Abstract
PEGylation (the covalent binding of one or more polyethylene glycol molecules to another molecule) is a technology frequently used to improve the half-life and other pharmaceutical or pharmacological properties of proteins, peptides, and aptamers. To date, 11 PEGylated biopharmaceuticals have been approved and there is indication that many more are in nonclinical or clinical development. Adverse effects seen with those in toxicology studies are mostly related to the active part of the drug molecule and not to polyethylene glycol (PEG). In 5 of the 11 approved and 10 of the 17 PEGylated biopharmaceuticals in a 2013 industry survey presented here, cellular vacuolation is histologically observed in toxicology studies in certain organs and tissues. No other effects attributed to PEG alone have been reported. Importantly, vacuolation, which occurs mainly in phagocytes, has not been linked with changes in organ function in these toxicology studies. This article was authored through collaborative efforts of industry toxicologists/nonclinical scientists to address the nonclinical safety of large PEG molecules (>10 kilo Dalton) in PEGylated biopharmaceuticals. The impact of the PEG molecule on overall nonclinical safety assessments of PEGylated biopharmaceuticals is discussed, and toxicological information from a 2013 industry survey on PEGylated biopharmaceuticals under development is summarized. Results will contribute to the database of toxicological information publicly available for PEG and PEGylated biopharmaceuticals.
Introduction
Since the 1982 introduction of the first recombinant insulin, biotechnologically derived drugs (biopharmaceuticals) have become essential in the treatment of serious life-threatening and chronic diseases. These drugs are often recombinant forms of endogenous molecules. Suitable methods to modify these biotechnologically derived molecules, in particular their half-life are limited, since some properties of peptides or proteins may be changed and result in either loss of efficacy or increased immunogenicity. To date, PEGylation (the covalent binding of one or more polyethylene glycol [PEG] molecule to another molecule) has been the most successful strategy to chemically modify these biopharmaceuticals. The addition of PEG can extend their half-life, increase stability, and in some cases, ameliorate the immunogenicity of highly immunogenic proteins such as enzymes. There are currently 11 approved PEGylated biopharmaceuticals in the United States and Europe (10 peptides/proteins and 1 aptamer as of 2015) and many more are in nonclinical and clinical development.
PEG has a simple repetitive structure, is chemically inert, and has low toxicity (Webster, Didier et al. 2007; Schellekens, Hennink, and Brinks 2013). Findings including adverse effects observed in nonclinical studies of approved PEGylated biopharmaceuticals are usually related to the intended pharmacological actions of the active drug and not to the PEG moiety (Webster, Didier et al. 2007; Kang, Deluca, and Lee 2009; Jevsevar, Kunstelj, and Porekar 2010; Ivens et al. 2013). However, PEG-related histologic changes, characterized as cellular vacuolation in certain tissues and cell types, have been observed in nonclinical toxicology studies for approximately half of the approved PEGylated drugs (Tables 1 and 2). These changes were not associated with pathologic effects such as tissue degeneration, necrosis, and cellular distortion or changes in study end points including hematology, clinical chemistry, urinalysis, or organ weight. Assessment and characterization of tissue vacuolation continues to be an important component of the toxicology evaluation of PEGylated biopharmaceuticals.
BioSafe survey results of PEGylated biopharmaceuticals in development 2013: Tissue vacuolation reported in toxicology studies in relation to PEG size.
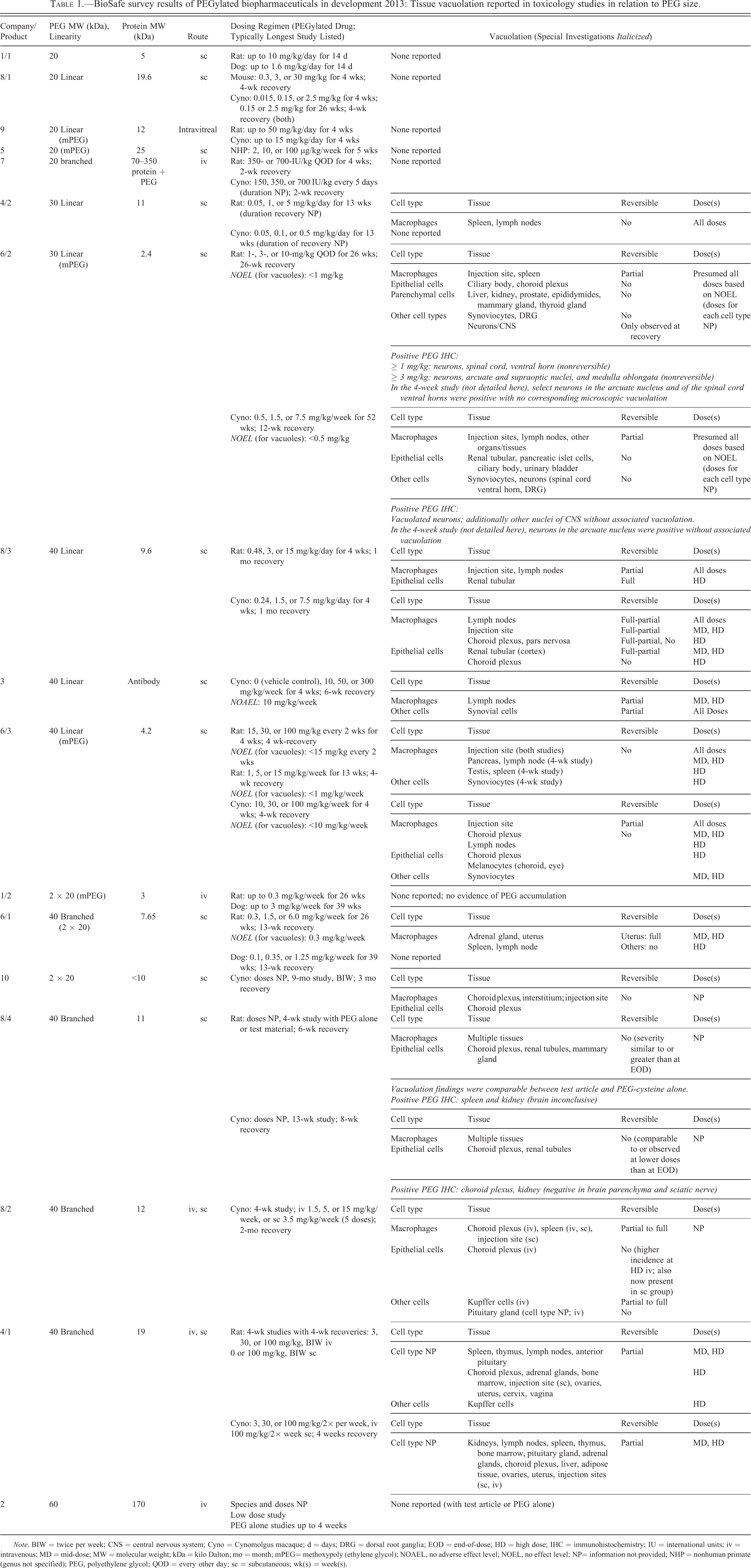
Note. BIW = twice per week; CNS = central nervous system; Cyno = Cynomolgus macaque; d = days; DRG = dorsal root ganglia; EOD = end-of-dose; HD = high dose; IHC = immunohistochemistry; IU = international units; iv = intravenous; MD = mid-dose; MW = molecular weight; kDa = kilo Dalton; mo = month; mPEG= methoxypoly (ethylene glycol); NOAEL, no adverse effect level; NOEL, no effect level; NP= information not provided; NHP = nonhuman primate (genus not specified); PEG, polyethylene glycol; QOD = every other day; sc = subcutaneous; wk(s) = week(s).
Summary of tissue vacuolation in pivotal toxicology studies with approved PEGylated products.
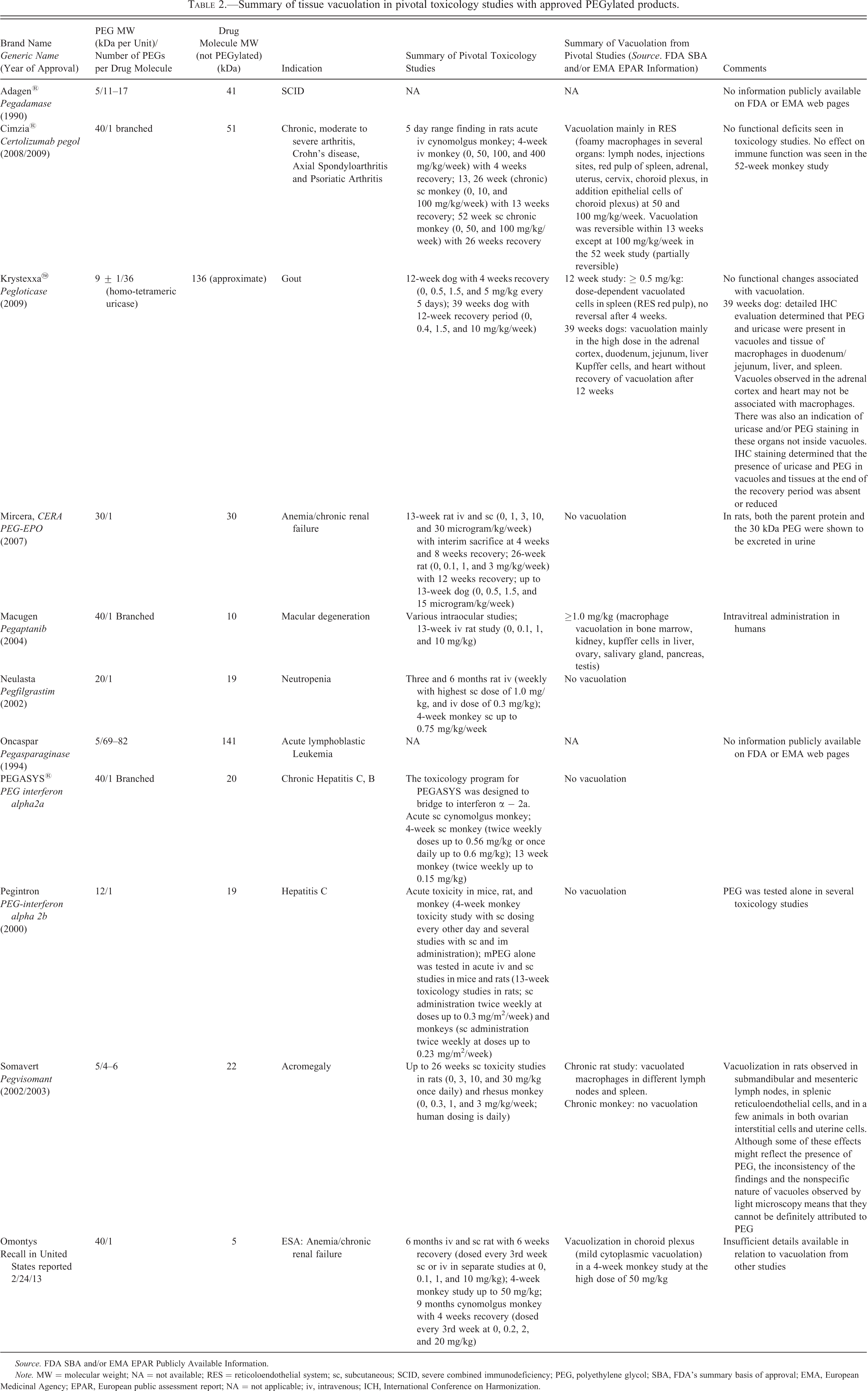
Source. FDA SBA and/or EMA EPAR Publicly Available Information.
Note. MW = molecular weight; NA = not available; RES = reticoloendothelial system; sc, subcutaneous; SCID, severe combined immunodeficiency; PEG, polyethylene glycol; SBA, FDA’s summary basis of approval; EMA, European Medicinal Agency; EPAR, European public assessment report; NA = not applicable; iv, intravenous; ICH, International Conference on Harmonization.
Many factors may impact the occurrence and degree of cellular vacuolation in toxicology studies of PEGylated biopharmaceuticals (Yamaoka, Tabata, and Ikada 1994, 1995; Webster, Didier et al. 2007) including: Pharmacological activity of the protein, Distribution of drug target, Drug dose, Dose frequency and duration of treatment, Distribution of the drug, Amount of nonspecific uptake versus specific drug receptor mediated uptake, Potential immunogenicity, Overall molecular weight of the drug, The molecular weight of PEG itself, and Types of clearance/removal mechanisms for drug or PEG.
Location, severity, and reversibility of vacuolation, as well as the presence or absence of any secondary morphologic changes such as tissue degeneration, cellular distortion, and inflammation or necrosis, should be carefully considered in risk assessment along with the dose/dose frequency at which such changes were observed in relation to the human therapeutic dose and dosing frequency. While common risk assessment strategies can be applied to effects related to the pharmacologically active drug component(s) of the molecule, determination of the potential adversity (or lack thereof) of cellular vacuolation attributed to PEG and extrapolation of potential risks for the patient poses a challenge since much about the mechanism and functional consequences of tissue vacuolation remains unknown.
This article summarizes experience with 11 approved PEGylated biopharmaceuticals and 17 PEGylated biopharmaceuticals in development and the impact of cellular vacuolation on development and product approval. Another emerging area of concern with PEGylated biopharmaceuticals is the potential for generation of antibodies to PEG, the potential for anti-PEG antibodies to cross-react with other PEGylated biopharmaceuticals, and the existence of preexisting anti-PEG antibodies of unknown origin in humans (Schellekens, Hennink, and Brinks 2013; Verhoef et al. 2014). The following points will be addressed specifically: Regulatory guidance and health authority recommendations to support clinical development, PEG disposition/metabolism, and excretion; Immunogenicity of PEG; Brief review of publicly available toxicology information for 11 approved PEGylated biopharmaceuticals; Toxicological relevance of cellular vacuolation observed in nonclinical toxicology studies and their potential risk for the patient population; 2013 BioSafe survey of pharmaceutical industry of 17 products from 10 companies before approval, including specific nonclinical case studies; and Considerations for nonclinical safety evaluation of PEGylated biopharmaceuticals.
Regulatory Guidance for PEGylated Biopharmaceuticals
The current guidance for nonclinical safety evaluation of biopharmaceuticals is ICHS6 R1 (International Conference on Harmonization [ICH] S6 R1, 2011). It also applies to PEGylated biopharmaceuticals but no specific guidance regarding the assessment of PEGylated biopharmaceuticals or observations related to systemic PEG exposure, such as cellular vacuolation, is included. Two additional guidance documents, which specifically address PEGylated drug products, have been published recently: Food and Drug Administration (FDA) Guidance for Industry: Immunogenicity Assessment of Therapeutic Protein Products (Part V.B.4 Glycosylation/PEGylation [FDA 2014]) and the Committee for Medicinal Products for Human Use (CHMP) Safety Working Party’s response to the European medicinal agency (EMA) Pediatric Committee regarding the Use of PEGylated drug products in the pediatric population intended for long-term treatment.
The CHMP response acknowledged that, while vacuolation in cells of the reticuloendothelial system (RES) may represent the body’s normal response to remove a foreign body, there was a concern for other cells and tissues such as the renal tubular epithelium or the choroid plexus epithelium (ependymal cells). The CHMP summarized that vacuolation in these cells was observed following certain conditions including PEG molecular weight ≥30 kilo dalton (kDa), toxicology study duration of at least 4 weeks, and a cumulative PEG dose of ≥0.4 µmol/kg/month. It was concluded: “until more data are available, the risk for ependymal cell vacuolation needs to be addressed before conducting longer term clinical trials in the paediatric population.” However, neither the definition of what constitutes an adverse finding nor the magnitude of the safety margin was defined in the CHMP response. It is also not known whether demonstrating evidence of partial or full reversibility of vacuolation may be sufficient for progression of development of the PEGylated drug candidate.
Currently, there is no clear understanding if vacuoles that occur with prolonged exposure to PEG may lead to any functional consequences. However, the body of evidence available for approved PEGylated biopharmaceuticals has not identified any clinically reported functional consequences for compounds where vacuolation was observed in toxicology studies. This is reflected in the lack of significant impact on the risk evaluation management (REMS) plans following PEGylated product approvals.
PEG Disposition, Metabolism, and Excretion
The aspects of biodistribution, metabolism, and excretion of PEG, relevant for nonclinical toxicology assessment of PEGylated biopharmaceuticals, are briefly summarized below. More detailed reviews are available in the literature (Yamaoka, Tabata, and Ikada 1994, 1995; Webster, Didier et al. 2007; Webster, Elliott et al. 2009; Cheng et al. 2012; Ivens et al. 2013; Longley et al. 2013; Rudmann et al. 2013; Baumann et al. 2014).
A core assumption for PEGylated biopharmaceuticals that are associated with the presence of tissue vacuolation attributed to the presence of intracellular PEG is that at some point after repeated administration, a steady state will be achieved, in which the rate of tissue distribution is equilibrated with the rate of catabolism and excretion. Demonstrating this, however, may be difficult.
Limited data are available on distribution, metabolism, and excretion of conjugated PEG, particularly for PEGs with molecular weights ≥20 kDa. One reason for this may be that the amount of PEG linked to biopharmaceutical drugs is usually relatively low (Webster, Didier et al. 2007), requiring sensitive methods to quantify free and/or conjugated PEG in tissues and body fluids. Such methods have advanced only recently (Cheng et al. 2012), although there are no published studies to the best of our knowledge of their application in PEG detection in tissue beyond immunohistochemistry (IHC), radioactive labeling, or ELISA methods. The bioanalytical detection of PEG (conjugated or unconjugated) is challenging because it has repetitive subunits; therefore, chromophore and radiolabeling may be confined to the terminal structure, which is more available to metabolism. For a review of PEG pharmacokinetics (PK), metabolism, and distribution, see Baumann et al. (2014). Even with the availability of recent, more sensitive analytical methods such as nuclear magnetic resonance (NMR), it is still difficult to distinguish between different PEG sizes that might result from (albeit limited) metabolism of the PEG chain (Cheng et al. 2012) or from the inherent polydispersity of the PEG products used in PEGylation. Method development for PEG localization in cellular structures like vacuoles beyond IHC may pose even greater challenges.
Using 125I-labeled PEG of different molecular weights administered intravenously (iv) to mice, Yamaoka, Tabata, and Ikada (1994, 1995) concluded that a two-compartment model could describe the distribution of PEG in the body. The time dependence of tissue accumulation was based on vascular permeability, with larger PEG translocating slowly from the circulation. The half-life increased from 18 min for a 3-kDa PEG to 24 hr for a 190-kDa PEG. Liver clearance, at least in part due to Kupffer cell uptake, was enhanced at higher molecular weights.
Using 14C labeling, Wang et al. (2012) reported a new labeling approach to assess tissue distribution of a PEG-40 kDa protein in mice. Their results show that the lowest amount of PEG-related radioactivity is seen in the brain relative to the other tissues (tissues evaluated in mice: blood, tumor, brain, bone marrow, heart, liver, lung, kidney, and muscle).
There are few literature reports of the use of IHC to identify PEG in tissue (Ton et al. 2007; Sundy et al. 2011; Rudmann et al. 2013). Rudmann et al. showed that the molecular weight of the PEG influenced both the tissue distribution and the vacuolation observed with high iv doses of unconjugated PEG (10, 20, or 40-kDa, 100 mg/kg once daily, every other day, and twice weekly, respectively, for 3 months). With 10- and 20-kDa PEG, PEG immunoreactivity was most prominent in the renal tubule epithelium and in alveolar macrophages, and hepatic Kupffer cells, while cellular vacuolation was absent. In contrast, rats given 40-kDa PEG had strong PEG immunoreactivity in splenic subcapsular red pulp macrophages, renal interstitial macrophages, and choroid plexus epithelial cells frequently associated with cellular vacuolation. These studies with PEG alone also demonstrated that PEG can be found immunohistochemically in cells without vacuolation. It is not known how far these data can be accurately extrapolated to PEGylated biopharmaceuticals or how much their distribution patterns depend on PEG dose versus drug receptor–mediated uptake and/or ligand-specific transport mechanisms. Although anti-PEG antibodies can be used to evaluate specific cellular and subcellular locations of PEG in tissues by IHC and may identify a correlation between presence of PEG and morphological changes, IHC is not quantitative and cannot show the entire sequence of events in catabolism of PEGylated biopharmaceuticals.
There are very few published studies specifically addressing potential mechanisms of cellular uptake of PEG, either alone or conjugated to other molecules (Yu et al. 2004). The predominant mode of cellular uptake (nonspecific vs. drug target mediated) and tissue distribution of PEGylated biopharmaceuticals and/or their free PEG catabolites likely depends on many factors, including dose, dose frequency and duration of treatment, the product’s pharmacological activity, distribution of relevant receptors, potential immunogenicity, overall molecular weight, PEG molecular weight, and types of clearance/removal mechanisms for drug or PEG (Yamaoka, Tabata, and Ikada 1994, 1995; Webster, Didier et al. 2007; Webster, Elliott et al. 2009; Baumann et al. 2014; Bendele et al. 1998). The frequent presence of PEG and PEG-associated vacuoles in Kupffer cells and macrophages as reported in the BioSafe survey (Tables 1, 2 and Table S2 in the Online Appendix) suggests that mechanisms of cellular uptake unique to phagocytic cells are major determinants of tissue reactions to high molecular weight PEG. As has been pointed out by the CHMP working party’s paper (CHMP Safety Working Party of the European Medicinal Agency 2012), this is likely related to the normal removal functions of macrophages.
It can be assumed that irrespective of the cellular uptake mechanisms of a biopharmaceutical due to nonspecific removal or in relation to its biological drug target or function, the protein part of the PEGylated biopharmaceutical will be catabolized into amino acids, and the PEG molecule or parts will be separated from the active drug part since there are few PEG metabolizing possibilities (Webster, Didier et al. 2007; Ivens et al. 2013; Baumann et al. 2014).
Renal clearance appears to be the most common route of excretion for small and large unconjugated PEG molecules. For large PEG molecules, there is evidence that long linear PEG molecules with molecular weights greater than the glomerular filtration rate cutoff for globular proteins can still pass through the glomerulus and can be excreted by the kidney, as seen in studies in mice (Yamaoka, Tabata, and Ikada 1994, 1995; Caliceti and Veronese 2003). This shape-changing behavior is called reptation (Nakaoka et al. 1997; Caliceti and Veronese 2003). The mechanism allows PEG molecules that exceed the glomerular filtration rate cutoff for proteins still to be excreted by the kidney.
PEG has been localized within the proximal convoluted tubules of the kidney (Bendele et al. 1998; Webster, Didier et al. 2007; Rudmann et al. 2013), which is consistent with renal excretion followed by reuptake via mechanisms (such as pinocytosis) that are common to other solutes that are freely filtered at the level of the glomerulus.
Immunogenicity of PEG
PEG has been regarded as a nonantigenic, nonimmunogenic moiety and has been used in some settings to protect proteins from the host’s immune system by reducing their immunogenicity (Dreborg and Akerblom 1990; Roberts, Bentley, and Harris 2002; Saenger et al. 2011). Historically, most PEGylated biopharmaceuticals have proven to be safe and efficacious and have not been associated with the development of anti-PEG antibodies. Recently, a number of reports have documented the development of anti-PEG antibodies following treatment with PEGylated therapeutics, which have been associated with reduced efficacy and hypersensitivity (Ganson et al. 2006; Judge et al. 2006; Armstrong et al. 2007; Hershfield et al. 2014). Some of these reports have evoked controversial discussion (Schellekens, Hennink, and Brinks 2013). It is not yet understood why some PEGylated biopharmaceuticals induce a clinically relevant anti-PEG antibody response, whereas others do not. Some important factors may involve the molecular weight of the PEG moiety, linker-type, number of attached PEG strands, and the immunogenicity of the protein part of the PEGylated biopharmaceutical. One additional variable may also be the assay format and assay sensitivity to detect anti-PEG antibodies (Schellekens, Hennink, and Brinks 2013).
Antibodies against PEG were first documented in animal studies following immunization with PEGylated proteins (Richter and Akerblom 1983). In these studies, PEG was found to be non-immunogenic when administered alone, but acquired immunogenic properties when conjugated to an immunogenic protein. The immunogenic potential of PEG was described as being directly related to the immunogenic nature of the protein. In these situations, PEG was considered to be a polyvalent hapten, a small molecule that can elicit an immune response only when attached to a large carrier molecule, such as a protein. Similar data were recently reported for PEGylated factor VIII (van Helden et al. 2011; Reipert et al. 2012). Two different mouse models were used to assess the immunogenicity of PEGylated factor VIII: a transgenic mouse model that expresses human factor VIII, and therefore recognizes it as self-protein, and a conventional hemophilic mouse model that recognizes human factor VIII as foreign protein. Only those mice that recognized factor VIII as a foreign protein developed anti-PEG antibodies. These data support the hapten concept for the immunogenicity of PEG. The immunological principles of anti-hapten antibody responses were first described by Mitchison (1971) and more extensively studied by Rock, Benacerraf, and Abbas (1984). An additional explanation for the development of anti-PEG antibodies could be cross-linking of B cell receptors that recognize repetitive epitopes within the PEG moiety. This type of antibody response requires multiple regularly spaced identical epitopes to allow simultaneous engagement of multiple B cell receptors with each epitope. The collective signal is sufficient to induce T cell–independent antibody responses (Mond, Lees, and Snapper 1995; Bachmann and Zinkernagel 1997). IgM is the major isotype resulting from T-cell independent antibody responses, although antibody class switching can also occur. IgM immune complexes are efficient activators of the complement system and can rapidly clear antigen from the blood stream, as well as induce hypersensitivity through buildup of the complement anaphylatoxins C3a, C4a, and C5a (Kindt, Osborne, and Goldsby 2006).
T cell–independent mechanisms might also be responsible for the induction of anti-PEG antibodies observed in patients following hyposensitization with PEGylated allergens. Richter and Akerblom (1984) described the development of IgM anti-PEG antibodies in approximately 50% of patients undergoing hyposensitization with PEGylated allergens.
Formation of anti-PEG antibodies has been associated with rapid blood clearance and loss of treatment effect in a subset of patients treated with PEG-uricase (Krystexxa®) and PEG-asparaginase (Oncaspar®; Ganson et al. 2006; Armstrong et al. 2007; Hershfield et al. 2014). Krystexxa is a PEGylated mammalian enzyme used for the treatment of chronic refractory gout (Kelly et al. 2001; Ganson et al. 2006; Sundy et al. 2011). The enzyme is covalently attached to multiple strands of 10-kDa monomethoxy PEG. Low titer anti-PEG IgM and IgG antibodies were detected at 3 to 7 days and at 7 to 14 days after the initial dosing, respectively. Anti-PEG antibodies were associated with rapid blood clearance of the drug and reduced treatment effect in a subset of patients. Oncaspar is a bacterial enzyme covalently linked to multiple 5-kDa methoxy-PEG molecules that is used for the treatment of acute lymphoblastic leukemia in pediatric patients (Schrappe, Reiter, Ludwig, et al. 2000; Schrappe, Reiter, Zimmermann, et al. 2000; Schrappe, Camitta, et al. 2000). Anti-PEG antibodies have been associated with rapid blood clearance and reduced treatment efficiency in up to one-third of patients treated with Oncaspar (Muller et al. 2000; Armstrong et al. 2007).
Interestingly, there have been several reports on the detection of anti-PEG antibodies in healthy blood donors, who have not received PEGylated biopharmaceuticals, suggesting that environmental PEG exposure may lead to the development of anti-PEG antibodies in a subset of the population (Richter and Akerblom 1984; Garratty 2004, 2008; Armstrong et al. 2007; Garay et al. 2012). Moreover, PEG is used during manufacturing of certain vaccines and plasma-derived biopharmaceuticals, for example, certain influenza virus vaccines (Polson 1976), hepatitis B virus vaccines (Takao et al. 1986), and plasma-derived FVIII products (Chtourou 2012). Residual PEG in these products could induce anti-PEG antibodies in individuals. It remains to be determined whether preexisting anti-PEG antibody responses affect the efficacy or safety of PEGylated biopharmaceuticals in drug-naive patients. Recently, Armstrong et al. (2007) speculated that preexisting anti-PEG antibodies might be the reason for reduced circulating asparaginase activity in patients treated with PEG-ASN.
Most PEGylated biopharmaceuticals have not been associated with the development of anti-PEG antibodies. There are some recent reports describing the development of anti-PEG antibodies, following treatment with some PEGylated biopharmaceuticals, that have been associated with reduced treatment effect and hypersensitivity (Mehvar 2000; Caliceti and Veronese 2003; Veronese and Pasut 2005; Hamidi, Azadi, and Rafiei 2006). In response to an increasing number of reports of PEG-related immunogenicity for products in development, the FDA has recommended assessing for anti-PEG antibodies in all subjects treated with experimental PEGylated protein biopharmaceuticals and to determine whether clinically significant changes in efficacy or safety are associated with these antibodies (FDA 2014). The FDA reports that anti-PEG antibodies have been associated with changes in PK, loss of product efficacy, and related adverse events. In addition, the FDA cautions that anti-PEG antibodies have been found to cross-react between PEGylated products (FDA 2014), suggesting that treatment with one PEGylated product might inadvertently sensitize an individual to other, unrelated PEGylated biopharmaceuticals.
In conclusion, anti-PEG antibodies should be monitored in clinical trials involving novel PEGylated protein biopharmaceuticals. Potential effects of anti-PEG antibodies on safety and efficacy should be carefully assessed.
PEG-related Cellular Vacuolation
Cellular vacuolation has been reported for 5 of the 11 approved PEGylated biopharmaceuticals (Table 2) and for 10 of the 17 products currently in nonclinical or early clinical development from the BioSafe survey (Table 1). For those PEGylated biopharmaceuticals associated with tissue vacuolation, the occurrence, incidence, and severity of vacuolation increase with dose, dosing frequency, and duration in toxicology studies as seen from the survey data. The assumption that vacuoles represent the presence of PEG is generally supported by dose dependency, but also the similarity between tissue patterns of vacuolation and the pattern of tissues that are immunohistochemically positive for PEG. The vacuoles associated with PEG are morphologically consistent with lysosomes by light microscopy, but these observations have not been confirmed by ultrastructural studies.
It is important to note that currently there are no widely accepted standardized histological criteria to grade the severity of PEG-related cytoplasmic vacuolation in animal tissues. Establishing standardized severity grading criteria for PEG-related cytoplasmic vacuolation may facilitate or enhance comparison across biopharmaceuticals from different sponsors, different study pathologists, and different types of PEGs.
Since anti-PEG antibodies suitable for IHC procedures have only become commercially available relatively recently, the data looking at correlation between vacuolation and IHC are sparse (Krystexxa FDA summary basis of approval [SBA]; Ton et al. 2007; Sundy et al. 2011; Rudmann et al. 2013). IHC studies using anti-PEG antibodies have also demonstrated that PEG-related cytoplasmic immunoreactivity occurs in cells that were not vacuolated in hematoxylin and eosin histological stain (H&E) sections (survey results for company 6, compound 2; Bendele et al. 1998; Rudmann et al. 2013) and, although the presence of vacuoles in toxicology studies of PEG or PEGylated therapeutics generally correlates with PEG dose, the vacuoles are not always immunoreactive for PEG (Bendele et al. 1998). This may reflect different sensitivity and specificity of available reagents, and/or duration of fixation prior to conduct of IHC, but also suggests that morphologic observations of vacuolation may not reflect PEG tissue burden. Other analytic techniques, and/or standardized anti-PEG reagents with higher sensitivity currently not available, may have to be developed to address this.
PEG-related vacuolation has been observed more frequently at PEG molecular weights ≥30-kDa (Table 1 and Table S2 in Online Appendix; Rudmann et al. 2013). One reason may be that smaller PEG molecules of 5-kDa or less are excreted readily by glomerular filtration and therefore systemic exposure is shorter and/or lower (Yamaoka, Tabata, and Ikada 1994, 1995). Vacuolation has also been observed in toxicology studies with PEGylated therapeutics with lower molecular weight PEG when, for example, with Krystexxa the product contains a number of PEG molecules (8 to 10, 10-kDa PEG molecules attached to each subunit of the tetramer protein uricase resulting in a large PEG dose). Bendele et al. (1998) demonstrated that after administration of PEG-conjugated tumor necrosis factor binding protein (TNF-bp) constructs with different PEG molecular weights (20-kDa PEG vs. 50-kDa PEG), the severity of renal tubular vacuolation was inversely related to molecular weight and PEG complexity (Bendele et al). In this study, a 50-kDa PEG-protein dimer (total molecular weight 86-kDa) was associated with the least severe vacuolation, and the 20-kDa PEG-protein monomer (total molecular weight 38-kDa) was associated with the greatest severity. Renal tubular vacuolation was not observed when the PEG molecules alone were administered. The observed differences may be related to exposure/reuptake due to the protein moiety and differences of glomerular filtration of the PEGylated monomer versus dimer.
Despite the fact that most PEG-related vacuolation is noted with PEGs at least 30-kDa, the exceptions described earlier illustrate the difficulties in making generalizations about PEG-related vacuolation and the contribution of PEG size since exposure and pattern of vacuolation are influenced by multiple factors including overall molecular size, dose, dosing frequency and pharmacological properties including nonspecific or drug target receptor mediated uptake, PK, and physiologic filtration barriers (Tables 1 and 2). Currently, there are insufficient published data regarding biopharmaceuticals utilizing PEG molecules larger than 40 kDa to understand PEG at the high end of the molecular weight spectrum.
Since cellular vacuolation is a nonspecific finding that can occur in response to a variety of influences and stimuli, the occurrence of vacuolation in toxicology studies of PEGylated biopharmaceuticals may not always be due to the presence of PEG in tissues. The actual presence of PEG in vacuoles can currently only be confirmed by IHC with anti-PEG detection antibodies.
The most commonly vacuolated cell types related to high molecular weight PEG are phagocytic cells like macrophages (Tables 1 and 2). Vacuoles have also been observed in non-phagocytic cell types including renal tubular epithelium, endothelium, synovial epithelium, adrenal cortical cells, choroid plexus epithelium, and in one case in neurons (Tables 1 and 2).
In summary, an understanding of the details of vacuole formation, their association with cellular organelles, the content of these vacuoles, and their relationship to PEG remains a gap in our understanding of PEG-related effects and their potential impact on organ function.
Vacuolated Cell Types
Phagocytic cells
Phagocytic cells containing vacuoles after administration of PEG or PEGylated biopharmaceuticals include Kupffer cells in the liver, reticuloendothelial cells in the spleen, and macrophages in a variety of tissues (lung, skin after subcutaneous [sc] injection, lymph nodes, bone marrow, thymus, adipose tissue, ovary, testis, lamina propria of the urinary bladder, vagina, cervix, endometrial stroma of the uterus, brain, adrenal cortex, pituitary, and the choroid plexus). Since removal of foreign bodies or materials from circulation is a normal function of these macrophages and histiocytes, the resulting vacuolation is considered a normal physiological response in phagocytic cells.
Whether the dominant mechanism of PEG uptake by phagocytic cells is via engulfment of free PEG, phagocytosis of damaged/senescent cells containing PEG or other potential mechanisms unique to phagocytic cells is currently not clear. Endocytic uptake into phagocytic cells has been classified as pinocytosis or phagocytosis (large particles > 250 nm) based on particle size, energy requirements, and morphologic attributes observed by electron microscopy. In the cells, these phagosomes (Ton et al. 2007; Hirota 2012; Oh and Park 2014) and endosomes fuse with lysosomes where the ingested materials are recycled or degraded. Indigestible materials are exocytosed or remain within the lysosome forming residual bodies.
Metabolism of PEG in phagosomes and lysosomes is likely limited since the enzyme repertoire for PEG metabolism in the body is limited (Webster, Didier et al. 2007; Ivens et al. 2013; Caliceti and Veronese 2003; Baumann et al. 2014). Details of the fate of PEG once ingested by phagocytic cells are currently not known. PEG may be exocytosed to be removed from the body by organs such as liver, kidney, and spleen, or it may remain in macrophages over their life span (Oh and Park 2014). Different types of macrophages have different life spans. While monocyte-derived macrophages are short lived in peripheral tissues and die within 2 weeks, other tissue macrophages have a longer life span and can survive for months, although life span data reported are variable (e.g., for Kupffer cells 5 weeks to 14 months) and may vary from tissue to tissue (Takahashi 2001; Parihar, Eubank, and Doseff 2010). This may be one underlying cause for long recovery times for certain tissues with vacuolated macrophages as seen in toxicology studies (see survey Table S1 for recovery times).
Non-phagocytic cells
PEG-associated vacuoles have been observed by light microscopy in H&E-stained sections in a limited spectrum of non-phagocytic cell types including proximal tubular epithelial cells, glomerular podocytes, and glomerular endothelial cells within the kidney, adrenal cortical cells, choroid plexus epithelial cells, and endothelial cells in cerebral cortical capillaries and the aorta (Bendele et al. 1998; Rudmann et al. 2013; Table 1). Vacuolation has also been observed in parenchymal (epithelial) cells of other tissues including joints (synovium), liver, pancreatic islets, prostate, epididymides, urinary bladder, mammary gland, thyroid gland, and ciliary body, as well as neurons in the dorsal root ganglia (DRG) and brain.
Vacuolated cells in the nervous system
Choroid plexus and/or pituitary gland vacuolation was observed in 7 of the 17 products in development in the BioSafe survey, respectively. In only 1 of the 7 molecules, PEG (2.4-kDa peptide with linear 30-kDa methoxy PEG; see company 6, product 2 below, indicated for type 2 diabetes in Appendix for more details) was detected by IHC in the choroid plexus vacuoles and in neurons (with and without vacuolation). The observations may be specific to this molecule and the nature of the peptide moiety, which was linked to brain penetration, potentially due to a protein-specific transporter (see also Tables 1, S1, and S2). It is assumed that this mechanism impacted the disposition of the PEG moiety in the nervous system. This product was discontinued from further development.
Because most neurons are nonrenewable or slow renewable cells, the observation of vacuoles in neurons is of concern in safety assessments. But the presence of PEG-related vacuolation in the choroid plexus cells does not indicate the presence or transport of PEG into neuronal or glial tissue.
Since vacuolation of the choroid plexus has been seen repeatedly, it might be important to understand its relation to the central nervous system (CNS). There are multiple barriers between the CNS and the circulation. The barrier between the blood and the brain or spinal cord parenchyma, referred to as the blood–brain barrier (BBB), is formed by the endothelium and pericytes of the capillaries in the neuropil. This barrier is distinct from the blood-cerebrospinal fluid (BCSF) barrier (BCSFB). The BCSFB is formed by the tight epithelium of the choroid plexuses and is present in all 4 ventricles of the mammalian brain. The layers of the BCSFB include a single cuboidal cell layer facing the CSF, derived from the ependyma; a layer of taller cuboidal choroidal epithelial cells, derived from the leptomeninges; and an inner core comprised of highly vascularized connective tissue with permeable blood vessels. This vascular core, where vacuolated macrophages presumed to contain PEG are sometimes observed, is outside of the CNS. Choroidal epithelial cells differ fundamentally from the adjacent ependymal cells by the presence of tight junctions and therefore form the anatomic basis for the restrictive BCSFB (Strazielle and Ghersi-Egea 2013). They also differ from cell types of the BBB in having a strong endocytotic and metabolic character with a large number of related vesicles (Abbott, Ronnback, and Hansson 2006; Strazielle and Ghersi-Egea 2013; Liddelow 2015). In view of the permeability of the blood vessels in the core, it is unclear whether the hydrophilic PEG filters through the vasculature into the core and is kept there by the choroidal epithelial tight junctions and is subsequently phagocytosed by resident macrophages, or whether circulating macrophages containing PEG filter out in this high blood flow area. It can be concluded that the choroid plexus has a barrier function for PEG, even when vacuolation in choroidal epithelial cells is present.
After iv administration of high doses of unconjugated 10-, 20-, and 40-kDa PEG over 3 months, PEG immunoreactivity and vacuolation were observed only with 40-kDa PEG in choroid plexus macrophages and choroid plexus epithelial cells but not in brain parenchyma, suggesting that the choroid plexus acts as a barrier between the blood and the ventricular CSF for unconjugated PEG (Rudmann et al. 2013).
While unconjugated PEG does not appear to enter the CNS, PEG may reach areas inside the BBB when it is conjugated to a biotherapeutic that can bind to a drug transporter or drug target receptor that facilitates crossing either the BBB or the BCSFB. In rats, sc recombinant human IGF-1 conjugated to a 40-kDa PEG was reported to reach higher steady state concentrations in brain tissue and CSF than non-PEGylated IGF-1, though it entered the CNS more slowly. In mice, administration of the same PEGylated IGF-1 sc resulted in dose-dependent increases in human IGF-1 in hippocampal CA1 neurons as assessed by IGF-1 immunoreactivity in frozen brain sections (Saenger et al. 2011). Immunoreactivity for localization of PEG and microscopic analysis for assessment of vacuolation were not performed in these studies, so the presence of PEG or potentially PEG-associated vacuolation was not evaluated. Kronenberg et al. (2013) describe briefly neuronal vacuolation after administration of a different PEGylated polypeptide, with confirmation of PEG content in the vacuoles using IHC that was only partially reversible. No further details are available.
The cases described above have used IHC for PEG detection. Wang et al. (2012) reported a new labeling approach for a PEG-40 kDa protein to assess tissue distribution in mice (40-kDa branched PEG-Adnectin). The results from this technique show the lowest amount of PEG-related radioactivity in the brain relative to other tissues (tissues assessed in mice: blood, tumor, brain, bone marrow, heart, liver, lung, kidney, and muscle). To the best of our knowledge, no other methods have been published that are sensitive enough to detect PEG in brain tissue.
It appears that while unconjugated (free) PEG does not enter the CNS, there may be transport of PEG across the BBB when conjugated to a therapeutic peptide, probably depending on the drug transporter.
Reversibility of PEG-related vacuolation
PEG-associated vacuolation may partially or completely reverse after a treatment-free period, although often the recovery periods used in nonclinical toxicology studies as seen from the survey results were too short to demonstrate reversibility (Tables 1 and S2). In the study by Bendele et al. (1998), cytoplasmic vacuolation in renal tubular epithelial cells remained after a 2-month recovery period, despite resolution of immunoreactivity for the protein to which the PEG was conjugated, suggesting that lysosomal enzymes may be able to process the protein but not the PEG. Larger vacuoles or multiple vacuoles may take longer to resolve during a treatment-free period, and additionally, retention of vacuoles may reflect the slow turnover of the cell population in which the vacuoles reside. As discussed earlier, for example, tissue macrophages may have a life span of up to 14 months (Takahashi 2001). In a retrospective review of approval information from FDA and EMA, for 11 marketed PEG conjugated biologics, there was indication that increasing dose and dosing duration was associated with reduced reversibility (Table 2). Nonetheless, the weight of evidence of published data from the survey suggests that the vacuoles are reversible, given sufficient recovery time.
Impact of PEG-associated vacuoles on cellular or organ function
In general, published studies indicate that despite significant cytoplasmic vacuolation of cells such as macrophages or renal tubular epithelial cells, adverse effects on organ function were not seen (Bendele et al. 1998; Sundy et al. 2011; Ivens et al. 2013). Rudmann et al. (2013) proposed that the cytoplasmic vacuolation seen after PEG administration is an adaptive rather than a toxic response and related to the increased demand for clearance of PEG.
In vitro or ex vivo effects on macrophage function are reported for two approved PEGylated drugs, certolizumab pegol (Cimzia®) and pegloticase (Krystexxa) (European public assessment reports [EPARs] EMA; FDA SBAs). In vitro macrophage function assays indicated that the PEG moiety alone reduced phagocytosis of bacteria and fungi at high concentrations above the intended pharmacological use (no effect level [NOEL] 1.0 mg/mL). Some inhibition of T cell proliferation to a toxoid challenge in a human cell system was also seen (NOEL 1.0 mg/mL; EMA EPAR). Administration of pegloticase to rats or dogs results in vacuolation of macrophages (EPAR EMA, FDA SBA), and IHC methods confirmed that PEG was present within the vacuoles of macrophages from dogs. Although the conclusion was that the vacuolation did not result in adverse effects to the health of the animals, vacuolated macrophages isolated from spleen of rats administered pegloticase had a reduced response to lipopolysaccharide challenge (FDA SBA). To the best of our knowledge, no in vivo host resistance studies have been conducted with certolizumab pegol or pegloticase to determine if the in vitro results translate in vivo.
As discussed earlier, administration of a PEGylated TNF-bp to animals resulted in marked vacuolation of renal tubular epithelial cells at the mid-doses and HDs, but no measurable effect on renal function as assessed by blood urea nitrogen (BUN), creatinine, urinalysis, and specific more sensitive renal markers (urinary N-acetyl-beta-
As previously mentioned, questions have been expressed by regulators regarding the potential impact of PEG-related vacuolation on the function of epithelium of the choroid plexus, particularly in pediatric populations (CHMP Safety Working Party of the European Medicinal Agency, 2012) and on the ability of the choroid plexus to generate CSF or transport hormones. It is unclear how to reliably evaluate effects on the function of the choroid plexus, since there are no readily available established biomarkers. It can be postulated that impairment of the choroid plexus ependymal function would lead to clinical symptoms in these animals. However, nonclinical toxicology studies of PEGylated drugs up to chronic duration did not report behavioral abnormalities during standard clinical observations in the toxicology studies in animals presenting choroid plexus vacuolation (FDA SBA or EMA EPAR for marketed products, see Table 2). None of the companies participating in the BioSafe survey reported special studies assessing animal behavior, for example in a functional observational battery for the 17 compounds.
In an overview paper, Kronenberg et al. (2013) briefly summarized that the presence of PEG in neurons did not affect function or viability as assessed by nerve conduction velocity and Fluoro-Jade staining, although no details of these studies are available.
Review of Publicly Available Toxicology Information of Approved PEGylated Biopharmaceuticals
Since 1990, 11 PEGylated biopharmaceuticals have been introduced into the market place as drugs for human use (Table 2). Ten are PEGylated protein biopharmaceuticals and 1 is a PEGylated aptamer (Macugen®) administered intravitreally. The PEG components of these biopharmaceuticals vary widely in size (Table 2), branching structure, and attachment type.
For Adagen® and Oncaspar, no information on toxicology studies was publicly available on the FDA or EMA web pages. For the other 9 biopharmaceuticals, the publicly available information related to vacuolation in the toxicology studies summarized by regulatory agencies is presented in Table 2. The table lists pivotal toxicology studies and information to date without details of the entire toxicology programs.
Biosafe Survey of PEGylated Biopharmaceuticals
A pharmaceutical industry survey was conducted by BioSafe (committee within Biotechnology Industry Organization industry organization, BIO) in 2013. Ten companies provided information on 17 PEGylated biopharmaceuticals currently in development to gather toxicological data. The objective of the survey was to collate recent company experiences with PEGylated biopharmaceuticals, to provide a forum for sharing knowledge, and to assess the basis for the apparent increase in industry and regulatory concerns around PEGylated biopharmaceuticals. PEG-related tissue vacuolation was seen in toxicology studies for 10 of the 17 compounds in the survey. No other PEG-related effects were reported.
The survey (blank questionnaire provided in the supplementary information, see Table S3) is summarized in the following and also in Tables 1, 2, and Tables S1, S2, S3 in the Online Appendix. Anonymity of the respondents and the products was maintained throughout.
Type of PEGylated Protein Products
The 17 products were heterogeneous, ranging from replacement molecules to receptor or protein targeting products with molecular weights ranging from 25 kDa to 350 kDa, although most products were less than 60 kDa. For 14 of the described molecules, the molecular weight of the PEG moiety exceeded the protein weight. The molecular weight of the PEG moiety ranged from branched or linear 20 kDa to 60 kDa (further details regarding the nature of the PEG moiety are given in Table S2, Online Appendix).
Clinical Indication and Patient Populations, Therapeutic Dosing Regimen, and Stage of Development
The clinical indications (e.g., rheumatoid arthritis, type 2 diabetes, age-related macular degeneration, muscle wasting, and multiple sclerosis) generally required chronic treatment and included both elderly and pediatric (for 6 products) populations (Table S2). Information about the human PEG intake per kg bodyweight and dosing schedule was provided for only a few of the products since most were still in early development and the clinical dose was not yet determined (see Table S1). The dosing regimens of the toxicology studies varied widely from microgram/kg to milligram/kg, with dosing frequency of daily to weekly, and study durations from 2 to 52 weeks (Table 1).
Four of the 17 products in the survey were terminated during development, 2 for reasons unrelated to PEG. Ten products were in clinical development but not yet approved for marketing. No information was provided about the development/approval status for 3 products.
Assessment of Disposition of PEGylated Protein and PEG Moiety
For 5 of the 17 products, there was some evidence for an impact on disposition related to drug target receptor-mediated uptake of the PEGylated protein. For the remaining 12 products, it is not known whether there was receptor-mediated uptake or whether it was not evaluated (Table S1).
For 7 of the 17 products, no specific studies were conducted to assess the disposition of the PEG moiety, while information on PK and/or disposition was gathered from 10 products (Table S1). The disposition of 3 of these 10 products was assessed using an assay for the drug only (radio immune assay or ELISA) with protein capture/anti-PEG detection. For 2 products (companies 5 and 7, see appendix for details), disposition was assessed using either a single iv dose of 125I-labeled product (information about site of radiolabel or whether on PEG moiety was not provided) or a single HD of tritium-labeled PEGylated recombinant coagulation protein with the label in the PEG.
The remaining 6 products reported IHC methodology (4 specifying PEG) without much detail. For 1 product, only exploratory studies were performed, as the method was insufficiently sensitive to detect low levels of PEG (Table S1). Only 2 of the 6 products discussed results of the IHC, but the correlation to PEG immunostaining of vacuoles was inconsistent.
Immunogenicity Assessment
For 11 products, there were no specific assays for measurement of anti-PEG antibodies in nonclinical studies. One stated that no anti-PEG antibodies were seen in a single-dose clinical study and others stated that clinical assays were available or planned (Table S1). Assays to detect antibodies to PEG in nonclinical studies were developed for 6 products, but either no positive responses were observed or there was little impact on the interpretation of the study. One company (7) stated that the development of IgM and IgG anti-PEG antibodies was observed in mouse models and macaques, but results were not considered to have any impact on safety considerations for humans.
Cellular Vacuolation in Toxicology Studies
Types of toxicity studies ranged from repeated iv or sc dose studies in mice, rats, dogs, and cynomolgus monkeys of 2 to 52 weeks duration. The dosing frequency varied from daily to weekly (Tables 1 and S1). For 1 product (Company 9), iv and intravitreal toxicology studies were conducted. No cellular vacuolation was observed for 7 of the 17 products (Tables 1 and S2). The dose levels and duration of dosing for these products were generally low and/or short term (up to 4–5 weeks). For 2 of the products, studies of 26 to 39 weeks in duration were conducted. None of the 4 products with a single 20-kDa PEG showed evidence of vacuolation (company 1 product 1, company 5 product 1, company 8 product 1, and company 9 product 1). In general, the maximum study duration for these 4 products was only 5 weeks, although for 1 product there was a 26-week study in cynomolgus monkeys. However, cellular vacuolation was observed in some of the 4-week studies with 30- to 40-kDa PEG products.
For several products, health authority interactions were not disclosed (Table S2). Company 5 did not disclose whether they had received any health authority questions, and company 8 stated that they had received questions from the EMA but no details were provided. Company 9 received questions from 1 health authority on their intravitreal product relating to the fate of PEG after intravitreal dosing and the potential impact of long-term dosing/accumulation of PEG by retinal pigmented epithelial cells.
Toxicology Studies with the PEG Moiety Alone
No specific studies on the PEG moiety were conducted for 14 of the 17 products, and for 1 additional product the response to this question was “undisclosed” (Table S1). Studies on the PEG moiety were conducted for 2 products: no evidence for PEG-related vacuolation was observed in a 4-week rat study (company 2). Company 8 included a PEG-only arm in the 4-week rat study for product 4. The 40-kDa 2-branched PEG was conjugated to cysteine (to block the reaction site of linker) and dosed to give PEG concentrations comparable with respect to the affected tissues and overall magnitude of vacuolation to the HD of the PEGylated protein. The observed vacuolation was comparable between the PEGylated protein and the PEG-cysteine.
Special Investigations
The survey asked to describe any special methodology used to evaluate potential adversity of PEG presence and/or cell vacuoles (e.g., IHC, electron microscopy, functional evaluations such as nerve conduction velocities, or in vitro studies). No special investigations other than IHC were conducted for 16 products. Additional veterinary examinations or neurological function included for 1 product (company 4) received the following comment from health authorities: “We are also aware of data that PEG can accumulate in neurons with other investigational PEGylated neurologically active peptides and that this has been correlated with impaired nerve function. The distribution of PEG-accumulating neurons is consistent with binding and internalization of the PEGylated peptide by neurons that bear specific receptors for the peptide. Notably, PEGylation does not appear to preclude transfer of brain-permeable peptides across the blood-brain barrier” (Tables 1 and S1).
Health Authority Interactions
No questions were received from health authorities for 6 products and the response to this question for an additional company was undisclosed. Health authority questions were raised for 10 products, and a summary of the questions raised and company responses is given in Table S2 and discussed earlier. However, based on the findings in the survey, the questions raised by health authorities relating to the disposition and fate of the PEG moiety and the impact of PEG vacuolation, including functional effects, are scientifically relevant.
Considerations for Nonclinical Toxicology Evaluations of PEGylated Biopharmaceuticals
The development of new PEGylated biopharmaceuticals has triggered the need for the formulation of a strategy to address the impact of PEG-related cytoplasmic vacuolation when seen in nonclinical toxicology studies. It should be considered that histologically observed cellular vacuolation alone without other changes in the surrounding cells (e.g., no cellular distortion, necrosis, or inflammation) should not by default be considered adverse as the tissue/cellular vacuolation can be adaptive and not a sign of a toxic response (see also Rudmann et al. 2013). Other factors such as the type of cell affected as well as the margins between observations of vacuolation in animal studies compared to clinical dose, dosing frequency, and duration of treatment, and the intended indication and target population should be considered when determinating the risk/benefit assessment of PEGylated biopharmaceuticals. The determination of a NOAEL as well as its application may be influenced by the underlying definition of adversity and the data available (for a discussion on NOAEL definition, see Dorato and Engelhardt 2005).
The following provides information for considerations to interpret PEG-related cytoplasmic vacuolation in nonclinical toxicity studies: Not adverse: vacuolation observed in phagocytes (RES) in the liver, spleen, muscle, kidneys, or other organs as long as the phagocytes are not distorting the normal tissue architecture, and there is no evidence of histopathological changes such as degeneration, inflammation, or necrosis, and there are no changes in biomarkers of injury and no impaired physiological function. A presence of vacuolated phagocytes in these tissues allows the determination of a no adverse effect level (NOAEL), supported by a lack of impairment of function. These changes may be adaptive and related to the normal physiological function of removal of macromolecules and particles by phagocytic cells. Not adverse: vacuolation observed in parenchymal or interstitial cells of organs such as the liver, spleen, muscle, kidneys, or other organs as long as there is no evidence of histopathological adverse changes such as degeneration, inflammation, or necrosis, and there are no changes in biomarkers of injury and no impaired function. A presence of vacuolation in these cells allows the determination of an NOAEL since there was no impact on function and no adverse effects.
Vacuolation in major organs or secondary changes in tissues associated with changes such as degeneration, inflammation, or necrosis may require further assessment and/or additional experiments or inclusion of new study end points (see considerations below) to determine adversity. Risk assessment needs to consider, for example, location, severity and reversibility/partial reversibility of vacuolation, NOAEL or NOEL, clinical indication (chronic or short term), and human PEG dose. In addition, it may be considered if application of a higher safety margin is warranted.
The interpretations above may change as additional understanding of the significance or mechanisms of PEG-related vacuolation and the possible impact, if any, on cellular function is gained.
Progression and Reversibility of Cellular Vacuolation
The evaluation of PEG-related cellular vacuolation has generally been limited to the histological detection of vacuoles at the relatively small number of necropsy time points that are typically included in toxicology studies. The evaluation of time of onset, progressivity, or lack thereof, at a given dose and treatment regimen, as well as recovery of vacuolation is usually not studied for most PEGylated biopharmaceuticals. On a case-by-case basis, it may be supportive to determine that vacuolation is not progressing beyond a certain point. When toxicology studies of different duration are available for a specific PEGylated biopharmaceutical, it may be possible to determine the potential progressivity and also recovery of PEG-related vacuolation by comparing the findings from different studies without conducting additional toxicology studies, especially when the nonhuman primate is the only relevant species.
It is important to consider that the PEG dose levels used for determination of any progression or reversibility should be in the range of the human clinical dose of PEG. Doses of PEGylated biopharmaceuticals several fold higher than the intended clinical dose and differences in dosing schedule of the toxicology studies may show higher severity and also cellular vacuolation in organs/tissues not representative for the human scenario. For example, recovery from high PEG loads may take longer than at clinically relevant doses.
Evaluation of Biomarkers of Organ Function
When vacuolation is seen in parenchymal or interstitial cells of organs such as the liver, spleen, muscle, or kidneys, biomarkers of organ and tissue functions may support demonstrating the nonadversity of the finding. Routine biomarkers of liver and kidney function are usually already included in toxicology studies as part of the biochemical evaluation of serum/plasma (i.e., “clinical chemistry profile” with urinalysis). In select instances, adding additional biomarkers of tissue injury or function may add value and should be considered on a case-by-case basis.
Toxicology Program Design for New PEGylated Biopharmaceuticals
In order to facilitate development of new PEGylated drug candidates, the following points may be considered during nonclinical development concerning PEG-related vacuolation.
Vacuolation is not seen with all PEGylated biopharmaceuticals and if present is not typically an indication of an adverse effect. The human risk of potential tissue vacuolation on physiological function can be determined by evaluating the type of vacuolation and tissue location in relation to human drug dose. The following summarizes experiences gained from nonclinical development of PEGylated drugs. These points should be considered on a case-by-case basis based on product attributes as well as clinical indication. The demonstration of partial or full reversibility of potential vacuolation may need longer than standard recovery time (in particular whether uptake is anticipated in cells with slow turnover). Reversibility data obtained in the clinical dose range are more relevant for assessment of potential clinical risk. Genotoxicity studies of PEG with linker may need to be done (small molecule linkers only; dependent on information available related to the linker used; see also ICH S6 R1, 2011, guidance on linkers). The stability of the covalent binding between protein/aptamer and PEG may need to be considered if a potential for PEG released from the drug molecule exists (this could result in differences in biodistribution of the PEGylated biopharmaceuticals and the free PEG). The presence of tissue-specific, drug target–related, or drug receptor–mediated uptake or the possibility of active transport of drug may impact toxicology findings and their interpretation. Biomarkers of organ/tissue function may support safety assessment and may be included in toxicology or safety pharmacology studies to support risk assessment when technologically feasible.
Considerations when vacuolation has been observed in toxicity studies: The type of vacuolated cells, phagocytic (consider that phagocytic cells/macrophages are present in many tissues/organs) versus non-phagocytic may impact safety assessment. The relation and degree (severity) of vacuolation in relation to treatment (dose, duration and frequency of dosing, potential accumulation, steady state of PEG, degree of vacuolation, presence, or absence of inflammation or distortion in vacuolated tissue) may be important to consider. Define and determine NOEL versus NOAEL (considering that vacuolation may be a natural removal mechanisms). Information on PK, distribution, excretion of PEG, or the PEG protein can support interpretation. In certain cases, the addition of a PEG-only arm in one of the repeated administration toxicology studies may be a tool to support separating receptor-mediated cellular uptake from “nonspecific” uptake. However, it has to be recognized that the PK and biodistribution of PEG alone are different from that of the PEGylated biotherapeutic. IHC can be used to determine if vacuoles contain PEG.
In specific cases, the following may be taken into consideration for risk assessment: For biotechnology-derived drugs with a potential for drug target receptor–mediated uptake or active transport across the BCSFB, consider showing lack of histological findings and PEG presence by IHC in the ventricular ependyma and other parts of the brain. Additional studies on immune function can be of value, if immune-related tissues like RES show a high grade of vacuolation, for example,
ex vivo macrophage function assay and
in vivo host resistance studies in rodents. The assessment of vacuoles in circulating monocytes could provide a biomarker for use in humans in some cases.
Discussion
PEGylation is a widely accepted approach to improve the pharmaceutical properties of biopharmaceuticals and has been used increasingly in development of new large molecule drug candidates. Cellular vacuolation seen histologically in toxicology studies with PEGylated biopharmaceuticals is the only effect related to PEG alone. Vacuolation has been reported in a number of organs and tissues for approximately half of the PEGylated biopharmaceuticals. In macrophages (fixed and infiltrating macrophages, including Kupffer cells) of the RES, vacuolation is seen most frequently (Rudmann et al. 2013) and may be an adaptive response to remove PEG. Uptake of PEG into these phagocytic cells is due to mechanisms not related to the drug target. Other cellular uptake mechanisms in non-phagocytic cells may be related to the active drug part of a PEGylated biopharmaceuticals through their cellular drug target receptor (target mediated uptake), or by scavenger receptors removing proteins, present for example in hepatocytes.
Experience with PEG and PEGylated biopharmaceuticals shows that no functional changes related to PEG for organs and tissues where cellular vacuolation was seen have been reported with the 11 approved drugs in animal toxicology studies. No functional changes were described for the 17 candidates in development in the BioSafe survey when tissue vacuolation was seen microscopically in toxicology studies.
It has been implied that vacuoles contain PEG; however, the only method to detect PEG with sufficient sensitivity in individual tissues that discriminates cytoplasmic PEG from PEG in vacuoles to date is IHC utilizing anti-PEG detection antibodies. The survey results showed that PEG has been detected in vacuoles when IHC was employed. Further, PEG positive staining by IHC was detected in some cases without vacuolation. These reports indicate that the limits of detection of PEG in tissue may vary based on the methodology and PEG-detection antibody employed. On a case-by-case basis, an integrated assessment with functional end points may support the positioning of the significance of the presence of vacuoles.
Published studies indicate that despite significant cytoplasmic vacuolation of cells such as macrophages and renal tubular epithelium, adverse effects on organ function do not commonly occur. In 2 cases, the presence of PEG within phagocytes has been associated with demonstrated effects on function in in vitro assays at high concentrations above the intended pharmacological use (certolizumab pegol and pegloticase, EMA EPAR 2012, FDA SBA 2010). There are also 2 published examples of renal tubular degeneration without impact on kidney function markers reported from specially designed research studies in rats. Bendele et al. (1998) included sensitive markers for proximal and overall renal tubular functions. Tubular degeneration was associated with renal tubular vacuolation after administration of PEG alone or PEGylated protein (Bendele et al. 1998; Rudmann et al. 2013). In the study conducted by Rudmann PEG alone was dosed iv and rats received high doses of 3 g/kg/month (300 µmol/kg/month) of PEG 10-kDa PEG, 1.5 mg/kg/month of 20-kDa PEG (75 µmol/kg/month), and 0.8 g/kg/month of 40-kDa PEG (20 µmol/kg/month; Rudmann et al. 2013). Overall, in vivo evaluation of the functional consequences of PEG presence has not identified any adverse effects on the most consistently affected organ systems (kidney and lymphoid organs).
The review of publicly available toxicology information for the 11 approved PEGylated biopharmaceuticals indicates that their toxicologic effects were derived from the active part of the drug molecule rather than the PEG moiety (Webster, Didier et al. 2007; Kang, Deluca, and Lee 2009; Ivens et al. 2013). The only effect attributed to PEG seen in these nonclinical toxicology studies was cellular vacuolization observed with 5 of the 9 approved PEGylated biopharmaceuticals for which toxicology information is publicly available. Two of the 5 products where vacuolation was seen, Somavert® and Krystexxa, are linked to several small PEG molecules (5 and 10 kDa, respectively), while 3 have a single 40-kDa PEG molecule (Omontys®, Macugen, and Cimzia) covalently bound. Vacuolation is seen mainly in phagocytic but sometimes also in non-phagocytic cells. Macrophages likely contribute to the clearance of larger PEG conjugates following phagocytosis and cellular vacuolation as an adaptive change due to uptake and removal of PEGylated biopharmaceuticals or PEG alone. No functional impact of vacuolation was reported in toxicology studies and no indication of PEG-related adverse effects has been reported from clinical trials or post-marketing surveillance with any of the approved drugs.
PEG-related tissue vacuolation is time and dose dependent as seen from the survey results. Importantly, there were no examples of toxicology studies where PEG-related cellular vacuolation resulted in degenerative changes. One case where there was evidence of PEG presence in neurons with and without vacuolation was within the 17 compounds reported in the survey. These observations may be specific to this product and the nature of the peptide moiety linked to brain penetration potentially due to a protein-specific transporter. The survey response did not include details of the peptide/protein to verify this mechanism. The other compounds in the survey as well as the approved drugs did not report presence of vacuolation in neurons, although vacuolation attributed to PEG was present in choroid plexus for 6 of the products.
There was some evidence that PEG-associated cellular vacuolation may be progressive in terms of the range of tissues affected with longer-term dosing. Nonreversibility or even increased PEG immunostaining can occur in some tissues during the treatment-free recovery period. The survey provides little information on the time to “steady state” for PEG-associated vacuolation. This is likely due to the limitations in time points of histological evaluation.
Recovery periods of studies reported in the survey range from 2 to 13 weeks increasing with the study duration. Some of the vacuolation has been reversible within the recovery times provided, but in other studies vacuoles were still seen after 13 weeks. There is also indication that vacuolation in certain tissues may have shorter recovery times than others, for example, renal tubular epithelium.
Recovery times may depend on differences in PEG removal mechanisms and/or cell turnover in tissues. For example, the reported life span of macrophages varies greatly (Takahashi 2001; Parihar, Eubank, and Doseff 2010). Takahashi reports that monocyte-derived macrophages die within 2 weeks, while tissue macrophages can survive for months (e.g., Kupffer cell life span is reported as 5 weeks to 14 months). Therefore, longer lived cells including macrophages may be responsible for the observation of limited or no recovery of cellular vacuolation seen in some toxicology studies with recovery periods of 4 or 6 weeks. Studies with longer recovery times may be advised for some of the longer-duration studies, although the recovery time may also be specific to the individual scenario of severity and location of cellular vacuolation.
It is important to consider that the PEG dose levels used for determination of any progression or reversibility should be in the range of the human clinical dose of PEG. Doses of PEGylated biopharmaceuticals several fold higher than the intended clinical dose and differences in dosing schedule of the toxicology studies may show higher severity and also cellular vacuolation in organs/tissues not representative for the human scenario. For example, recovery from high PEG loads may take longer than at clinical doses.
There do not appear to be any examples in the survey of products resulting in choroid plexus vacuolation at doses below the 0.4 µmol/kg/month limit outlined in the EMA position paper.
From the survey results and published literature, it can be concluded that there are certain gaps in our knowledge about tissue vacuolation related to PEG or PEGylated biopharmaceuticals: There are no widely accepted standardized histological criteria to grade the severity of PEG-related cytoplasmic vacuolation in animal tissues. Although vacuoles are visible by light microscopy, there is no knowledge regarding the ultrastructural changes associated with PEG presence, particularly in nonphagocytic cell types. Currently, the impact of vacuolation in the choroid plexus and potential effects on production of CSF are not known. There is only limited knowledge on turnover of vacuolated cells and the fate of PEG thereafter. Some toxicology studies have evaluated the reversibility of vacuolation, but tissue specific recovery times are not known. It is not known how PEG is removed from vacuoles.
We suggest that establishing standardized severity grading criteria for PEG-related cytoplasmic vacuolation may facilitate or enhance comparison across therapeutic products from different sponsors, different study pathologists, and different types of PEGs.
Another potential option to address information gaps regarding distribution of PEG or associated functional effects is to integrate specialized analyses into routine nonclinical toxicity studies on a case-by-case basis.
PEGylated therapeutic proteins can induce antibodies to PEG under certain conditions when the protein itself is immunogenic or other facilitating properties are present, as shown from coadministration of adjuvant in animal studies. Since this may be mediated through a hapten effect of the protein or peptide, results from animal studies likely do not predict the occurrence of anti-PEG antibodies in humans if the drug protein/peptide has a human sequence. There are other reports proposing that the shielding effect of PEG can reduce the immunogenicity of the parent molecule (Veronese and Mero 2008). Anti-PEG antibodies should be monitored in clinical trials involving novel PEGylated protein therapeutics and potential effects on safety and efficacy should be carefully assessed.
Conclusions
The evaluation of the impact of PEG-related cellular vacuolation observed in toxicology studies on risk assessment has been a challenge for industry and regulatory authorities. There is indication that PEG-related cellular vacuolation as seen histologically is absent below a certain dose of PEG per month (0.4 µmol/kg/month). When observed, vacuolation increases with PEG dose and dosing time, likely until a steady state between removal/excretion and drug dosing is reached.
The distribution of vacuolation in cells and tissues depends on many factors including pharmacological activity of the drug, biodistribution, amount of endocytic uptake versus receptor/target mediated uptake, potential immunogenicity, overall molecular weight of the PEGylated biopharmaceutical, the molecular weight of PEG itself, types of clearance/removal mechanisms for PEGylated drug or PEG alone, dose, dose frequency, and duration of treatment. Additional factors may influence the distribution of PEGylated biopharmaceutical including removal of the PEGylated biopharmaceutical by protein scavenger receptors, and differences in the endocytotic, phagocytic, and metabolic capacity of cells exposed to PEG (Rudmann et al. 2013), including a potential capacity to release PEG from the drug molecule either by cleaving the linker or by degrading the protein from its free terminus (Webster, Didier et al. 2007). Although all these factors may be variables in toxicology testing, well-planned toxicology studies should allow overall risk assessment and the determination of the risk/benefit of a PEGylated biopharmaceutical. If needed the risk/benefit assessment for PEGylated biopharmaceuticals may be supported on a case-by-case basis by additional supporting data, particularly when biopharmaceuticals are proposed for treatment in chronic indications or in pediatric patients.
It can be concluded from data in the public domain, all published data and information provided by the BioSafe survey that tissue vacuolation can be nonadverse when vacuoles are in phagocytes as long as the phagocytes are not distorting the normal tissue architecture, there is no evidence of impaired function, and there are no changes in biomarkers of injury. This may also apply to other cell types as long as surrounding tissue architecture and functional markers are not impaired. There needs to be an alignment of the definition of an NOAEL in toxicology studies where vacuolation is reported, away from assigning adversity to any microscopic finding not present in control tissue.
When PEG-induced vacuolation is seen, the assessment should take factors like tissue location, dose and duration of toxicology study, reversibility, severity of vacuolation, and functional consequences into consideration and determine the risk/benefit in relation to indication. Given the diversity of marketed PEGylated biopharmaceuticals and new PEGylated drug candidates in development, a careful tailored risk assessment is important.
Footnotes
Acknowledgments
We are grateful to Marietta Putz and the BioSafe Leadership Committee for critical review and their helpful comments to the manuscript. We thank everybody who summarized and provided the survey data.
Author Contribution
All authors (II, WA, AB, ABB, JC, DM, BD, AI, LD, JL, BR, TS, BS, LT, PT, JS) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
The authors are employed in organizations that are engaged in the development of a variety of innovative therapeutic products which may include PEGylated biopharmaceuticals. The authors have no other relevant involvement (financial or otherwise) in organizations or entities that would represent a financial interest or conflict relative to the issues discussed in this article. The authors are experts in the health care sciences, who are offering guidance on important issues to consider in the design of nonclinical safety assessment programs for PEGylated biopharmaceuticals. Several of the authors serve on a subcommittee of BioSafe, a committee composed of member volunteers from within the Health Section of the Biotechnology Industry Organization (BIO).
Supplemental Material
Table S1.—BioSafe survey results: molecular weight of the products, types of toxicology studies conducted, methods to determine PEG in tissue, and estimated molar PEG intake per months.
Table S2.—BioSafe survey results: key findings from 17 PEGylated biotherapeutics in nonclinical development (BioSafe Survey) tissue vacuolation estimated no effect level (NOEL), clinical indication, and health authority interactions.
Table S3.—Blank survey conducted by (BioSafe 2013).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
