Abstract
AV7909 is a next-generation anthrax vaccine candidate indicated for post-exposure prophylaxis of exposure to Bacillus anthracis. AV7909 consists of the Anthrax Vaccine Adsorbed (AVA) bulk drug substance and the immunostimulatory Toll-like receptor 9 agonist oligodeoxynucleotide adjuvant, CPG 7909. Safety testing for pediatric population is warranted to support the potential emergency use of AV7909 in children. This study was conducted to investigate the local tolerance and potential systemic toxicity and their reversibility in juvenile rats by repeat intramuscular injections of the AV7909 vaccine candidate. Animals were dosed on postnatal day (PND) 21 (at weaning), PND 28, and PND 35, with the test article (AV7909), the adjuvant alone (Alhydrogel + CPG 7909), or sterile water for injection. Core group animals were necropsied on PND 37 and recovery group on PND 49. Study end points included survival, clinical observations, injection site observations, body weights, clinical pathology (hematology, coagulation, and clinical chemistry), pro-inflammatory biomarker analysis (alpha-2 macroglobulin [A2M] and alpha-1 acid glycoprotein [AGP]), and anatomic pathology. Immune response to vaccination was measured using the high-throughput anthrax lethal toxin neutralization assay (htpTNA). The AV7909 vaccine candidate produced no apparent systemic or local toxicity. The AGP and A2M levels were elevated in both the adjuvant-alone and AV7909 groups at the end of treatment but were comparable to control levels by the end of the recovery period. All animals in the AV7909 group demonstrated a robust neutralizing antibody response. The results indicate that AV7909 has a favorable safety profile in juvenile rats.
Introduction
Anthrax is an infectious disease caused by the Bacillus anthracis bacteria. Infection can be highly lethal and poses a major biological threat. The AV7909 vaccine candidate is intended for post-exposure prophylaxis (PEP) for the general population. It is designed to provide an enhanced immune response, compared to the approved BioThrax (Anthrax Vaccine Adsorbed; AVA) vaccine, while requiring fewer dose administrations for protective immunity. 1,2 AV7909 consists of the AVA bulk drug substance and the immunostimulatory Toll-like receptor (TLR) 9 agonist oligodeoxynucleotide (ODN) adjuvant, CPG 7909.
The AVA drug substance is similar in composition and manufacturing process to the commercial BioThrax product (US License number 1755, STN 103821). BioThrax is prepared from cell-free filtrates of cultures of an avirulent B anthracis strain, which are adsorbed to the Alhydrogel adjuvant. 3 Anthrax protective antigen (PA) is the primary immunogen in AVA. 4 CPG 7909 is a ligand of choice for human TLR9 because it strongly activates B cells, improves antigen presentation, and facilitates generation of vaccine-specific cellular and humoral responses. 5 Vaccination of rats with rPA7909, a vaccine candidate based on the recombinant PA protein (rather than the native PA such as that contained in the culture filtrates used to manufacture AVA), which is also adjuvanted with CPG 7909, produced robust immune stimulation with no systemic toxicity. 6 Clinical safety of AV7909 was subsequently demonstrated in several clinical studies, which showed that AV7909 vaccination was not associated with an increase in adverse events as compared to AVA, with the most common events being injection site reactions. 2,7,8
Young animals are considered appropriate models for pediatric assessments. Thus, animal juvenile toxicity studies may need to be conducted to support safety assessment for products intended for use in children. 9 Although the proposed PEP indication for AV7909 does not presently include the pediatric population (<18 years), safety testing in a juvenile animal model for this vulnerable demographic is warranted to support the potential emergency use of AV7909 in children. Presently, safety data on AV7909 in the juvenile population are limited, although the AVA component of the vaccine candidate was evaluated in a developmental and reproductive toxicity study in rabbits where AVA vaccination of dams (twice prior to mating and once during gestation) produced anti-PA immunoglobulin G antibodies that were transferred to their offspring in utero. 10 While a robust immune activation was observed in dams, fetuses and kits, no adverse effects were observed. 10 The available safety data in adult animals and humans suggest that AV7909 has a good safety profile and is well tolerated. In the absence of an immediate threat of anthrax exposure, it would not be feasible to conduct a clinical study to evaluate the safety of AV7909 in pediatric populations. Therefore, to support the potential emergency use of the vaccine candidate in a pediatric population, a toxicology study in juvenile animals was conducted.
The study was designed to investigate the injection site tolerance and potential systemic toxicity induced by multiple intramuscular (IM) injections of the AV7909 vaccine candidate (test article, including the adjuvant) or adjuvant (Alhydrogel + CPG 7909) alone. The control group received sterile water for injection. Survival, clinical, and injection site observations; body weights; clinical pathology (hematology, coagulation, and clinical chemistry); and anatomic pathology (organ weights, gross observations at necropsy, and microscopic evaluation of selected tissues) were evaluated. Blood was collected for the analysis of pro-inflammatory biomarkers (alpha-2 macroglobulin [A2M] and alpha-1 acid glycoprotein [AGP]) in serum and immunogenicity using the high-throughput toxin neutralization assay (htpTNA).
Materials and Methods
Experimental Design
The US Food and Drug Administration Center for Biologics Evaluation and Research Division of Vaccines and Related Products Applications published a final guidance providing general considerations for developmental toxicity studies of preventative and therapeutic vaccines in 2006, 9 and the 2013 World Health Organization (WHO) guidelines on nonclinical evaluation of adjuvanted vaccines stated that additional nonclinical toxicology studies may be necessary to support the use of vaccines in special populations. 11 However, the specific design requirements of juvenile toxicology animal studies are not explicitly defined and remain largely at the discretion of the investigational product developer. Thus, the present study was designed to model a repeat-dose toxicity study in adult rats but targeted to assess potential toxicity in juvenile animals.
The rat model was selected for safety toxicity evaluation because it is an acceptable and widely used species for this type of study. Rats generate a robust antibody response to PA as a main immunogen of AV7909. 12 Rats also respond well to CPG 7909 ODN as a vaccine adjuvant component. 6
The study was conducted under Good Laboratory Practices 13 . The housing and animal care practices met current standards of Association for Assessment and Accreditation of Laboratory Animal Care, and current requirements stated in the Guide for the Care and Use of Laboratory Animals 14 and Animal Welfare Act regulations. The protocols were approved by Battelle Memorial Institute’s Institutional Animal Care and Use Committee.
Female Sprague Dawley (CD: SD) rats and their litters were obtained from Charles River Laboratories (Charles River Nomenclature: CD rats). Pups were kept with their dams until weaning on postnatal day (PND) 21. All rats had ad libitum access to certified feed (Purina Certified Rodent Diet, LabDiet 5002) except when fasted prior to the scheduled blood collection and necropsy.
One pup/sex/litter were assigned to each dose group. In total, the core and recovery phases each had 10 males and 10 females per dose group.
Test and Control Material
The test article, AV7909, was an engineering batch manufactured by Emergent BioSolutions Inc. The CPG 7909 adjuvant was obtained from Nitto Denko Avecia Inc. The aluminum adjuvant (Alhydrogel) was purchased from InvivoGen. Sodium chloride (0.9%) for injection and sterile water for injection were purchased from Hospira.
The adjuvant material was prepared by combining CPG 7909 and Alhydrogel at the final measured concentrations of 0.48 mg/mL of bound CPG 7909 (unbound = 0 mg/mL) and 1.3 mg/mL of aluminum in 0.85% sodium chloride.
Dose Administration
The appropriate test article, control article or adjuvant-alone, was administered at a maximum dose volume of 0.1 mL to each animal via bolus IM injection into the anterior portion of the quadriceps muscle on PND 21, 28, and 35. The accelerated dosing schedule was employed in order to ensure that the dosing is completed within the juvenile development stage of the animals; namely, from weaning to the periadolescent stage. 15 The injection site was alternated between the right and left quadriceps muscle for each dose administration. The dose volume, site of administration, and dose rotation were selected to minimize the possibility of nerve damage, to consider the occurrence of inflammation and its sequelae in accordance with a good practice guide to the administration of substances. 16
Clinical Observations
Moribundity and mortality observations were performed twice daily for all animals during quarantine and study periods, except on the day of necropsy when animals were observed once. Cage-side clinical observations were performed for dams and litters at least once daily on the days of body weight measurements prior to weaning and/or study start. Cage-side clinical observations for pups were performed at least once daily starting at weaning and continuing throughout the study. For detailed clinical observations performed outside of the cage, rats were removed from their primary enclosure and examined for general health and signs of toxicity. Detailed clinical observations were performed approximately at 1 to 2 hours, 24 hours, and 48 hours postdose.
Injection Site Observations
Test and control administration sites were observed for erythema or edema and graded according to the modified method of Draize (Table 1). 17 The observations occurred 1 to 2 hours following each dose administration, and daily for up to 1 week following each vaccination (at approximately the same time as the post-dose observation), until either a score of 0 was obtained or termination.
Dermal Draize Observations Scoring.

Body Weights
Body weights by sex within a litter were measured on PND 10 and 14. Individual body weights were recorded for all pups at randomization, on each day of dosing, 48 hours after dosing, and at least weekly until necropsy.
Clinical Pathology
Specimens for clinical pathology evaluations were collected at the scheduled termination on PND 37 for core animals and on PND 49 for recovery animals. Animals were fasted overnight prior to scheduled termination.
Animals were anesthetized with carbon dioxide/oxygen (70% CO2/30% O2). Blood for standard hematology and clinical chemistry panel analysis was collected via the retro-orbital venous plexus and blood for coagulation analysis was collected from the vena cava or aorta.
Assessment of Pro-Inflammatory Biomarkers
Specimens for AGP and A2M assessments were collected at the scheduled termination on PND 37 for core animals and on PND 49 for recovery animals. Animals were anesthetized prior to collecting blood via the retro-orbital venous plexus. Blood specimens were allowed to clot for at least 30 minutes and then processed to serum using at least 10 minutes of centrifugation at a relative centrifugal force of at least 1,800 g. The serum was kept on dry ice and stored in a freezer set to maintain −85 °C to −65 °C. Biomarker concentrations in rat serum samples were assessed using a commercially available vendor-validated kit, MILLIPLEX MAP Rat Vascular Injury Magnetic Bead Panel 3—Toxicity Multiplex Assay (Merck KGaA).
Immunogenicity Evaluation
Specimens for immunogenicity evaluations were collected at scheduled termination on PND 37 for core animals and on PND 49 for recovery animals. Prior to blood collection, the animals were fasted overnight (approximately 15 to 24 hours). Blood was collected via the retro-orbital venous plexus, allowed to clot for at least 30 minutes, and then processed for serum collection. To measure the neutralization of anthrax lethal toxin (LT), 50% effective dilution (ED50) and 50% neutralization factor (NF50) were determined using htpTNA. 18
Necropsy
A necropsy was performed on all core and recovery phase animals at scheduled termination (PND 37 and 49, respectively). Prior to necropsy, animals were fasted overnight, sedated with 70% CO2/30% O2, and exsanguinated after blood collection. Necropsy included examination of external surface of the body and all orifices; the cranial, thoracic, abdominal and pelvic cavities, and their contents; and the collection of standard tissues recommended by the WHO. 11 Fixed tissues from all core and recovery animals along with any gross lesions from all necropsies were processed to slides and stained with hematoxylin and eosin for histopathologic examination.
Statistical Analysis
All appropriate quantitative in-life data were analyzed for test-article effects by a parametric or nonparametric analysis of variance (ANOVA). For all data, normality was determined by the Shapiro-Wilks test and homogeneity of variances was determined by Levene test. When necessary, data were log-transformed to meet parametric assumptions. For parametric data determined to be normally distributed and homogeneous among groups, an ANOVA F-test was used to determine whether there were differences among the group means. If the ANOVA F-test was significant, then tests for differences between the control and each of the comparison groups were conducted using Dunnett test, which adjusted for multiple comparisons. For nonparametric data that were not normally distributed and/or nonhomogeneous, a Kruskal-Wallis test was used to determine whether there were differences among the group means. If the Kruskal-Wallis test was significant, then tests for differences between the control and each of the comparison groups were conducted using Wilcoxon tests and the Bonferroni-Holm method to correct for multiple comparisons. All statistical tests were performed at the 0.05 level of significance (P < 0.05), after accounting for multiple comparisons.
Results
Clinical Observations
All animals survived until the scheduled removal from the study. Swelling of one or both hind limbs was commonly observed in animals in the adjuvant-alone and AV7909 groups (Table 2). Swelling was also noted for a single control female on a single day (Table 2). Most adjuvant-alone and AV7909-treated animals exhibited a firm nodule on the top of the anterior quadricep of one or both legs. These corresponded to nodules within the subcutis that were found during the macroscopic observation and to inflammatory lesions observed during the histopathological examination. The nodules were considered attributable to the adjuvant-alone and AV7909 exposure. There were no other relevant clinical signs noted.
Swelling Incidence and Duration.
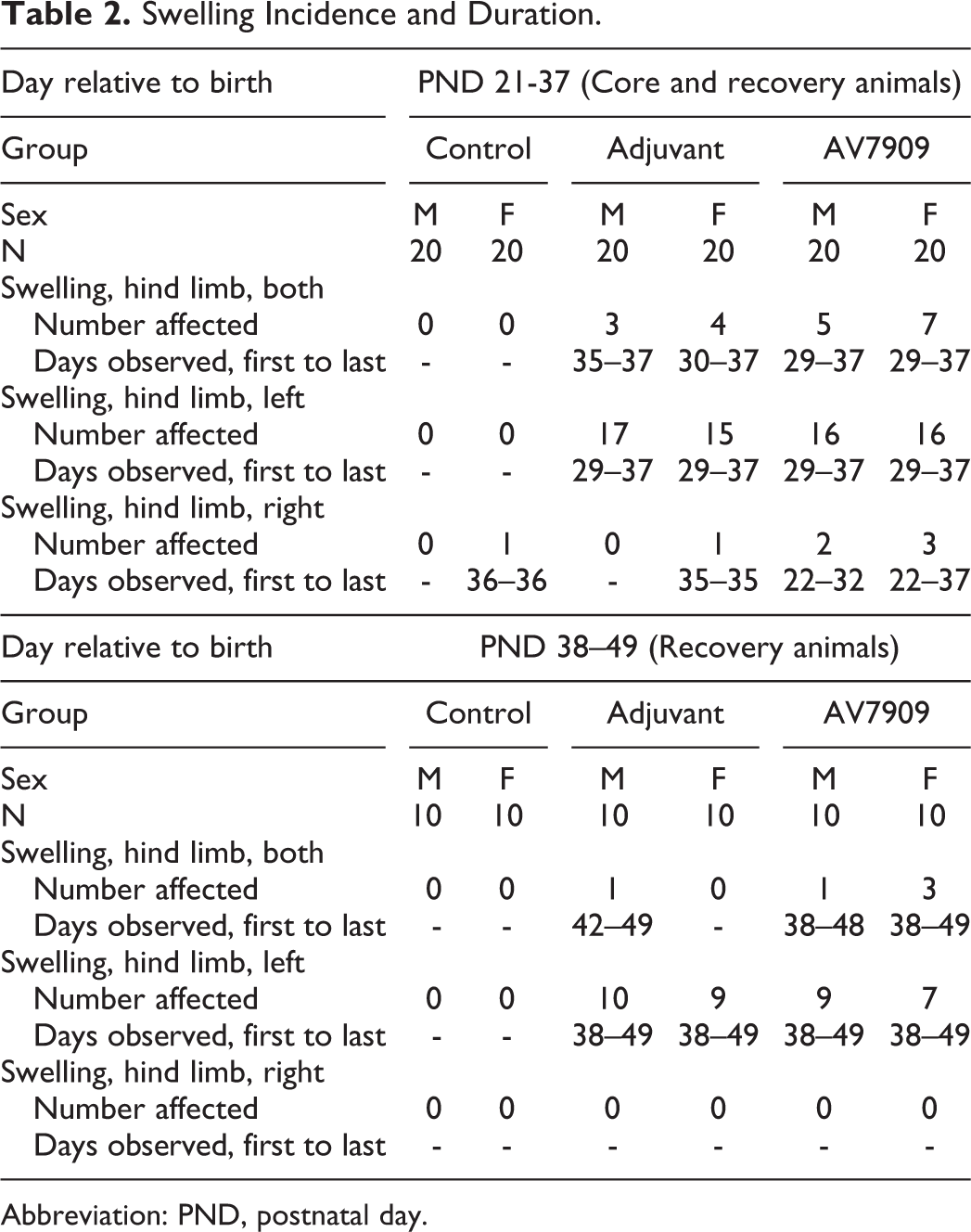
Abbreviation: PND, postnatal day.
Injection Site Observations
Erythema
Minimal erythema was noted for a single male in the adjuvant-alone group and a single male and female in the AV7909 group (all score 1). In all instances, erythema was noted on PND 29 only and no instances of erythema were observed in control animals.
Edema
Two adjuvant-alone-treated females had minimal edema on PND 22 that resolved by the following day (Table 3). Five males and 4 females had mild edema starting on PND 29. All instances of edema had resolved by PND 33. In the AV7909 group, 6 males and 2 females had minimal edema on PND 22 (score 1). All resolved by the following day. On PND 29, 7 males and 8 females had minimal (score 1) or mild (score 2) edema. In all cases, no edema was seen by PND 34. No edema was noted in any control animal (Table 3).
Summary of Mean Edema Scores.
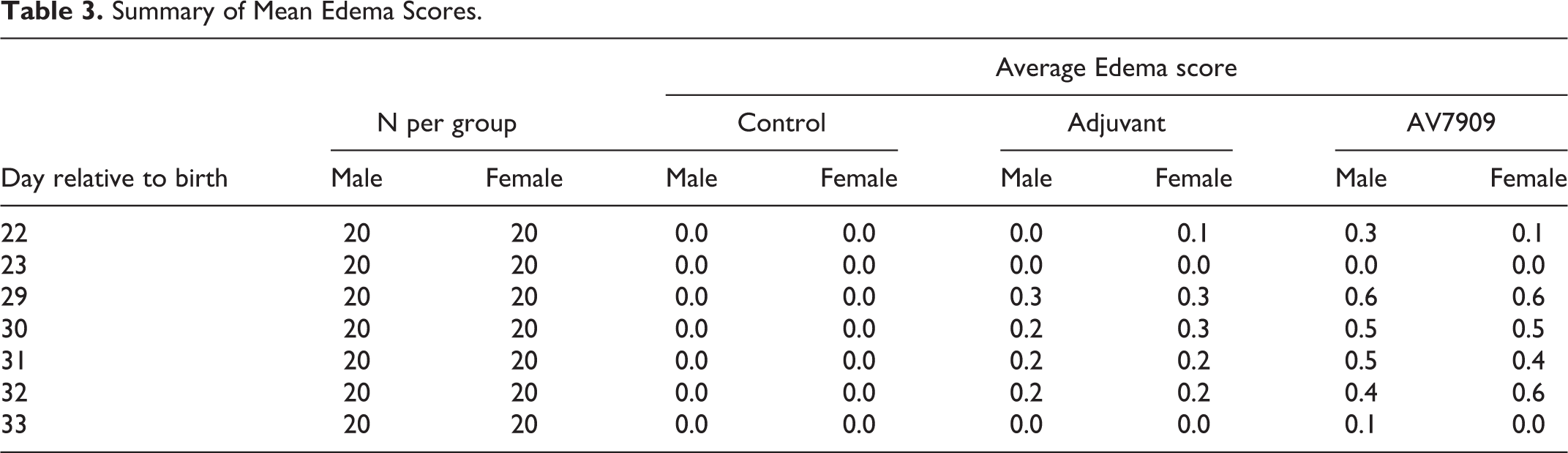
Body Weights
There were no statistically significant differences in group mean body weights. In general, animals in the adjuvant-alone and AV7909 groups had lower body weight gains compared to controls but were mostly not statistically significant (Table 4). Mean body weight gains were significantly lower for adjuvant-alone males on PND 23 to 28 (−8%) and for AV7909 males (−41%) PNDs 35 to 36 and 35 and 37 (−27%) compared to control males (P < 0.05). Males in the adjuvant-alone group also had lower weight gain on PND 35-36 (−31%) though the difference from controls was not statistically significant (Table 4). No other statistically significant differences in absolute body weights or weight gains were observed at any time point examined.
Summary of Mean Body Weights.
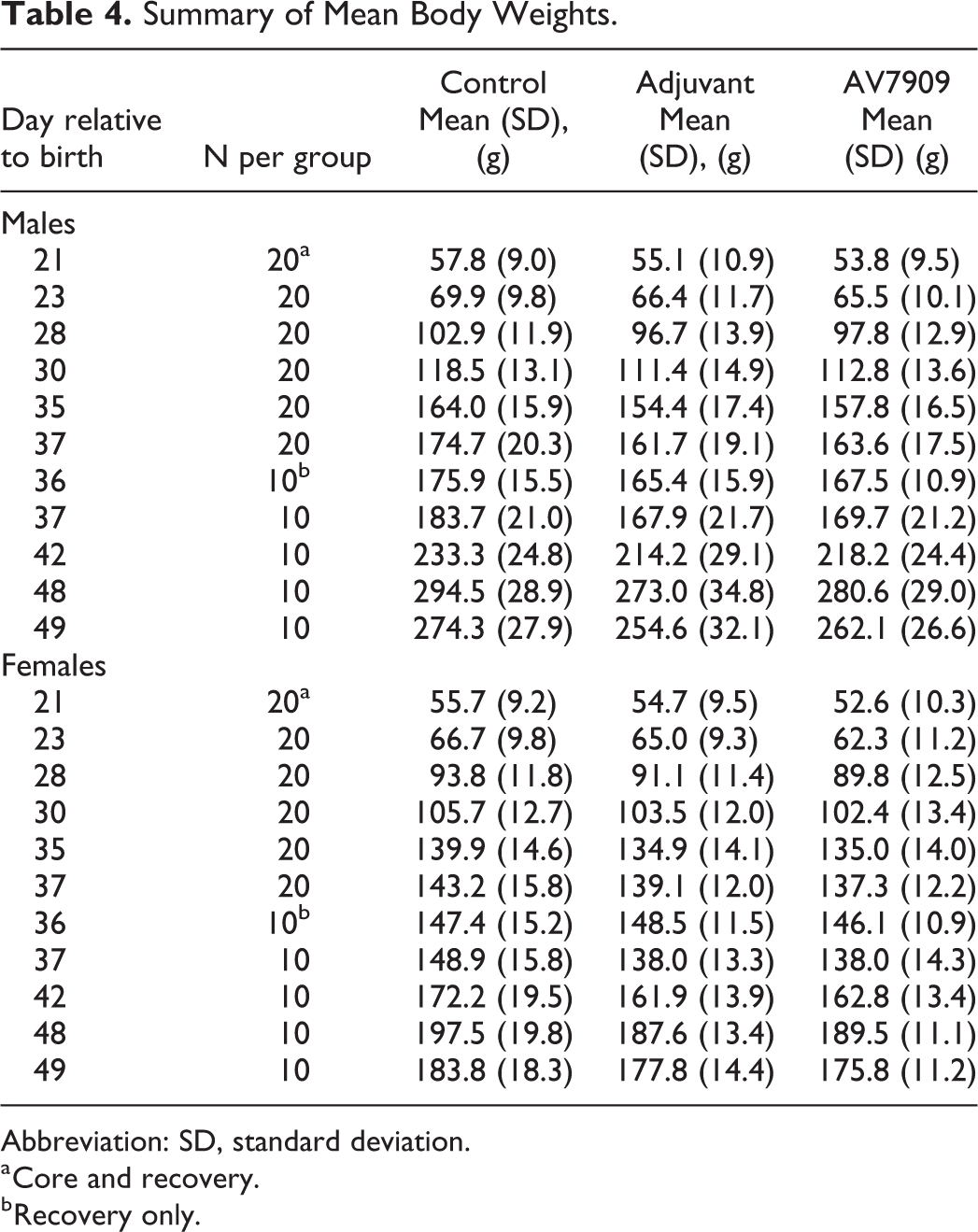
Abbreviation: SD, standard deviation.
a Core and recovery.
b Recovery only.
Hematology and Clinical Chemistry
Changes in clinical pathology parameters were indicative of inflammation and were observed in both adjuvant-alone and AV7909-exposed animals independent of sex. Findings included increased neutrophil, monocyte, large unstained cell count, fibrinogen, and globulin, and decreased albumin and albumin to globulin (A/G) ratio on PND 37 (Table 5).
Notable Changes in Hematology and Clinical Chemistry on PND 37 (Core Animals).
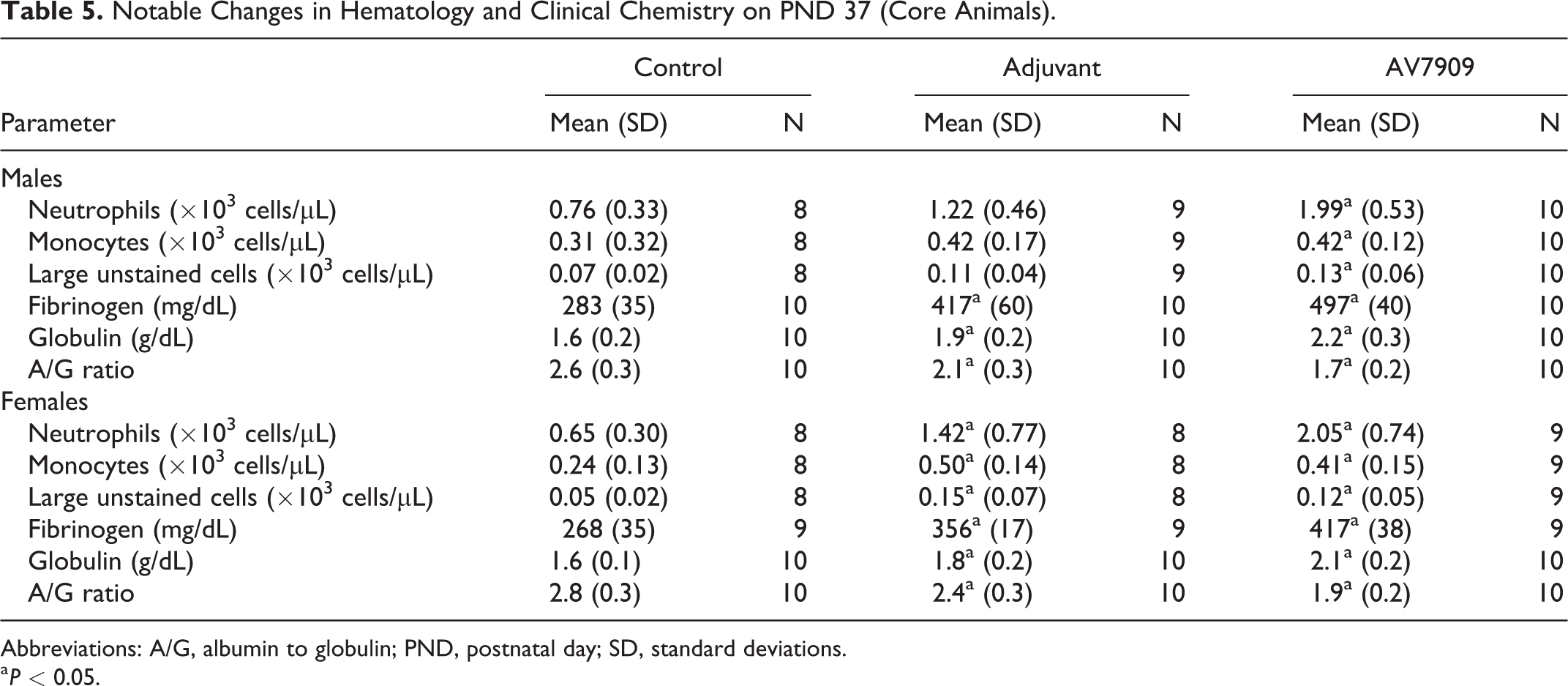
Abbreviations: A/G, albumin to globulin; PND, postnatal day; SD, standard deviations.
a P < 0.05.
All hematology and clinical chemistry findings were reversible and largely resolved completely by the end of the recovery period (PND 49).
Assessment of Pro-Inflammatory Biomarkers
On PND 37, AGP and A2M levels were systematically elevated in the adjuvant-alone and AV7909 groups relative to controls at the end of treatment (PND 37) (Table 6). Increased subject-to-subject variability was also observed in AGP response at PND 37. However, at the end of the recovery period (PND 49), A2M and AGP serum concentrations from animals in the AV7909 and adjuvant-alone groups were comparable to controls for both sexes (Table 6). In general, changes in pro-inflammatory biomarkers correlated with the hematology and clinical chemistry findings described above.
Group Average Serum Biomarker Concentrations.
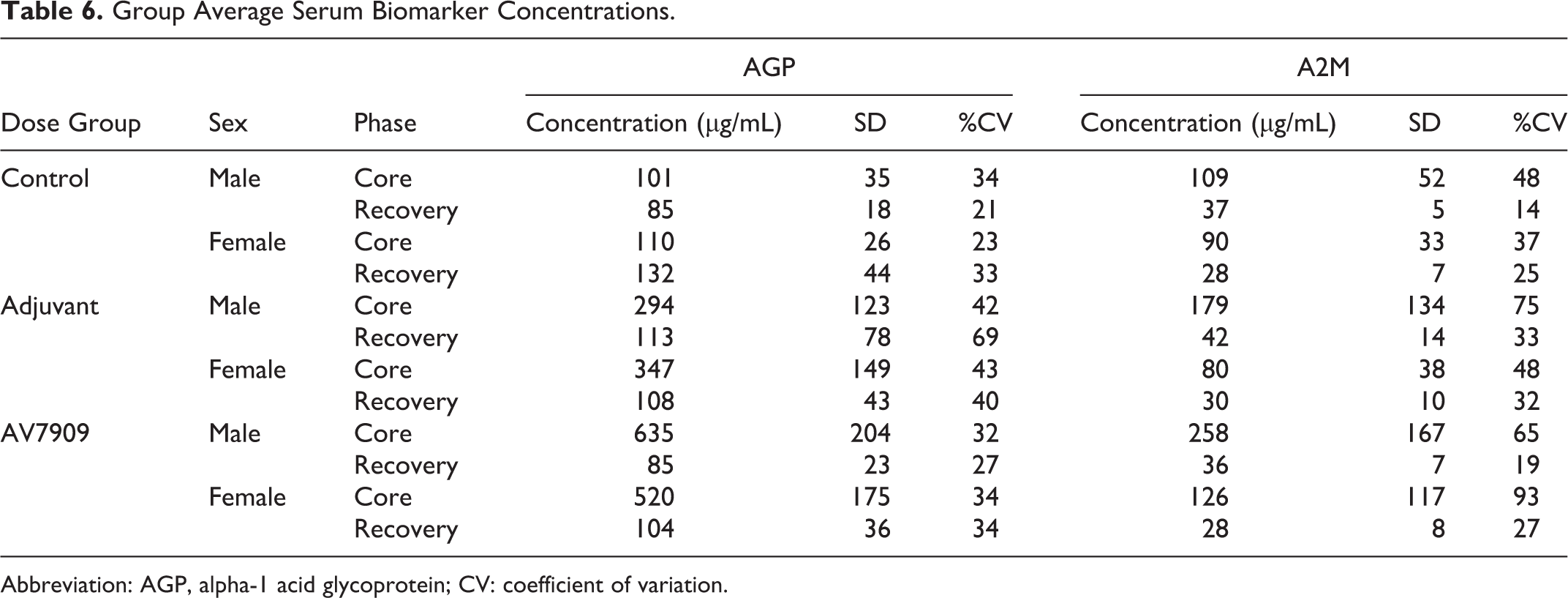
Abbreviation: AGP, alpha-1 acid glycoprotein; CV: coefficient of variation.
Immunogenicity
The purpose of the immunogenicity testing was to evaluate juvenile rat serum collected at PND 37 and 49 for levels of functional anti-LT neutralizing antibodies produced in response to vaccination with AV7909. The results of TNA titers (NF50) are summarized in Figure 1. As expected, all animals vaccinated with AV7909 demonstrated a robust immune response at PND 37 and PND 49 as measured by htpTNA (ED50 and NF50). All rats in the control and adjuvant-alone groups tested negative at PND 37 and PND 49.
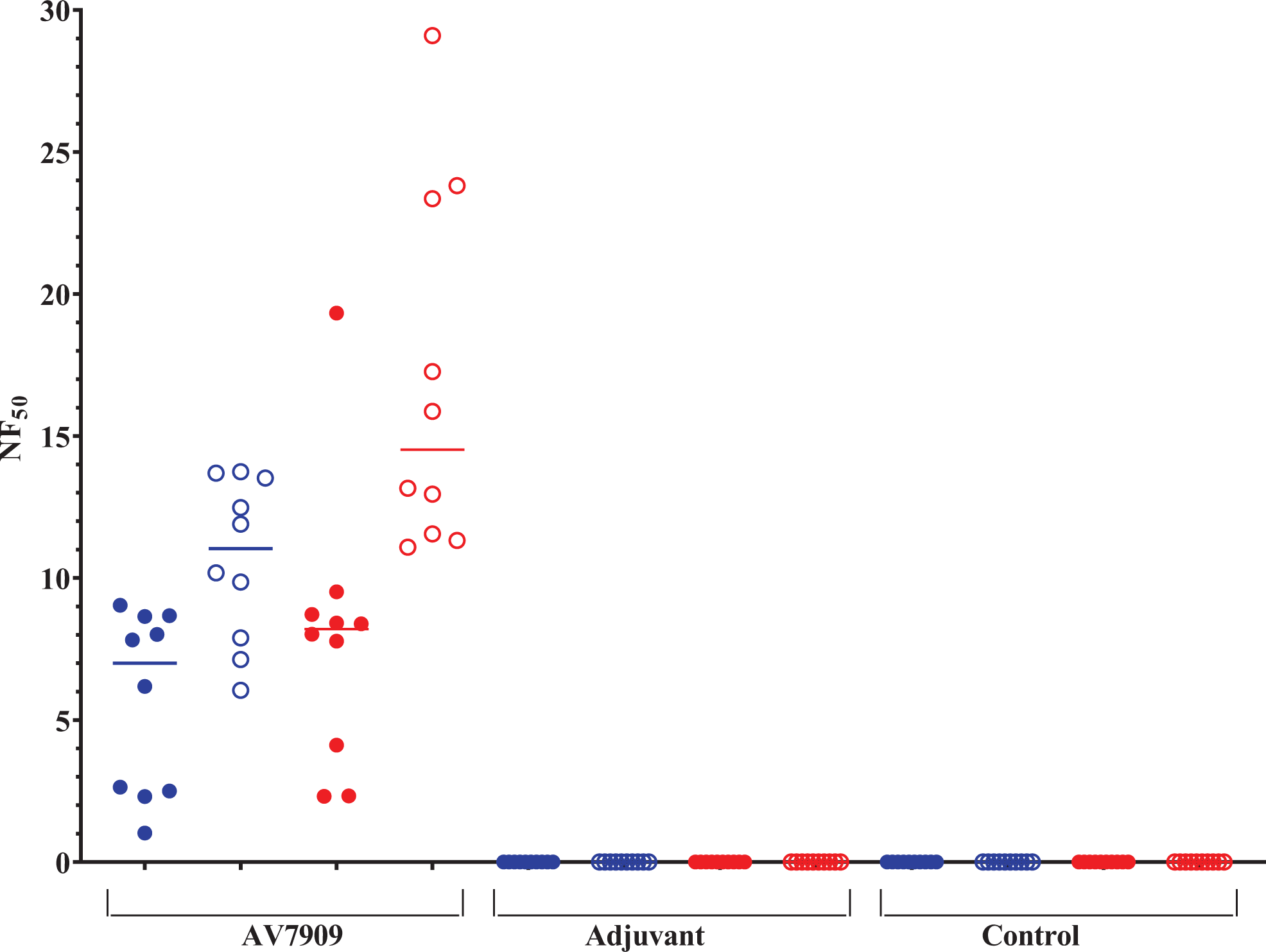
Group mean 50% neutralization factor (NF50). The NF50 is calculated as the ratio of the 50% effective dilution (ED50) of the test serum (the dilution that neutralizes 50% of the cytotoxic effect of the anthrax lethal toxin) and the ED50 of a serum reference standard. In the AV7909-vaccinated group, median NF50 values were 5.69 (males, blue) and 7.89 (females, red) at PND 37 (filled symbols) and 10.64 (males, blue) and 16.95 (females, red) at PND 49 (open symbols). In the control and adjuvant-alone groups, TNA was not detected or was below the limit of detection.
Anatomic Pathology
Terminal body and organ weights
Core and Recovery
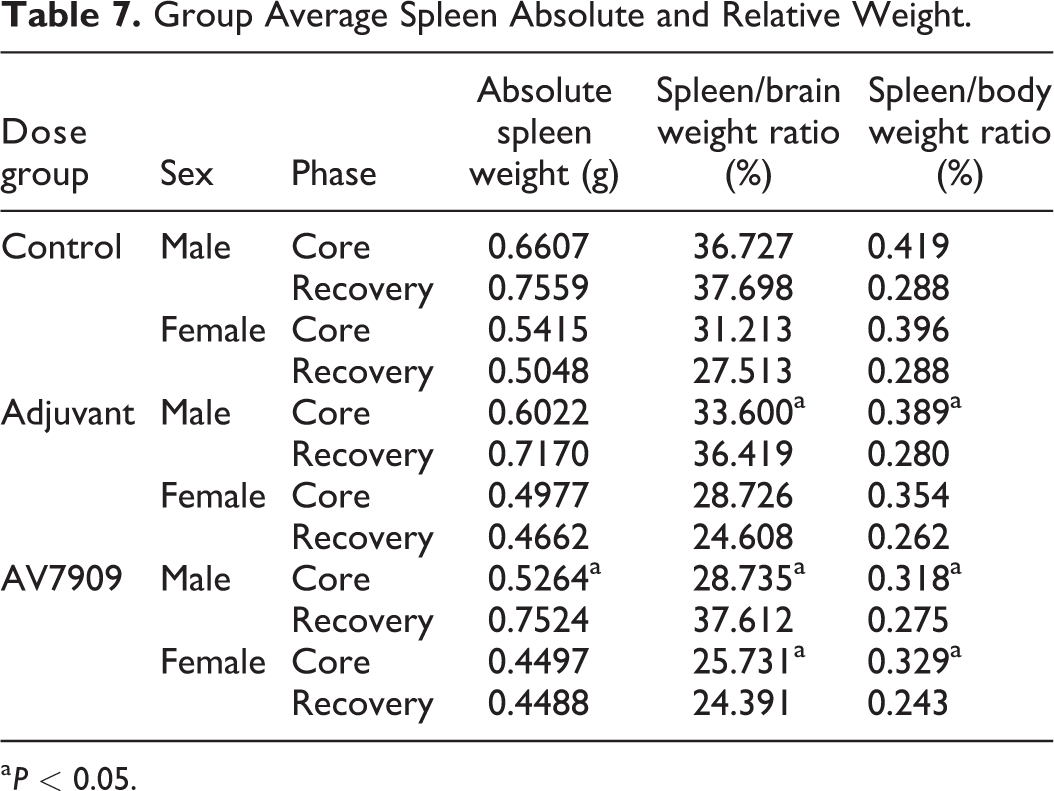
Mean spleen weights were increased in core male AV7909 animals (+25.5%; Table 7). Mean spleen to brain weight ratios and spleen to terminal body weight ratios were increased in AV7909 (+27.8% and 32.1%, respectively) and adjuvant-alone males (+16.9% and 22.4%, respectively) core males. Mean spleen to brain weight ratios (+21.3%) and mean spleen to terminal body weight ratios (+20.5%) were also increased in core AV7909 females. These increased spleen weights likely represent immune stimulation by the adjuvant with and without AV7909 vaccine candidate material. Unlike in core animals, spleen weights and spleen weight ratios were not significantly elevated in either the AV7909 or adjuvant-alone groups when measured at the end of the recovery period. There were no statistically significant differences in mesenteric lymph node weights between adjuvant-alone (males: −7.4%, females: +12.9%) or AV7909 animals (males: −15.4%, females: −1.6%) and controls. There were no relevant changes in terminal body weights or in any other measured organ weights in core or recovery adjuvant-alone and AV7909 animals (Table 7).
Group Average Spleen Absolute and Relative Weight.
a P < 0.05.
Gross findings
Core
Treatment-related gross lesions were confined to the injection sites, iliac lymph nodes, and the femur. Masses and nodules were noted in left and right injection sites within the subcutis. The masses and nodules were noted in AV7909 and adjuvant-alone animals only and corresponded to inflammatory lesions seen microscopically. Iliac lymph nodes were enlarged in core AV7909 animals. Enlargement corresponded to minimal lymphoid hyperplasia. One animal in the core adjuvant-alone group had a thickened right femur which corresponded to periosteal inflammation.
Recovery
Treatment-related gross lesions were confined to injection sites and iliac lymph nodes. Masses and nodules were noted in left and right injection sites within the subcutis. The masses and nodules were noted in AV7909 and adjuvant-alone animals and corresponded to inflammatory lesions seen microscopically. Iliac lymph nodes were enlarged in recovery male AV7909 and female adjuvant-alone animals. Enlargement corresponded to minimal lymphoid hyperplasia.
Histopathology
Core
Inflammation within the skeletal muscle or subcutis of injection sites were noted in the AV7909 and adjuvant-alone groups, which is consistent with the expected immunostimulatory effects of the test materials (Table 8). Inflammation in core AV7909 and adjuvant-alone animals sacrificed on PND 37 was primarily chronic active inflammation characterized by central deposit of amphophilic material with infiltrating foamy macrophages and cellular debris and surrounded by lymphocytes, plasma cells, and variable numbers of degenerate neutrophils. A few animals (see Table 8) had granulomatous lesions which were characterized by a central deposit of amphophilic material surrounded by foamy macrophages, lymphocytes, plasma cells, and variable amount of fibrosis. These were identified as resolving chronic-active lesions. A few animals (see Table 8) had chronic inflammation consisting of fibrosis, variable numbers of lymphocytes, and plasma cells and neovascularization.
Incidence and Severity of Granulomatous Inflammation and Chronic Lesions.
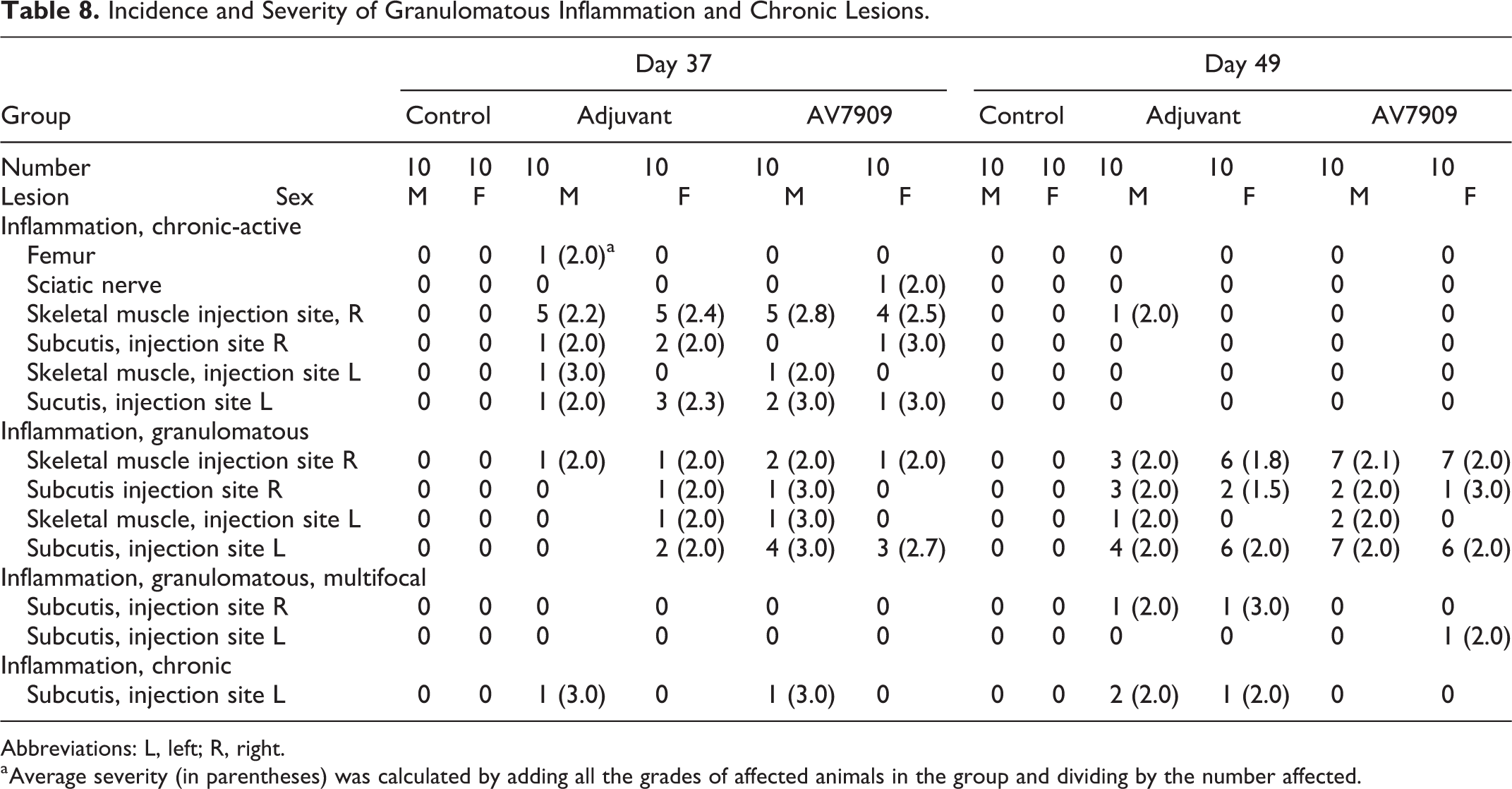
Abbreviations: L, left; R, right.
a Average severity (in parentheses) was calculated by adding all the grades of affected animals in the group and dividing by the number affected.
Iliac lymph nodes were enlarged due to minimal lymphoid hyperplasia in one core AV7909 male and one female. This is likely due to the expected inflammatory response induced by the adjuvant and vaccine candidate material.
In addition to inflammation at the injection sites, one core adjuvant-alone animal exhibited chronic-active inflammation of the right femur and one core AV7909 animal had chronic-active inflammation of the axon nerve sheath (Table 8). These lesions were likely the result of the weekly injections administered deep inside the femur, rather than a specific effect of the vaccine or adjuvant.
Control animals had lesions in the injection site skeletal muscle only. At PND 37, skeletal muscle necrosis and histiocytic inflammation were most prominent. Muscle degeneration and regeneration were also identified in skeletal muscle.
Recovery
Test article-related microscopic lesions consisted of inflammation within the skeletal muscle or subcutis of injection sites. No remarkable differences between the AV7909 and adjuvant-alone groups were noted, which is consistent with the expected immunostimulatory effects of the adjuvant. By PND 49, most lesions in the AV7909 and adjuvant-alone groups consisted of granulomatous inflammation in skeletal muscle and subcutis of injection sites. There were a few chronic-active lesions, as well as a few chronic lesions. Iliac lymph nodes of one AV7909 male and two adjuvant-alone females were enlarged due to minimal lymphoid hyperplasia, suggesting continuation of the inflammatory response. Control animals had lesions in the injection site skeletal muscle only. At PND 49, microscopic changes indicated general recovery and were limited to fibrosis and minimal skeletal muscle degeneration.
Discussion
As a result of the postnatal and prepubescent growth and developmental changes that occur during maturation, structural and functional characteristics of various organ systems differ between children and adults. Consequently, extrapolation of adult toxicological data to pediatric populations does not always accurately describe the safety profile of a product in the developing child, nor does it serve as an adequate model to provide guidance on the appropriate pediatric dose levels. Due to the potential off-label use of products in children and the previously lacking safety data of approved products in the pediatric population, regulatory authorities now mandate the pediatric assessment of new drugs in an appropriate animal model or justify why these data are not generated. In general, juvenile animals exhibit growth and developmental characteristics similar to pediatric patients and are, therefore, considered appropriate models for pediatric assessments, either preceding pediatric clinical trials or when pediatric safety trials cannot be adequately or safely performed. 19
While guidelines for the nonclinical safety testing for drug products intended for pediatric use exist, 19 there is no specific requirement for such studies for vaccine candidates. As a result, regulatory guidance for the design of vaccine juvenile animal studies is limited. Sponsors are ultimately left to design fit-for-purpose juvenile animal studies on a case-by-case basis. 10,19,20 In the case of the present study, it was modeled on a repeat-dose toxicity study in adult animals but evaluated potential toxicity in a juvenile population. 21
The adjuvant-alone and AV7909 vaccine candidate were both well tolerated, and all animals in the AV7909 group mounted a robust immune response at PND 37 and PND 49. The safety and immunogenicity response in the present study was consistent with clinical studies conducted with the AV7909 vaccine candidate. 1,2
As expected, findings were indicative of generalized immune system stimulation and were observed in both the adjuvant-alone and AV7909 groups. Erythema was noted in a limited number of animals in the adjuvant-alone and AV7909 groups only. Edema was noted in a few adjuvant-alone animals and was observed at a greater frequency in AV7909 animals. Edema was primarily observed starting at PND 22, with all instances resolved by PND 34. Swelling of the hind limbs and subcutaneous nodules near the injection sites were seen in most animals in the adjuvant-alone and AV7909 groups, which corresponded to inflammatory lesions seen during the histopathological examination.
Changes in clinical pathology parameters were also indicative of inflammation and were observed in both adjuvant-alone and AV7909-exposed animals at the end of treatment on PND 37 and had resolved completely by the end of the recovery period. Serum AGP and A2M are sensitive biomarkers of an acute phase response in rodents. 22 Both biomarkers were elevated in both the adjuvant-alone and AV7909 groups at the end of treatment and returned to levels comparable to control levels at the end of the 2-week recovery period (PND 49).
Increased spleen weights were seen in adjuvant-alone and AV7909 animals at the end of treatment but were not evident in recovery animals at the end of the 2-week recovery period. The spleen plays an important role in immune function and systemic inflammation, 23 -25 and the increased spleen weights likely reflect immune stimulation induced by the adjuvant. Histopathologic lesions in the AV7909 group were indistinguishable from lesions produced by the adjuvant-alone and were considered attributable to the immunostimulatory effect of the adjuvant. Microscopic findings were limited to local inflammatory lesions of the injection site subcutis or skeletal muscle or the adjacent sciatic nerve or femur. Alhydrogel is known to induce strong immune system reactivity, and localized inflammatory effects are commonly seen. 26 -29 Lesions at PND 37 were mainly chronic-active inflammation characterized by a central pool of amphophilic material with infiltrating foamy macrophages and surrounding lymphocytes, plasma cells, and variable number of degenerate neutrophils. At PND 49, there was partial resolution and lesions were more granulomatous with loss of neutrophils and replacement by fibrous connective tissue. The noted systemic effects were slight decreases in the body weight gains and increases in spleen weights of AV7909 and adjuvant-only animals at PND 37, compared to the control animals. In addition, enlargement of iliac lymph nodes due to minimal lymphoid hyperplasia was observed in a limited number of AV7909 and adjuvant-only animals. The spleen and lymph node changes were likely due to nonspecific immune stimulation.
Overall, the study demonstrated that AV7909 was well tolerated in juvenile animals. Effects of AV7909 and the adjuvant alone were consistent with robust immune system stimulation. Importantly, AV7909-vaccinated animals generated a strong anthrax lethal toxin-neutralizing antibody response, which was not seen in the adjuvant-alone or control animals. The results of this study support potential emergency use of this vaccine candidate in the pediatric population.
Footnotes
Acknowledgments
The authors would like to acknowledge all the Battelle staff involved in the support and completion of this work and would like to especially thank Mathew Robinson, Megan Bell, Javelyn Evans, and Ekai Moritz for all their hard work and contribution.
Author Contributions
A. Zmarowski contributed to conception, design, and drafted the manuscript; J.D. Ballin contributed to acquisition, analysis, interpretation, and drafted the manuscript; J. Sharits contributed to acquisition, analysis, and drafted the manuscript; K. Carrico contributed to acquisition, analysis, and drafted the manuscript; J. Novak contributed to acquisition, analysis, and drafted the manuscript; J. Shearer contributed to conception, design, interpretation, and critically revised the manuscript; B. Blauth contributed to conception, design, interpretation, and critically revised the manuscript; B. Ionin contributed to conception, design, interpretation, and critically revised the manuscript; J. Reece contributed to conception, design, interpretation, and critically revised the manuscript; V. Savransky contributed to conception, design, interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded, in whole or in part, with federal funds from the Department of Health and Human Services, Biomedical Advanced Research and Development Authority, under contract HHSO100201600030C.
