Abstract
Calcium, a component of approved human vaccines administered via systemic routes, has a good safety profile. Recently, intranasally administered vaccines containing calcium have shown promise in generating mucosal immune responses in animal models. However, the safety of intranasally administered calcium is unknown. This study evaluates the safety of intranasally administered calcium at 2- to 13-fold higher doses than used in experimental vaccines. At a calcium dose of 22 mg/kg, 80% of the Balb/c and 20% of the C57BL/6 mice die within the first 24 hours. At 11.0 mg/kg, there is no overt toxicity in either strain, based on body weight, clinical scores, blood chemistry, and histopathology of major organs at 7 days post administration. In C57BL/6 mice, apart from acute and subacute inflammation in the lungs at up to 3 days post administration, especially at the 22-mg/kg dose, there is no overt toxicity. Doses of calcium up to 11 mg/kg appear to be safe in a mouse model.
Calcium, usually in the form of calcium phosphate, has been approved and used as an adjuvant in systemically administered vaccines for many decades, especially in Europe. 1-3 Vaccines such as diphtheria, tetanus, pertussis (DTP) adsorbed onto calcium phosphate have been successfully administered via systemic routes without overt safety issues in humans or experimental animal models. 4-6 More recently, calcium has been used as a component of intranasally administered vaccines, but so far only in animal model studies. Calcium phosphate nanoparticles have been evaluated in animal models, as adjuvants in intranasally administered vaccines for generating mucosal immune responses. 7,8 Cochleate cylinders, devoid of any aqueous compartments, formed by the addition of calcium chloride to liposomes made from ester lipids, have also been evaluated in animal models, for intranasal vaccine administration. 9-12 We recently demonstrated that archaeal lipid mucosal vaccine adjuvant and delivery (AMVAD) formulations (aggregated lipid structures containing aqueous compartments), obtained by the interaction of calcium chloride and archaeosomes (liposomes made from archaeal polar lipids), elicited strong, sustained, and memory-boostable antigen-specific mucosal and systemic immune responses upon intranasal administration to mice. 13,14 In these studies, the amount of calcium chloride required for making the AMVAD formulations varied, depending on the source of the archaeal species for the polar lipids.
Whereas the safety and reactogenicity of calcium-containing vaccines administered systemically, such as by intramuscular and subcutaneous routes, have been extensively investigated in animal models and humans, 2,5,15,16 there is very little information on the safety or tolerability of calcium per se administered by the intranasal route. Both the efficacy and the safety of a vaccine are affected by the specific administration route. 17,18 Furthermore, from a regulatory perspective, the safety data of a vaccine administered by one route cannot be directly applicable for the same vaccine administered by a different route. 3 Therefore, the objective of this study was to evaluate the tolerability or the safety profile of intranasally administered calcium chloride in Balb/c and C57BL/6 mice, at doses that were 2- to 13-fold greater than those that had been used previously, 13,14 in evaluating the efficacy of intranasally administered AMVAD vaccines in mouse models.
Materials and Methods
Animals
Specific-pathogen-free, female, Balb/c or C57/BL6 mice were purchased from Charles River Laboratories (Montreal, Quebec, Canada) and entered the experiments at 6 to 8 weeks of age. The mice were housed (5 per cage) and used as per the Canadian Council on Animal Care Guide to the Care and Use of Experimental Animals, and all animal care and use protocols were approved by the Institutional Animal Care Committee.
Preparation of Calcium Formulations
An autoclaved (121°C, 20 minutes) solution of 200 mM CaCl2 in 0.85% NaCl (pH 7.1) was aseptically diluted to 25 to 100 mM CaCl2 using autoclaved 0.85% NaCl (saline, pH 7.1). The calcium formulations, and the control formulation consisting of only 0.85% NaCl (saline), were stored at 4°C until use. All formulations were prepared aseptically, using pyrogen-free glassware and sterile MilliQ water.
Experimental Design
Groups of preweighed mice (n = 5-20) were intranasally administered 50 µL of either saline, or one of the 25- to 200-mM CaCl2/saline formulations at 0 day, to give an effective dose of 0 to 22 mg of Ca2+ per 1 kg of body weight. For intranasal administration, the mice were anesthetized by intraperitoneal injection of ketamine and xylazine at 0.1 mg and 0.05 mg/g body weight, respectively, in 0.25 mL injectable saline. The calcium formulations were administered to both sides of the nasal cavity of the mice, by drop-by-drop addition to alternate nares of the mice. Mice were observed at least once daily for clinical signs and symptoms such as hyperactivity, lethargy/sedation, aggressive behavior, piloerection (ruffled or erect fur), alopecia, dehydration, salivation, nasal discharge, and diarrhea. The overall clinical symptoms for each mouse were scored (treatment groups were blinded) on a sliding scale of 0 to 4, where 0 represented the normal, active, healthy mouse and 4, the moribund mouse. To derive the average clinical score of a group of mice, we decided that any mouse that had died would be included for averaging the score of the group on the day of death but would not be included for calculating the average score on the subsequent days for the surviving mice in the group. The body weight of mice was monitored just prior to formulation administration and daily thereafter. The change in body weight was expressed as percentage change in body weight from that at 0 days.
At day 1, 2, 3, or 7 post administration, 5 mice from each group were euthanized by CO2 asphyxiation. Blood samples were collected by cardiac puncture and sera were separated and stored at –20°C until used for clinical blood chemistry analyses. Spleen, lungs, liver, heart, kidneys, and thymus from each mouse were collected and observed macroscopically. These organs, and the brain, were fixed in 10% neutral-buffered formalin (pH 7.0). To assess the potential effect of intranasal calcium administration on the nasal mucosal epithelium, additional groups of C57BL/6 mice (n = 20) were intranasally administered Ca2+ at 22.0 mg/kg or saline (control group) on day 0. Clinical signs and body weight changes were monitored. Five mice per each group were euthanized 1, 2, 3, and 7 days later. For histopathology of the nasal tissue, the skin from the head of the euthanized mouse was removed carefully prior to immersing the head in formalin. After the head was washed with 60% ethanol, it was immersed in 5% formic acid for decalcification (2 weeks at room temperature, replaced with fresh formic acid solution every 2 days). The nasal tissue and organs were sectioned and stained by hematoxylin and eosin (Department of Pathology and Laboratory Medicine, University of Ottawa, Ottawa, Ontario, Canada) for histopathology examination. The brain was not examined macroscopically in preference to fixing it in formalin before extracting from the cranium, to avoid potential damaging of the tissue for histopathology observations.
Clinical Blood Chemistry
The sera were assayed for the levels of alkaline phosphatase (ALP), serum glutamic pyruvic transferase (SGPT), blood urea nitrogen (BUN), glucose, creatinine, bilirubin, albumin, globulin, and total protein using the Roche Hitachi 917 Analyzer (Vita-Tech, Markham, Ontario, Canada).
Statistical Analyses
All data are reported as mean ± standard deviation (SD) for each group. Differences between treatment groups were assessed by 2-tailed Student’s
Results
Mice are one of the small laboratory animal species that have been extensively used in preclinical evaluation of the efficacy and safety of experimental vaccines. Balb/c and C57BL/6 mouse strains are most commonly used in vaccine research because they express predominantly different immune responses (Th2 vs Th1, respectively). To evaluate calcium toxicity, we used Balb/c and C57BL/6 strains of mice because both strains were used in our previous studies evaluating the efficacy of intranasally administered calcium-containing AMVAD formulations in eliciting mucosal and systemic immune responses. 13,14
The doses of calcium chloride evaluated in the current safety study were selected to exceed the maximal doses of Ca2+ that were administered to mice in our earlier evaluation of the efficacy of intranasally administered AMVAD vaccine formulations. 13,14 We calculated that the maximal potential amount of Ca2+ per intranasal immunization in these cited studies would have been in the range of 1.7 to 11 mg per 1 kg of body weight, depending on the specific source of archaeal lipids used in each formulation. The amount was calculated taking into consideration the maximal amount of Ca2+ that would potentially be associated with the amount of lipid administered (based on the starting wt/wt ratio of lipid/Ca2+ used for making the formulation and assuming no disproportionate loss of lipid or Ca2+ during preparation) and the amount of Ca2+ that was contributed by 50 µL of the final 0.85% saline/14.9 mM CaCl2 buffer in the vaccine dose diluted for immunization. Therefore, the tolerability of Ca2+ in the current study was evaluated at single bolus doses that were 2- to 13-fold in excess of those used in our previous AMVAD vaccine efficacy studies.
Upon intranasal administration of a single CaCl2 dose equivalent to a Ca2+ dose of 0 to 22 mg/kg body weight, 4 of the 5 Balb/c mice in the group given the highest dose of 22 mg/kg were found dead between 12 and 24 hours. The average clinical score for the group at 1 day post administration (inclusive of the dead mice) was 3.4, with the sole surviving mouse having a score of 1.0 (slightly sick, slight ruffled fur, otherwise normal). The surviving Balb/c mouse in the group had a normal score of 0 at 2 days after administration and thereafter (Figure 1). The scores of the dead mice are not included for averaging beyond the day of death, so as to indicate the health of the surviving mice only. In the corresponding C57BL/6 group given the highest dose of Ca2+ (22 mg/kg), 1 of the 5 mice died between 12 and 24 hours. The average clinical score of this group was 2.2 at 1 day (sick, ruffled fur, some hunching, slow movement), with the scores of the living mice ranging between 1 and 1.5. The scores of the surviving mice in this group gradually improved with each subsequent day, achieving a normal, healthy score of 0 by day 5 (Figure 1). Apart from the indicated symptoms, there were no other clinical symptoms such as hyperactivity, aggressive behavior, alopecia, dehydration, salivation, nasal discharge, or diarrhea in any group. At the lower Ca2+ doses of 3.0, 5.5 (clinical scores not shown), or 11.0 mg/kg (Figure 1), the clinical scores of both the Balb/c and C57BL/6 groups were normal and comparable to the respective control groups receiving saline only (0 mg Ca2+). None of the other mice in Balb/c or C57BL/6 groups died or had to be euthanized because of poor clinical scores.
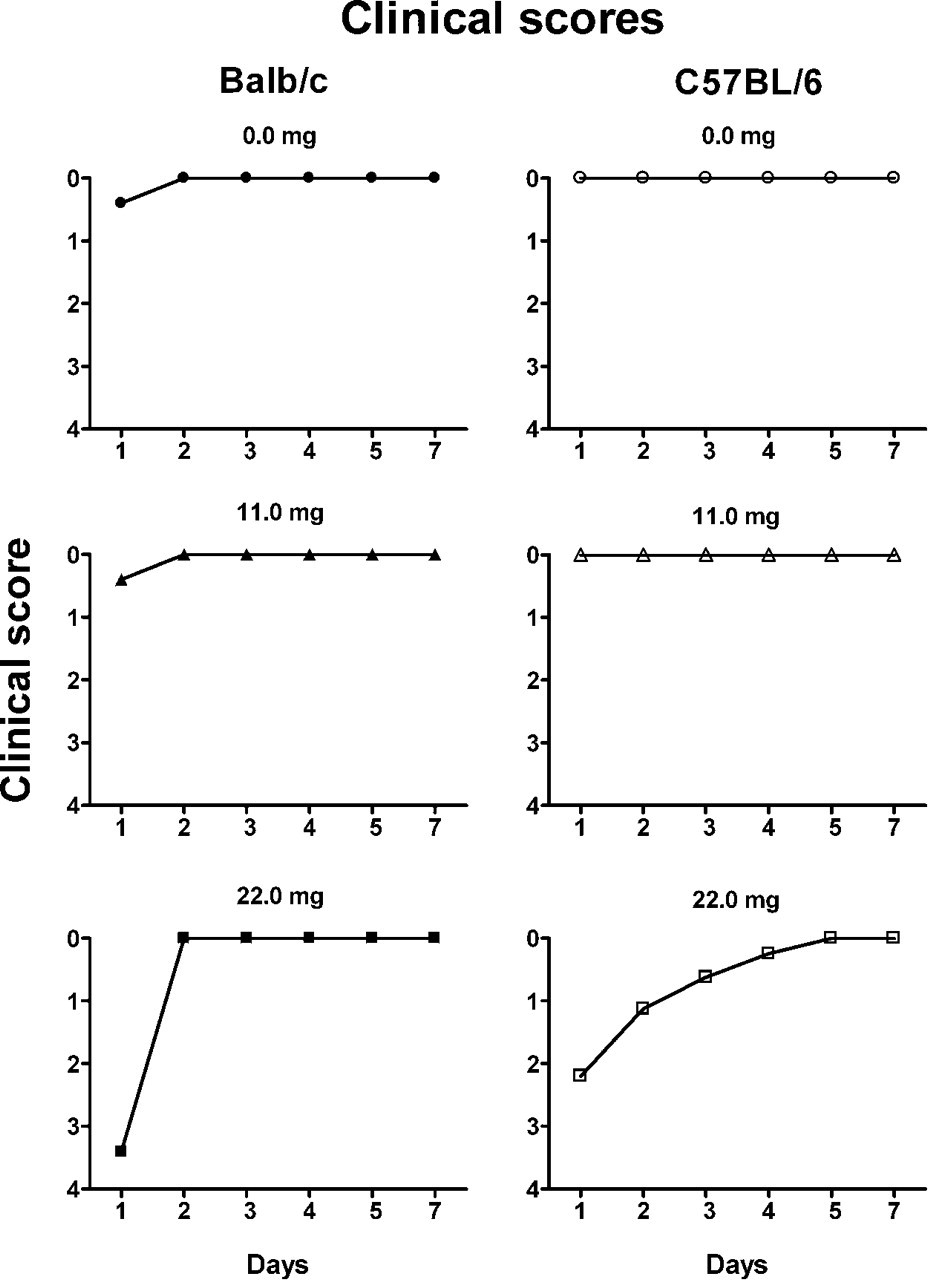
Average clinical scores of groups (n = 5) of Balb/c (left column) and C57BL/6 (right column) mice at 1 to 7 days after intranasal administration of the indicated calcium dose of 0 to 22 mg/kg body weight. The individual clinical scores were assigned as 0 (normal, active, healthy), 1 (slightly sick, slightly ruffled fur, otherwise normal), 1.5 (moderately sick, some ruffled fur), 2 (sick, ruffled fur, slow movement, hunching), 3 (very sick, ruffled fur, very slow movement, hunched, eyes shut), or 4 (moribund). For any mouse that had died, the individual score of 4 was included in deriving the average clinical score for the group for the day of death, but this individual score was not weighted in calculating the next day’s average clinical score for the surviving mice in the group. At the highest dose, 1 of 5 Balb/c mice and 4 of 5 C57BL/6 mice survived after 1 day of administration. The groups of mice administered saline only (0 mg calcium) were used as the control group for comparative purposes.
The change in body weight of all surviving mice in groups of Balb/c mice receiving Ca2+ doses of 11.0 mg/kg and lower was not statistically significant from the control group (Figure 2). The sole surviving Balb/c mouse receiving the highest Ca2+ dose of 22 mg/kg had lost about 7% of body weight by day 2, but at 7 days it had almost returned to its starting weight. In the C57BL/6 mice, the 4 surviving mice in the group administered the highest Ca2+ dose (22 mg/kg) had lost about 16% body weight at 2 days post treatment (Figure 3), and this loss was significant compared with the control group (
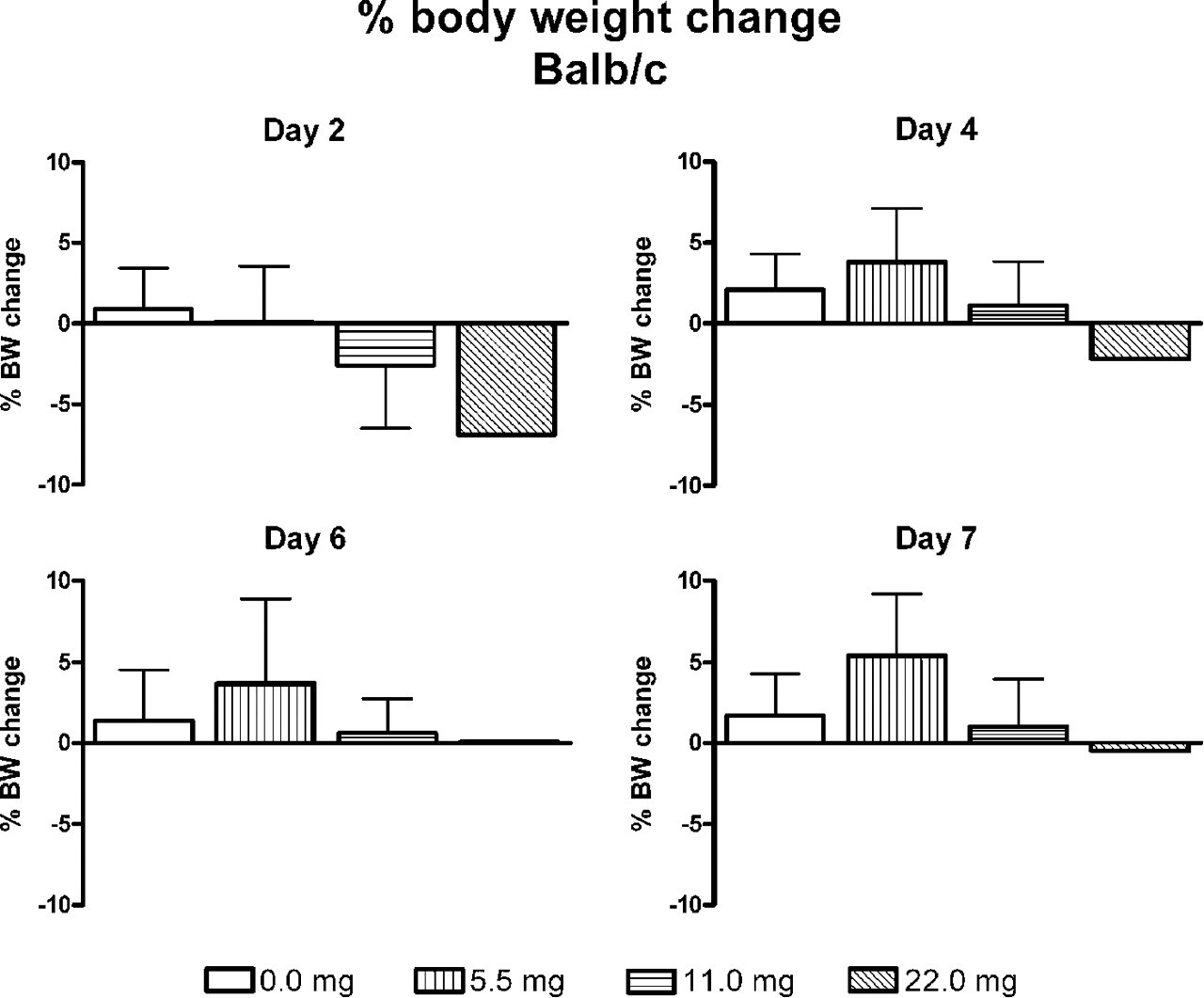
The percentage change in body weight of groups (n = 5) of Balb/c mice at 2, 4, 6, and 7 days after intranasal calcium administration of 0 to 22 mg/kg. At the highest dose of calcium, the change represents that of the single surviving mouse. The change in body weight of none of the groups was significantly different (
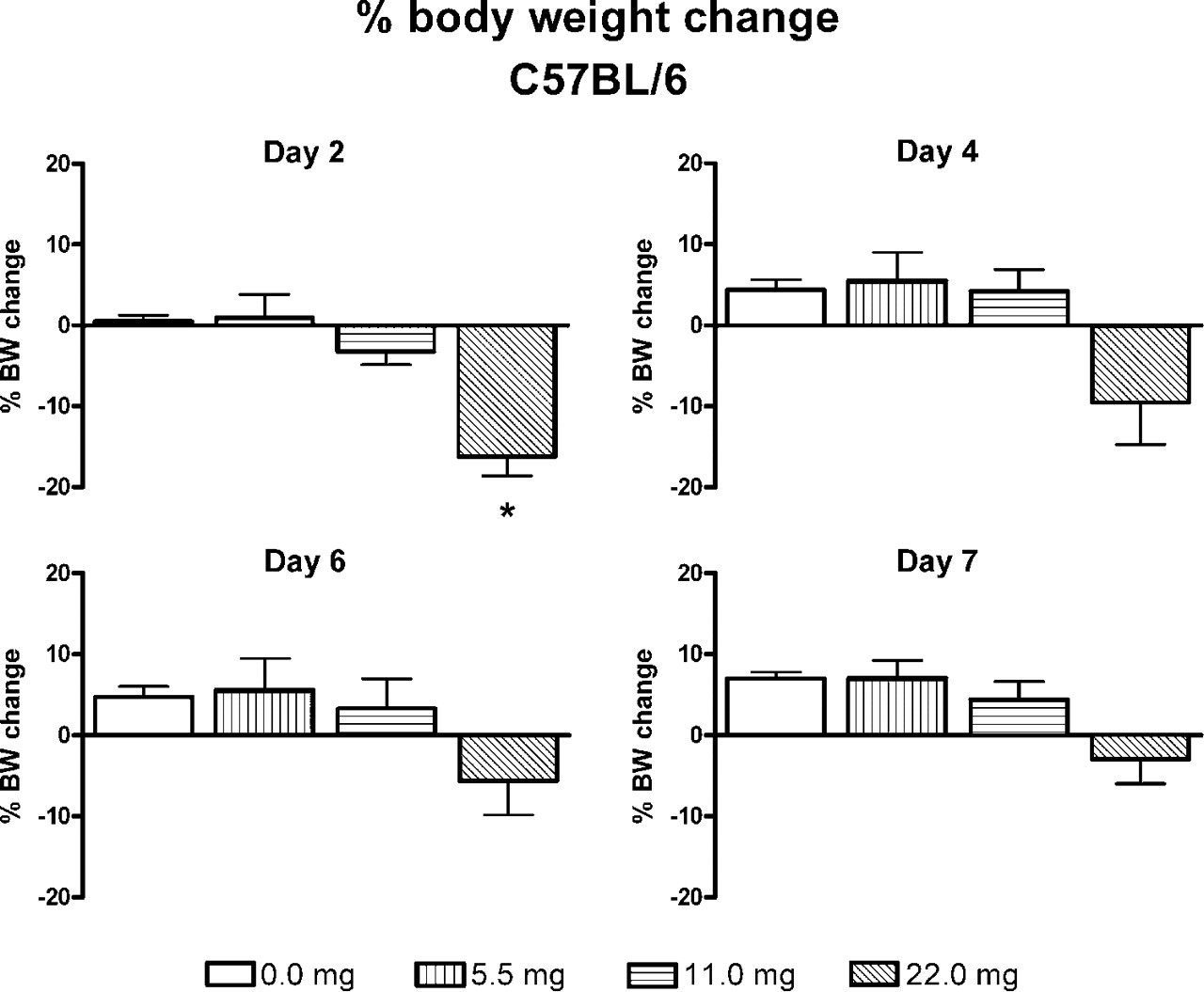
The percentage change in body weight of groups (n = 5) of C57BL/6 mice at 2, 4, 6 and 7 days after intranasal calcium administration of 0 to 22 mg/kg. At the highest dose of calcium, the change represents the average of the 4 surviving mice. The change in body weight of none of the groups was significantly different (
The ALP, SGPT, glucose, BUN, creatinine, and bilirubin in sera obtained at 7 days post administration of Ca2+ were comparable and not statistically different from the values observed in the respective Balb/c and C57BL/6 control groups receiving saline only (data not shown).
The gross necropsy of spleen, lungs, liver, thymus, kidneys, and heart at 7 days post treatment was normal and comparable to that of the respective organs in the control groups. Histopathologically, the lungs from the surviving Balb/c (n = 1) and C57BL/6 (n = 4) mice administered calcium at 22 mg/kg showed mild, chronic bronchopneumonia consisting of the infiltration of the alveolar spaces by small to medium numbers of large alveolar macrophages and the hypertrophy and hyperplasia of the type II alveolar epithelial cells. The lungs from mice receiving calcium at doses of 11 mg/kg and lower showed no remarkable changes. The histopathology of liver, spleen, heart, thymus, kidney, and brain from all groups appeared to be normal.
The results from the blood chemistry and histopathology observations at 7 days post administration, and clinical signs at up to 7 days, showed that intranasal administration of calcium at doses up to 11 mg/kg was relatively safe and did not cause any severe systemic adverse effects in mice.
Based on the above observations, we decided to investigate whether any acute toxic effects of Ca2+ could be observed, especially based on blood chemistry and histopathology, within a shorter 3-day postadministration period. This was evaluated at Ca2+ doses of 0.0, 5.5, 11.0, and 22.0 mg/kg in C57BL/6 mice rather than the more sensitive Balb/c, because it would potentially allow us to identify specific toxic events even at the highest dose where the Balb/c died rapidly but the C57BL/6 mice generally became sick but survived. None of the mice in any treatment group died, including those given the highest dose of calcium. At 1 and 2 days post administration, the clinical scores of the group of mice receiving the highest dose were similar to those shown for the C57BL/6 mice in Figure 1 for the previous experiment, and all mice had normal scores at 3 days (data not shown). The clinical scores of mice receiving all of the lower doses were normal (score of 0) and similar to the scores of the control group receiving saline only.
All mice, including the saline-only control group, lost between approximately 3% and 8% of their initial body weight at 1 day post administration (Figure 4). The loss was greatest (8.1%) in the highest dose group, but all mice were regaining the lost weight by 3 day post administration. The body weight change in none of the treatment groups was significantly different from that in the saline control group at any of the days.
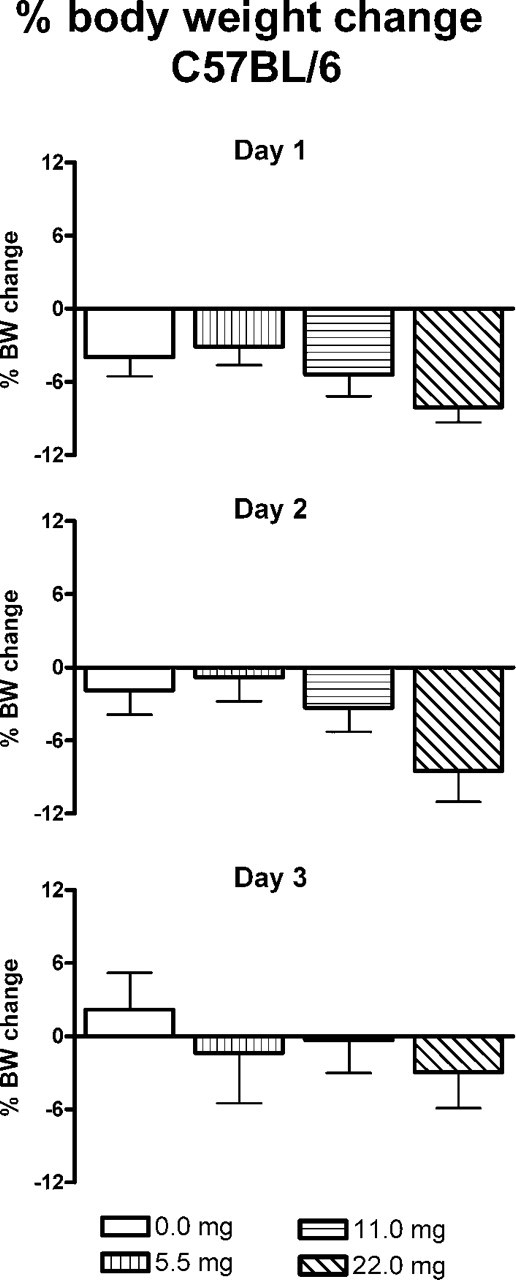
The percentage change in body weight of groups (n = 5-15) of C57BL/6 mice at 1, 2, and 3 days after intranasal calcium administration of 0 to 22 mg/kg body weight. None of the mice in any group died or had to be euthanized because of poor clinical outcome. The change in body weight of none of the groups was significantly different (
The total protein, glucose, ALP, BUN, SGPT, and creatinine in sera obtained at 1, 2, and 3 days post administration from all treatment groups are shown in Figure 5. These parameters in the different treatment groups were not significantly different from the corresponding values observed in the saline-only control group, except for the ALP and creatinine levels at the highest dose and ALP at the 11.0 mg/kg dose at 1 day post administration. At the subsequent days, there were no significant differences in any of the measured serum chemistry parameters.
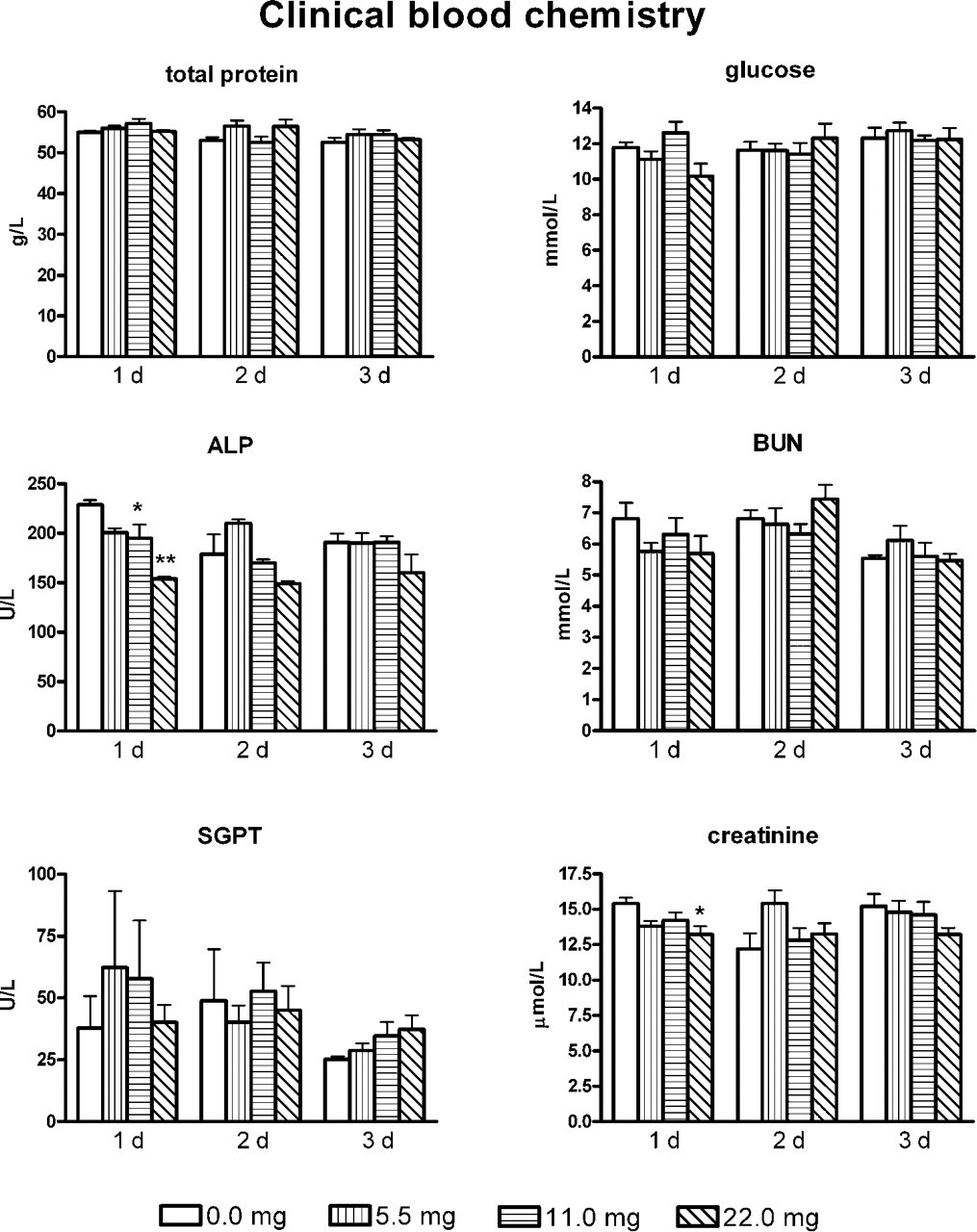
The average total protein, glucose, alkaline phosphatase (ALP), blood urea nitrogen (BUN), glutamic pyruvic transaminase (SGPT), and creatinine in sera of C57BL/6 mice at 1, 2, and 3 days post intranasal calcium administration of 0 to 22 mg/kg body weight. *
The gross necropsy of all major organs (spleen, lungs, liver, thymus, and heart) from the calcium treatment groups, at 1 to 3 days post treatment, was normal and comparable to that of corresponding organs from the saline-only control group. For the group receiving the highest dose of calcium, 22.0 mg/kg, the histopathology of lungs at 1 day post administration showed the presence of occasional neutrophil infiltration in the bronchial lumens and alveolar spaces. Such infiltration was more extensive in the lungs of the mice at 2 days post administration, where some lung sections also showed the fibrinous or proteinous exudates in the alveolar spaces. At 3 days post administration, the lungs from this group showed severe subacute bronchopneumonia in some areas where the alveolar spaces were occluded by medium to large numbers of alveolar macrophages, and some macrophages were large in size and contained foamy cytoplasm (Figure 6). The bronchial and type II alveolar epithelial cells in the affected areas showed some degree of hypertrophy and hyperplasia. The perivascular and peribronchial spaces were highly dilated and contained eosinophilic exudates, and occasionally the small blood vessels in these areas were occluded by inflammatory cells. The lungs from the mice receiving calcium at 11.0 mg/kg showed the occasional presence of small amounts of basophilic cellular debris in the lumens of some airways at day 1 but not at subsequent days (data not shown). The lungs from the mice receiving 5.5 mg/kg showed no remarkable changes at any time point. In all sections examined, the liver, spleen, kidneys, thymus, heart, and brain showed no remarkable changes. Moreover, there were no remarkable histopathological changes in the nasal mucosal epithelium of any of the mice administered the highest dose of calcium, 22.0 mg/kg (data not shown).
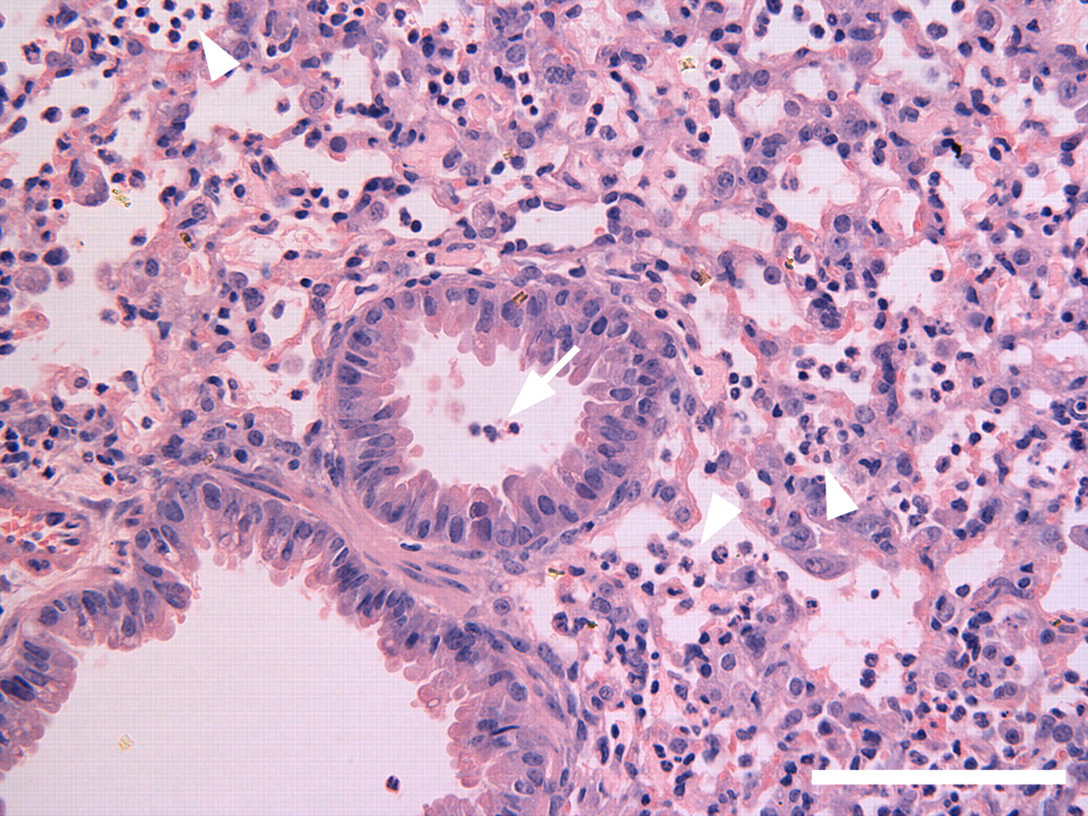
Subacute bronchopneumonia seen in a representative lung from the group of C57BL/6 mice sacrificed at day 3 after intranasal Ca2+ administration of 22 mg/kg. The lung shows the presence of small numbers of mononuclear cells in a small airway (arrow) and large numbers of mononuclear cells (arrowheads) mixed with large alveolar macrophages in the alveolar spaces. Hematoxylin–eosin stain, bar = 100 µm.
Discussion
Calcium phosphate has been used for decades as an adjuvant in vaccines administered systemically to humans. 1,3,4 Recently, calcium phosphate nanoparticles, 7,8 and cochleate cylinders made by the interaction of ester lipid liposomes with calcium chloride, 9,12 have been evaluated in animal models as adjuvants for generating mucosal immune responses via intranasally administered vaccines. More recently, we demonstrated the efficacy of intranasally administered AMVAD formulations containing calcium chloride in eliciting long-lasting mucosal and systemic immune responses in mouse models. 13,14 Compared with the abundant availability of animal model and human data on the use and tolerability of calcium phosphate in systemically administered vaccines, 2,5,15,16 there are no comparable data on the safety or tolerability of calcium per se used as a component of intranasal experimental vaccines. There are no published reports on the intranasal administration of calcium in humans in the context of vaccine applications. Because the safety of a vaccine can be influenced by the specific route of administration, 3,17,18 the objective of this study was to evaluate the tolerability/safety of intranasally administered calcium chloride using Balb/c and C57BL/6 mouse strains.
For assessing the safety of a vaccine component, it is prudent to include dosages that encompass and exceed those expected to be administered for efficacy. However, from the information in published literature,
8,9,11,12,14
it was difficult to precisely assess the amounts of calcium that would have been administered as a component of the various vaccines administered intranasally in animal models. Based on the descriptions of the published information
13,14
and our unpublished records of the body weights of the 6- to 12-week-old female Balb/c and C57BL/6 mice entering the experiments described, we calculated (as described in the Results section) that the maximal amount of calcium administered per intranasal immunization would have been in the range of 1.7 to 11 mg/kg body weight, depending on the type and source of the archaeal lipid used for making the AMVAD vaccine formulation. The evaluation of safety of calcium at up to 22 mg/kg body weight in the current study was therefore 2- to 13-fold higher than the calcium present in various AMVAD formulations previously used in AMVAD vaccine studies.
13,14,19
The 22 mg/kg dose of Ca2+ was 9- to 13-fold in excess of that (1.7-2.5 mg/kg body weight) present in
This study demonstrated that intranasal administration of Ca2+ (administered as CaCl2) at the highest tested dose of 22 mg/kg was toxic to mice. The toxicity was more evident in Balb/c mice than in C57BL/6, where 80% of the mice died within 24 hours of administration. One of the 5 C57BL/6 mice died at the 22-mg/kg dose, but the 4 surviving mice gradually recovered to normal clinical scores and regained most of the lost body weight by 7 days post administration. The death of the mice within 24 hours of administration was too quick to be attributable to a lack of appetite (starvation) or dehydration. There was no evidence of dehydration in the dead or the surviving mice. Calcium plays a critical role in many mammalian biological functions such as neurotransmission, cell–cell communication, biomembrane functionality/permeability, enzyme and protein activation/regulation, muscle functioning, blood clotting, and bone and tooth formation.
20
However, excess calcium can be a cellular toxin, and the body has evolved various means for maintenance of cellular calcium homeostasis.
21
Currently, we do not know the mechanism of the observed toxicity or the reason for the lethality of the 22-mg/kg dose of calcium. However, direct toxicity to the lower respiratory tract could be a contributing factor, because the damage in the liver and kidneys, as reflected by histopathology and clinical pathology, was minimal, as discussed below. At Ca2+ doses of 11 mg/kg and lower, no mice in any group died, and no adverse clinical signs or reactions were noted in either mouse strain. In contrast, intranasal administration of 5 µg of mLT (a mutant form of heat-labile enterotoxin from
Histological examination of the nasal tissue from C57BL/6 mice given the highest calcium dose (22 mg/kg) showed no abnormalities at 1 to 3 days or 7 days post administration, and these appeared similar to the nasal tissues from the saline-treated control mice. However, the lungs from the mice given 22 mg/kg showed acute and subacute bronchopneumonia with the infiltration of neutrophils and macrophages in the pulmonary tissues between days 1 and 3. The lung inflammatory responses were less severe and less extensive at the 11-mg/kg dose of Ca2+ and were hardly evident in the 5.5-mg/kg group. The lung inflammation, even at the highest dose, was not evident or was only minimal in a separate group of mice observed at day 7, indicating that the inflammation is self-resolved in a few days. Apart from the inflammatory responses seen in the lungs, there were no remarkable changes in the other tissues (liver, spleen, heart, thymus, kidneys, or brain) examined. Mild inflammation localized to the administration sites has also been reported with calcium phosphate nanoparticles administered systemically or via inhalation (about 1.2 mg of calcium phosphate per kilogram of body weight) in animal models. 7 Infiltration of deformed neutrophils and accumulation of macrophages with foamy cytoplasm, which occurred at the injection site upon intramuscular calcium injection of about 3.3 mg/kg (as calcium phosphate gel) in guinea pigs, was reported to be completely resolved by 8 weeks. 5 The presence of self-resolved inflammatory responses in the lungs, following intranasal administration of calcium in the current study, was probably essential for the recruitment and activation of antigen-presenting cells and other immune cells. The presence of these lymphoid tissues is believed to be important for the induction of mucosal immune responses. 22
The tolerability of calcium when administered alone may be different than when given intranasally as one of the several components of a vaccine. In this context, we recently evaluated the safety of a vaccine comprising AMVAD formulation prepared from
The results of the current study show that a single, intranasal administration of Ca2+ at a 22-mg/kg dose results in moderate to severe toxicity in mice. This study suggests that intranasal administration of such high amounts of calcium could potentially pose severe safety concerns. However, at a Ca2+ dose of 11.0 mg/kg, a dose that was 4- to 6-fold in excess of the maximal amount of Ca2+ present in previous
Footnotes
Acknowledgments
The technical assistance of Rhonda KuoLee in collecting organs, and Tom Devecseri in preparing the figures, is highly appreciated. This paper is NRCC 50003.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
This research was funded by the National Research Council of Canada.
