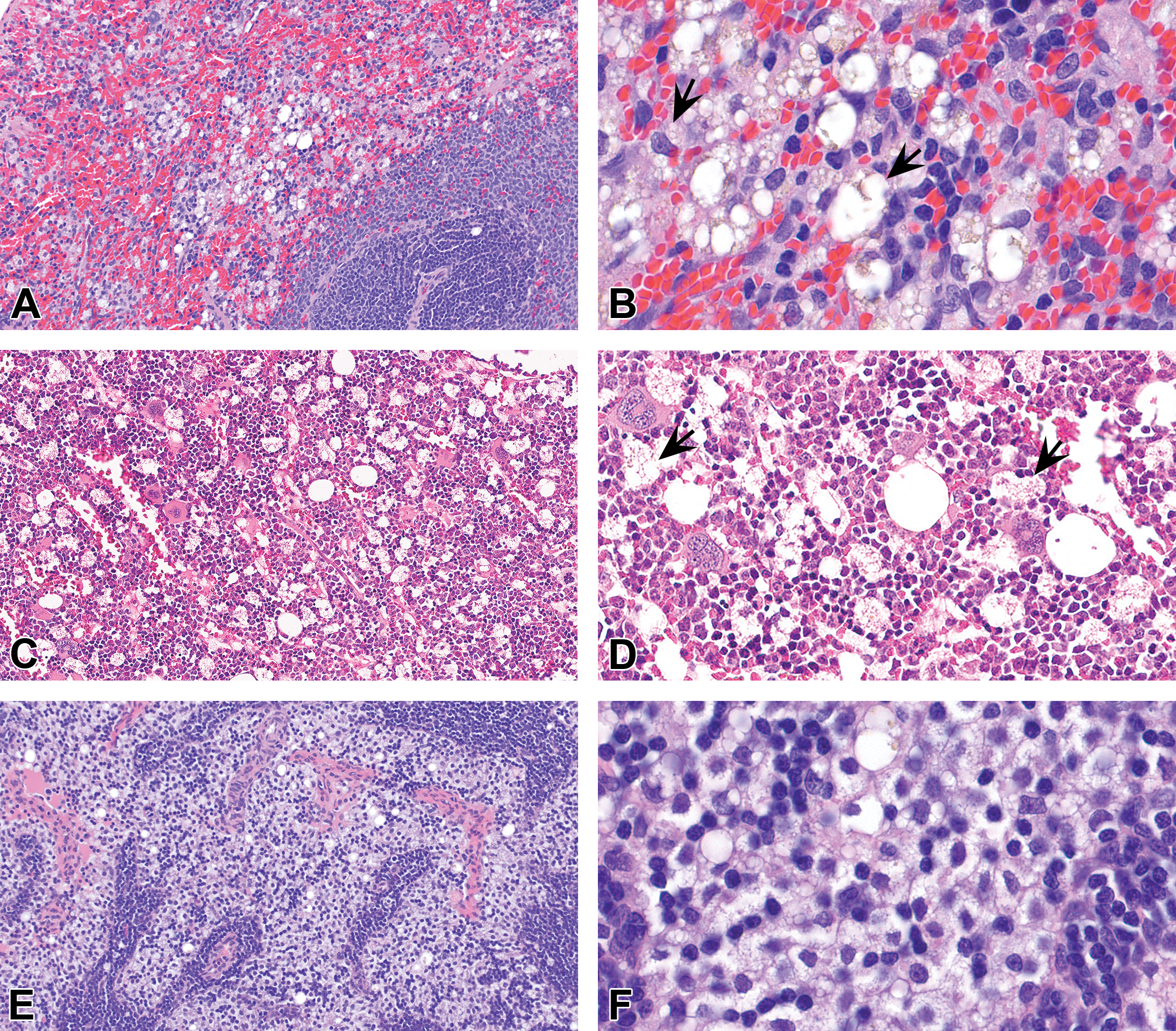
Spleen (A–B), bone marrow (C–D), and lymph node (E–F) from rats (A–B) or cynomolgus monkeys (C–F) administered PEGylated biopharmaceuticals. Vacuolation in these sites typically manifests as vacuolation of macrophages (arrows). B, D, and F are higher magnifications of A, C, and E respectively. Hematoxylin and eosin stain. PEG = polyethylene glycol.
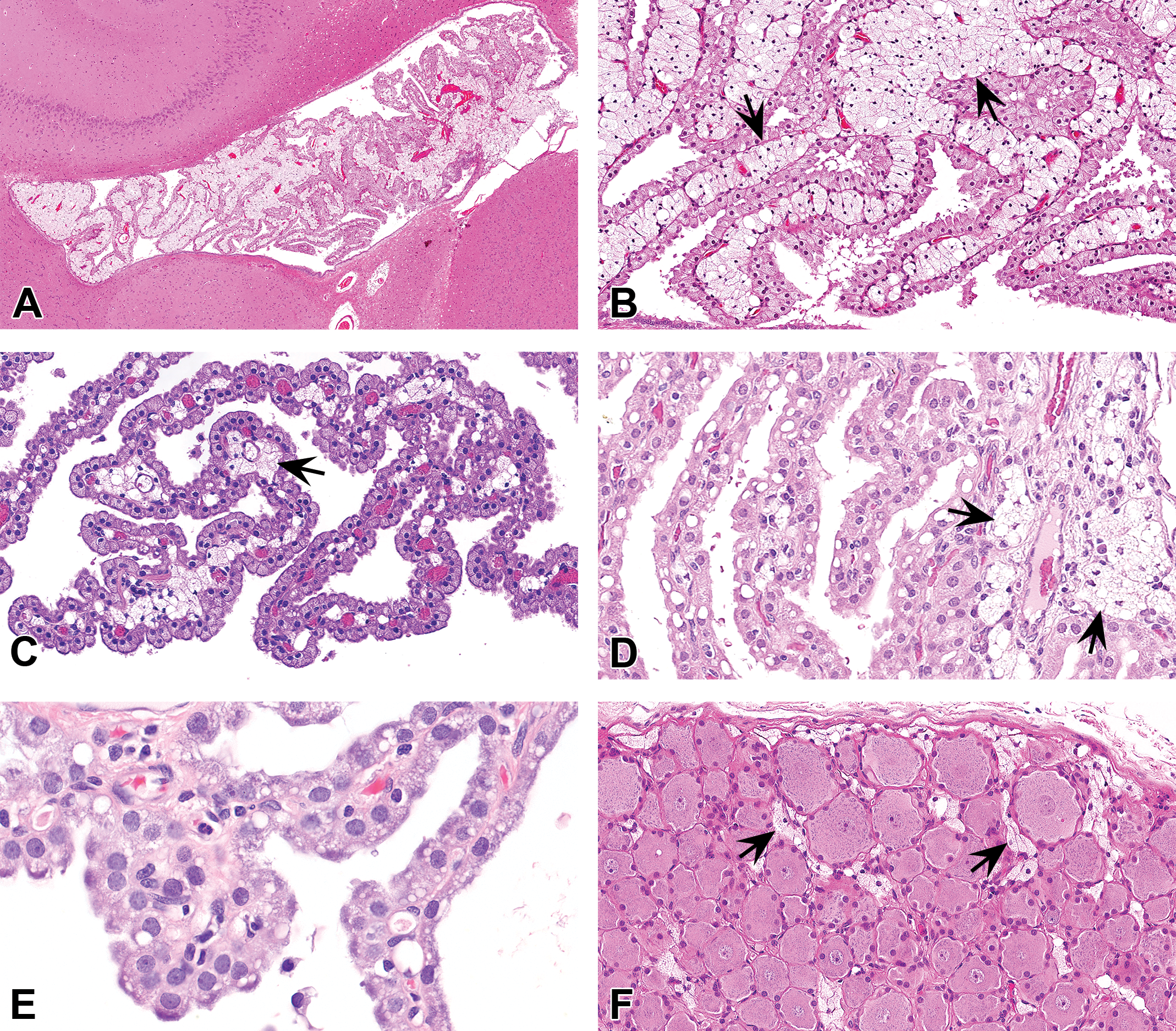
Brain (A–E) and dorsal root ganglion (F) from cynomolgus monkeys (A–D, F) and rats (E) administered PEGylated biopharmaceuticals. In the brain, PEG-related vacuolation typically manifests as vacuolation of the choroid plexus epithelium (CPE), sometimes with concurrent infiltration of vacuolated macrophages (arrows) in the interstitium of the choroid plexus (CP; A–D) or as vacuolation of the CPE alone (E). Vacuolated macrophages may occur in sufficient numbers to expand the interstitium of the CP (B) or ganglia (F). B is a higher magnification of A. Hematoxylin and eosin stain. PEG = polyethylene glycol.
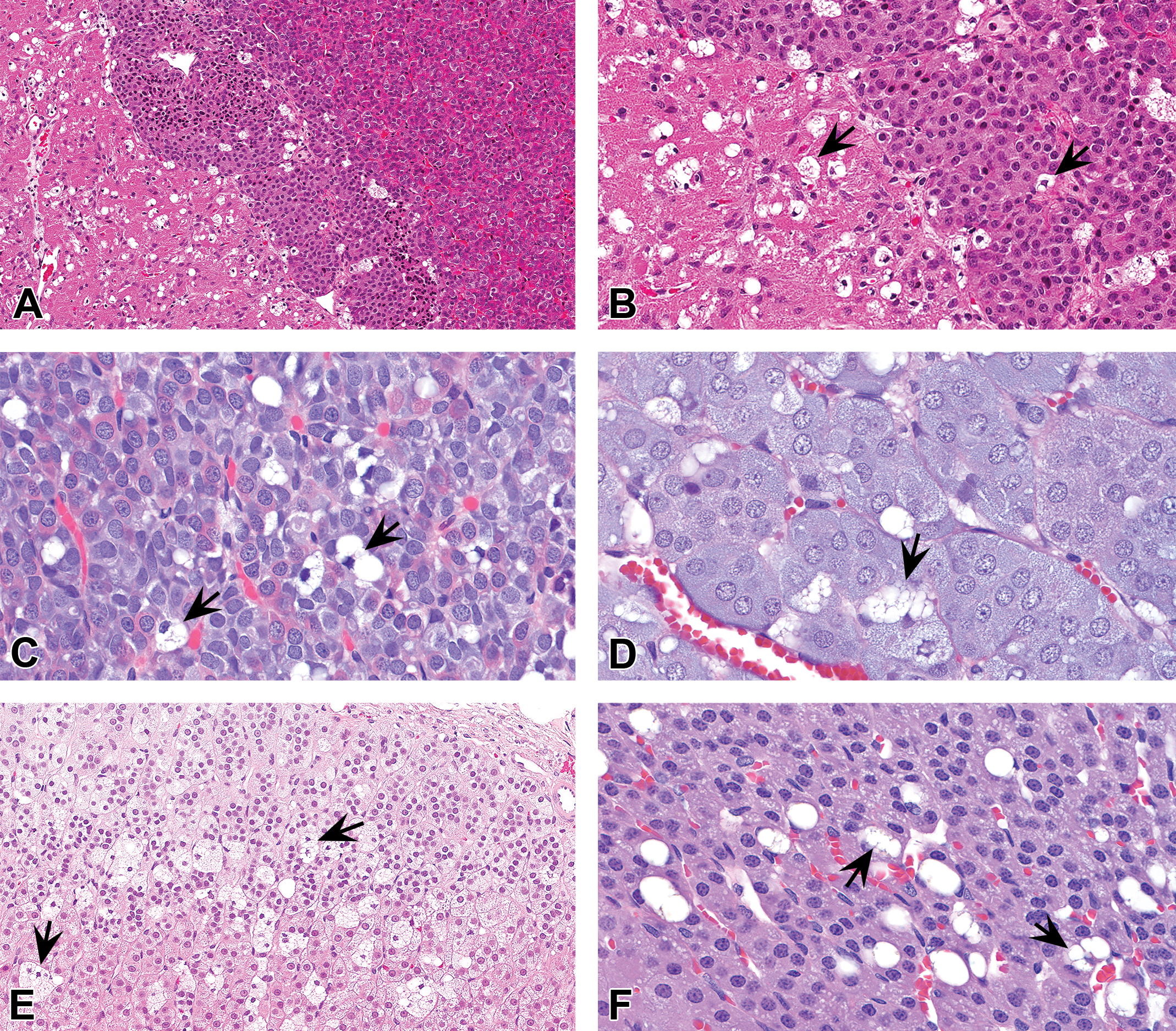
Pituitary gland (A–C) and adrenal gland (D–F) from cynomolgus monkeys (A–B, E) or rats (C–D, F) administered PEGylated biopharmaceuticals. Vacuolation (arrows) may manifest as infiltrates of vacuolated macrophages in these organs and/or vacuolation of parenchymal cells. B is a higher magnification of A. Hematoxylin and eosin stain. PEG = polyethylene glycol.
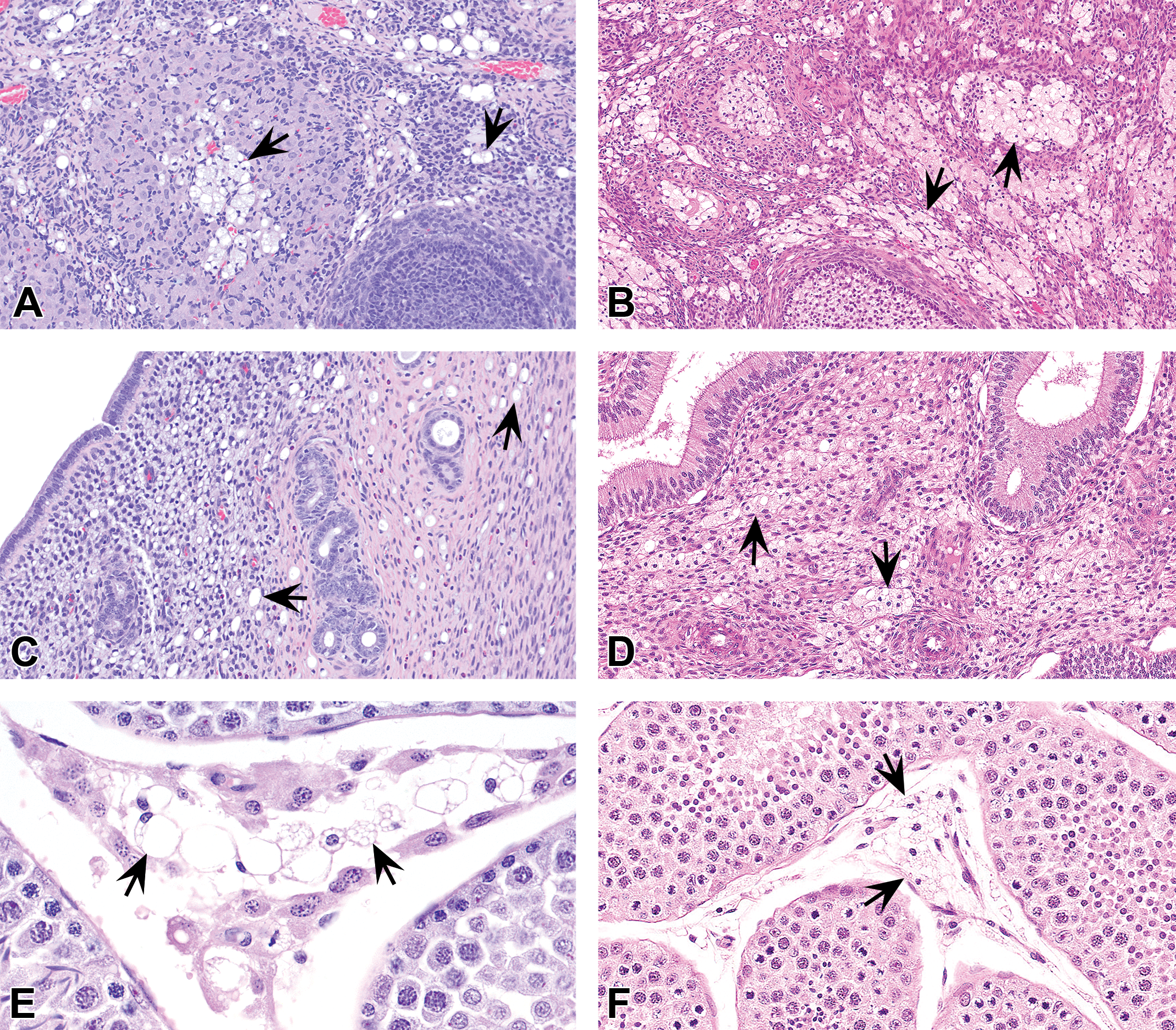
Ovary (A–B), uterus (C–D), and testes (E–F) from rats (A, C, and E) and cynomolgus monkeys (B, D, and F) administered PEGylated biopharmaceuticals. Vacuolation (arrows) may manifest as either infiltrates of vacuolated macrophages in these organs and/or vacuolar changes in other cells such as the parenchymal epithelium (F; vacuolated macrophages or Leydig’s cells). Hematoxylin and eosin stain. PEG = polyethylene glycol.
Eye (A), urinary bladder (B), or heart (C–D; atrium and valve, respectively) from cynomolgus monkeys (A–C) and a rat (D) administered PEGylated biopharmaceuticals. Vacuolation may manifest as infiltrates of vacuolated macrophages in these organs. In panel A, vacuolated cells are present (arrows) in the choroid. In panel B there are vacuolated macrophages in the submucosal interstitium of the urinary bladder (arrow). In panel C, vacuolation is present in the epicardial mesothelium (arrowheads) concurrently with many vacuolated macrophages in the myocardial interstitium (arrows). In panel D, vacuolated macrophages (arrow) have accumulated in an atrioventricular valve. Hematoxylin and eosin stain. PEG = polyethylene glycol.
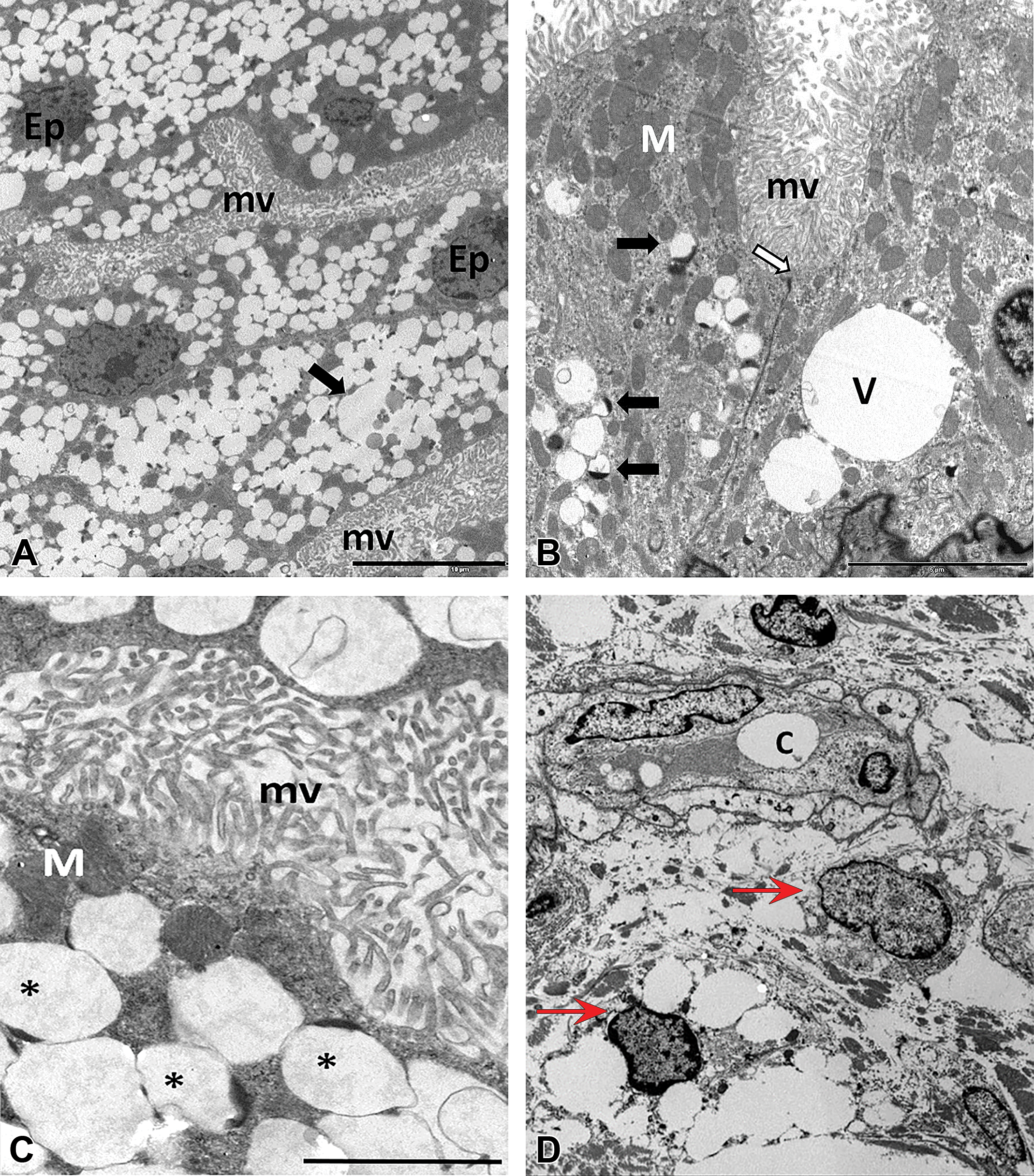
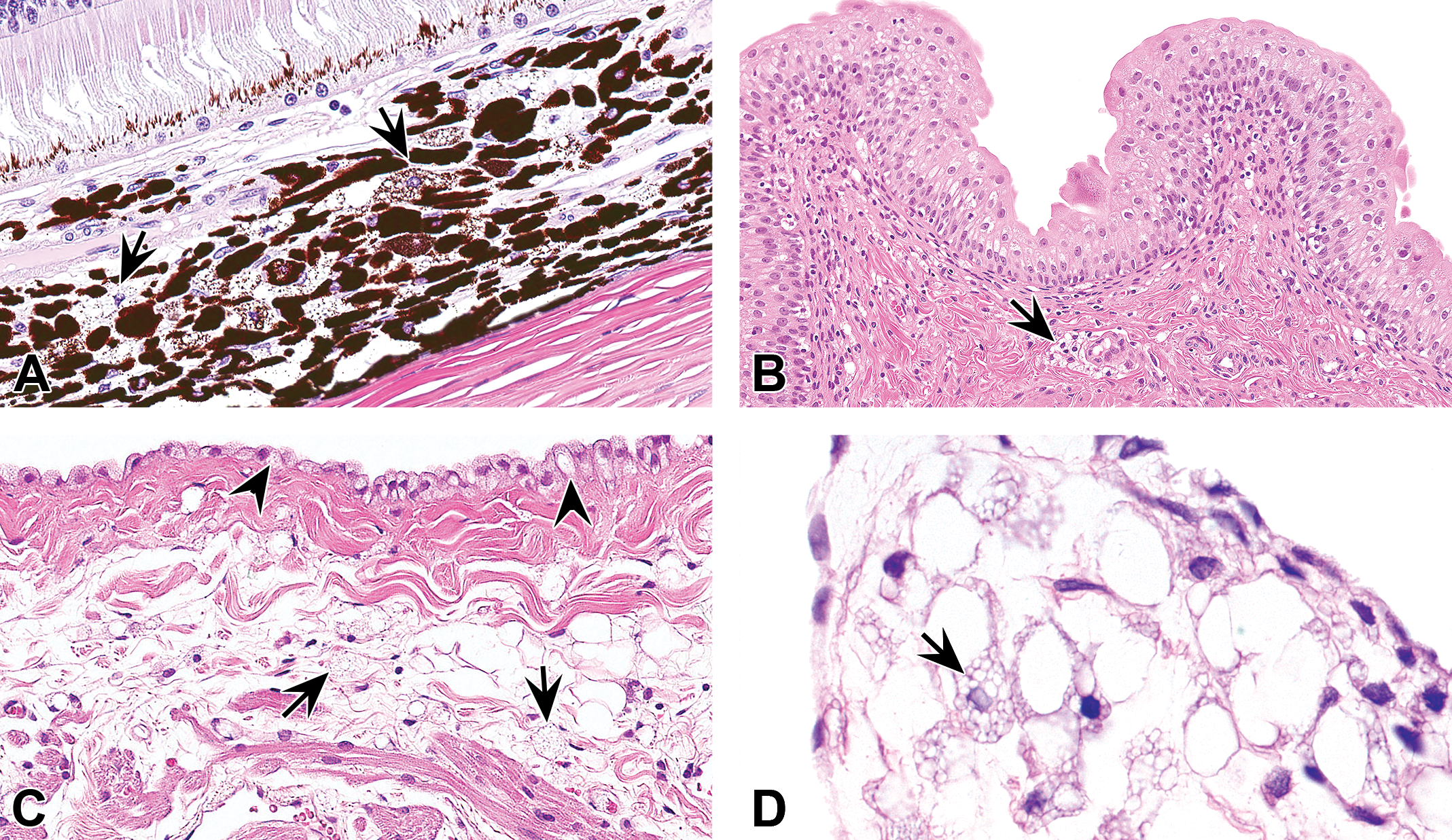
Transmission electron micrographs of choroid plexus epithelium (CPE) of cynomolgus monkeys (A–C) or subcutaneous tissue of a rat (D) administered PEGylated biopharmaceuticals. PEG-related TEM changes were characterized by electron-lucent and membrane-bound small and large cytoplasmic vacuoles (V) in the CPE cells (Ep). Occasionally, vacuoles contained highly electron-dense material eccentrically displaced to one side (black arrows or *). Bar size in A = 10 µm, B = 5 µm, C = 2 µm. Ep = CPE, mv = microvilli, V or C = vacuole, M = mitochondria, white arrow = cell–cell junction; red arrows = nuclei of vacuolated macrophages. 2.5% glutaraldehyde fixation, postfixation in 2% osmium tetroxide in cacodylate buffer, and embedding in epon-araldite. Uranyl acetate and lead citrate. PEG = polyethylene glycol; TEM = transmission electron microscopy.
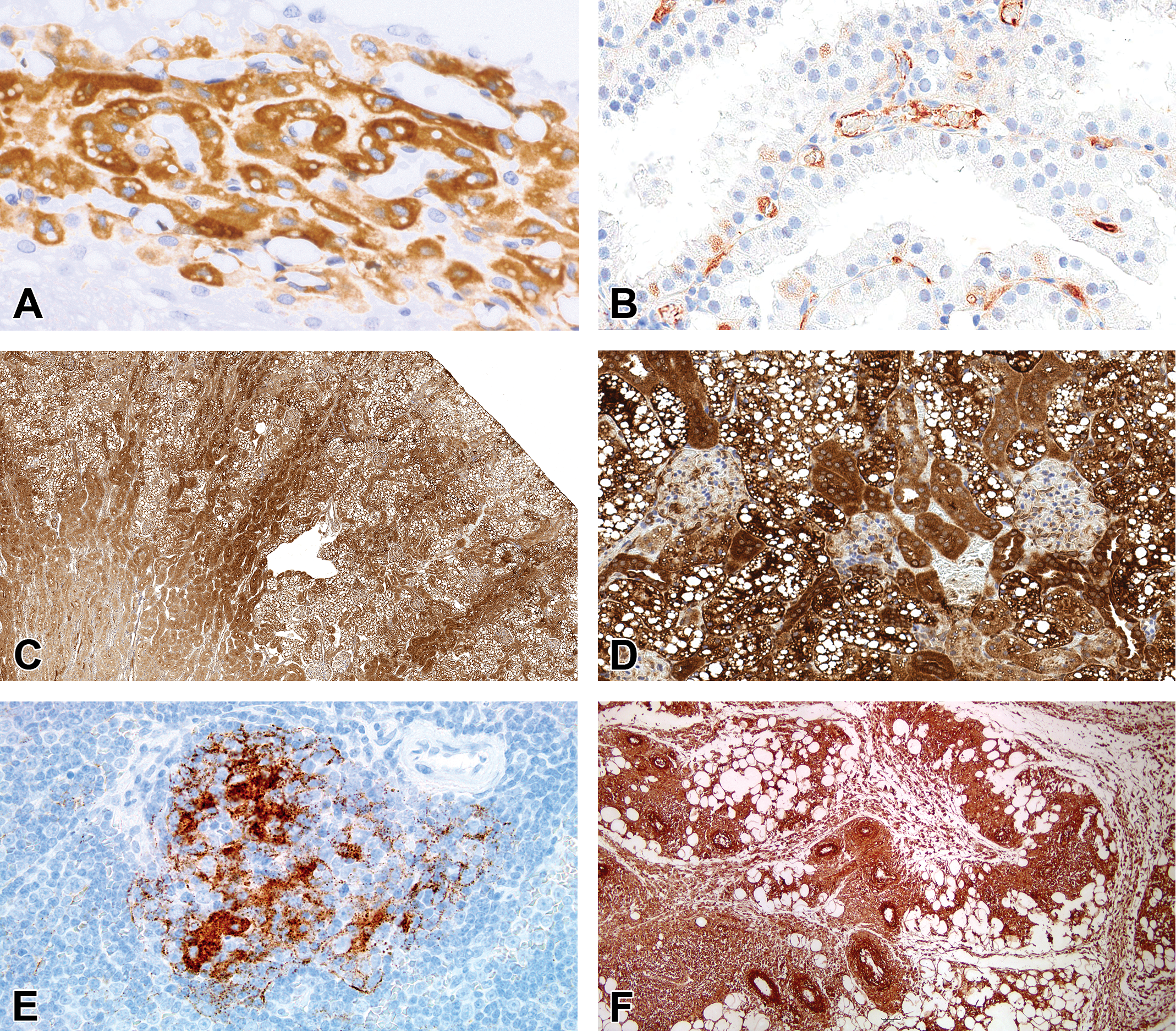
IHC evaluation of PEG in tissues from rat (A, C-D), cynomolgus monkeys (B), and Beagle dog (E–F). Choroid plexus (A–B), kidney (C–D), spleen (E), and subcutaneous injection site (F). A and D show that the cytoplasm of the choroid plexus epithelium (A) or renal tubular epithelium (D) is immunoreactive for PEG; however, the vacuoles are not immunoreactive for PEG. FFPE. DAB with hematoxylin counterstain. DAB = 3,3’-diaminobenzidine; FFPE = formalin-fixed, paraffin-embedded; IHC = immunohistochemistry; PEG = polyethylene glycol.
PEG-associated vacuoles usually present as few to many clear, colorless cytoplasmic vacuoles. These may range in size from small (<3 µm) to quite large (up to 30 µm; Bendele et al. 1998; Bolon et al. 2001). In epithelial tissues, most cells in a given region may be affected, although the magnitude of the effect may differ among functionally distinct regions. For example, in the kidney, early accumulation of PEG-associated vacuoles occurs in tubules located in the outer third of the cortex, while continued exposure results in extension of the change deeper into the cortex (personal observations of the authors).
PEG-associated vacuoles do not stain with standard histochemical stains used in assessing formalin-fixed, paraffin-embedded (FFPE) tissues. Besides H&E, vacuoles remain uncolored when treated with periodic acid–Schiff, alcian blue, or toluidine blue (data not shown). Despite strong cytoplasmic immunoreactivity for PEG using anti-PEG antibodies (Figure 10), the actual vacuoles are not immunoreactive for PEG or have variable levels of PEG immunoreactivity (Rudmann et al. 2013). The vacuoles are membrane-bound and consistent with lysosomes by transmission electron microscopy (TEM, Figure 9; Rasmussen et al. 2016). Chaperone-mediated autophagy may be involved in the pathogenesis of the vacuolar changes (McDuffie et al. 2016).
Current Practices for Microscopic Evaluation and Review of the Existing Terminology Used in Nonclinical Safety Studies to Describe the Range of PEG-associated Cellular Alterations
A review of the peer-reviewed literature and publicly available regulatory reviews for various PEGylated biopharmaceuticals indicated that the terminology used most often to capture PEG-associated vacuolar changes in nonclinical safety studies was “vacuolation” with the actual affected cell type or suborgan structure used in the table and/or text of the document as a modifier (summary presented in Table 2). Additional terminology used in some reports includes “foamy macrophages” and “vacuolated.” Vacuolated macrophages were typically captured as “histiocytic vacuolation,” “vacuolated macrophages,” or as “foamy macrophages” (Table 2). Anecdotally, other diagnostic terminology to describe vacuolation of cells may include “vacuolation, increased, [suborgan structure],” “Kupffer cells, foamy,” “clear cytoplasm,” or “foamy cytoplasm.” Often there was no information regarding whether the vacuolation was microvesicular or macrovesicular in nature or a mixture of both large and small vacuoles. In one instance, the use of the term “foamy macrophages” was referring to “finely vacuolated” per the regulatory reviewer, which would suggest that in that particular case, the vacuolation could be consistent with “microvesicular” (see FDA 2008). In the references presented in Table 2, clarifying statements were often included to indicate the lack of any cellular and/or presumed functional damage in the cells or tissues. Vacuolation was typically given a severity grade; however, details of the breadth of the severity scale (e.g., 4-point or 5-point scale) and the precise criteria for assigning the severity grades usually were not described. Table 3 summarizes examples of published severity scoring criteria. Other grading criteria are likely in use in the toxicologic pathology community. For example, some of the authors (Fleurance, Bolon) are aware of another grading system in which “grade 5” was attributed to the most affected animal during the treatment phase, irrespective of the “real” severity of the vacuolation, in order to enhance the differences in discriminating degrees of vacuolation between animals or treatment groups and to better assess the reversibility of the change.
Summary of the Terminology Used to Describe PEG-associated Vacuolation of Tissues in the Peer-reviewed Literature and Publicly available Regulatory Reviews.
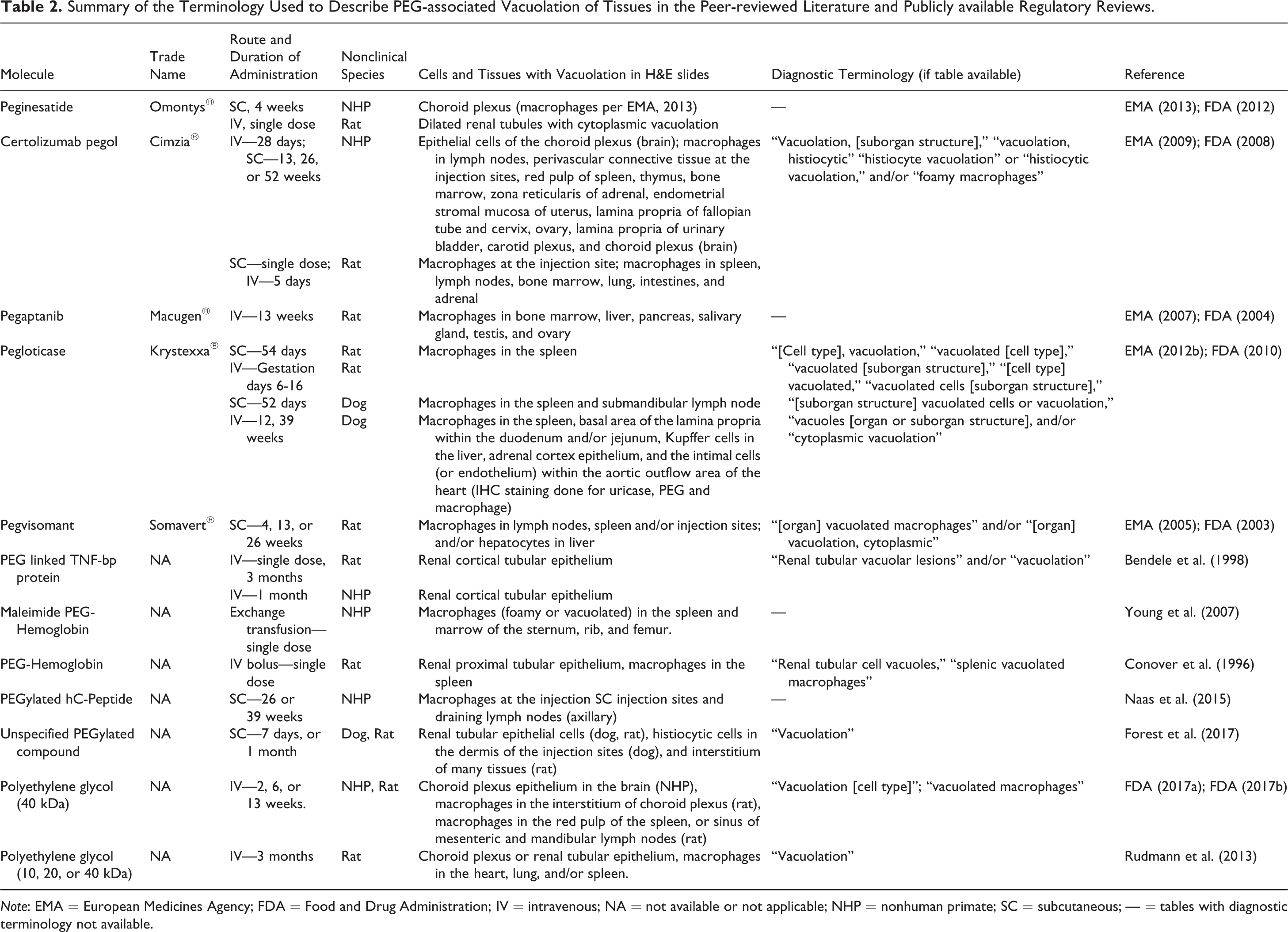
Note: EMA = European Medicines Agency; FDA = Food and Drug Administration; IV = intravenous; NA = not available or not applicable; NHP = nonhuman primate; SC = subcutaneous; — = tables with diagnostic terminology not available.
Examples of Severity Grading Systems Used for Recording PEG-associated Vacuolation in Tissues.

a Bendele et al. (1998) utilized two grading systems. The less detailed 4-point grading system was used for studies with milder changes such as those occurring in studies that utilized one or two doses of the PEGylated biopharmaceutical.
Relationship of the Chemical Nature of PEG to the Range of PEG-associated Cellular Alterations
Recent surveys of marketed PEGylated drugs and PEGylated biopharmaceuticals in development show evidence that PEG-associated vacuolation in parenchymal organs is noted most frequently with PEGs of at least 30 kDa (Ivens et al. 2015; Turecek et al. 2016), while PEGs of smaller size (15–20 kDa) tend to accumulate more specifically in renal tubular epithelium (Bendele et al. 1998; Bolon et al. 2001). It has been hypothesized that this variation is due to the reported higher glomerular filtration of smaller PEG molecules and shorter and/or lower systemic exposure, while large PEGs with slower renal clearance have a higher potential to accumulate in parenchymal cells (Bendele et al. 1998; Baumann et al. 2014; Ivens et al. 2015). Exceptions to the more limited distribution of cellular vacuolation with PEGylated therapeutics containing PEG of lower MW are believed to be a result of the larger total PEG dose or other factors such as complexity of the PEG moiety. Examples of these exceptions include marketed products (e.g., Krystexxa; 8–10, 10 kDa PEG molecules attached to each subunit) and other investigative PEGylated biopharmaceuticals (20 kDa PEG attached to FGF21 variants; Xu et al. 2013). In addition, despite the trend for larger PEG to be associated with more widespread vacuolation, an increasing number of publications on the pharmacokinetics and tissue distribution of PEG alone, metabolized PEG and PEGylated proteins, convincingly demonstrates that apart from the PEG MW, numerous other factors may contribute to the location, extent, and pattern of cellular vacuolation. These include the complexity of the PEG moiety (e.g., branching PEGs are filtered less effectively by the kidney relative to linear PEGs of the same mass; Bolon et al. 2001), dose size, dosing frequency and duration, the balance of nonspecific uptake (the presumed mechanism for uptake into choroid plexus epithelium [CPE] and macrophages) versus receptor-mediated uptake, potential immunogenicity, and turnover kinetics of cells accumulating PEG (Xu et al. 2013; Baumann et al. 2014). Due to the complex interplay of several factors above and beyond PEG size, it is not possible to make generalizations or predictions about the relationship between the chemical nature of any particular PEGylated xenobiotic and corresponding characteristics of PEG-associated vacuolation (e.g., cell type, tissue location, size, number per cell).
Only a few published studies address mechanisms of cellular binding, uptake, and trafficking of PEG or PEGylated proteins (Bendele et al. 1998; Yu et al. 2004; Markovsky et al. 2012). The most valuable reviews on tissue distribution of PEG-associated cellular vacuolation have been provided by Baumann et al. (2014) and Ivens et al. (2015). In brief, PEGylated proteins are thought to be removed from the circulation through nonspecific protein-associated mechanisms, target/receptor-mediated uptake, and pinocytosis. The protein moiety of PEGylated biopharmaceuticals appears to be degraded by lysosomal enzymes following denaturation in matured endosomes. It is not clear whether protein degradation occurs while still being coupled to PEG or after cleavage. The PEG moiety is not metabolized due to the limited enzyme repertoire to digest PEG in eukaryotic lysosomes. PEG, metabolized PEG, and possibly unmetabolized PEGylated protein then may be released from the cell by exocytosis or following cell turnover (Baumann et al. 2014). The PEG moiety likely is returned to lymphatics and the blood where it may be eliminated by either renal filtration or undergo reuptake into cells by endocytosis. As a consequence, gradual accumulation of hygroscopic PEG leads to engorged vesicles, likely lysosomes, which become visible as cellular vacuoles microscopically. The combination of the rate of exocytosis, dose and frequency, cell turnover rate, and potential reuptake rate will determine the extent of PEG accumulation in cells (Baumann et al. 2014).
Review of IHC Procedures and Available Reagents to Detect PEG in Tissues
Detection of PEG immunoreactivity in cells and tissues using PEG-specific antibodies and standard IHC procedures is used to characterize the presence and cellular localization of PEG (Rudmann et al. 2013; McDuffie et al. 2016; Rasmussen et al. 2016). In some of the authors’ experience, the reagents PEG-B-47 and PEG-2-128 work well for IHC procedures in tissues from rats and monkeys. Table 4 summarizes examples of IHC procedures reported by investigators. Using IHC reagents, PEG immunoreactivity can be detected in tissues with or without vacuolation (Ivens et al. 2015). This methodology allows the pathologist to correlate the presence of PEG in the cell with PEG-associated vacuolation and may also demonstrate potential distribution of PEG in other unaffected cells and tissues. Some commercially available anti-PEG antibodies are listed in Table 5. Commercial antibodies are mostly of the IgM or IgG isotype and are produced in rabbits, mice, or rats. These antibodies recognize either the terminal methoxy group or the PEG backbone. Currently, there are no standardized approaches with respect to tissue fixation, reagents, or staining protocols when employing such procedures (Table 4). Since PEG is a polyethylene-based polymer, it would be expected to be vulnerable to extraction with xylene and ethanol during conventional tissue processing and deparaffinization. Despite this, most of the currently published IHC results appear to have used FFPE materials, which raises the possibility that evaluating the deposition of PEG in frozen sections may yield different results. As reported by Ivens et al. (2015), vacuoles may contain PEG-immunoreactive contents. However, in the experience of some of the present authors and the reports of others (Forest et al. 2017), vacuoles observed in the tissue after the administration of PEGylated compounds, for various study durations, often are not immunoreactive for PEG despite the cytoplasm being immunoreactive for PEG (Figure 10). Rasmussen et al. (2016) demonstrated immunoreactive vesicles in CPE of rats; however, the vesicles were not visible on standard H&E tissue sections. Inconsistent anti-PEG immunoreactivity using some commercial antibodies may be a consequence of the existing tissue preservation and processing methods, variability in IHC procedures and/or reagents, and/or the different MWs, conformations, and/or distribution of PEG in the tissues. It is not clear whether the PEG in the vacuoles is removed during histologic processing (Forest et al. 2017) or whether vacuolation instead may result from fluid distension of the lysosomes due to the hygroscopic nature of the PEG contained therein (as suggested by Bendele et al. 1998). The latter hypothesis is supported by TEM evaluation, which shows that PEG-associated cytoplasmic vacuoles mostly contain electron-lucent contents (Figure 9; Forest et al. 2017; Rasmussen et al. 2016).
Summary of Selected Immunohistochemical Procedures to Assess PEG Immunoreactivity in Tissues.
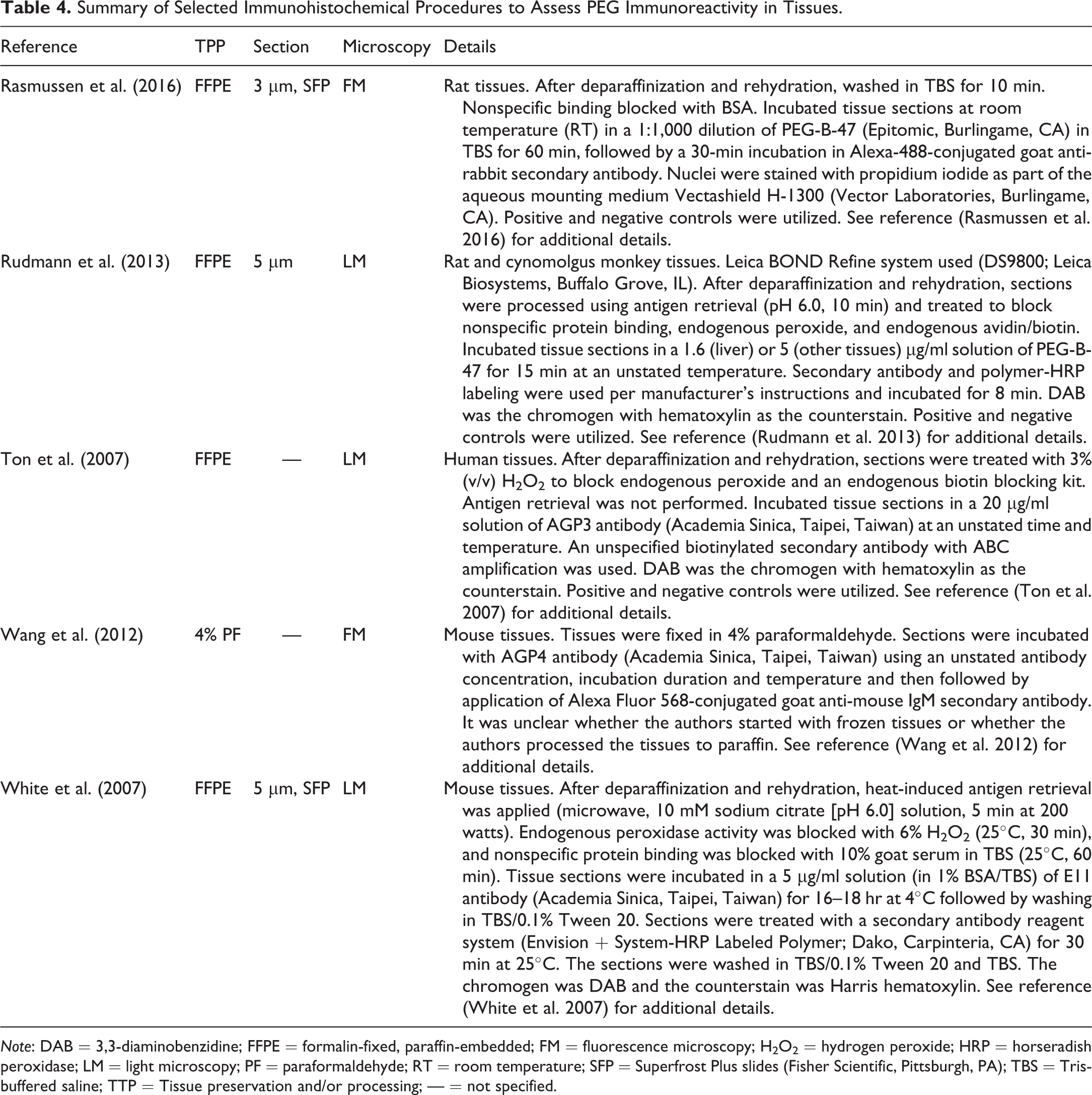
Note: DAB = 3,3-diaminobenzidine; FFPE = formalin-fixed, paraffin-embedded; FM = fluorescence microscopy; H2O2 = hydrogen peroxide; HRP = horseradish peroxidase; LM = light microscopy; PF = paraformaldehyde; RT = room temperature; SFP = Superfrost Plus slides (Fisher Scientific, Pittsburgh, PA); TBS = Tris-buffered saline; TTP = Tissue preservation and/or processing; — = not specified.
Types and Examples of Antibodies Used for IHC Detection of PEG in Tissues.

a AGP3, AGP4, and E11 were generated by immunizing mice with a conjugate of E. coli β-glucuronidase-RH1 mAb-sPEG Where the sPEG is a hydroxyl-terminated eight-arm poly(ethylene glycol).
b AGP4 is claimed to bind the backbone of PEG allowing for wider detection of PEGs.
c PEG-B-47, reported as being specific for the terminal methoxy group of methoxyPEG, was generated with a 5 kDa mPEG-KLH. Some vendors (Abcam) claim that it only recognizes conjugated forms of PEG by ELISA and recommend using PEG-2-128 for recognizing the PEG backbone in Western blots and enzyme-linked immunologic assays. However, Rudmann et al. (2013) used PEG-B-47 successfully in experiments using unconjugated PEG in their studies. The current authors did not identify publications that used PEG-2-128 in IHC procedures.
Value of Further Characterizing the Biodistribution/Accumulation of PEG through IHC, Immunochemical, or Other Techniques
Regulators and others have expressed hypothetical concerns regarding potential long-term risks associated with PEG exposure and related vacuolation in certain vital tissues/structures such as central nervous system neurons, circumventricular organs, or the choroid plexus (Kronenberg et al. 2013; Ivens et al. 2015). To date, these concerns have been based entirely on morphologic observations upon routine histomorphologic evaluation of H&E-stained, FFPE sections. However, the short-term safety of PEG has been studied extensively without identification of toxicity beyond reports of renal tubular cell vacuolation and degeneration at very high dose levels (reviewed in Fruijtier-Pölloth 2005; Webster et al. 2007, 2009). The duration of the toxicity studies in which these assessments have been performed has been dictated by nonclinical development guidance (typically ≤ 6 months; less commonly up to 12 months). In some instances, the severity of vacuolation has been marked or severe, occasionally leading to tissue distortion, but yet without demonstrated adverse functional outcomes (Ivens et al. 2015; unpublished observations). Extended recovery periods typically lead to complete or at least partial reversal depending on the tissues/organs, the degree of which likely reflects the basal turnover rate for cells within a given organ (Ivens et al. 2015; unpublished data). For example, complete or partial recovery in lymphoid tissues can occur within 3 to 6 months, whereas the same recovery duration may result in a one-grade improvement in the CPE (unpublished data).
Cumulative published data from multiple drug products indicate that drug target distribution and pharmacology may modulate the distribution and extent of PEG accumulation in tissues (Baumann et al. 2014). However, based on IHC evaluation for unconjugated PEG of various MWs, it is also clear that the MW of the PEG appears to have the greatest influence on the quantity and tissue distribution of PEG, independent of protein conjugation (Rudmann et al. 2013). In addition to the existing IHC methods used to understand the distribution of PEG conjugates, increasingly sophisticated approaches to ADME (absorption, distribution, metabolism, excretion) assessment have been reported in recent years. These approaches have provided an enhanced understanding of how protein-conjugated PEG is distributed and cleared from tissues after parenteral administration. For example, comprehensive characterizations of the pharmacokinetics, tissue distribution, excretion, and metabolite profiling have been reported recently in rats for PEGylated rFVIII (glycoPEGylated FVIII, N8-GP; Bjørnsdottir et al. 2016) and rFIX (nonacog β pegol, N9-GP; Sternebring, Christensen, and Bjørnsdottir 2016) using a combination of PEG radiolabeling, enzyme-linked immunosorbent assay, high-performance liquid chromatography, and electrophoresis. Their data and associated simulations support the growing body of literature indicating that protein-specific clearance pathways can contribute to the initial tissue distribution and disposition of the intact PEGylated product, while recirculation of PEG after metabolism of the protein backbone results in a subsequent prolonged disposition. While these studies are extremely valuable in the characterization of product-specific influences on the ADME of PEG, they typically represent the outcome of single-dose evaluations. It is likely that saturable mechanisms may be involved in ADME processes related to PEG, and therefore, patterns of PEG accumulation may shift under steady-state dosing conditions. Consequently, these standard ADME approaches cannot address potential broadening of PEG tissue distribution in chronic treatment settings. Defining the biodistribution and rates of elimination for PEG under steady-state dosing conditions may, therefore, be an important gap in current assessments.
Furthermore, conventional ADME methods, while providing important quantitative tissue distribution information, do not provide high-resolution topographic information and therefore may fail to identify accumulation of PEG in small but highly perfused vital structures such as the choroid plexus or tissues of the hypothalamic–pituitary axis. Methods such as IHC at the light microscopic and electron microscopic (immunoEM) levels employing antibodies that can differentiate unconjugated PEG and its degradation products from the intact protein-PEG product may effectively fill these distribution, metabolism, and elimination gaps when applied to tissues obtained under conditions in which a PEGylated product has been given for varying amounts of time. In addition, IHC may be employed to resolve diagnostic dilemmas in which vacuoles associated with PEG overlap or cannot be differentiated from artifactual vacuoles (commonly observed in neural tissues) or vacuoles that are associated with normal physiologic processes (e.g., in macrophages, hepatocytes, adrenocortical epithelial cells).
Multimodality ADME assessments (including steady-state evaluations) coupled with anti-PEG IHC evaluations will likely be key components in the effective modeling of PEG absorption and clearance in tissues of special concern and may be critical in the overall assessment of products employing higher MW PEGs under chronic-use conditions. Therefore, it would be of great value to optimize and standardize anti-PEG IHC methodologies to complement the growing sophistication of ADME techniques utilized in the safety assessment of PEGylated products. Recently, Forest et al. (2017) demonstrated the use of magnetic resonance spectroscopy and nuclear magnetic resonance spectroscopy to study the distribution and elimination of PEG in dogs after multiple doses in relation to morphologic changes in tissues. Additional investigation using the combination of these techniques may increase our understanding of the relationship of the biodistribution of PEG conjugates, morphologic changes, and risk for PEG-related adverse functional events with chronic administration.
Collectively, the information presented above highlights that a variety of methods, including identification of vacuoles via light microscopy, have been utilized to infer or characterize the biodistribution of PEG in tissues. These techniques offer valuable insight into the distribution of PEG. The continued evaluation and utilization of these techniques together with standard safety end points may provide additional understanding of the pathogenesis and impact of PEG-associated morphologic changes in nonclinical toxicity studies.
Points to Consider in the Documentation, Description, and Communication of PEG-associated Findings in Safety Assessment Study Reports Submitted to DRAs
The clear communication of test article–related pathology findings to DRAs is of critical importance to drug development programs. The STP has published various “best practice” and “points to consider” manuscripts that pathologists may consult when determining how to document, describe, and communicate tissue findings in nonclinical toxicity studies (Crissman et al. 2004; Morton et al. 2006; Mann et al. 2012; Kerlin et al. 2016; Schafer et al. 2018). These general-purpose documents have relevance when addressing the implications of PEG-associated vacuoles. Below are more specific points to consider based on the recommendations from these publications. These points will facilitate the documentation, description, and communication of PEG-associated microscopic changes in nonclinical studies. In general, vacuolation can be an artifact of suboptimal fixation or an artifact due to other causes (Li et al. 2003). Therefore, it is imperative that tissue fixation in nonclinical studies of PEGylated pharmaceuticals be of high quality to avoid interference with histopathologic evaluation and facilitate the subsequent documentation, broader description and communication. Neutral-buffered 10% formalin is a suitable choice for standard light microscopic assessments included in nonclinical safety study reports submitted to DRAs. A review of the literature and the authors’ collective experience indicate that this fixative and processing to paraffin block should also be sufficient for IHC detection of PEG in the cytoplasm of cells with some reagents. However, it is unclear whether these methods are optimal for demonstrating PEG in vacuoles. Utilize consistent descriptive terminology to record PEG-associated microscopic findings and grade the severity of the findings. Our review suggests that “vacuolation [cell type]” and “vacuolated macrophages” are commonly used descriptors and are consistent with the use of descriptive terminology proposed by International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) publications for various organs and systems (Mann et al. 2012). Specific affected cells and the character of the vacuoles (e.g., size or relative size, shape) should be included in the narrative portion of the report. Consider including in the pathology report (e.g., in the methods section) a description of the specific criteria that were used to assign the histopathologic severity grades for PEG-associated cytoplasmic vacuolation. Representative photomicrographs of each severity grade may also be helpful when included in the report. This is particularly important in situations where understanding reversibility or progression, over time or across studies of increasing dose and/or duration, is critical to the assessment of risk. Adequately describe and interpret all PEG-associated microscopic findings, when possible correlating these to other findings in the study, and articulate clearly the affected dose groups. PEG-associated findings should be discussed together in the pathology report; this would be particularly helpful when there are many organs affected. Consider providing summary tables of PEG-associated findings and their severity scores in the pathology report. This presentation may facilitate communication of all affected organs, relevant microscopic findings, and simplify the discussion of their significance or lack of significance. Clearly articulate any PEG-associated finding that was considered to be adverse, including a rationale supporting this judgment. Pathologists may need to consider whether or not it is appropriate in their pathology report to provide a brief justification regarding why PEG-associated findings were or were not considered adverse. Provide references that help support the interpretation of the findings as being PEG-associated, assist in the understanding of the distribution of the findings, and/or help provide context regarding the adversity or lack of adversity for these findings.
Conclusion
Recent comprehensive reviews have addressed the basic challenges of developing PEGylated pharmaceutical products and have highlighted gaps that remain in our current understanding of PEG-associated tissue vacuolation. These gaps include lack of widely accepted standardized histological terminology and criteria to record and grade the severity of vacuolation (Ivens et al. 2015). Although the Working Group did not generate unique standardized terminology or severity grading criteria, we provide an expanded pictorial review of representative PEG-associated light microscopic findings in tissues, review published terminology and grading criteria for this change, and provide points to consider for toxicologic pathologists tasked with evaluating and reporting PEG-associated morphologic findings in nonclinical studies. Collectively, our reviews highlight that PEG-associated tissue vacuolation is generally consistent in appearance (e.g., clear round cytoplasmic vacuoles of variable size) across a variety of PEGs and PEGylated biopharmaceuticals and thus should be amenable to the utilization of uniform descriptive terminology. Our review suggested that the terms “vacuolation [cell type]” and “vacuolated macrophages” are commonly used descriptors, and these choices are consistent with the use of descriptive nomenclature proposed in INHAND publications of standard diagnoses for rodent tissue lesions (Mann et al. 2012). Above and beyond recording the occurrence of vacuolation, toxicologic pathologists should consider providing additional descriptive information about the character (e.g., size or relative size, shape). The use of adequate descriptions and uniform terminology, particularly across studies, will greatly facilitate the toxicologic characterization of a PEGylated biopharmaceutical and the communication of these findings to DRAs.
Establishing an industry-wide, standardized severity grading system for any morphologic finding associated with a xenobiotic is a challenging task due to the inherent subjective nature of microscopic evaluation and other factors (Schafer et al. 2018). Similarly, establishing a standardized severity grading system for PEG-associated vacuolation will be challenging. Additional challenges included “tradition” or resistance to changing existing approaches and the use of different (“purpose-crafted”) grading systems depending on the severity (as exemplified by Bendele et al. 1998; Table 3). The severity grading systems summarized in Table 3 highlight variations in complexity or detail of several published systems. These variations speak to the challenge that pathologists face when generating a severity grading system that is not too simple nor excessively complex, particularly for findings that are critically important (Schafer et al. 2018). It is unclear what the appropriate level of detail or complexity should be in setting a severity grading system for vacuolation. The potential gap generated by the lack of a standardized grading system may complicate interstudy comparisons, for example, as in a series of studies of increasing duration for a given PEGylated biopharmaceutical, as differences in severity scores recorded across studies may or may not be meaningful, given the lack of functional consequences. The lack of standardization, or lack of communication of the grading system used, may also complicate comparisons across compounds or laboratories, thereby causing difficulties in review of nonclinical data sets by DRA scientists. An ideal severity grading system for deployment in routine nonclinical toxicity studies will need to be simple to use, lead to repeatable results, yet include sufficient guidance to ensure persistent and consistent use by practitioners of various backgrounds. To address these challenges, the Working Group proposes that toxicologic pathologists should consider describing in their pathology reports the specific criteria used to assign the histopathologic severity grades for PEG-associated changes. These criteria can be included as text in the methods section of the report and if warranted, as representative images.
Understanding the pathogenesis of PEG-associated findings, the relationship of these findings to tissue distribution of the PEGylated biopharmaceutical or the PEG moiety, and the implications of risk to humans continue to be areas of concern to biopharmaceutical scientists and DRAs. Unfortunately, a comprehensive understanding of the nature and pathogenesis of PEG-associated vacuolation will require further study. Special methods such as IHC, TEM, and other novel analytical techniques now available to biopharmaceutical scientists may help answer these questions with continued investigation. The Working Group’s review of published information on IHC procedures and reagents highlights considerable variability in the reagents, tissue preservation, tissue processing, and other procedures. Perhaps due to these differences, IHC has not provided conclusive information on the contents of vacuoles other than suggesting that these may contain variable, yet low, amounts of PEG. Standardization and sharing of optimized tissue preservation, tissue processing, and IHC procedures, together with exploration of different preservation and processing methods, may generate further insights. As part of the pictorial review, the Working Group shared TEM images of cells with PEG-associated vacuolation (Figure 9) and reviewed limited information (including images) from publications. Taken together, the available information suggests that vacuoles are consistent with endosomes and/or lysosomes and contain material that is mostly electron lucent or of low electron density. However, these assessments have not, so far, included methods such as electron immunochemistry, biochemical procedures, or other methods that may be useful in elucidating further the contents of the vacuoles and thus provide more information about the pathogenesis of PEG-associated vacuolation.
The expanded pictorial review of PEG-associated microscopic findings combined with the points to consider provided above do not definitively answer the gaps previously identified by others. However, the information presented herein will help practicing toxicologic pathologists grapple with several key analytical and reporting issues, as they evaluate the qualitative nature, extent, and significance of cytoplasmic vacuolation in various organs consequent to the repeated administration of PEGylated biopharmaceuticals during nonclinical safety studies.
Footnotes
Acknowledgments
The technical and/or image assistance provided by Kelly Credille and James Alston (Experimental Pathology Laboratory, Eli Lilly and Company) and Kathleen Heinz-Taheny (Pathology, Eli Lilly and Company) are greatly appreciated.
Author Contribution
All authors (AI, BMB, BB, AB, SC, RF, RG, DH, JL, AR, AS, BZ) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The authors declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
