Abstract
AV7909 is a next-generation anthrax vaccine under development for post-exposure prophylaxis following suspected or confirmed
Introduction
Anthrax is a zoonotic infectious disease, caused by
Preclinical studies have been conducted with CPG 7909-adjuvanted vaccine candidates against diseases caused by
Preclinical vaccine evaluation in relevant animal models includes all aspects of testing, product characterization, and proof of concept/immunogenicity studies. In particular, safety testing in animals is an essential prerequisite to moving a candidate vaccine to the clinic. 13 Repeat-dose toxicity studies in animals can help establish the maximum tolerated dose in an animal model and aid in selecting dose levels in the subsequent clinical studies. If no major safety signals are revealed, a single repeat-dose toxicity study in 1 relevant species may be sufficient. 14
The purpose of this preclinical toxicology study was to evaluate potential local and systemic toxicity of a novel anthrax vaccine AV7909 in a rat model following 3 intramuscular (IM) administrations on Days 1, 15, and 29 followed by a 2-week recovery period.
Materials and Methods
Test and Control Articles
Two liquid formulations of AV7909 lots containing different amounts of CPG 7909 were manufactured at Emergent BioSolutions. AV7909 Lot 1 contained 0.5 mL AVA and 0.5 mg CPG 7909 per dose. AV7909 Lot 2 contained 0.5 mL AVA and 0.25 mg CPG 7909 per dose. Both test articles were manufactured by Emergent BioSolutions (Lansing, MI). The control article 0.9% sodium chloride was obtained from Hospira, Inc. (Rocky Mount, NC). The CPG 7909 adjuvant control was purchased from Girindus America (Cincinnati, OH) as dry powder and was reconstituted on each day of dosing to provide 0.5 mg in 0.5 mL of saline.
Experimental Animals
A total of 160 (80 male and 80 female) Sprague Dawley rats (Crl: CD®), 11 weeks of age, were obtained from Charles River Laboratories (Portage, MI). Male (weight range 254.4 to 340.1 g) and female (weight range 178.5 to 228.4 g) rats were randomized into 4 groups. All rats had
The study was conducted in accordance with the provisions of the USDA Animal Welfare Act, the PHS Policy on Humane Care and Use of Laboratory Animals, and the US Interagency Research Animal Committee Principles for the Utilization and Care of Research Animals. The protocols were approved by Institutional Animal Care and Use Committee (IACUC). A minimum number of animals were used for the study to achieve biological significance based on the study design in accordance with industry standard. 15
The rat model was selected as previous studies in rats have shown strong immune response to PA 16 and CPG 7909 ODN. 17 This model is also widely used in toxicology studies.
Experimental Design
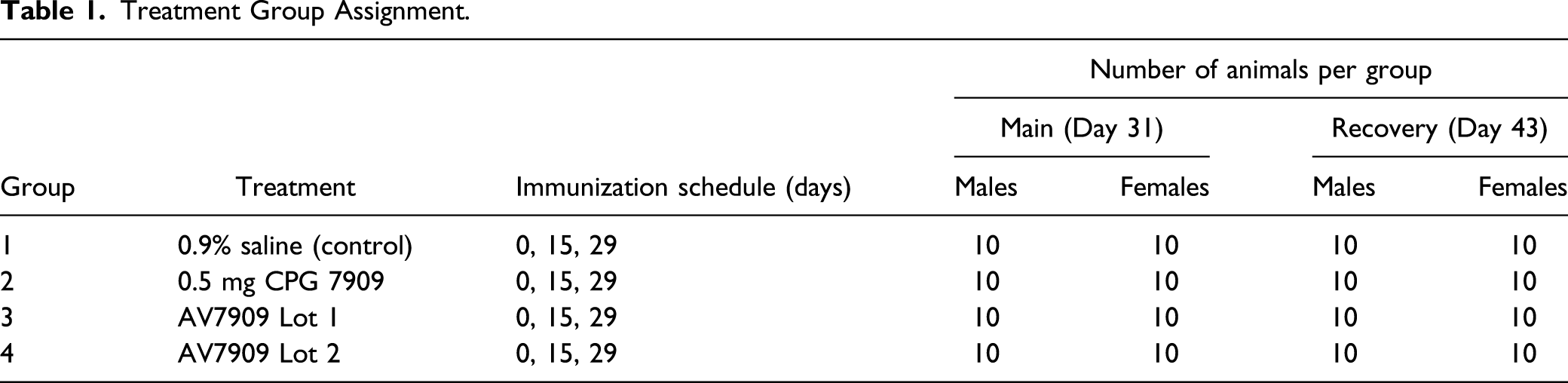
Treatment Group Assignment.
Dose Administration and Injection Site Observations
Dermal Draize Observations.

Clinical Observations, Body Temperature, and Body Weights
Cage-side observations were performed at least twice a day and included observation for mortality, moribundity, and general health and signs of toxicity. Full clinical observations were performed weekly, 1 day prior to termination and on the day of termination. These observations included evaluation of skin and fur characteristics, the injection site, eye and mucous membranes, respiratory, circulatory, autonomic and central nervous systems, somatomotor, and behavior patterns. The time of onset, location, dimensions, appearance, and progression of any grossly visible or palpable tumors were also observed. Ophthalmologic observations were conducted using indirect ophthalmoscopy and slit lamp biomicroscopy (as needed) following administration of 1% tropicamide® mydriatic solution. Individual body weights were recorded for all animals, on a weekly basis and were also performed 1 day prior and on the day of termination. Body temperature was recorded for all animals prior to dosing, 24 ± 2 hours after dose and 48 ± 2 hours after dose.
Clinical Pathology
Clinical pathology tests including clinical chemistry, hematology, blood coagulation, and urinalysis were performed as essential and standard parts of preclinical safety assessment of human pharmaceuticals. Prior to sample collection, animals were fasted overnight (with water available). The animals were anesthetized via CO2/O2 inhalation and blood was collected via puncture of the abdominal aorta followed by humane euthanasia. The core tests included chemistry, hematology, blood coagulation standard parameters described in literature, and relevant guidance documents.19,20
Necropsy
The main study animals were sacrificed on Day 31 and recovery animals were sacrificed on Day 43. On each sacrifice day, 10 animals/sex were euthanized via carbon dioxide inhalation followed by exsanguination. Animals were necropsied immediately after death. Necropsy included examination of the external surface of the body and all orifices; the cranial, injection sites, thoracic, abdominal and pelvic cavities, and their contents; and the collection of tissues. The organs were weighed as soon as possible after dissection at scheduled necropsies. Paired organs were weighed together, with the exception of iliac lymph nodes as noted below. Adrenal glands, brain, heart, kidneys, iliac lymph nodes (left and right separately), liver, lung (with bronchi), ovaries (with oviducts), spleen, testes (with epididymides), thymus, and uterus (with cervix) were all weighed. Tissues were preserved in 10% neutral buffered formalin (NBF), except eyes and testes that were first preserved in modified Davidson’s fixative and then transferred to NBF. Two bone marrow smears were prepared from the left femur of all animals. Slides were air dried, fixed in methanol, and stored at room temperature until disposal at the end of the study.
Histopathology
All protocol-required tissues were embedded in paraffin, sectioned, stained with hematoxylin and eosin, and examined by a board-certified pathologist. For evaluation of local and systemic toxicity, samples were collected from the standard list of tissues, as recommended by regulatory guidance documents.14,21
Immunogenicity Assessment
Blood samples were collected into serum separator tubes prior to necropsy on Days 31 (main) and 43 (recovery) via puncture of the abdominal aorta. Blood was allowed to clot at room temperature for at least 30 minutes and serum isolated by centrifugation. The serum samples were tested by enzyme-linked immunosorbent assay (ELISA) for antibodies against
Anthrax toxin-neutralizing antibody levels in rat serum samples were measured using anthrax lethal toxin (LT) neutralization assay (TNA) as described previously.
23
The assay is designed to measure and quantify the functional ability of serum to neutralize
Statistical Analysis
All appropriate quantitative in-life data were analyzed for test article effects by a parametric or nonparametric analysis of variance (ANOVA) using SigmaStatTM Statistical Software version 1.0. For all data, normality was determined by the Kolmogorov–Smirnov test, and homogeneity of variances was determined by Levene’s test and one-way ANOVA test. Nonparametric Kruskal–Wallis ANOVA was used to transform data rank-wise if the normality or equal variance tests failed. For parametric data, if the ANOVA indicated statistical significance among experimental groups, then the Dunnett’s t-test was used to delineate which groups (if any) differed from the control. For non-parametric data, if the ANOVA indicated statistical significance among experimental groups, then the Dunn’s test was used to delineate which groups (if any) differed from the control. All statistical tests were performed at the 0.05 level of significance (
Results
Clinical Observations
No treatment-related adverse effects or mortality were noted during cage-side or clinical observations following administration of AV7909. However, some low-occurrence observations not related to the treatment and common among laboratory rats were noted. These observations included 3 animals with alopecia, 1 animal with abrasion away from the injection site, 1 animal with mucoid feces, and 1 animal with hunched posture on Day 1 and with rough hair coat from Days 8 to 43.
Based on the results of the Draize scoring, none of the main or recovery group animals exhibited any signs of edema or erythema throughout the course of the study (data not shown).
The normal body temperature for rats is approximately 38–39°C and individual animals did not exceed the upper limit of this range, except female animals in groups 1 (39.50°C) and 2 (39.62°C) at 48 hours following the third immunization, which exhibited temperature above 39°C and was considered incidental and not treatment-related.
AV7909 treatment did not cause any ocular lesions in rats. However, 1 male rat in Group 4 showed hemorrhage in the fundus of the left eye on Day 31. The spontaneous lesions in the retina, including retinal hemorrhage, can be observed during normal ocular development and in naïve adult rats.24,25 Therefore, due to the low incidence and based on review of the concurrent control data, the finding was considered incidental and not related to the test article administration.
Food Consumption and Body Weights
Overall, there were no significant differences in the mean total food consumption in the treated groups (groups 2–4) relative to the control group (Group 1) within the main and recovery animals. All animals that were vaccinated with AV7909 test article showed gradual increase in weight over the course of the study, indicating normal gain in body weight and that the vaccine did not have any effect on food consumption. There were no significant differences in the mean total absolute body weight change for the Days 1–29 or Days 1–42 intervals (Figure 1). Body weight change. Body weight change is presented as mean and standard deviation. A steady increase in body weight over the course of the study in both male and female rats that were administered with the AV7909 test article was noted except on Days 29 and 43 where there was a slight decrease in body weights as the rats were fasting. Group 1 - 0 mg (control article) Group 2 - 0.5 mg CPG 7909. Group 3 - 0.5 mL AV7909 Lot 1 (0.5 mL AVA and 0.5 mg CPG 7909). Group 4 - 0.5 mL AV7909 Lot 2 (0.5 mL AVA and 0.25 mg CPG 7909). AVA, anthrax vaccine adsorbed.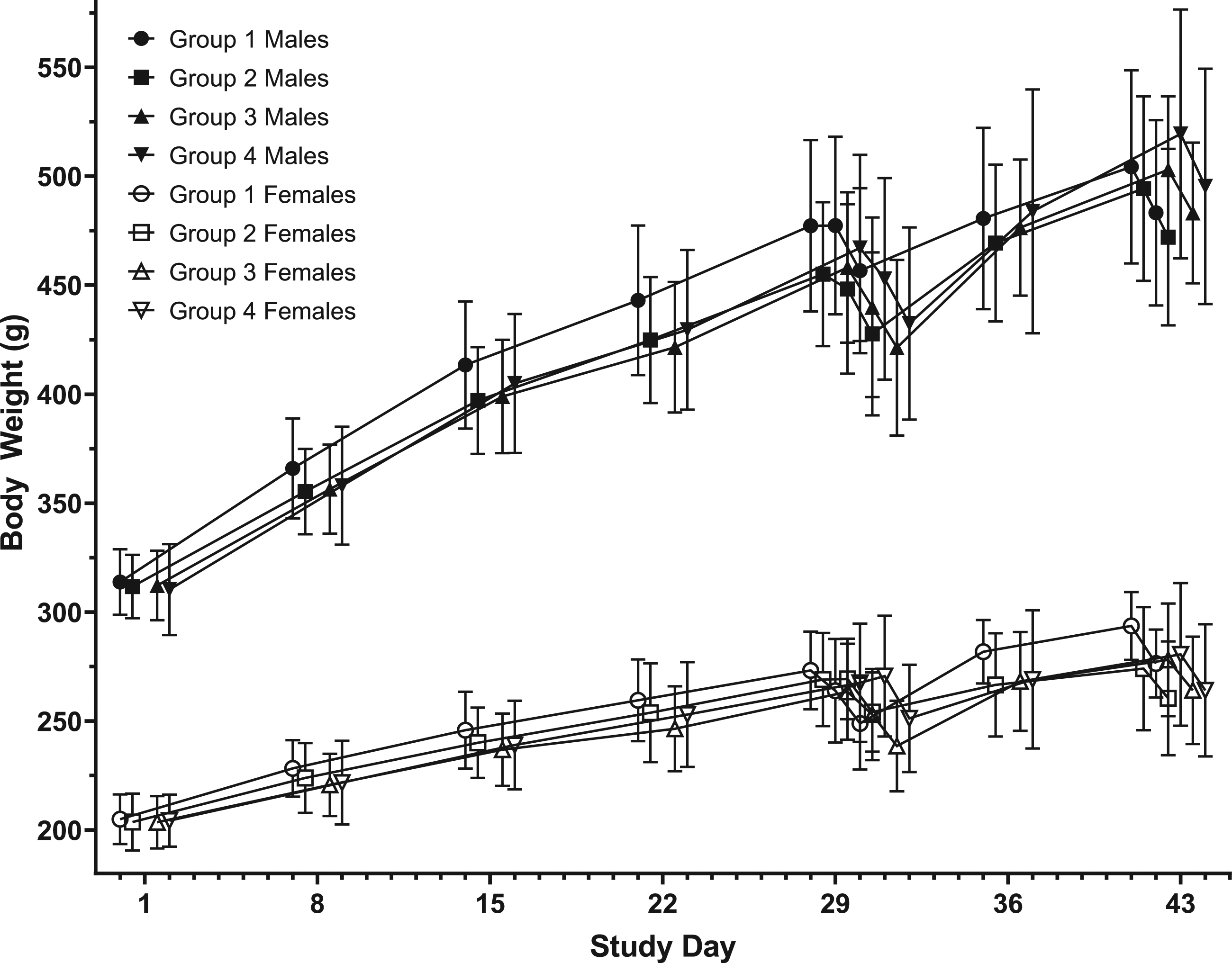
Clinical Pathology
Hematology
Hematology Results.
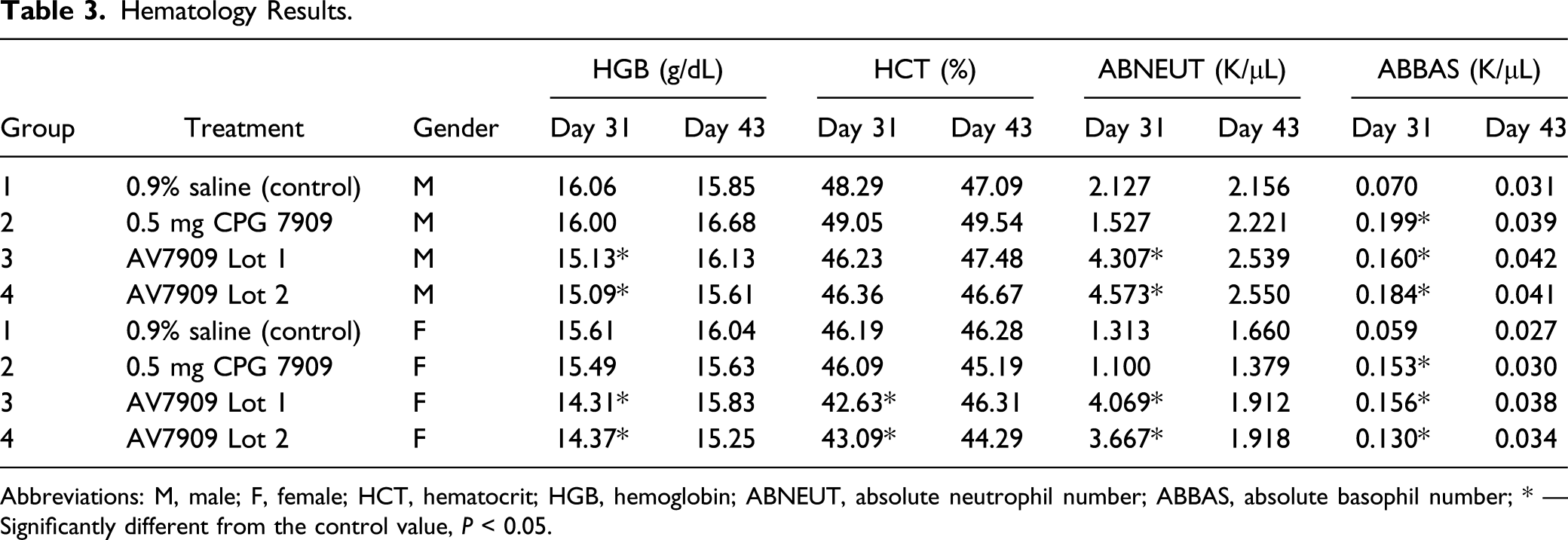
Abbreviations: M, male; F, female; HCT, hematocrit; HGB, hemoglobin; ABNEUT, absolute neutrophil number; ABBAS, absolute basophil number; * — Significantly different from the control value,
Clinical Chemistry
Key Clinical Chemistry Results.
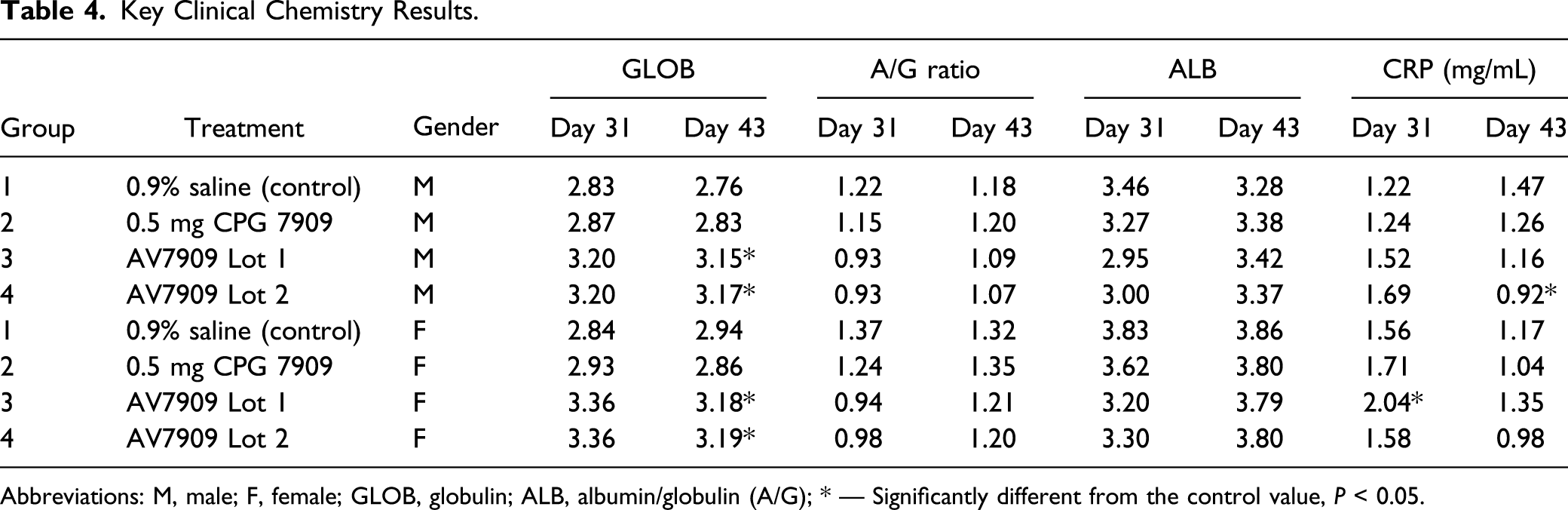
Abbreviations: M, male; F, female; GLOB, globulin; ALB, albumin/globulin (A/G); * — Significantly different from the control value,
Coagulation
Coagulation Results.
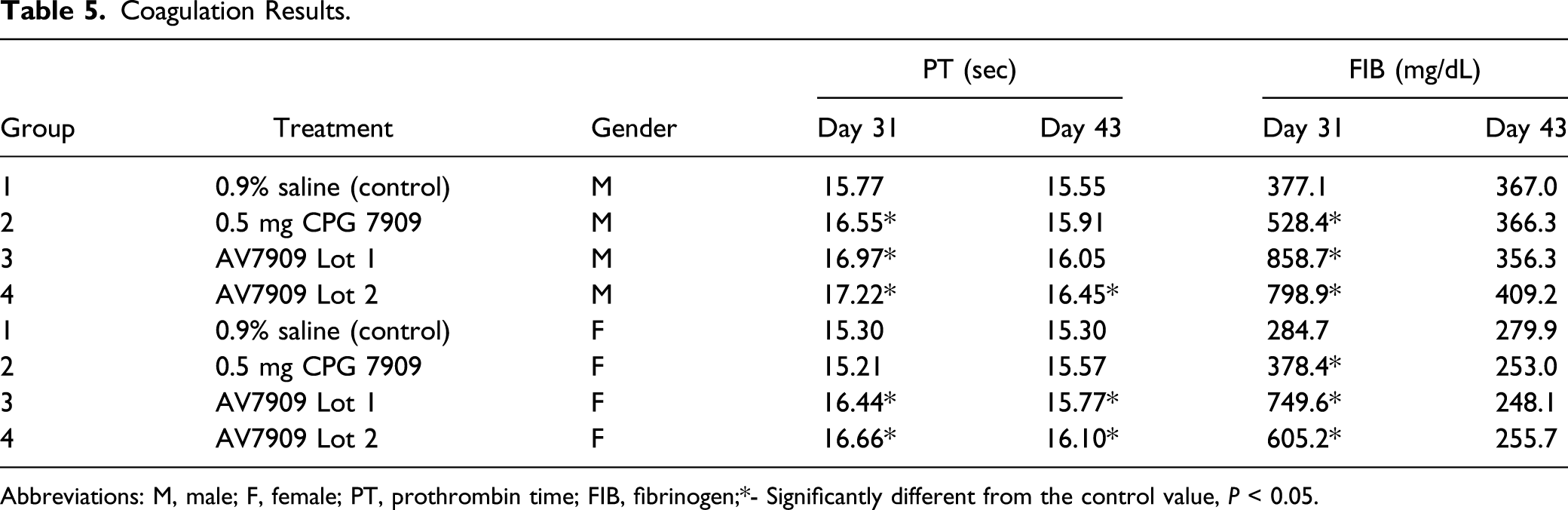
Abbreviations: M, male; F, female; PT, prothrombin time; FIB, fibrinogen;*- Significantly different from the control value,
Necropsy and Histopathology
Organ Weights
Organ Weights.
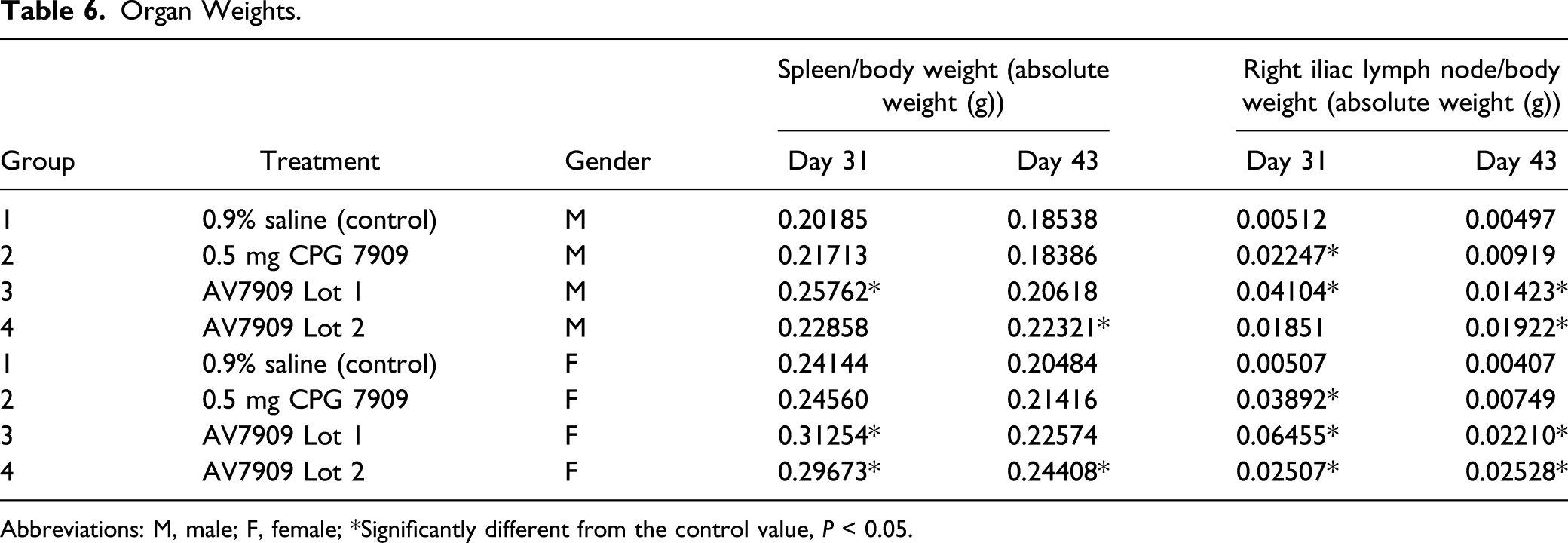
Abbreviations: M, male; F, female; *Significantly different from the control value,
Animals sacrificed at recovery necropsy also had treatment-related weight increases for right iliac lymph nodes and spleen. Absolute and relative right iliac lymph node weights were increased in animals in Groups 3 and 4, correlating with increased GLOB values and microscopically with lymphoid hyperplasia. This finding was consistent with immune stimulation and was non-adverse. Absolute spleen weight was increased in Group 4 males, and spleen weight relative to body weight was increased in Group 4 males and females. This change correlated microscopically with increased splenic hematopoiesis in Group 4 males and was non-adverse.
Macroscopic Findings
In animals sacrificed at main necropsy, test article–related changes occurred at the injection site in Groups 3 and 4 (9/20 animals), and in the inguinal lymph node in 1 Group 4 female. Masses at the injection site were intramuscular, tan, white, or yellow, firm, semi-firm, or soft (cut surface), often multilobular, and contained caseous material. These macroscopic changes correlated with microscopic necrosis and inflammation of the muscle and surrounding fascia, occasionally extending into the subcutis. Treatment-related enlargement of the right iliac lymph node occurred in Groups 2 (7/20 animals), 3 (16/20 animals), and 4 (9/20 animals), and 1 Group 3 female showed enlargement of the right inguinal lymph node. These lymph node findings correlated microscopically with lymphoid hyperplasia.
In animals sacrificed at the recovery necropsy, test article–related injection site masses were observed in Groups 3 (11/20 animals) and 4 (12/20 animals), and in a single animal in the adjuvant-treated Group 2 (1/20 animals) and the saline control Group 1 (1/20 animals). The morphology of these masses was similar to those observed in the main necropsy animals. The macroscopic masses observed in Group 1 and Group 2 animals were not present microscopically, while in Group 3 and 4, the masses correlated microscopically with necrosis and inflammation of the muscle and surrounding fascia, occasionally extending into the subcutis. Two Group 3 animals had test article–related masses in abdominal skeletal muscle, which were tan, firm, and contained caseous material. These masses correlated microscopically with necrosis and inflammation of the muscle and surrounding fascia. The lesions likely were an extension of changes at the adjacent injection site. Treatment-related enlargement of the right iliac lymph node also occurred in Groups 2 (3/20 animals), 3 (7/20 animals), and 4 (10/20 animals), and correlated microscopically with minimal to mild lymphoid hyperplasia.
Microscopic Findings
In animals sacrificed at the main necropsy, test article–related adverse changes occurred at the injection site in Groups 3 (19/20 animals) and 4 (14/20 animals). Mild to marked necrosis occurred within muscle at the injection site for Groups 3 and 4 (primarily Group 3), accompanied by mild to moderate chronic, chronic-active, or granulomatous inflammation which often extended to the surrounding fascia and occasionally into the subcutis. Necrosis and/or inflammation were occasionally observed in sections of skeletal muscle taken from areas adjacent to the injection site. These inflammatory changes correlated with alterations in peripheral leukocyte counts. Minimal to mild subacute inflammation at the injection site occurred in Groups 2, 3, and 4 (primarily Groups 2 and 4) without injection site necrosis. These inflammatory changes correlated with increase in peripheral leukocyte counts and were treatment-related that were considered adverse. Treatment-related minimal to moderate lymphoid hyperplasia occurred in iliac lymph nodes (primarily in the right node, ipsilateral to the injection site) in Groups 2, 3, and 4, and in the inguinal lymph node in 1 Group 3 animal. This change was consistent with immune stimulation, correlated with increased serum globulin values for Groups 3 and 4 and was non-adverse. Test article–related changes were also observed in the lymph nodes. Scattered accumulations of macrophages occurred in the iliac lymph nodes of animals in animals in Groups 3 and 4. These findings were non-adverse. One Group 4 animal had test article–related necrosis and chronic inflammation in an inguinal lymph node, a change that was adverse. Four Group 4 females had test article–related mild increases in macrophage numbers in the spleen, possibly correlating with an increased spleen weight. This finding was associated with a normal clearance mechanism and was non-adverse.
In animals sacrificed at the recovery necropsy, test article–related adverse changes occurred at the injection site of animals in Groups 3 (12/20 animals) and 4 (14/20 animals). These changes correlated with masses observed macroscopically and consisted of mild to marked necrosis accompanied by minimal to moderate chronic, or mild to moderate granulomatous, inflammation. Inflammation was occasionally observed in routine sections of skeletal muscle taken from areas adjacent to the injection site. In 2 Group 3 animals, areas of necrosis and inflammation in the abdominal skeletal muscle were similar microscopically to changes at the injection site and correlated with macroscopic masses. These lesions likely were an extension of changes at the adjacent injection site. Treatment-related minimal to mild chronic inflammation also occurred at injection sites in the absence of injection site necrosis in Groups 2, 3, and 4. This finding represented a normal progression from the subacute inflammation present on Day 31 observed at injection sites that lacked necrotic changes. These inflammatory changes were considered to be treatment-related and adverse. Treatment-related minimal to mild lymphoid hyperplasia also occurred in the right iliac lymph nodes (ipsilateral to the injection site) in Groups 2, 3, and 4. This change was consistent with immune stimulation, correlated with increased serum globulin values for Groups 3 and 4, and was considered non-adverse. Test article–related changes were also observed in the iliac lymph nodes. Scattered accumulations of foamy macrophages occurred in the iliac lymph nodes of animals in Groups 3 and 4. This finding was non-adverse. Two Group 4 females had test article–related minimal to mild increases in macrophage numbers in the spleen. This finding was related to a normal clearance mechanism, and non-adverse. Minimal to mild increased splenic hematopoiesis was observed in Group 3 and 4 males, likely a compensatory response to the decreased HGB and HCT levels noted on Day 31 and correlating with the increased spleen weight for Group 4 males. This change was test article–related but non-adverse.
Immunogenicity
Immunogenicity of the vaccine was evaluated using the anti-PA IgG ELISA and toxin neutralization assay as described below. Both assays demonstrated a robust immune response generated in rats by AV7909 vaccination, thus confirming the specific biological effect expected from the vaccine and immunostimulatory effect of the CPG 7909 adjuvant.
Anti-PA IgG ELISA
Serum samples from Days 31 and 43 were analyzed using anti-PA ELISA. As expected, the serum samples from the control group and CPG 7909 adjuvant (Group 1 and 2) did not have quantifiable levels of PA-specific IgG (below the limit of quantitation of 0.8 µg/mL). In contrast, animals in Groups 3 and 4 generated significant antibody levels with specificity to PA, indicative of a robust immune response to AV7909. The results are shown in Figure 2. Anti-PA IgG levels as measured by ELISA. Anti-PA IgG levels in rats are reported as mean and standard error for the group of animals that were administered with the AV7909 vaccine. No detectable levels of anti-PA IgG were observed in control group animals or the adjuvant-alone group and hence not included in the figure. Group 3 - 0.5 mL AV7909 Lot 1 (0.5 mL AVA and 0.5 mg CPG 7909). Group 4 - 0.5 mL AV7909 Lot 2 (0.5 mL AVA and 0.25 mg CPG 7909). AVA, anthrax vaccine adsorbed; ELISA, enzyme-linked immunosorbent assay.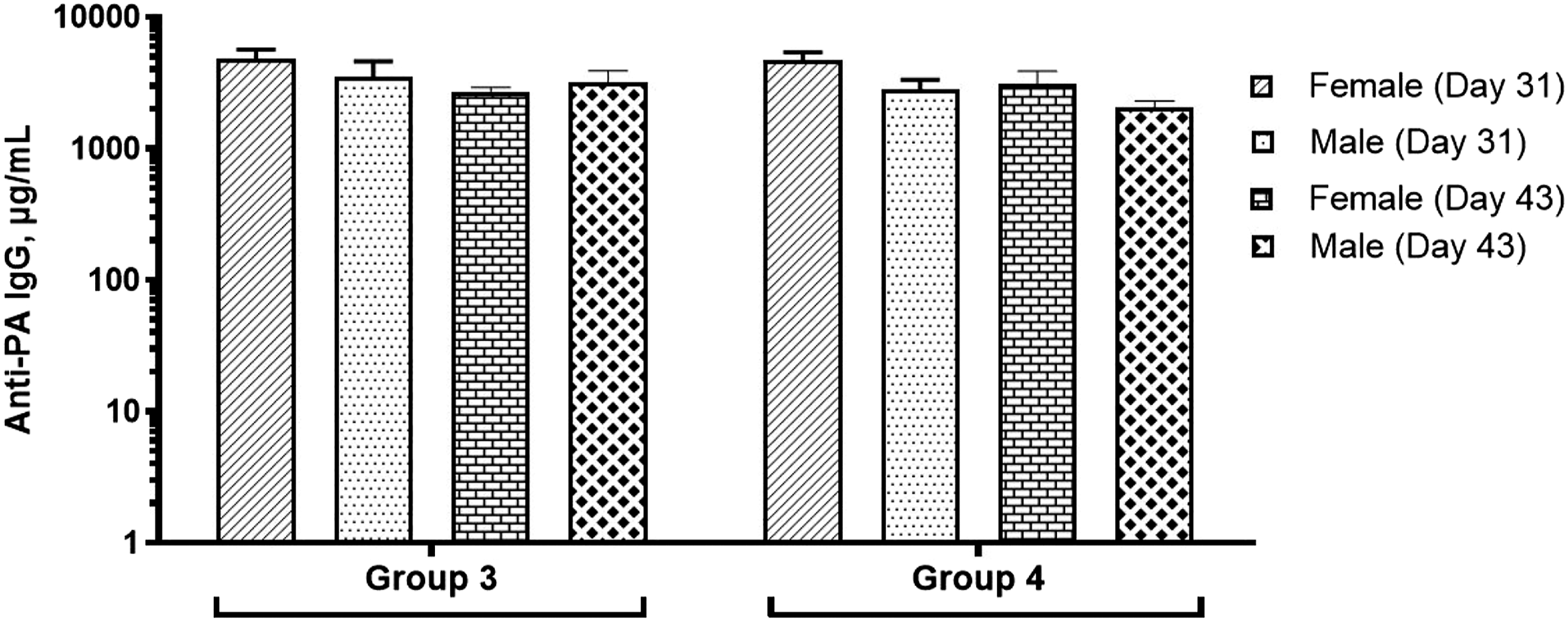
Toxin Neutralization Assay
In addition to the high anti-PA antibody levels, AV7909 induced a strong functional immune response in both groups of vaccinated animals (Groups 3 and 4), as measured by the toxin-neutralizing antibody levels (Figure 3). Consistent with the ELISA data, the serum effective dilution that neutralizes 50% of the cytotoxic effect of the anthrax lethal toxin (ED50) values in the control group and the group that was administered with CPG 7909 alone were not quantifiable (data not shown). Toxin-neutralizing antibody (ED50) levels as measured by TNA. TNA ED50 (the dilution that neutralizes 50% of the cytotoxic effect of the anthrax lethal toxin) levels presented as mean and standard deviation. ED50 values were obtained from animals that were immunized with 3 doses of AV7909 vaccine. In the AV7909-vaccinated group, median ED50 values were in the order of 10000 that suggests strong immune response generated by the vaccine. The toxin neutralizing antibodies were not detectable (below limit of quantitation) in the control and CPG 7909 alone groups and not shown in the figure. Group 3 - 0.5 mL AV7909 Lot 1 (0.5 mL AVA and 0.5 mg CPG 7909). Group 4 - 0.5 mL AV7909 Lot 2 (0.5 mL AVA and 0.25 mg CPG 7909). AVA, anthrax vaccine adsorbed; TNA, toxin neutralization assay.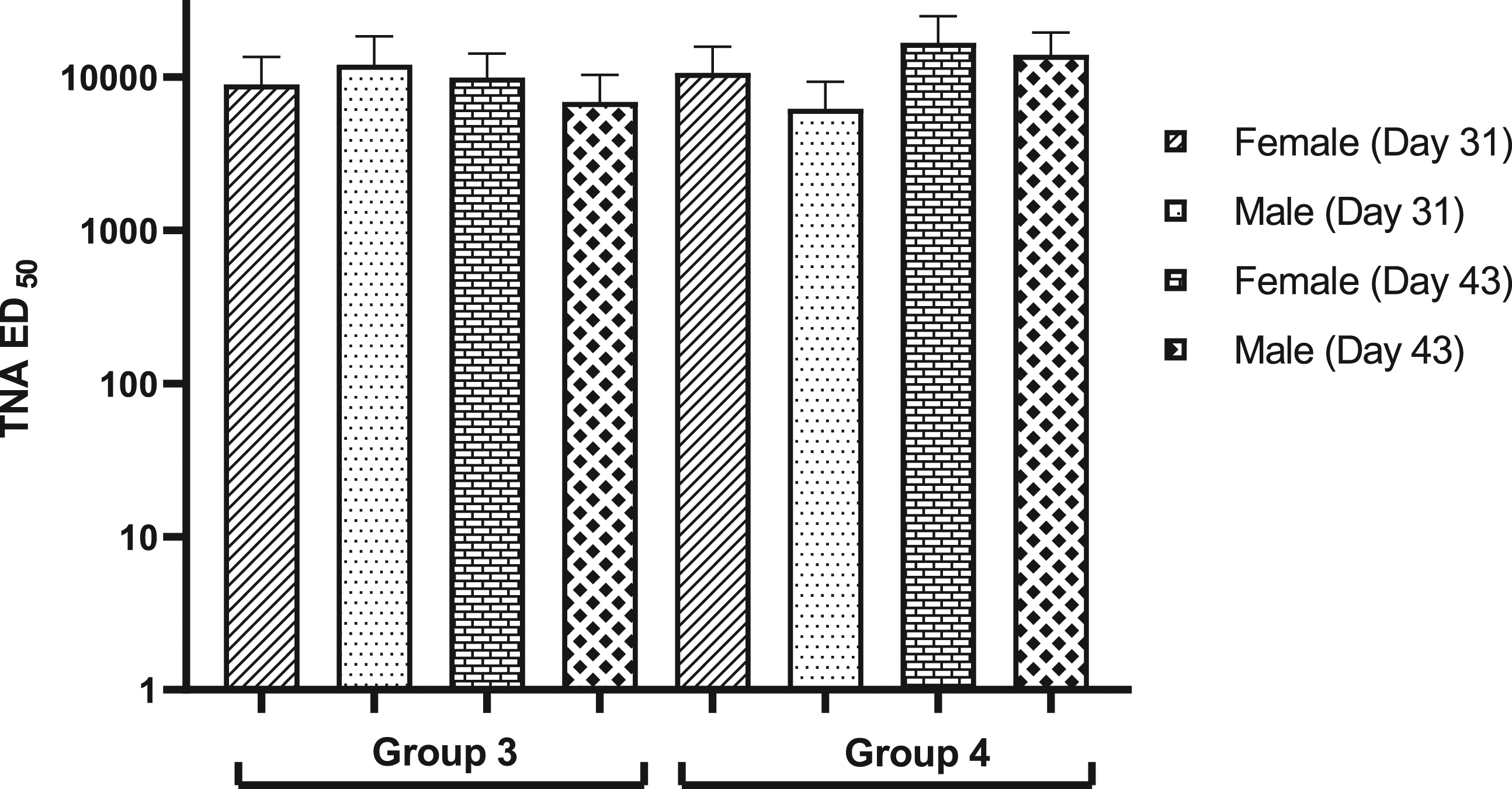
Discussion
Identifying the appropriate dosage of a vaccine plays a vital role in balancing its ability to be effective within safety limits. Assessing systemic toxicity and local reactogenicity of a vaccine in animals can help predict these effects in human subjects and define the appropriate safe dose levels. A repeat-dose toxicity study in animals, in which the number of vaccinations exceed that intended in the clinic, is, therefore, an important tool for evaluating the safety of the vaccine and can help identify potential safety signals that should be closely monitored in the clinic.
In this repeat-dose toxicity study, the maximum dose levels that could be safely tolerated in adult rats was determined to be 0.5 mL of AVA with 0.5 mg of CPG 7909. The study demonstrated that the AV7909 vaccine was safe and well tolerated in adult rats. AV7909 was able to induce a robust immune response without causing any significant effects on target organs or any systemic immediate or delayed adverse effects. Importantly, a strong antibody response, including the functional TNA response, which is known to be predictive of vaccine efficacy, 6 confirmed the appropriateness of using the rat model to evaluate preclinical safety of the AV7909 vaccine.
Although local reactogenicity associated with immune stimulation was noted, these observations are consistent with the findings in other toxicology studies with AV7909.26-28 Indeed, such effects are observed frequently, especially with adjuvanted vaccines. Furthermore, the 14-day recovery period, which is in line with current regulatory guidance for preclinical safety evaluation of vaccines, may not be sufficiently long to observe complete resolution of the local reactions. 29 Similar local reactogenicity observations were reported for other CpG ODN-adjuvanted vaccines, including those against hepatitis B,30,31 influenza, 32 malaria,33-35 pneumococcus,36,37 cytomegalovirus, and tetanus. 38 Of note, in the recent repeat-dose toxicity study in juvenile rats, 27 AV7909 was also well tolerated with no severe adverse effects or morbidity and induced a strong neutralizing antibody response. Taken together, these data indicate a favorable safety profile for CPG 7909 when used as a part of the vaccine adjuvant system.
A comprehensive review of repeat-dose toxicity studies in rat or rabbit models with a variety of vaccines administered IM reported acute and/or chronic inflammation at the injection sites, often accompanied by lymphoid hyperplasia and/or increased weight in draining lymph nodes and the spleen, as well as necrosis of muscle fibers within the injection sites. These findings were interpreted as an expected reaction associated with vaccine- and/or adjuvant-induced immune response, and the investigational vaccines were deemed to be well tolerated. 39 The author concluded that a recovery period of 2 to 4 weeks was sufficient to show evidence of reversibility of injection site effects and that the changes can be interpreted as related to an expected reaction to vaccination, associated with generation of an immune response augmented by the presence of an adjuvant. Myofiber necrosis at the injection site, with or without mineralization, was also observed in a rabbit repeat-dose toxicity study of 2 different formulations of a malaria vaccine candidate, 1 of which, selected based on a more favorable immunogenicity profile, subsequently progressed to Phase 2 and 3 clinical studies. 40 Necrosis of muscle fibers at the injection site, following IM administration, was also reported in repeat-dose toxicity studies with other vaccines, including inactivated enterovirus vaccine candidate 41 and nucleic acid–based HIV vaccine candidates, 42 both of which were found to be safe and well tolerated, and advanced to Phase 1 clinical studies.
Local reactions such as erythema, edema, inflammation, and pain have been described following CpG ODN administration in humans.4,30,43,44 However, CpG ODNs were generally well tolerated, with little and transient side effects. In most cases, local inflammatory lesions resolved in less than 24 hours 45 . The inflammatory effects observed in this study are also consistent with those commonly seen with Alhydrogel-adjuvanted vaccines. 46
Overall, the results of this study demonstrated that repeated IM administration of AV7909 to Sprague Dawley rats did not induce any systemic toxicity. The study, therefore, supports the use of the AV7909 vaccine in humans when administered as 2 IM injections.
Footnotes
Acknowledgments
The authors would like to thank the technical staff at Inotiv for the animal immunization, toxicological assessments, necropsy, and histopathology; Andrea Harris for the anti-PA ELISA testing; and Katalin Baranji for the htpTNA testing.
Author Contributions
Veena V. Rao, Vladimir Savransky, Boris Ionin, Joshua J. Reece, Sukioon Park, and Bruna Blauth contributed to the conception and design of the toxicology study as well as writing of the manuscript; Jon R. Inglefield and Michael J. Lacy contributed to the immunogenicity analysis; C. Steven Godin contributed toxicological evaluation. The content of this manuscript was approved by all authors who take responsibility for the overall integrity and accuracy of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This project was funded in whole or in part with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under Grant No. 5 U01 AI078169-02.
