Abstract
A recombinant protective antigen (rPA) anthrax vaccine candidate (rPA7909) was developed as a next-generation vaccine indicated for postexposure prophylaxis of disease resulting from suspected or confirmed
Introduction
Anthrax is caused by the gram-positive, spore-forming bacterium
Following a deliberate bioterrorist contamination of letters with anthrax spores, which were then distributed by the US Postal Service in 2001, 21 cases of anthrax were identified (11 had confirmed inhalational anthrax and 11 [7 confirmed and 4 suspected] had cutaneous anthrax); 5 deaths due to inhalational anthrax were reported.
9,10
The Federal Select Agent Program has classified
Novel anthrax vaccines are being developed to improve upon the currently licensed anthrax vaccine. These improvements include fewer immunizations and faster time to protection. rPA7909 is a recombinant protective antigen (rPA) anthrax vaccine adjuvanted with the immunostimulatory oligonucleotide CpG 7909 and developed for postexposure prophylaxis against anthrax. The vaccine is a sterile, lyophilized formulation that, upon reconstitution, has the appearance of a white to off-white suspension. The final reconstituted drug product contains 75 μg rPA, 750 μg aluminum (as Alhydrogel adjuvant), and 250 μg CpG 7909 in 0.5 mL (full dose) with other excipients.
CpG oligodeoxynucleotides (ODNs) are potent immunostimulants.
12
CpG ODNs activate antigen-presenting cells and B cells by binding to Toll-like receptor 9, precipitating a cascade of events resulting in the secretion of T-helper 1-type cytokines and chemokines.
13,14
CpG 7909 was initially developed by Coley Pharmaceuticals (Wellesley, MA, USA).
15
The molecule has been extensively characterized both in preclinical studies and in the clinic. Studies in guinea pigs and in non-human primates reported a correlation between anthrax lethal toxin neutralizing antibody levels and survival in animals vaccinated with Anthrax Vaccine Adsorbed (AVA) adjuvanted with CpG 7909 and challenged with
To support the Investigational New Drug Application and entry into clinical studies, preclinical assessments were required to determine whether the proposed route of administration and number of doses of rPA7909 would be reasonably safe in humans. Assessments were designed to identify any potential systemic and organ-specific toxicity. The approach used for the rPA7909 safety evaluation followed current regulatory guideline concepts for nonclinical safety assessments of vaccines and adjuvants. 20,21 Here, we report a toxicology study to investigate the local tolerance and potential local and systemic toxic effects induced by repeated injections of the lyophilized rPA7909 vaccine and reversibility of toxic effects over a period of 2 weeks after the last injection.
Materials and Methods
All studies were conducted at Noble Life Sciences, Inc (Sykesville, Maryland) in compliance with Good Laboratory Practices. The Noble Life Sciences Institutional Animal Care and Use Committee approved the use of animals in this study. The work was performed in compliance with the Animal Welfare Act and followed the principles outlined in the National Research Council’s Guide for the Care and Use of Laboratory Animals. 22 Sprague Dawley rats were selected for the study because they have been used successfully in previous studies with anthrax vaccine and CpG 7909 and have shown to be able to develop a strong immune response to vaccination. Further, the rat model was selected because this species is an acceptable animal model per current guidelines for nonclinical vaccine safety studies. 20
Animals
Male and female Sprague Dawley rats were purchased from Harlan Laboratories, Inc (Frederick, Maryland) and were used for the general toxicity and immunogenicity of the rPA7909-vaccinated candidate. One hundred twenty rats (equal numbers of males and females) were acclimatized/quarantined for 1 week. Rats were 11 to 12 weeks old upon initiation of dosing and weighed from 228 to 257 (males) and 185 to 211 g (females). Animals were housed individually in polycarbonate shoebox cages with wire lids with 7090 Teklad sani-chips certified hardwood bedding and provided with Certified Global Harlan Teklad Laboratory 2016C Diet and fresh water ad libitum throughout the study.
General procedures for animal care and housing were in accordance with the Guide for Care and Use of Laboratory Animals. 22 Male and female animals were housed in separate rooms. All animals were housed in environmentally controlled rooms with at least 10 air changes per hour. Temperature was maintained at 18°C to 26°C and relative humidity was maintained at 47% (20%; males) and 53% (29%; females) with a 12-hour light/dark cycle.
Test and Control Articles
The lyophilized rPA7909 vaccine test article and diluent were supplied by Emergent BioSolutions Inc (Gaithersburg, Maryland). Following reconstitution with diluent, the rPA7909 vaccine test article contained 75 μg rPA, 750 μg aluminum, and 25 μg CpG7909 per 0.5 mL. A solution without rPA containing the same concentrations of adjuvants and excipients as the reconstituted rPA7909 vaccine test article was prepared for use as adjuvant control material. All materials were maintained at 2°C to 8°C and prepared on the day prior to each of the 3 dose days. The vaccine antigens, adjuvant, and diluent used to formulate the test articles were manufactured according to Current Good Manufacturing Practices. Dose analysis was conducted to confirm that the identity, strength, and purity met established test article acceptance criteria.
Vaccination
Three groups of 40 Sprague Dawley rats per group (20/sex/group) received 3 doses of negative control (diluent, group 1), adjuvant control (excipients + CpG 7909 + Alhydrogel adjuvant, group 2), or test article (lyophilized rPA7909 test article reconstituted in negative control article, group 3; Table 1).
Study Group Descriptions.

Abbreviations: F, female; M, male.
a Administered as 2 intramuscular injections (0.25 mL in the right hind limb and 0.25 mL in the left hind limb) at each dose day.
Animals were administered vaccine or control articles via IM route of 0.5 mL (0.25 mL/site) in each hind limb on days 1, 15, and 29. Twenty animals from each group (10/sex/group) were euthanized on days 31/32 to evaluate possible acute effects after the last dose. The remaining animals in each group (10/sex/group, excluding any deaths prior to scheduled termination) were euthanized on days 43/44 to evaluate potential recovery of any acute effects and the immune response.
The IM route was selected as this was the proposed human clinical route and 0.5 mL the proposed volume. The number of administrations (3) and dosing volume (0.25 mL at 2 different sites) in this study was expected to exceed that expected in the clinic, with the duration of dosing being shorter than the expected clinical interval between doses to exaggerate any possibility of toxicity.
Evaluation of Clinical Signs of Toxicity
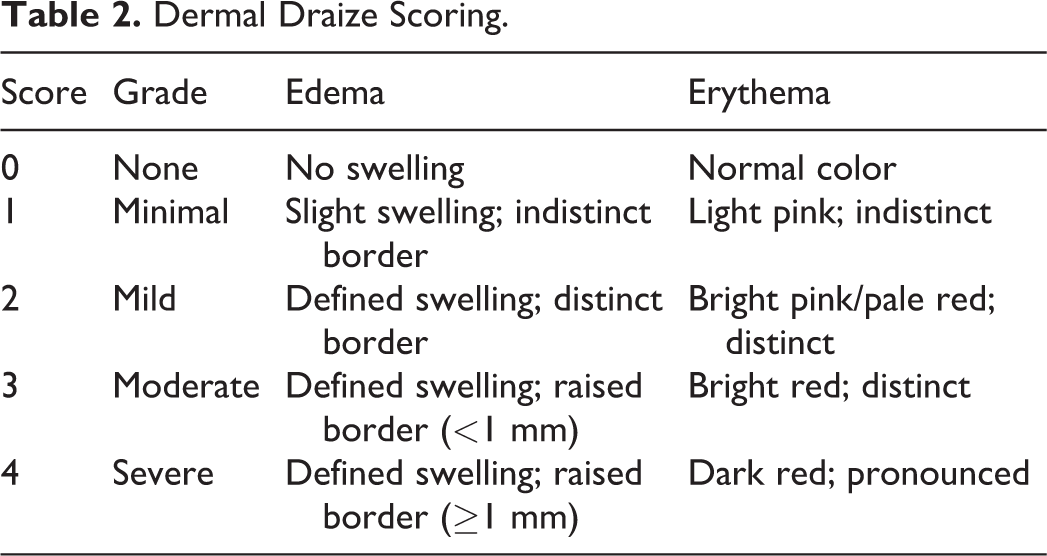
Cage-side observations were conducted daily prior to noon to evaluate animal general health and to qualitative assess water/food intake and urine/feces output. Physical examinations included evaluation of appearance and behavior and palpations of body surfaces and internal organs. Test and control injection sites were observed for erythema/eschar and/or edema and graded according to the modified method of Draize (Table 2). 23 Ophthalmic examinations of each animal included 1 slit-lamp biomicroscopy with transillumination examination, 1 retroillumination examination, and 1 indirect biomicroscopy examination on day −7 and prior to its scheduled termination on either day 30 (those animals scheduled for necropsy on days 31/32) or day 42 (those animals scheduled for necropsy on days 43/44). Tropicamide was used as a mydriatic agent.
Dermal Draize Scoring.
Clinical Pathology
Nonterminal blood samples were collected on day 1 prior to vaccination, and terminal blood samples were collected on days 31/32 and days 43/44 for hematology, coagulation, serum chemistry, and immunogenicity analyses. Prior to nonterminal blood collection, animals were anesthetized via inhalation of 3% isoflurane and blood was collected from the vena cava or tail vein. Prior to terminal blood collection, animals were anesthetized via IM injection of 100 mg/kg ketamine and 10 mg/kg xylazine and blood was collected from the vena cava. Food was removed from the cages of animals between 4 and 12 hours prior to blood collection on days 1, 31/32, and 43/44 for the subset 10 animals used for serum chemistry, serology, and C-reactive protein (CRP) analyses.
Hematology analysis was done in-house by Noble Life Sciences (Sykesville, MD, USA) using the Hemavet 950 FS Hematology Analyzer. Analyses of fibrinogen, prothrombin time, and activated partial thromboplastin time coagulation parameters were performed by Antech Diagnostics GLP (Morrisville, North Carolina) using a Sysmex CA1500 Coagulation system. Serum chemistry parameters were determined by Antech Diagnostics GLP using a Beckman/Olympus 640e clinical chemistry analyzer. The CRP analysis was performed by Antech Diagnostics GLP using a rat CRP enzyme-linked immunosorbent assay (ELISA).
Euthanasia and Postmortem Evaluation
Animals scheduled for acute necropsy on days 31/32 and recovery necropsy on days 43/44 were anesthetized with an IM injection of ketamine/xylazine anesthesia and exsanguinated. Death was confirmed by visual observation and cutting of the abdominal aorta. All animals euthanized on days 31/32 and 43/44 were subjected to a full gross necropsy, which included examination of external surface of the body, the injection sites, all orifices, and the cranial, thoracic, and abdominal cavities and their contents. Weights were obtained for the following organs: adrenal glands, brain, heart kidney, liver, lungs, spleen, testes/ovaries, iliac lymph nodes, and thymus. Paired organs were weighed together. Toxicity was assessed based on the results of clinical observations, physical examinations, body weights, ophthalmology, clinical pathology analysis (hematology, coagulation, and serum chemistry), organ weights, and macroscopic and microscopic pathology evaluation. Inflammation was assessed by evaluating levels of CRP along with changes observed in clinical chemistry, hematology, and coagulation end points.
Evaluation of Immunogenicity (Anti-PA Immunoglobulin Subtype G ELISA)
To verify vaccine administration, immune response was evaluated by determining antiprotective antigen immunoglobulin subtype G (IgG) levels measured by ELISA in the rPA7909-vaccinated group compared to the control groups at the time of termination. Purified rPA was used as the solid-phase immobilized antigen and an enzyme-conjugated anti-gamma chain secondary antibody was used as the reporter or signal system. The assay end point was reported as the mean serum concentration of anti-PA-specific IgG (µg/mL). Anti-PA IgG content of rat sera was calculated using binding curves obtained for each sample using a log-log linear fit in reference to a standard curve.
Statistical Analysis
Group means and standard deviations were determined, at a minimum, for injection site observations, body and organ weights, organ to body weight ratios, CRP, and coagulation, hematology, and serum chemistry parameters using Microsoft Excel. Descriptive statistics were presented by sex. Statistical testing was performed using 1-way analysis of variance (ANOVA) to compare the means of each group and gender conducted at the 0.050 level of significance. If either the normality or the equal variance test failed, then the parametric analysis was continued using the nonparametric Kruskal-Wallis ANOVA on rank-transformed data. For parametric data, if the ANOVA indicated statistical significance among experimental groups, then the Dunnett
Results
Mortality
Two rPA7909-vaccinated female animals were found dead during the study (one each on days 1 and 4). Based on the necropsy examination, both deaths were attributed to hemorrhaging likely due to vena cava blood collection. The deaths were not considered to be related to rPA7909 vaccination. No other deaths were observed during the study.
Clinical Observations
In the diluent control animals, findings were limited to discharge from one or both eyes in 1 male on day 22 and slight edema in the left hind limb of 1 female on day 10. One adjuvant control male had discharge from the nose on days 15 and 22, and another adjuvant control male had minimal discharge from the nose on day 22. One rPA7909-vaccinated male had slight brownish buildup in the right eye recorded on day 36. These findings were considered incidental and not related to test article administration. There were no additional findings or observations noted during daily cage-side observations or physical examinations during the study.
Body Weight, Body Weight Change
No significant effects on body weight or body weight change were observed during the study that could be attributed to rPA7909 administration. The mean body weight gain from day 1 to days 43/44 necropsy of adjuvant control females was decreased compared to the diluent control females (Figure 1).
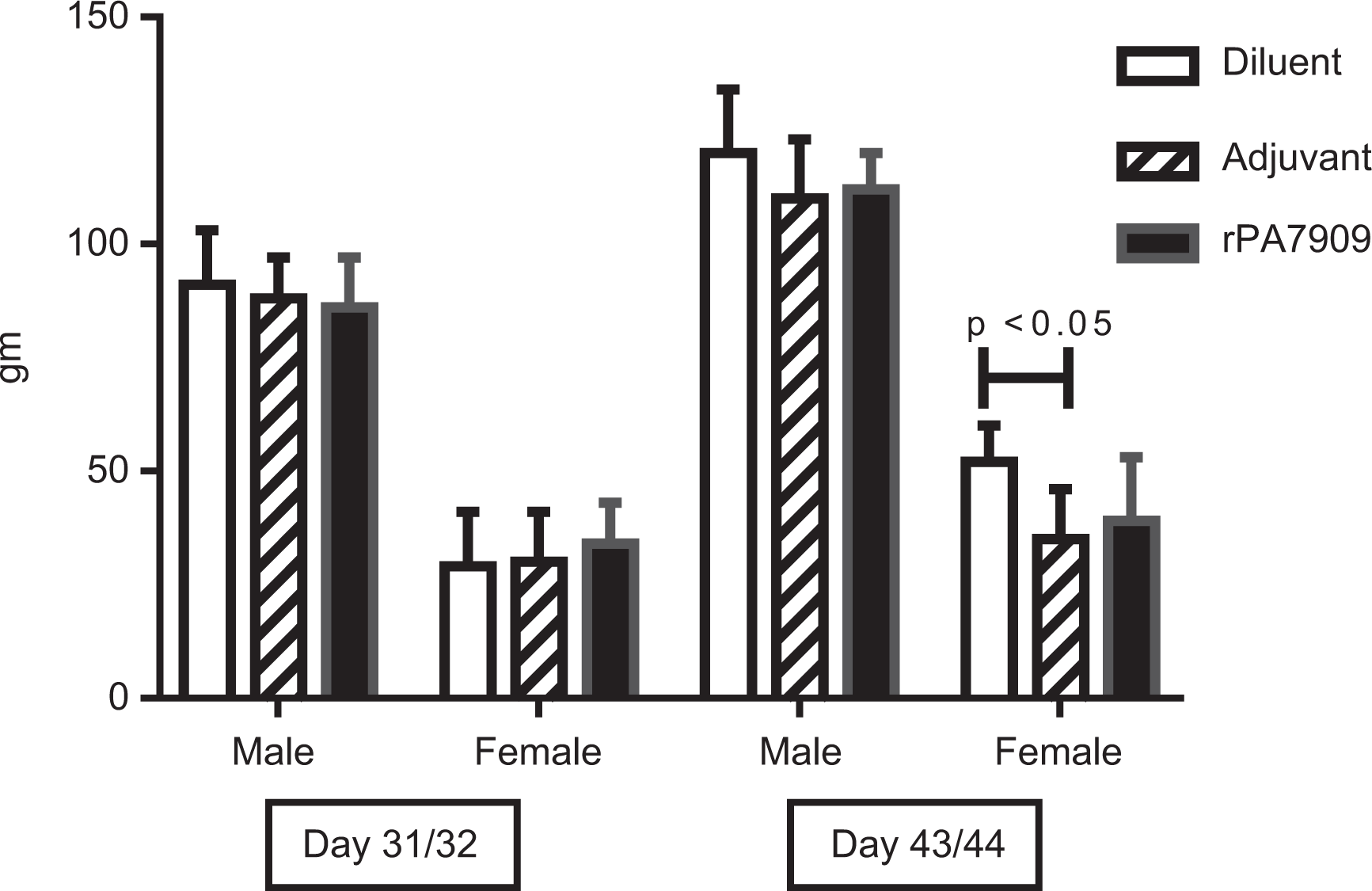
Body weight change. Change in body weight was determined based on the difference in baseline body weights measured on quarantine release and body weights measured on days 31/32 and 43/44 for both acute and recovery necropsy groups, respectively. The mean body weight gain from quarantine release to the days 43/44 necropsy of adjuvant control females was decreased compared to the diluent control females.
Local Reactogenicity
Several animals exhibited very slight or well-defined erythema at the injection sites, which was comparable in all groups (Table 3). Very slight or slight edema was also noted in some adjuvant control and rPA7909-vaccinated animals. The severity was comparable in both groups; incidence was slightly increased in the rPA7909-vaccinated animals receiving the test article, with a low incidence overall. However, none of these findings were significant, and signs of erythema and edema resolved to baseline levels within 4 days after each vaccination. Nodules noted at the injection sites of some adjuvant control and rPA7909-vaccinated animals were of minimal to moderate severity and of low incidence. These changes were considered an expected observation after IM injection of this type of vaccine material and were not considered adverse.
Dermal Draize Observations.a
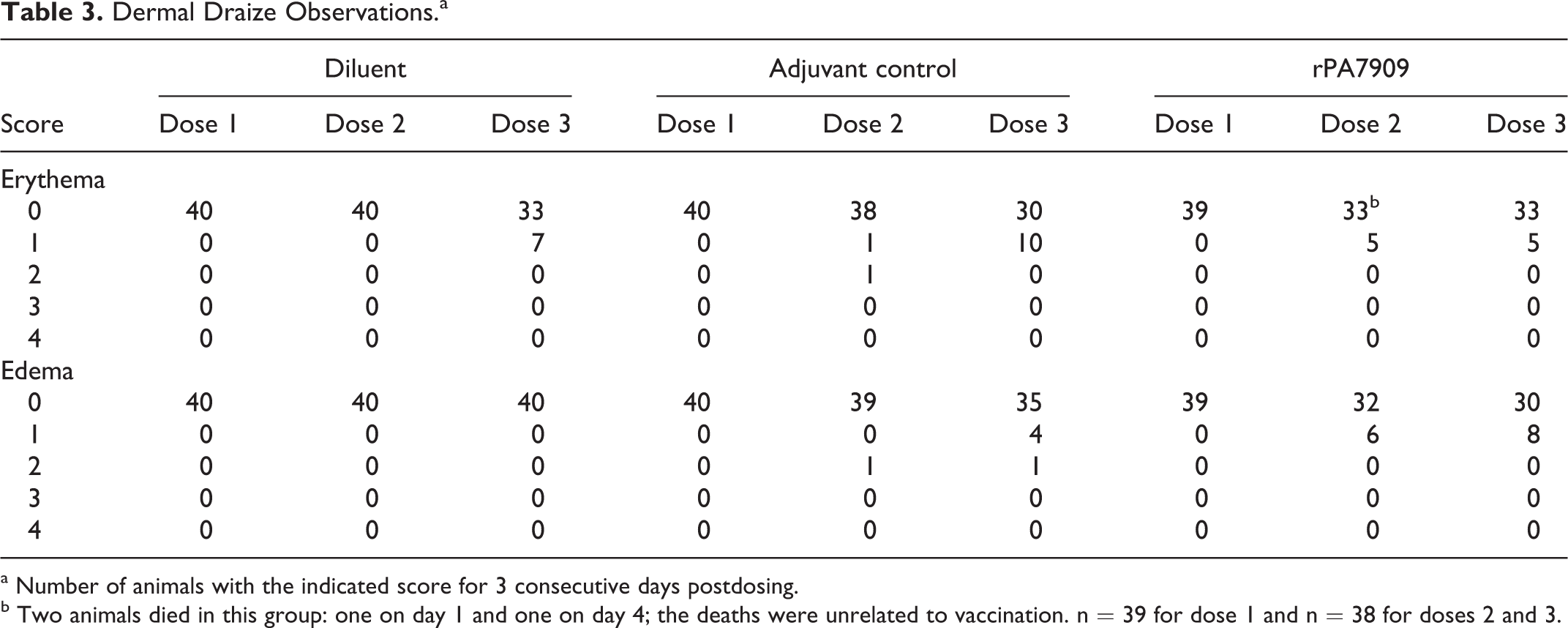
a Number of animals with the indicated score for 3 consecutive days postdosing.
b Two animals died in this group: one on day 1 and one on day 4; the deaths were unrelated to vaccination. n = 39 for dose 1 and n = 38 for doses 2 and 3.
Ophthalmology
Several animals included on study had corneal dystrophy in one or both eyes at all intervals. However, it was subtle for most animals. The presence of corneal dystrophy is common in Sprague Dawley rats, was observed in animals from all groups, and was not considered to be relevant to evaluation of the test article. There were no ophthalmic findings attributable to test article administration. A summary of ophthalmology data is presented in Table 4.
Summary of Ophthalmic Findings.
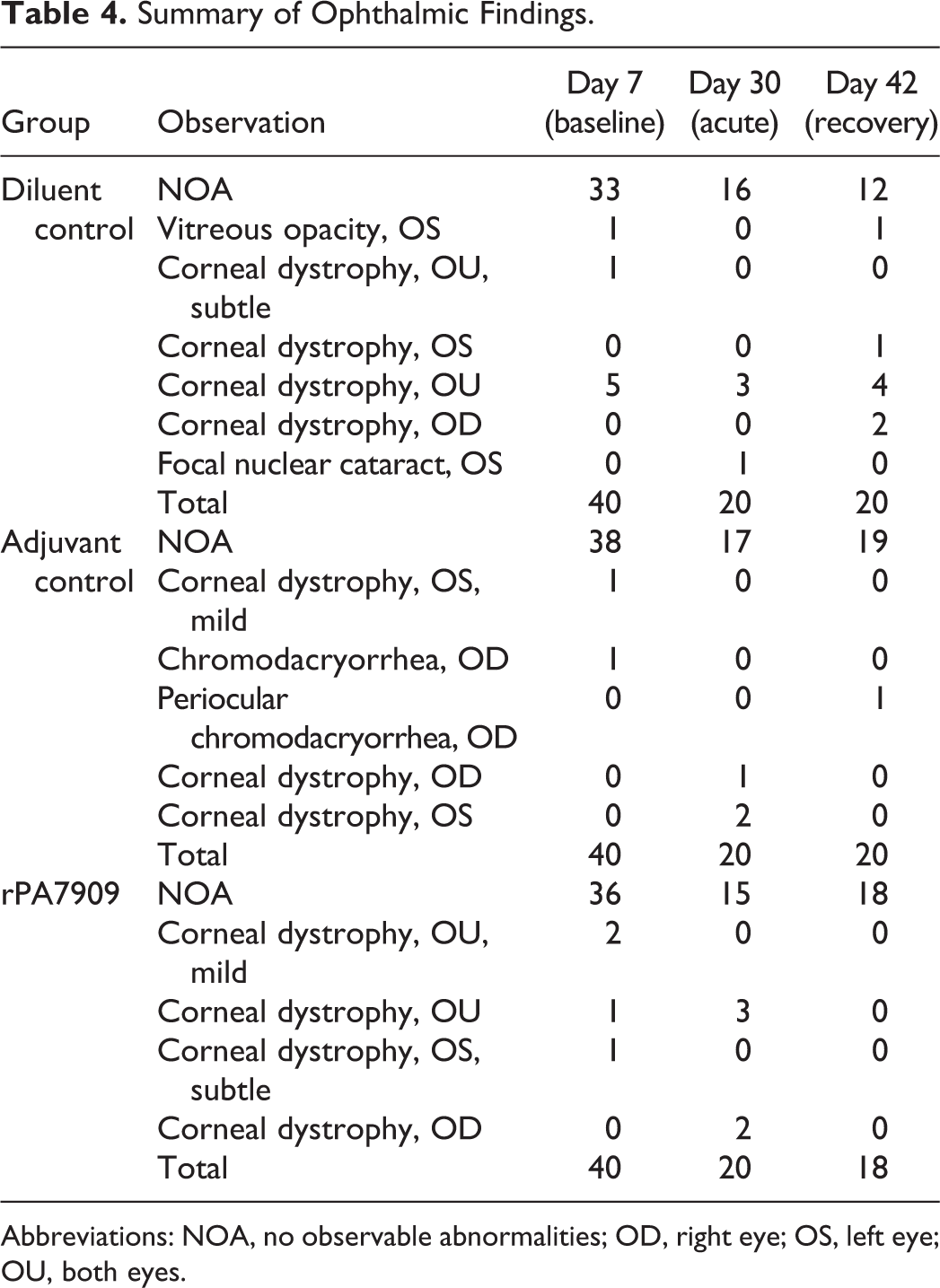
Abbreviations: NOA, no observable abnormalities; OD, right eye; OS, left eye; OU, both eyes.
Clinical Pathology
Hematology
The hematology data were generally unremarkable and similar among the groups at each collection interval, with one exception. Changes in the hematology data, likely associated with the administration of adjuvant control and rPA7909 vaccine, included slightly higher mean total leukocyte and lymphocyte counts on days 31/32 in adjuvant control males and rPA7909-vaccinated males and females relative to diluent controls (Table 5). By recovery days 43/44, the values for total leukocyte and lymphocyte counts were comparable among all groups.
White Blood Cell and Lymphocyte Counts.a,b
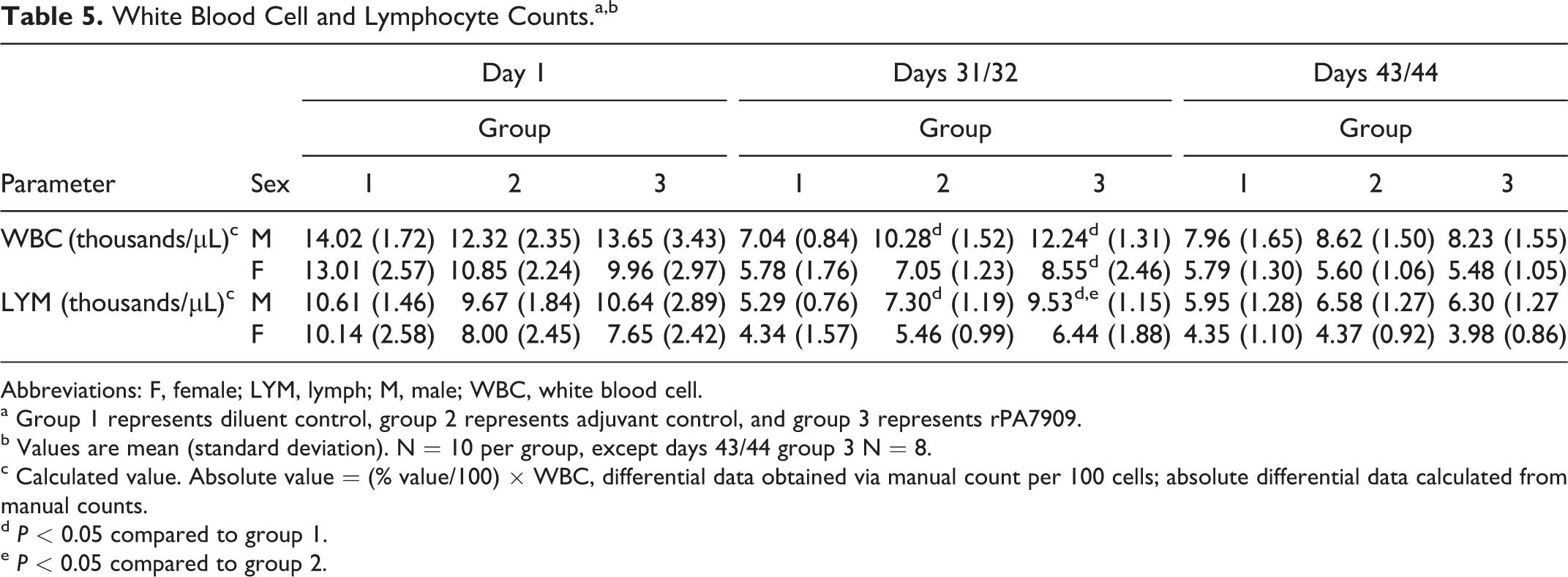
Abbreviations: F, female; LYM, lymph; M, male; WBC, white blood cell.
a Group 1 represents diluent control, group 2 represents adjuvant control, and group 3 represents rPA7909.
b Values are mean (standard deviation). N = 10 per group, except days 43/44 group 3 N = 8.
c Calculated value. Absolute value = (% value/100) × WBC, differential data obtained via manual count per 100 cells; absolute differential data calculated from manual counts.
d
e
Serum chemistry and CRP
Subtle, but significant, differences were observed in a few clinical chemistry parameters, suggesting an effect of the administration of the rPA7909 vaccine (Table 6). Although statistically different, the values for total protein, albumin, (calculated) globulin, and albumin to globulin ratio were generally unremarkable and not biologically relevant. Mean CRP values in rPA7909-vaccinated males were higher on days 31/32 relative to diluent and adjuvant controls. By recovery days 43/44, the CRP values were comparable between all groups, with the exception of one diluent control male animal that exhibited a CRP value >500 mg/dL; the mechanism behind this elevation in this male was not apparent from the data examined.
Total Protein, Albumin, (Calculated) Globulin, A/G Ratio, Fibrinogen, and CRP.a,b
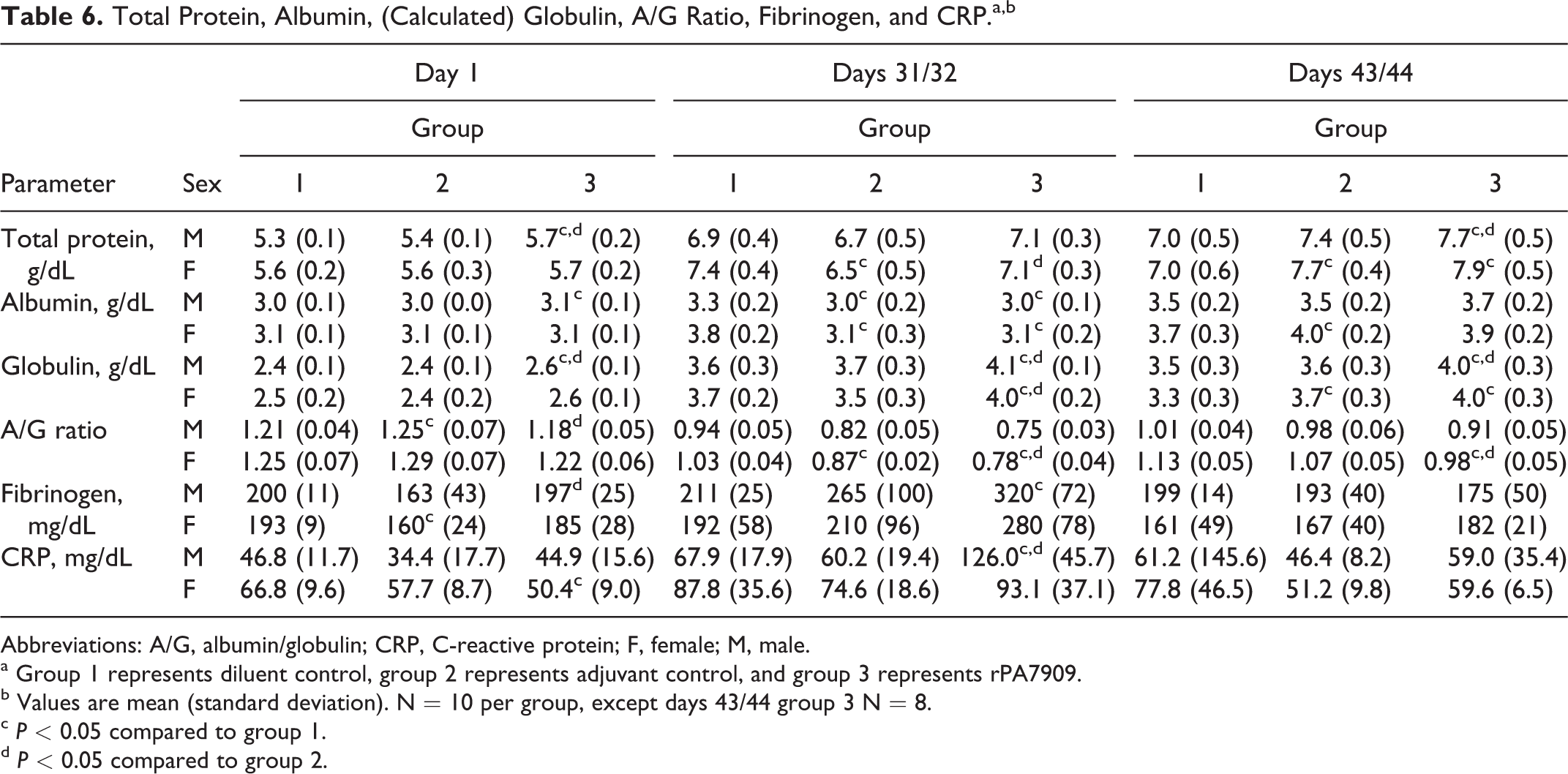
Abbreviations: A/G, albumin/globulin; CRP, C-reactive protein; F, female; M, male.
a Group 1 represents diluent control, group 2 represents adjuvant control, and group 3 represents rPA7909.
b Values are mean (standard deviation). N = 10 per group, except days 43/44 group 3 N = 8.
c
d
The mechanism behind the lower albumin values in the adjuvant control animals on days 31/32 was not obvious from the data examined (body weights, organ weights, and clinical pathology data). The persistently elevated globulin values in rPA7909-vaccinated males and females (and to a lesser extent for adjuvant control males and females) at recovery correlated with the inflammation noted at the injection sites as well as with the lymphoid hyperplasia observed microscopically in these groups. The slightly higher fibrinogen, globulin, and CRP values in rPA7909 animals on days 31/32 were consistent with acute inflammation associated with the final IM injection of rPA7909 vaccine on day 29. The lack of noteworthy differences in fibrinogen and CRP concentrations (acute-phase proteins) at recovery and the magnitude of the higher globulin concentrations in rPA7909-vaccinated animals at days 31/32 and 43/44 were suggestive of an immunologic response.
The remaining serum chemistry data were generally unremarkable and similar across the groups, except for a few statistically significant differences that were considered incidental to the administration of either adjuvant control or (rPA7909) vaccine because of the small magnitude of the difference and the lack of biologic or pathologic meaning.
Coagulation
The coagulation results were generally unremarkable and similar among the groups, but the mean fibrinogen concentrations were slightly higher in rPA7909-vaccinated males and females on days 31/32, suggesting a mild inflammatory response to IM injection of the vaccine on day 29 (Table 6). The mean fibrinogen values were unremarkable and comparable among all groups at days 43/44.
Organ Weights
At days 31/32 necropsy, all of the rPA7909-vaccinated animals right and left iliac lymph nodes were significantly increased in weight compared to diluent controls, while left iliac lymph node and male right iliac lymph node of adjuvant control group female were significantly increased compared to the corresponding lymph node from the diluent control group (Table 7). The increased iliac lymph node weights were attributed microscopically to lymphoid hyperplasia, macrophage aggregates in the paracortex, and in the rPA7909 group, medullary cord plasmacytosis. The absolute spleen weight of rPA7909 group female was significantly increased compared to that of the diluent and adjuvant control group. There were no histomorphologic correlates for the increased rPA7909 group spleen weights.
Organ Weights.a,b
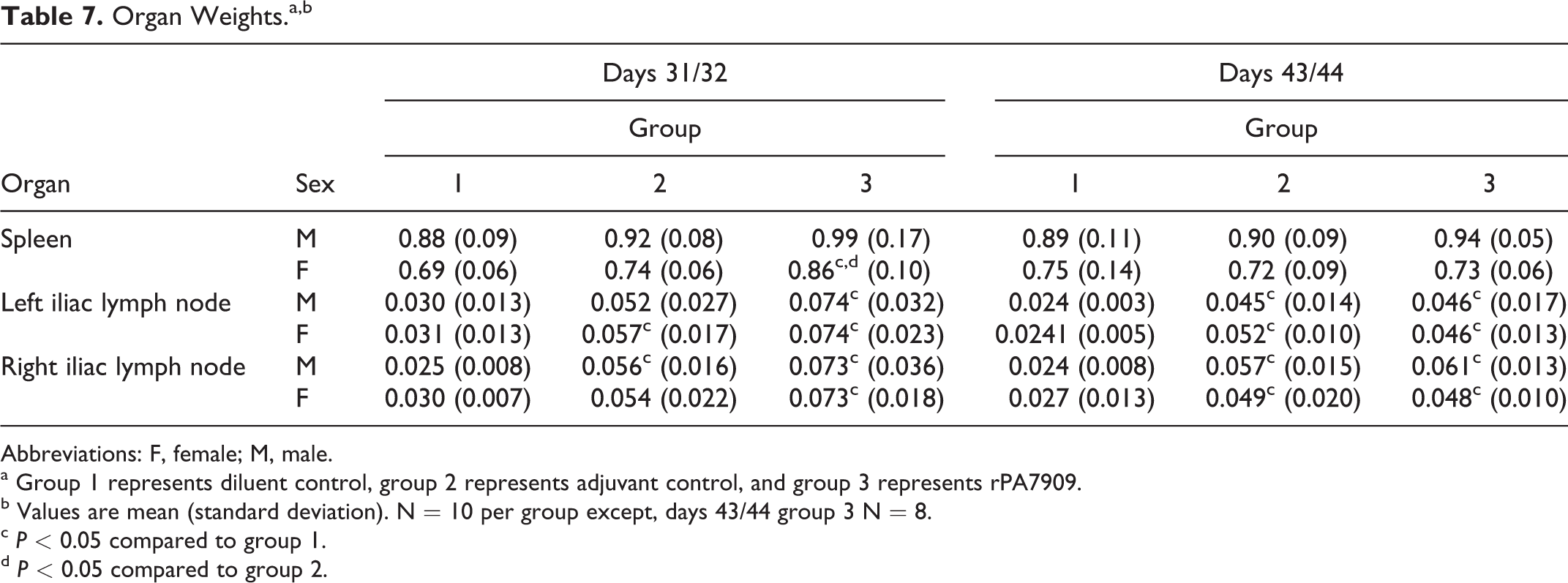
Abbreviations: F, female; M, male.
a Group 1 represents diluent control, group 2 represents adjuvant control, and group 3 represents rPA7909.
b Values are mean (standard deviation). N = 10 per group except, days 43/44 group 3 N = 8.
c
d
At days 43/44, the right and left iliac lymph node absolute weights of all the adjuvant control and rPA7909 groups were significantly increased compared to that of the corresponding diluent control (Table 7). The increased iliac lymph node weights were attributed microscopically to lymphoid hyperplasia.
Additionally, the adjuvant control group female relative (percent body weight) adrenal weight was significantly increased compared to the diluent control group female relative adrenal weight, and the adjuvant control group male absolute kidney weight was significantly decreased compared to the diluent control group male kidney weight (data not shown). These changes in the adrenal and kidney weights were attributable to individual animal variation and were not considered treatment related, since they lacked microscopic correlates, weren’t observed in both genders, lacked similar changes in the corresponding absolute (adrenal) or relative (kidney) weights, and were not observed in the adjuvanted vaccine group.
Anatomical Pathology
At both necropsy intervals, the primary macroscopic observations consisted of enlarged left and/or right iliac lymph nodes, which were correlated microscopically with lymphoid hyperplasia and/or plasmacytosis of the medullary cords. At both necropsy intervals, enlarged lymph nodes were seen more commonly in the rPA7909 group and slightly less so in adjuvant control group animals.
At the days 43/44 necropsy interval, an adjuvant control group female had a raised white mass on the caudal pole of the left kidney, which was correlated microscopically with unilateral nephroblastoma, a spontaneously occurring neoplasm of the rat kidney. This neoplasm was considered to be incidental.
All other findings, at both necropsy intervals, many of which related to distended or dilated uterine horns, were attributed to estrus cycling and considered to be incidental background findings, with the exception of red foci observed in the lung of a diluent control group female, which was correlated with acute hemorrhage and attributed to blood collection methods just prior to necropsy.
Histopathology
At days 31/32 and 43/44 necropsy intervals, changes were observed at the right and left injection sites, around the left and right sciatic nerves, and in the draining left and right iliac lymph nodes, for animals in all groups. For all injection sites, mononuclear cell inflammation, consisting primarily of macrophages and slightly fewer lymphocytes and plasma cells replacing often degenerated myofibers, occurred deep in the muscle and surrounding subcutis, although, occasionally, inflammation was also observed in the dermis. Central necrosis of the larger inflammatory cell aggregates was recorded as necrosis of the muscle and subcutis. Degeneration/regeneration of the myofibers was common in all groups around the areas of inflammation. Regeneration of myofibers predominated at days 43/44 compared to the degeneration more evident at days 31/32. Mononuclear cell infiltrates around the sciatic nerves were considered to be an extension of the inflammatory process at the IM injection site in some animals. At days 43/44, fewer sciatic nerves from any group had mononuclear cell infiltrates observed perineurally, also indicating dissipation of injection site changes with a 2-week recovery. The increased size of some of the iliac lymph nodes observed at necropsy and the increased iliac lymph node weights at days 31/32 were attributed microscopically to lymphoid hyperplasia, macrophage aggregates in the paracortex, and in rPA7909 group, medullary cord plasmacytosis. The increased size of some of the iliac lymph nodes observed at necropsy as well as the increased iliac lymph node weights at days 43/44 were attributed microscopically to lymphoid hyperplasia. Overall, the left and right iliac lymph node findings were similar at both necropsy intervals, although plasmacytosis of medullary cords was absent, with an additional 2-week recovery.
Immunogenicity
Anti-PA IgG levels were below the lower limit of quantitation in all diluent control and adjuvant control animals on days 1, 31/32, and 43/44 (data not shown). rPA7909-vaccinated animals also had anti-PA IgG levels below the lower limit of quantitation for the assay on day 1, as expected, but had measurable levels of anti-PA IgG in specimens collected on days 31/32 (average 1,703 µg/mL) and days 43/44 (average 3,331 µg/mL), indicative of a robust immune response to vaccination (Figure 2).

Quantification of anti-PA IgG levels in rats given rPA7909 vaccine (group 3). For each time point, plotted are the individual rat serum anti-PA IgG concentrations (•) as well as the group geometric mean concentrations ( ) and 95% confidence intervals. IgG indicates immunoglobulin G.
) and 95% confidence intervals. IgG indicates immunoglobulin G.
Discussion
Existing licensed prophylactic vaccines are heterogeneous products and may include inactivated bacterial and viral vaccines, live attenuated vaccines, recombinant proteins, polysaccharide and conjugated vaccines, and DNA vaccines. They often contain immunostimulatory components represented by various adjuvants. The adjuvants include inorganic salts (such as alum), lipid A fractions of lipopolysaccharide (eg, monophosphoryl lipid), saponin-based mixtures (eg, QS-21), oligonucleotides (eg, CpG sequences), and oil emulsions (eg, MF59). Publication on animal safety studies for vaccine and novel adjuvants is relatively rare since vaccines are generally safe and well tolerated.
The purpose of this study was to investigate the local tolerance and potential local and systemic toxic effects induced by multiple IM injections of the rPA7909-vaccinated candidate and to evaluate the reversibility of any toxic effects over a period of 2 weeks after the last injection. Comparisons were made with the diluent control and adjuvant control (excipients + CpG 7909 + Alhydrogel adjuvant) articles. A rat model was selected for the current study since rats were used in previous safety and immunogenicity evaluates of CpG 7909 and CpG 7909 adjuvanted vaccines. 24,25 Typically, only a single species is required for vaccine safety evaluations. 20
The rPA7909 vaccine was well tolerated and produced local and systemic effects that were attributed to the inflammatory process from the injected material. The animals that received the rPA7909 vaccine produced the expected antigen-specific antibody response that correlated with the macroscopic findings of enlarged lymph nodes in conjunction with lymphoid hyperplasia, macrophage aggregates in the paracortex, and the microscopic finding of medullary cord plasmacytosis. Increased weight and/or hyperplasia of draining lymph nodes near the vaccination site is a common finding in nonclinical vaccine safety studies. 26 Hematology changes returned to diluent control levels at the end of the 2-week recovery period. The elevated clinical chemistry parameters (total protein and globulin) that were attributed to the rPA7909 vaccine administration persisted at the end of the 2-week recovery period. Serum CRP level was included as a biomarker for acute inflammatory reaction in this study. However, measurement of serum levels of α1-acid glycoprotein, α2 macroglobulin, and haptoglobin as biomarkers for acute inflammatory reaction should also be considered when using rats as the toxicology species. 27,28
The results for rPA7909 were consistent with similar findings in the safety evaluation of single and repeated IM administrations of CpG 7909 containing MAGE-A3 cancer immunotherapeutic in rabbits and cynomolgus monkeys, which included enlargements of iliac, popliteal, axillary, and inguinal lymph nodes and increased incidence or severity of mononuclear inflammatory cell infiltrates observed in injected muscles.
29
Local reactogenicity associated with active inflammation at the injection sites is a well-recognized response in vaccines containing Alhydrogel adjuvant.
30,31
Other previously conducted preclinical safety studies also showed favorable safety profile of vaccines adjuvanted with CpG 7909 and enabled a number of clinical trials evaluating safety, immunogenicity, efficacy of malaria,
32
-34
hepatitis B,
35
pneumococcal conjugate,
36,37
influenza,
38
cytomegalovirus,
39
and anthrax.
17,18
Three doses of vaccine containing CpG ODN given at various intervals (eg, 0, 2, and 4 or 0, 4, and 8 or 0, 8, and 24 or 0, 8, and 36 weeks) used in human vaccine trials yielded a relatively benign toxicity profile limited to fatigue, rigors, myalgia and pyrexia, and mild injection site reactions.
40
The target population for the rPA7909 vaccine will primarily be healthy individuals who have been exposed or suspected to have been exposed to
Footnotes
Acknowledgments
The authors wish to thank Leslie Hopson and the staff at Noble Life Sciences for the technical execution of the toxicology assessments, John Inglefield for performance of the ELISA, and Annie Jones (Magus Strategic Communications) for editorial assistance with manuscript preparation.
Author Contributions
V. Savransky contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. M. Lacy contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. B. Ionin contributed to conception and design, contributed to interpretation, and critically revised the manuscript. M. Skiadopoulos contributed to conception and design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. J. Shearer contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are current or former employees of Emergent BioSolutions Inc, the developer of the rPA 7909 and AVA adjuvanted with CpG 7909 vaccine candidates.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This study was funded by BARDA Contract HHSO100201000059C.
