Abstract
Central nervous system (CNS)-targeted products are an important category of pediatric pharmaceuticals. In view of the significant postnatal maturation of the CNS, juvenile animal studies (JAS) are performed to support pediatric development of these new medicines. In this project, the design and results of juvenile toxicity studies from 15 drug compounds for the treatment of neurologic or psychiatric conditions were analyzed. Studies were conducted mostly in rats; sometimes in addition in dogs and monkeys. The study design of the pivotal JAS was variable, even for compounds with a similar therapeutic indication. Age of the juvenile animals was not consistently related to the starting age of the intended patient population. Of 15 compounds analyzed, 6 JAS detected more severe toxicities and 6 JAS evidenced novel CNS effects compared to their adult counterparts. The effects of CNS on acoustic startle and learning and memory were observed at high dosages. Reversibility was tested in most cases and revealed some small effects that were retained or only uncovered after termination of treatment. The interpretation of the relevance of these findings was often hampered by the lack of matching end points in the adult studies or inappropriate study designs. Detailed clinical observation and motor activity measures were the most powerful end points to detect juvenile CNS effects. The need for more detailed behavioral examinations in JAS, for example, on learning and memory, should, therefore, be decided upon on a case-by-case basis, based on specific concerns in order to avoid overloading the studies.
Introduction
With an increasing requirement for specific safety assessment of medicines for the pediatric population, regulatory authorities have published guidelines to be consulted in case of development of pharmaceuticals for use in the pediatric age range. 1,2 In various regions of the world, this has led to a new regulatory branch of safety testing, for example, nonclinical safety testing in juvenile animals. 3,4 Initially, a specific regulatory framework regarding the design of toxicology studies to support pediatric development was lacking. It was not clear what type of outcomes could be expected from the studies in juveniles, that is, a difference in targets of toxicity and/or a difference in sensitivity in juveniles compared to adults. Such differences might be due to organ/system immaturity or effects on organ/system development. 3,4
One area of concern is the central nervous system (CNS), as children might benefit from treatment through improvement of their neurological condition, but at the same time could suffer from adverse effects on the CNS, such as sedation or effects on cognition.
A high number of pharmaceuticals targeting the CNS are on the market. Because of signals that psychotropic drugs can have negative effects on cognition, it was discussed that for these CNS-targeted drugs relevant information could be gained from safety studies in young animals. 4 These studies may contribute to predicting whether the exposure to a drug in pediatric patients can lead to developmental and/or functional abnormalities and to identifying toxicological signals and/or possible biomarkers that are not seen in adults. 2,5,6
It is worthwhile to systematically evaluate the outcome of these studies. The aim of this European Union (EU) analysis is to evaluate juvenile animal safety data with respect to study design, end points (particularly those expected to detect neurological side effects), and results and to learn from juvenile toxicity approaches for CNS compounds. The outcome of this analysis may also be important for the discussion of the International Council for Harmonisation (ICH) regarding guidance on nonclinical safety testing in support of the development of pediatric medicines. 7
Methods
Product Selection
The virtual archives of the Pediatric Committee (PDCO) at the European Medicines Agency (EMA) were searched for data regarding compounds with a Pediatric Investigation Plan (PIP) in therapeutic areas of neurology, psychiatry, anesthesiology, and pain. Only those directly targeting the CNS (ie, CNS-targeted compounds) for which juvenile animal studies (JAS) were performed and/or requested for were considered for inclusion in the analysis. Data were available for PIPs which were agreed between 2008 and 2015, that is, between the coming into force of the EU Paediatric Regulation and the date of the analysis. 8 Complete data packages consisted of PIP summary reports for initial and modification procedures, the investigator brochure, and toxicology study reports both in juvenile and adult animals. The regulatory impact was determined by checking the PDCO assessment reports, and the decisions of the Committee for Medicinal Products for Human Use (CHMP) reflected in the Summary of Product Characteristics (SmPC).
A total of 83 compounds were retrieved from the EMA database in therapeutic areas of neurology, psychiatry, anesthesiology, and pain. Of these, 58 compounds were CNS-targeted and only 25 of these compounds JAS reports were available. Only 15 compounds corresponding adult nonclinical safety data were available and only those compounds were selected for further analysis. Indications targeted by compounds with available complete data packages were De la Tourette and restless legs syndrome (Tou), narcolepsy (Nar), schizophrenia (Schi), attention-deficit hyperactivity disorder (Adhd), depressive and anxiety disorders (Dep), and epilepsy and bipolar I disorders (Epi; Table 1). These compounds are anonymized to protect commercial interests using an alphanumerical code related to the target indication with the abbreviation as indicated.
Characterization of the Compounds Assessed (Pharmacology, Therapeutic Condition, and Age Range).

aCompounds are anonymized in association with the intended indication: Adhd, attention-deficit hyperactivity disorder; Dep, depressive and anxiety disorders; Epi, epilepsy and bipolar I disorders; Nar, narcolepsy; Schi, schizophrenia; Tou, de la Tourette and restless legs syndrome.
Data Evaluation
Once all available data were retrieved, an evaluation form was developed and used to make a full assessment of the juvenile animal data for the selected compounds. The following parameters were summarized and assessed for every compound and each juvenile toxicity study performed: clinical indication, youngest age of the patient population, intended human dose and exposure at this dose, species selection, age of animals at study start, study duration, dosing regimen in animal study, timing of CNS end points in relation to time of dosing as well as day of the study, animal exposure end points (area under the curve for 0-24 hours [AUC0-24]; Cmax, at different time points, if available), and main treatment-related effects. For each compound, an adult repeated-dose study in the same species was also assessed to directly compare the exposures and effects with the respective JAS, at similar doses and treatment duration, where available. In addition, the overall pharmacotoxicological data package was used to interpret the findings in relation to each compound’s mechanism of action (pharmacology) and to the effects in adult animals. Finally, regulatory evaluation and resulting inclusion of the information into the regulatory documentation by the EMA’s CHMP were assessed.
Results
Species Selection and Number of Animals
Of the 15 compounds rats were used in JAS with 14 out of these. A second species was used in 7 cases (dogs in 6 cases [Adhd2, Epi1, Epi2, Epi4, Epi5, Schi3], Rhesus monkeys in one case [Tou1]). For the remaining compound (Epi3), the dog was the single species used. For one case (Epi6) in which initially mice had been used, the mouse data were discarded in view of excipient toxicity (see later), and rat data were generated using another excipient.
The number of animals in the pivotal studies in rats and nonrodents is given in Table 2. The number of rats used per study varied between 250 and 760, and in one case up to 1,120 pups. Rats used for toxicokinetic (TK) assessment are included in these numbers. For nonrodents, the number of animals used per study is also variable and varied between 47 and 140.
Study Design Elements of the Juvenile Animal Studies Assessed.
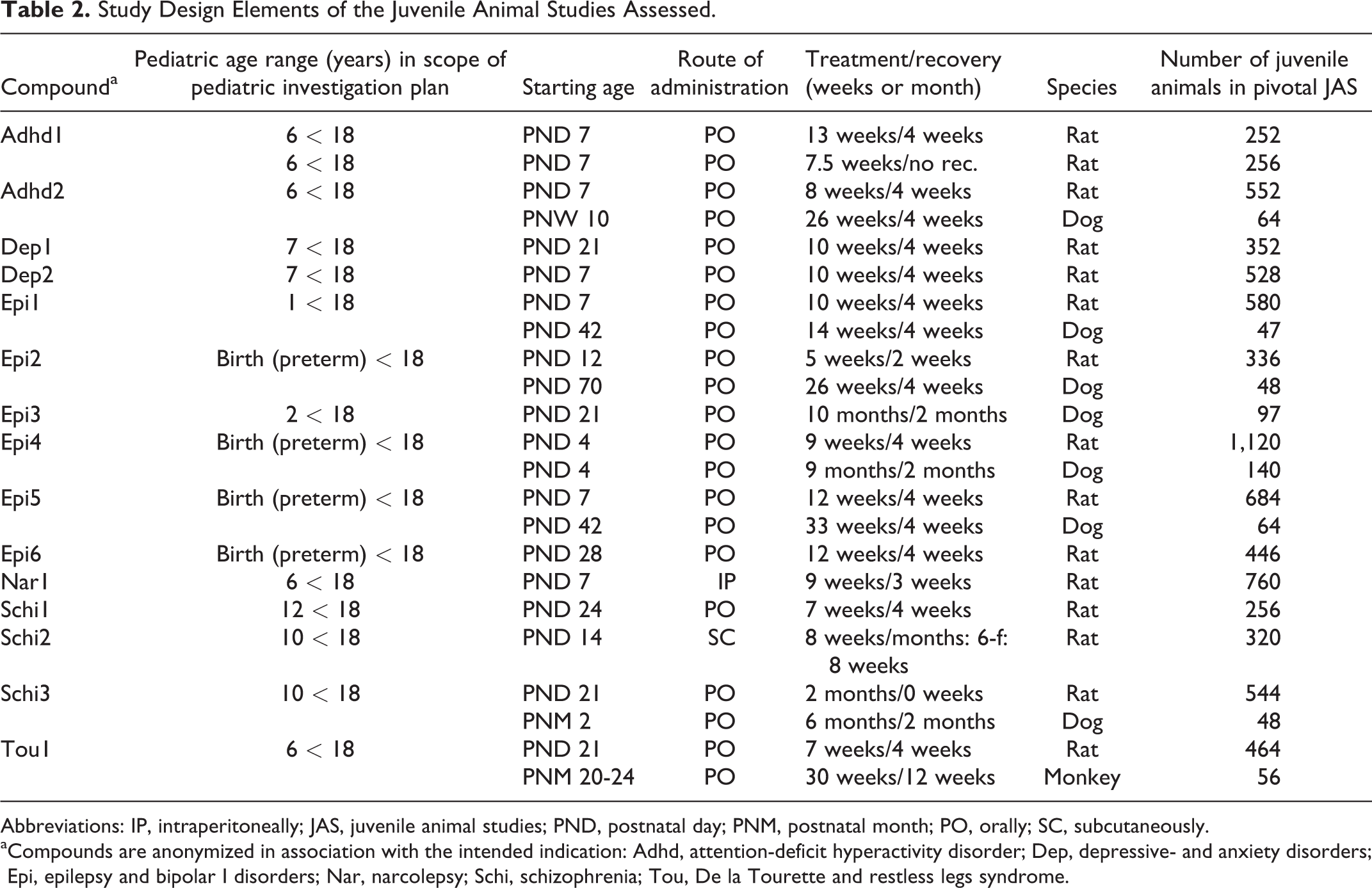
Abbreviations: IP, intraperitoneally; JAS, juvenile animal studies; PND, postnatal day; PNM, postnatal month; PO, orally; SC, subcutaneously.
aCompounds are anonymized in association with the intended indication: Adhd, attention-deficit hyperactivity disorder; Dep, depressive- and anxiety disorders; Epi, epilepsy and bipolar I disorders; Nar, narcolepsy; Schi, schizophrenia; Tou, De la Tourette and restless legs syndrome.
Starting Age
When comparing the age of the animals at study start (Table 2) with the youngest age of the intended patient population, a major heterogeneity was observed. The 6 antiepileptic compounds, most being developed to treat neonates including preterm newborns, showed significant differences in starting age. In rats, it ranged from postnatal day (PND) 4 to PND 28, in dogs from PND 4 to PND 70.
For a product for the treatment of Tourette syndrome, which can be diagnosed in patients from 6 years of age, rats PND 21 were studied. For the 2 antidepressants, with a similar patient age range, the rat JAS started at PND 7 and PND 21. Although the youngest target age for Adhd and Nar was also 6 years, the JAS for these products all started at PND 7 in rats. The 3 compounds to treat schizoaffective disorders and developed for children aged from 10 to 12 years had ages at study start in rats varying from PND 14 to PND 24.
Duration of Treatment and Recovery
The duration of treatment varied from 5 to 13 weeks in rats. For Epi2, the treatment lasted only up to PND 46, whereas for Epi6 it lasted until PND 112. Most studies lasted until PND 70 with a variance up to PND 91. This is in-line with guideline recommendations to treat animals up to reaching adulthood when the target organs have a long development period 2,3 and also takes into account that most of the compounds are intended to be used chronically.
Route and Frequency of Administration
In nearly all studies, daily administration by the oral route was applied in-line with the intended clinical route. For Nar1, the oral route was tried initially but did not result in a clinically relevant level of exposure. Therefore, the intraperitoneal (IP) route was applied in the juvenile rat study to enhance exposure. For Schi2, the subcutaneous route was used in toxicity studies as relevant to the clinical route (sublingual). For Nar1, Epi4, and Dep1, compounds were administered twice a day (BID administration). Of note, when oral BID administration started in very young rats (PND 4, Epi4), higher than usual mortality was noted mainly before weaning in all groups, including controls (19, 9, 20, or 69 unscheduled deaths at 0, 150, 300, or 600 mg/kg, respectively).
Use of Excipients
In the early studies with Epi6, propylene glycol was used as excipient (5 mL/kg of body weight), which led to mortality at all doses including controls in the juvenile animals of choice (in this case, mice). These mouse studies were discarded in the evaluation because of this confounding mortality. Later on, rats were tested with hydroxymethylcellulose to aid a stable suspension and these data were used for the current evaluation.
Exposure: Comparison Between Juvenile and Adult Animals
All cases were selected to have adequate determination of systemic exposure in the pivotal JAS. Maximum concentration (Cmax) was given and the AUC during 24 hours (AUC0-24) had been calculated by the sponsor. In a majority of the JAS, blood sampling to determine plasma levels of the test article was performed on 2 occasions: close after study start and at the end of the study. Sometimes, an additional sampling was included around the time of weaning. We have chosen to focus on the AUC0-24 as a measure to compare exposure between juvenile and adult animals, although it is acknowledged that in some cases, the Cmax would represent a more relevant measure of exposure in relation to specific CNS effects. For some compounds, exposure in adult animals had not been determined, and in those cases the “adult exposure data” were taken from the JAS latest time point, usually from young adult animals. Brain concentrations were not determined in any of the reviewed studies.
In Table 3 (third column), the detailed outcome of the comparison between juvenile and adult animals is presented for all individual compounds. Generally, in rats, when treatment was started preweaning (PNDs 7-14), exposure was initially high, followed by a decrease around weaning time or even up to PND 35, while increasing again at later ages, although not as high as in the preweaning period (eg, Epi1 and Epi5). Differences in exposure were usually only 1.5- to 3-fold. When administration started around or just after the time of weaning, the exposure was usually found to increase toward the end of the treatment period (PNDs 70-98).
Comparison of Exposure and Toxicity in Juvenile and Adult Animals in the CNS Compounds That Were Assessed.

Abbreviations: AUC, area under the curve; BID, twice daily; CNS, central nervous system; f, female; LOAEL, lowest-observed-adverse-effect level; H&E, hematoxylin and eosin; JAS, juvenile animal studies; m, male; Nar, narcolepsy; NOAEL, no-observed-adverse-effect-level; NOEL, no-observed-effect-level; PND, postnatal day; PO, orally; TK, toxicokinetic.
aCompounds are anonymized in association with the intended indication: Adhd, attention-deficit hyperactivity disorder; Dep, depressive- and anxiety disorders; Epi, epilepsy and bipolar I disorders; Nar, narcolepsy; Schi, schizophrenia; Tou, De la Tourette and restless legs syndrome.
In dogs, exposure comparisons were possible for 5 compounds, as in one case no data in adult dogs were available. In 4 cases, the exposure was 2- to 3-fold higher in juvenile animals (starting age PNDs 4-70), whereas in one case the exposure was similar in juvenile and adult animals. In the one case of monkeys being used, there was similar exposure in juvenile (2-4 years of age) and adult animals.
Comparison of General Toxicity Between Juvenile and Adult Animals
Toxicity end points such as mortality, detailed clinical observations (see later), body weight and food consumption, clinical chemistry, and behavioral observations were included, as well as macroscopic examinations and common histopathology at necropsy. Reproductive end points (such as sexual development, hormone analysis, and reproductive assessments) were included in the majority of rat studies.
Supplementary end points such as bone measurements, ophthalmoscopy, and extended neuropathology were less frequently reported in the study reports. In Table 3 (fourth column), a conclusion on the comparison of exposure and toxicity in juvenile versus adult animals is presented for each individual compound. Differences between juvenile and adult rats in terms of increased severity of the effects were observed for 7 compounds (Tou1, Epi2, Epi3, Epi4, Epi5, Epi6, Schi3) and not for 8 other compounds (Nar1, Epi1, Adhd1, Adhd2, Schi1, Schi2, Dep1, Dep2).
In 8 cases, no novel toxicity was observed in juvenile animals as compared to adults. Effects seen in juvenile rats only, but secondary to pharmacological effects on growth, such as decreased food consumption and body weight (eg, delayed sexual maturation, femur length), were not considered as “new toxicity.” In 6 cases, novel CNS toxicities were observed, which are discussed later on.
In one case, Epi3, novel toxicity consisted of a bone effect, that is, reduced mineral bone content, area, and density of the femur and lumbar vertebrae in juvenile dogs. However, to our knowledge, bone mineral density had not been assessed in adult dogs, hence making a direct comparison impossible.
In all cases, the results of the JAS have been reported in the SmPC (with the exception of one case where the marketing authorization was suspended). The review of PDCO assessment reports did not identify any significant amendments of the clinical pediatric development caused by JAS outcomes for the products analyzed.
Screening of CNS Effects in Juvenile Animals
Examinations intended to detect CNS pharmacological or toxicological side effects were performed in most cases during the treatment period, usually also during the off-treatment “reversibility” period, and in one case, behavioral examinations were done during the reversibility period only.
The following qualitative observations were generally recorded in the rat studies: Clinical observations intended to detect evidence of ill-health and/or mortality, cage observations performed twice or sometimes once daily. Detailed clinical observations intended to detect pharmacological or toxicological side effects. Often, these observations were timed in relation to time of administration: prior to administration, just after administration, a couple of hours later on, and sometimes at the end of the afternoon. They generally consisted of cage observations, handling observations, and observations of reaction to manipulation. Physical examinations performed routinely once a week.
Detailed clinical observations have also been recorded in all the nonrodent studies. In addition, in some of the nonrodent studies, operant behavior was assessed.
Behavioral examinations in the rat studies, which were usually assessed a few weeks after start of treatment (sometimes just before daily dosing), generally consisted of: Open field observation/functional observatory battery (FOB), or modified Irwin Auditory (or acoustic) startle reflex testing Learning and memory testing: Water maze tests were used for these purposes.
9
These tests were conducted at ages >PND 50, usually once during treatment, and once during off-treatment. Morris
10
or Cincinnati
11
water maze tests were most commonly used (Tou1, Adhd1, Adhd2, Epi1, Epi5, Epi6, Schi2, Schi2, Dep1, and Dep2), while in 2 cases (Nar1, Schi3), the type of water maze was not specified. In 2 cases, other water mazes were used, including Biel
12
or multiple T-maze
13
(Epi2, Epi4). Motor activity monitoring
In juvenile rats, various extended neuropathology evaluations (eg, colorations intended to detect pharmacologic or neurotoxic effects on neurons, apoptosis, myelinization) of peripheral nervous system and CNS were performed in 8 of 15 cases.
Effects on CNS Measures Monitored During JAS
An overview of the CNS end points included in each study and their respective outcomes is presented in Tables 4 (rats) and 5 (nonrodents). The effects of CNS detected through detailed clinical observations were noted for nearly all compounds during the treatment phase (eg, salivation, increased/decreased activity, stereotypic behavior, convulsions, abnormal gait, ptosis, tenseness, vocalization, aggressivity) but not during the off-treatment (reversibility) phase.
Effects on CNS Measures in Rodents.a

Abbreviations: BID, twice daily; Clin obs, clinical observations; +, CNS treatment effects; D, study day; f, female; FOB, functional observational battery; HD, high dose; IP, intraperitoneally; m, male; MD, mid dose; ND, no data/not done; −, no difference from controls; Phys ex, physical examinations; PND, postnatal day; PNW, postnatal week; SC, subcutaneously; W, study week.
aFor Epi3, no rodent studies were available.
bCompounds are anonymized in association with the intended indication: Adhd, attention-deficit hyperactivity disorder; Dep, depressive and anxiety disorders; Epi, epilepsy and bipolar I disorders; Nar, narcolepsy; Schi, schizophrenia; Tou, De la Tourette and restless legs syndrome.
Effects on CNS Measures in Nonrodents.a

Abbreviations: Adhd, attention-deficit hyperactivity disorder; CNS, central nervous system; Dep, depressive and anxiety disorders; Epi, epilepsy and bipolar I disorders; ND, no data/not done; +, CNS treatment effects; −, no difference from controls; Nar, narcolepsy; Schi, schizophrenia.
aFor Adhd1, Dep1, Dep2, Epi6, Nar1, Schi1, and Schi2, no nonrodent studies were available.
In 7 of the 8 nonrodent JAS, effects were observed through detailed clinical observations. For operant behavior, only effects were observed in the monkey study with Tou1. The operant test battery aimed at assessing learning, short-term memory, color discrimination, and motivation. Other compounds, such as Adhd2, Epi1, and Epi5, did not induce any effect in these end points.
During FOB/Irwin testing, no effects were noted for any compound during the treatment period. An effect on rotarod performance, most probably related to an increased motor activity, was recorded for Schi2 during the off-treatment period (not tested during the treatment phase).
Acoustic startle reflex (Table 4, third column) was affected by Tou1, Adhd2, and Epi1 as well as by Dep1. Effects were mainly seen at high dosages. Statistical difference from controls was seen in startle reflex data for Epi4 during the off-treatment period, but not during the treatment period.
Effects on water maze were reported for Tou1, Epi2, Schi1 (only females), and Schi3. For Epi2, learning alterations and memory deficit (eg, worse water maze performance) were noted during the treatment period. The 4 studies with water maze findings also included a water maze assessment during the off-treatment period, but none revealed adverse alterations during the off-treatment period.
Motor activity was affected by Tou1 (increased), Adhd1 (increased), Adhd2 (decreased), Epi4 (decreased), Schi3 (increased), Dep1 (increased) during the treatment phase while not being an end point for Schi2, and Dep2 during the treatment phase. Motor activity differences from control were still seen during the off-treatment phase with Tou1 (decreased motor activity, possibly a rebound effect) and Schi3 (increase persisted in females). Motor activity was measured during the off-treatment period only and found to be different from control data for Schi2 (increased signs of recovery in males, but not in females, also observed in adult study). Extended neuropathological histopathology investigations did not show signs of neurotoxicity for any of the compounds.
Comparison of the behavioral profiles between juvenile and adult animals is impossible as behavioral end points used in JAS were not addressed during the adult toxicity studies. Nevertheless, detailed clinical observations were recorded for different ages of animals during the JAS, and the evolution of these observations from the youngest age to the young adult rats was informative in this respect.
Age-related novel CNS effects were observed for 6 compounds: Tou1, Epi4, Adhd1, Dep2, Schi1, and Schi2. For example, in male rats of the rat JAS for Tou1 underactivity predominated, while in females initially tending to be underactive, overactive behavior increased as treatment progressed. Increased motor activity was seen afterward, during the off-treatment period, possibly a rebound effect. This was not described for adult animals. In the monkey study with Tou1, different behavioral effects were observed in juvenile animals compared to adult monkeys, as evidenced by toxicity (severe agitation resulting in wounds and bruises at the 2 higher doses) occurring in adult animals only. In juvenile monkeys, alteration of the ability to learn complex behavioral tasks was noted. These tests were not performed in adult monkeys. Similarly, the rat JAS for Adhd1 revealed behavioral effects at the end of the dosing period (increased agitation, tenseness, vocalization, aggressiveness, followed by decreased activity/no movement/fixed gaze starting on PND 80), whereas no similar clinical CNS signs were reported either in younger rats or in the adult rat study.
Juvenile animals exposed to Tou1, Epi3, and Epi5 appeared more sensitive to the pharmacological activity or CNS clinical signs. In the case of Epi2, Epi4, and Schi3, mortality, possibly linked to exaggerated pharmacology, was observed in young animals at an exposure that was not lethal in their adult counterparts.
Regarding Epi 6, the pivotal rat JAS (PND 28) showed a similar toxicity profile as observed in adult animals. However, in the dose-range finding study performed in younger animals (PND 21), notwithstanding similar exposures, more severe clinical signs inducing preterminal euthanasia were reported in these animals.
Discussion
The present article summarizes the review of the juvenile safety studies conducted with 15 CNS-targeted compounds. The selection of compounds was conditioned by the availability of adequate data in the EMA archives. We have chosen to focus on compounds with a direct CNS target and to leave out compounds that only have an indirect effect on CNS conditions (eg, compounds with a target in the immune system for the treatment of multiple sclerosis). The studies performed for the 15 compounds reveal a number of issues with regard to the study design.
Species Selection
In the case of antiepileptic drugs, the choice of 2 species for 4 of the 6 compounds is not fully understood. In general, neither the choice of the species nor the number of species used for toxicity testing in juvenile animals was justified in the available documents. The choice of a second species might have been related to the outcome of some rodent studies in previous programs for pediatric antiepileptics, that is, with an observed decrease in brain weight leading to further questions regarding the relevance of this effect (Epi1).
Regulatory guidelines indicate that usually one (rodent) species would be sufficient, and the rat is seen as the default species. Selection of an adequate animal species, no matter whether rodent or nonrodent, should be scientifically justified whenever possible. This is especially important considering the limitations of JAS in nonrodents for CNS drugs: difficulty to treat very young animals up to adulthood, difficult access to appropriate tests, and ethical and practical challenges of obtaining and treating preweaning nonhuman primates.
Starting Age
There was notable variety of the age of juvenile rats and dogs at study start, which did not always correspond to the youngest age of the intended patient population. Of particular interest was Epi6, where the single pivotal JAS was conducted in 28-day-old rats, which is too old to represent the stage of CNS development in the intended population (preterm neonates) as a 28-day-old rat would rather correspond to a 4-year-old in terms of brain development. 14 In addition, apparent increased severity of toxicity was observed in rats given the drug from PND 21 compared to rats that were given the drug from PND 28 and older, indicating higher sensitivity in younger rats, based on the severity of the clinical signs leading to preterminal euthanasia occurring at lower doses in the youngest rats. Consequently, the critical window of vulnerability of the developing brain and any deleterious effects of Epi6 associated therewith could have been missed. Therefore, it remains uncertain to what extent the observation of a similar toxicity profile in the pivotal JAS compared to adult animals can be extrapolated to the youngest children.
On the contrary, PND 7 as starting age for the juvenile rat studies with Adhd1, Adhd2, Dep2, and Nar1 with a target population from 6 or 7 years of age onward is considered too young. Before PND 10, the rat is comparable to a human fetus or a preterm neonate. Therefore, safety signals in such young rats may be irrelevant for humans based on differences in developmental stages. 15 In the case of Adhd1, the clinical relevance of the novel behavioral effects at the end of the juvenile rat study (increased agitation, tenseness, vocalization, and aggressiveness, followed by decreased activity/no movement/fixed gaze) is questionable in view of the immature rat brain at the start of the study compared to the target age in children. However, these findings might be related to the neuropharmacological properties of Adhd1, which may potentially affect neurodevelopmental processes in early phases of brain development, leading to changes in neuropharmacological response and behavioral outcome at later stages in life. Hence, these findings may also reflect a particularly sensitive period in these young adult animals and be relevant for adolescent patients.
In very young rats, excessive systemic and brain exposure related to physiological and metabolic immaturity may induce severe toxicities of little clinical relevance. For Dep2, the very early dosing could indeed be an explanation for the sharp increase in systemic exposure at the study start. Dosing on PND 7 led to 37-fold higher AUC values compared to PND 76. At the lowest dose, changes in body weight, physical development, and pituitary weight were observed in males, and a no-observed-adverse-effect-level (NOAEL) could not be determined. The effects did not reverse during the recovery period and are novel toxicities compared to previously available nonclinical data for this compound. However, at the lowest dose in the JAS, at PND 7, the exposure (in terms of AUC) was still twice the exposure at the NOAEL of the comparative adult rat study, and more than 700 times the AUC at the clinical dose (5 mg/d in 6-17-year-old patients), putting the relevance of these novel findings into question.
In conclusion, whereas in regulatory guidelines, the starting point is that the age range of juvenile animals should cover the window of brain development corresponding to the children’s treatment period, this principle was not respected in several of the reviewed JAS and as a result, the clinical relevance of these study results is questionable. Animal age at study start should be scientifically justified to improve the clinical relevance of the studies. The choice of 2 species and the variety in starting age might be related to the time period in which the studies were conducted, before US Food and Drug Administration (FDA) and EU guidelines 2,3 had been published and before the EU PIP procedure came into force in 2007.
Study Duration
In spite of the variability in the starting age of JAS mentioned above, the study duration was more harmonized as most studies covered the full developmental trajectory of the brain with a majority of the rat studies including dosing up to at least PND 70. Short-term studies focusing on juveniles as compared to adult periods of sensitivity to the pharmacological or toxicological effects are not very common. This may be due to the assumption that CNS behavioral effects could be delayed or long lasting even after cessation of compound administration throughout brain development up to adulthood.
In one case, severe CNS effects were seen in rats aged PND 80 and older. These effects were not detected in younger or adult rats pointing out to the need to cover brain development up to adulthood.
Number of Animals Used
The number of animals used in this type of safety studies is an important concern, in relation to the information gained. In this analysis, the number of juvenile animals on study is quite variable and in some cases very high. The highest numbers were observed in complex studies which covered multiple end points requiring additional cohorts, particularly pup’s reproductive capacity in rat JAS.
In an effort to reduce the number of animals needed, study designs should be streamlined and investigated end points scientifically justified and reduced to the most relevant ones. In addition, when studies are started with rats preweaning, additional pretest animals are needed in order to avoid confounding factors (eg, genetics, group-size during fostering) between groups susceptible to interfere with study results. Usually, groups are built from as many litters as are present in the study (ICH, S5 16 ). It has, however, to be established whether these principles should be applied stringently or if a more flexible approach may be adopted in the interest of reducing the number of animals used for these studies.
Use of Excipients
Usually, excipients used in JAS are similar to those used in the adult formulations. Nevertheless, they may have a different safety profile in very young animals. The use of propylene glycol as vehicle for Epi6 led to excessive mortality in young mice. These data are in-line with the results of a juvenile (PND 7) mouse study, 17 which showed that propylene glycol produces ethanol-like apoptotic neurodegeneration in the developing mouse CNS starting at doses of 2 mL/kg. At 1 mL/kg, no apoptotic effects were observed. At 10 mL/kg, it induced mortality (unpublished results). Although excipient toxicities may seriously hamper the interpretation of the effects caused by the active ingredient, using a clinical formulation for the JAS can be useful to assess the safety of the clinical formulation and the need for a specific pediatric formulation.
Toxicokinetics
Toxicokinetic data are important to understand whether the test results occurred in a similar exposure range as the intended patient exposure. Moreover, they are critical to assess the impact of changes in systemic exposure across age ranges on the observed toxicities. Therefore, it is desirable to measure TK shortly after the study start and at the end of the study. An additional sampling around the time of weaning could also be useful in some cases. It should be kept in mind, however, that age-related changes in pharmacokinetics cannot be directly extrapolated from animals to humans. Estimation of kinetic properties in humans rather stems from knowledge of developmental changes of enzymes and organs influencing pharmacokinetics in humans, as well as modeling. The need to evaluate brain exposure should be properly addressed taking into account potential differences in brain penetration between species, although it has to be admitted that for none of the compounds included brain exposure was determined.
CNS Effects in Juvenile Animals
Effects have been observed during detailed clinical observations, but generally no effects were detected during specific behavioral tests, such as the FOB. The timing of the detailed observations in relation to dosing (before and several times following dosing) might explain why these effects were not observed during the other behavioral examinations (usually performed once/twice at the end of the study; often just before dosing and during the off-treatment period). This apparent discrepancy may also be due to the kind of end points evaluated. During detailed clinical observations, a broad spectrum of mostly qualitative end points was evaluated. During behavioral examinations, specific end points were measured. As effects were rarely seen just before dosing (unless at high doses) or during the off-treatment period, results indicate that most effects are related to drug exposure and are not long lasting.
Effects on acoustic startle and water maze tests were only seen with a low number of compounds, mostly at mid and/or high doses. These effects were often not consistent between doses and sexes. In one case, a change in auditory startle response was detected only during the off-treatment period. In another case, learning deficits were detected in groups previously treated with the high dose, whereas memory task errors were noted at all doses, which were not, however, consistent regarding dose dependency or between sexes. This may indicate a low sensitivity of these tests. As these tests were not performed during adult toxicity studies, it is also not possible to determine whether these effects are specific to juvenile animals.
Effects on motor activity were seen in 6 of 12 compounds during the treatment period and in 3 cases during the off-treatment period. The effects observed during the off-treatment period are not easy to interpret. For Tou1, a decrease in motor activity might have been a rebound effect caused by the stimulation of motor activity during treatment. In the case of Schi2, off-treatment effects were observed on rotarod performance and on motor activity (high locomotor and rearing activity), at all doses and dose related. These effects tended to decrease in male but persisted in females until the end of the recovery period. As no tests were carried out during the treatment period, it is again difficult to interpret these effects. For Schi3, nondose related but statistically significant increases in motor activity were observed both in males and females at all doses tested during the treatment period. In males, the effect on motor activity resolved during the off-treatment period while in females the effect on motor activity persisted during the drug-free recovery period at the highest dose tested. Long-term effects of antipsychotics, such are tardive dyskinesia, are also well-known in humans, but their relevance for children will have to be confirmed by clinical data.
Impact of Ontogeny of Target Systems
Various receptors are being involved with these 15 compounds, including dopamine receptors (covering 5 compounds: Adhd2, Tou1, Schi1, Schi2, and Schi3), α2-adrenergic receptors (Adhd1), sodium channels (Epi1, Epi2, and Epi3), potassium channels (Epi6), AMPA glutamate receptor (Epi5), and histamine H3 receptors (Nar1). Other compounds have a less clearly defined receptor interaction.
There is a lot of published literature regarding the ontogeny of dopamine receptors in rats. D1 and D2 receptors follow similar developmental patterns, showing a peak between PND 28 and PND 42 in the caudate and putamen, and decreasing thereafter. 18,19 However, the picture is more complicated as dopamine also occurs in the ventral striatum. The pattern of maturation of the dopaminergic innervation is, however, less clear in the frontal cortex. 20 For other receptors and channels, identified literature 21 -23 was not very helpful to understand the relevance of the observations.
Overall, in the absence of a discussion by the sponsors on the findings in relation to the ontogeny of the receptors or the relationship between the observations in juveniles as compared to their effects in adults, an in-depth elaboration on the behavioral findings observed with the various dopaminergic (agonists and antagonists) or other compounds was not considered feasible within the remits of this study.
Lessons Learned With Regard to Screening of CNS Effects in Juvenile Animals
Detailed Clinical Observations
Behavioral observations, although only qualitative, are important because they may help to detect severe abnormalities, such as convulsions, aggressiveness, or catalepsy. As they are performed at several time points, including just before daily administration of the drug and during the off-treatment period, they allow to differentiate high-dose or secondary short-term pharmacological effects from long-term or delayed pharmacological effects and/or neurotoxicity. In most cases, these observations can also be bridged to the adult animal data.
To reach these objectives, it would suffice to perform detailed clinical observations at the time of the highest exposure (peak exposure) and shortly before the next dosing (trough level). The severity of the effects should be related to exposure data.
Behavioral Examinations (Rat Studies)
Quantitative instrumental behavioral examinations whenever tested (not all were evaluated for each drug) showed lower incidences of treatment effects (as compared to the detailed clinical observations), and as these tests were not performed during adult toxicity studies, it is difficult to determine whether the observed effects were specific to juvenile animals.
During FOB testing, treatment effects were detected in only 1 case of 11 studies. Compared to the high rate of positive effects detected during detailed clinical examinations, this seems to indicate a low sensitivity of the FOB test and endorses a recent similar observation for pesticides 24 . Considering that FOB is not quick or simple, the need to include them in JAS is thus questioned.
Treatment effects on acoustic startle and water maze tests were also seen in few cases, mostly at mid and/or high doses, often not consistently between doses and sexes. Of note, different maze tests were performed and their choice was not justified. They are all aimed at detection of learning and memory deficits, while testing for the brain networks that mediate higher cognitive functions need to include assessments of working memory, attention, long-term memory, and executive functions such as cognitive flexibility, not all being detected by one test. 24 If these tests are thought to be needed to assess the hazard for impacting cognitive function in children and adolescents, the most relevant test should be selected and justified.
During (loco)motor activity testing, treatment effects were more frequently detected, including during the off-treatment period. As hyperactivity and hypoactivity are easy to detect, it may sometimes reflect rebound effects and—as was shown in 2 studies—may also reflect long-term effects during the off-treatment period, it may be wise to measure motor activity in complement to detailed clinical observations.
The need for more detailed behavioral assessment should, therefore, be decided on a case-by-case basis, based on specific concerns, in order to avoid overloading the studies. On the other hand, more detailed evaluation during the recovery phase may be needed if CNS effects are seen during treatment or are suspected to bear potential delayed effects, in order to assess reversibility versus long-term/delayed toxicity.
These data are consistent with the results of an Environmental Protection Agency review 24 of 69 neurodevelopmental toxicity studies on pesticides, mostly neurotoxicants. Across the 69 studies, 2 types of neurobehavioral tests showed multiple positive results, locomotor activity and auditory startle response, whereas 2 other behavioral tests FOB and Learning and Memory (L&M tests) underperformed. Although Vorhees et al recommend dropping the FOB, they also suggest possible explanations for the apparent low sensitivity of L&M tests and propose reasons why they should still be performed, and how the selection of relevant neurocognitive tests could be improved taking into account the different cognitive functions covered by the Morris, Cincinnati, Radial Water Maze tests, and the Conditioned Fear test. Moreover, they suggest running these tests in young adult animals, as this test age is expected to result in better performance. The authors insist on the use of validated, standardized, automated, and scientifically chosen tests.
The incidence of positive findings in our project is also roughly consistent with recently published data from a similar research project conducted by the FDA Divisions on Psychiatry and Neurology Drugs, 25 particularly the low incidence of positive findings for the FOB. Effects were observed during off-treatment for 5 of 14 drugs, that is, in 36% of the studies, compared to the 54% reported by FDA Divisions just mentioned.
The slightly lower scores of positive findings for some of the behavioral tests observed in our project are difficult to interpret. The Center for Drug Evaluation and Research project analyzed a number of studies twice as high as in the present study, that is, a different data set, although a partial overlap of a number of compounds may be possible. In addition, the timing of the tests has not been discussed; therefore, it is not known whether the observations were always done just prior to the daily treatment (trough exposure to the drug) or at the Cmax (highest exposure to the drug).
Conclusions
This limited data set does not allow us to make clear recommendations regarding which concerns should trigger JAS for CNS compounds. It is important to have a strong scientific justification for each study, and dialogue with regulatory authorities before the conduct of this type of studies is recommended.
New toxicities or higher sensitivity to the treatment were detected in JAS compared to adult studies, but it was not possible to relate these effects to clinical situations. Nevertheless, multiple pediatric epidemiological studies showing convergence are usually needed to properly assess neurotoxicity in the clinic. The evaluation of CNS in daily practice is often difficult, as the methodology required for systematic results is difficult to implement in children with a high variety of confounders. From this point of view, it is important to study the behavioral effects in animals as it might be the only way to learn whether these compounds have adverse effects on daily functioning.
The evaluation of the designs and results of these studies highlighted which end points are more susceptible to yielding relevant data, particularly to detecting pharmacological and/or neurotoxic effects. Clearly, detailed clinical observations at specific time points in relation to time of administration are the most informative. Behavioral examinations, on the other hand, should be done only if scientifically justified and using well-standardized procedures.
Finally, the evaluation of the study designs stressed the high variability of these; sometimes not supporting the patient population to be treated (with respect to starting age and from an organ/systems maturation point of view), overly complicated, and therefore susceptible to induce irrelevant effects and/or to confound the readout, and using very high number of animals. It is important, based up experience, to streamline these study designs in order to obtain the optimal level of information. The companies responsible for development of the compounds reviewed in this article kindly agreed to publish the information on their products in its present format.
Footnotes
Authors’ Note
The views expressed in this article are the personal views of the authors and may not be understood or quoted as being made on behalf of or reflecting the position of EMA or one of its committees or working parties.
Acknowledgments
The authors thank Benedetta Polsinelli for her help in retrieving the data used in this study. Furthermore, the authors appreciated the help of David Jones, who kindly gave linguistic suggestions.
Author Contributions
Van der Laan, J.W. substantially contributed to conception or design, contributed to acquisition, analysis or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content; Van Malderen, K. contributed to acquisition, analysis or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content; Duarte, D. contributed to acquisition, analysis or interpretation of data, and critically revised the manuscript for important intellectual content; Egger, G. substantially contributed to conception or design, contributed to acquisition, analysis or interpretation of data, and critically revised the manuscript for important intellectual content; Lavergne, F., Roque C., Vieira, I., and Wiesner, L. contributed to acquisition, analysis, or interpretation of data and critically revised the manuscript for important intellectual content; Carleer, J. substantially contributed to conception or design, contributed to acquisition, analysis or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. J.W.L. initiated the project in collaboration with the Non-clinical Working Group (NcWG; supporting the Pediatric Committee [PDCO] at the European Medicines Agency [EMA]). N.d.J. did the analysis of 8 compounds. D.D., G.F.E., F.L., C.G.R., I.V., and L.W. (all members of the NcWG with the exception of G.F.E. and C.G.R.) contributed to the analysis of at least 1 compound and to the writing of the article and are listed alphabetically according to surname. K.V.M. and J.C. are the senior authors as the present and past chair of the NcWG and alternate member of the PDCO at the EMA, contributing by analysis/review of compounds analysis and writing of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
