Abstract
Cationic amphiphilic drugs (CADs) can induce phospholipidosis (PLD) in organs/tissues. Several ophthalmic pharmaceuticals containing CADs are marketed and used in children. To investigate the effect of PLD on the developing cornea, chloroquine and amiodarone, which are representative CADs, were applied topically to the eyes of juvenile rabbits, and the effects in juvenile rabbits were compared with those in young adult rabbits. Diffuse corneal cloudiness was observed in chloroquine- and amiodarone-treated eyes. Histopathologically, vacuolation was observed in the corneal epithelium and keratocytes. On ultrastructural examination, these vacuoles contained multilamellar inclusion bodies, which are a characteristic of PLD. The size of the vacuoles in the corneal epithelium was reduced in juveniles compared with young adults. Cytoplasmic lamellar bodies and exocytosis in the corneal endothelium were observed in young adult rabbits but not in juvenile rabbits. This study revealed that topical application of chloroquine or amiodarone induces corneal PLD in juvenile and young adult rabbits. Corneal endothelial changes occurred only in young adult rabbits, but ophthalmological changes were similar between juveniles and young adults. The results of the study suggest that the effects of corneal PLD were similar among age groups based on risk assessment.
Drug-induced phospholipidosis (PLD) is recognized as an accumulation of intracellular phospholipids in the organs/tissues affected by the xenobiotic drugs or chemicals (Halliwell 1997; Nonoyama and Fukuda 2008). Treatment with cationic amphiphilic drugs (CADs) leads to alterations in organs/tissues, including changes in cytoplasmic morphology characterized by the appearance of cytoplasmic myelin-like multilamellar structures called lamellar bodies on ultrastructural analysis. The eye is one of the target organs of drug-induced PLD. After systemic administration of CADs, such as chloroquine, amiodarone, and tamoxifen, corneal deposits appear as manifestations of vortex keratopathy, which represents a typical clinically abnormal corneal adverse event of drug-induced PLD (Li, Tripathi, and Tripathi 2008; Raizman et al. 2017). Topical subconjunctival administration of gentamicin, one of the aminoglycosides antibiotic ophthalmic drugs, also induced vortex keratopathy (Hollander and Aldave 2004). Several marketed ophthalmic pharmaceuticals also contain CADs as active ingredients, such as antimicrobial aminoglycosides (Raizman et al. 2017). No studies have assessed the effects of ocular medication on corneal PLD in humans; however, drug-induced corneal PLD also develops after topical ocular application of chloroquine and amiodarone in rats and rabbits (Drenckhahn, Jacobi, and Lüllmann-Rauch 1983; Bockhardt, Drenckhahn, and Lüllmann-Rauch 1978; Yamagiwa et al. 2017). These results suggest that the topical application of CADs containing ophthalmic pharmaceuticals has the potential to induce PLD. Ophthalmic antibiotics are also applied to children to treat corneal/conjunctival infections (Chung and Buhr 2000; Coulter 2004; Fortinguerra, Clavenna, and Bonati 2012). Although CADs containing ophthalmic formulations can be used to treat children, no information exists about the effect of drug-induced PLD on the developing cornea. These formulations may affect the eyes because of their morphological or functional immaturity in children and influence the postnatal ocular development. Rabbits are generally used for safety evaluation of ophthalmic drugs (Attar et al. 2013; Kurata et al. 2017). An increase of Na+/K+-ATPase pump density in the rabbit corneal endothelium occurs for up to 30 days after birth (Stiemke, Edelhauser, and Geroski 1991). Another study reported that the density of the corneal endothelium in rabbits continues to decrease until 16 weeks of age (Bahn et al. 1986). These facts suggest that the rabbit cornea continues to develop even after eye-opening and can be used to assess the potential for corneal PLD in a juvenile animal model.
The purpose of this study was to investigate the age differences in the effect of drug-induced PLD on rabbit eyes when chloroquine or amiodarone is topically administered and to characterize corneal changes induced by these drugs in juvenile rabbits. More specifically, the aim of this study was to evaluate the effect of topical ocular application of chloroquine or amiodarone in the eyes of juvenile rabbits of postnatal age ranging from 11 to 25 days, and the results of ophthalmological, histopathological, and ultrastructural findings are compared with those in young adult rabbits (9–11 weeks old) with the same treatment.
Material and Method
Test Articles
Chloroquine diphosphate was purchased from Sigma-Aldrich (Saint Louis, MO) and stored at room temperature. Amiodarone hydrochloride was purchased from Tokyo Chemical Industry (Tokyo, Japan) and refrigerated.
Animals
Rabbits used in this study were housed, treated, and cared for in a facility fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All animal experimentation procedures were performed in accordance with the animal welfare guidelines of Senju Pharmaceutical Co., Ltd. Prior to the experimentation, the protocol was reviewed and approved by the Institutional Animal Care and Use Committee.
Two male and ten female 5- to 6-day-old Japanese white Kbl:JW rabbits were purchased from Kitayama Labes Co., Ltd. (Nagano, Japan) with their dams. They were housed in plastic cages (Tecniplast SpA, Buguggiate, Italy) with a wooden box (Kitayama Labes Co., Ltd., Nagano, Japan) for lactation in air-conditioned conventional animal rooms at 22°C ± 3°C temperature and 55% ± 10% relative humidity with a 12-hr light/dark cycle. Dams were fed a commercial diet (Lab R stock; Nosan Corporation, Tokyo, Japan) once daily, and all animals were supplied with tap water
Experimental Procedures
Chloroquine diphosphate or amiodarone hydrochloride was dissolved or suspended in a vehicle (1.0% hydroxypropyl methylcellulose, 0.5% polysorbate 80, 0.1% sodium citrate, 0.1% sodium edetate, 1.0%
Summary of the Groups in This Study.
One day after the termination of dosing, juvenile rabbits at postnatal day (PND) 18 or 25 were separated from their dams, anesthetized via sevoflurane inhalation (Mylan Inc., Canonsburg, PA), and euthanized by exsanguination through the abdominal aorta. Following euthanasia, the eyes were enucleated and collected. Young adult rabbits at 10 or 11 weeks old were sacrificed, and the eyes of each animal were enucleated in the same manner. Collected eyes were placed in 2.5% glutaraldehyde-0.1 M phosphate buffer fixative, which was replaced the next day with 10% neutral-buffered formalin (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). After fixation, the tissues were embedded in paraffin and sectioned at approximately 3 μm. The sections were stained with hematoxylin and eosin and examined microscopically. For ultrastructural examination, one of each eyes treated with vehicle, chloroquine, and amiodarone for 14 days in both juvenile and young adult rabbits was selected. From each eye, a portion of the cornea was collected from enucleated or formalin-fixed eyes, minced to approximately 1 mm2, and placed in 2.5% glutaraldehyde-0.1 M phosphate buffer fixative for 1 hr. The samples were rinsed with 3% sucrose-0.1 M phosphate buffer several times and then placed in 1% osmium tetroxide fixative. Following fixation, tissues were embedded in epoxy resin, ultrathin sectioned, and stained with lead citrate and uranyl acetate. A transmission electron microscope (H-300, Hitachi Ltd., Tokyo, Japan) was used to observe corneal changes.
Results
Anterior Ocular Observation
Diffuse corneal cloudiness was observed at 14 days of treatment in both chloroquine- and amiodarone-treated eyes in juvenile rabbits and at 7 and 14 days of treatment in chloroquine-treated eyes and at 14 days of treatment in amiodarone-treated eyes in young adult rabbits (Table 2). As symptoms of ocular irritation, swelling of the eyelid and corneal fluorescein–stained spots were observed in chloroquine-treated eyes of juvenile and young adult rabbits, respectively. In amiodarone-treated eyes, corneal fluorescein–stained spots, including corneal erosion and conjunctival congestion, were observed in both juvenile and young adults. Furthermore, swelling of the eyelid, reddening of the eyelid margin, conjunctival edema, and subsequent corneal leukoma and corneal neovascularization were observed in juvenile rabbits (Table 2).
Summary of the Results of Slit-lamp Biomicroscopy with Ocular Instillation of Chloroquine or Amiodarone.
a Associated with moderate focal corneal cloudiness.
Histopathological and Ultrastructural Examination
Vacuolation was observed in the corneal epithelium and keratocytes in both chloroquine- and amiodarone-treated eyes in juvenile rabbits and young adult rabbits (Table 3; Figures 1 and 2). In chloroquine-treated eyes of juvenile rabbits, the cytoplasm of the corneal epithelium exhibited a foamy appearance due to filling with microvacuoles (Figure 1A and C), whereas the corneal epithelium of young adult rabbits contained relatively large vacuoles (Figure 1B and D). In the ultrastructural examination, multilamellar bodies were sparsely observed in the corneal epithelial cytoplasm of the juvenile rabbit (Figure 2A), whereas degraded multilamellar bodies were densely observed in the lysosomes in young adult rabbit (Figure 2B). Keratocytes were filled with multilamellar bodies in juvenile rabbit (Figure 2C). In contrast, keratocytes of young adult rabbit exhibit less multilamellar bodies, and their cytoplasm were filled with other cellular materials (Figure 2D). In the corneal stroma, keratocytes were hypertrophic due to the emergence of cytoplasmic vacuoles in both juvenile and young adult rabbits (Figure 1A and B). In the ultrastructural examination, keratocytes were filled with multilamellar bodies in juvenile rabbit (Figure 2C). In contrast, young adult rabbits exhibit less multilamellar bodies, and their cytoplasm was filled with other cellular materials (Figure 2D). Multilamellar bodies and exocytosis were observed in the corneal endothelium only in chloroquine-treated eye in the young adult rabbit but not in juvenile rabbit (Figure 2E and F). Cell infiltration in the corneal stroma and inflammation in the filtration angle were observed only in the chloroquine-treated eye of one young adult rabbit (Table 3). Vacuolation of the bulbar conjunctiva and necrotic/inflammatory responses, such as single-cell necrosis of the corneal epithelium, cell debris/inflammatory cell infiltration of the corneal stroma, or corneal neovascularization, was observed in the amiodarone-treated eyes of both juvenile and young adult rabbits (Table 3; Figure 3). In the ultrastructural examination, vacuoles in the corneal epithelium and keratocytes were enlarged lysosomes that contained multilamellar bodies (Figure 4A–D). In the corneal endothelium, these lysosomal lamellar bodies were present in young adult rabbit (Figure 4F) but not in juvenile rabbit (Figure 4E).
Summary of the Results of Histopathological Examination of the Eyes with Ocular Instillation of Chloroquine or Amiodarone.
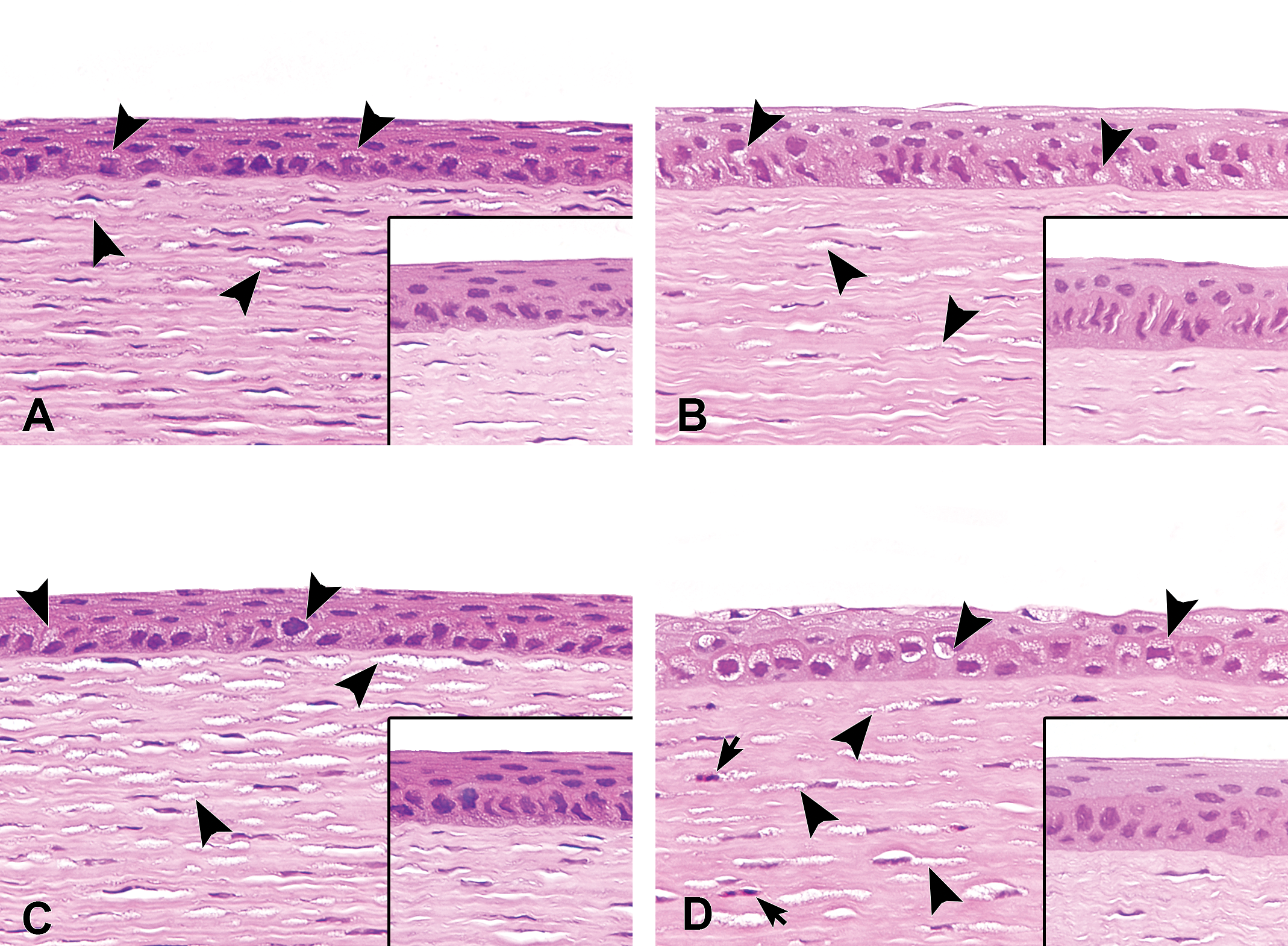
Representative hematoxylin and eosin–stained sections of corneas treated with ocular instillation of chloroquine for 7 (A and B) or 14 days (C and D) in juvenile (A and C) and young adult rabbits (B and D). Vacuolation of corneal epithelium and keratocytes (arrowheads) is observed in both juvenile and young adult rabbits. Vacuoles in the corneal epithelium of juvenile rabbit are relatively small compared with those in young adult rabbits. Vacuolation of keratocytes is more apparent after 14 days of treatment (C and D). Inflammatory cell infiltration (arrows) is also observed in the corneal stroma of young adult rabbits treated with chloroquine for 14 days. Each insert image presents vehicle-treated cornea.
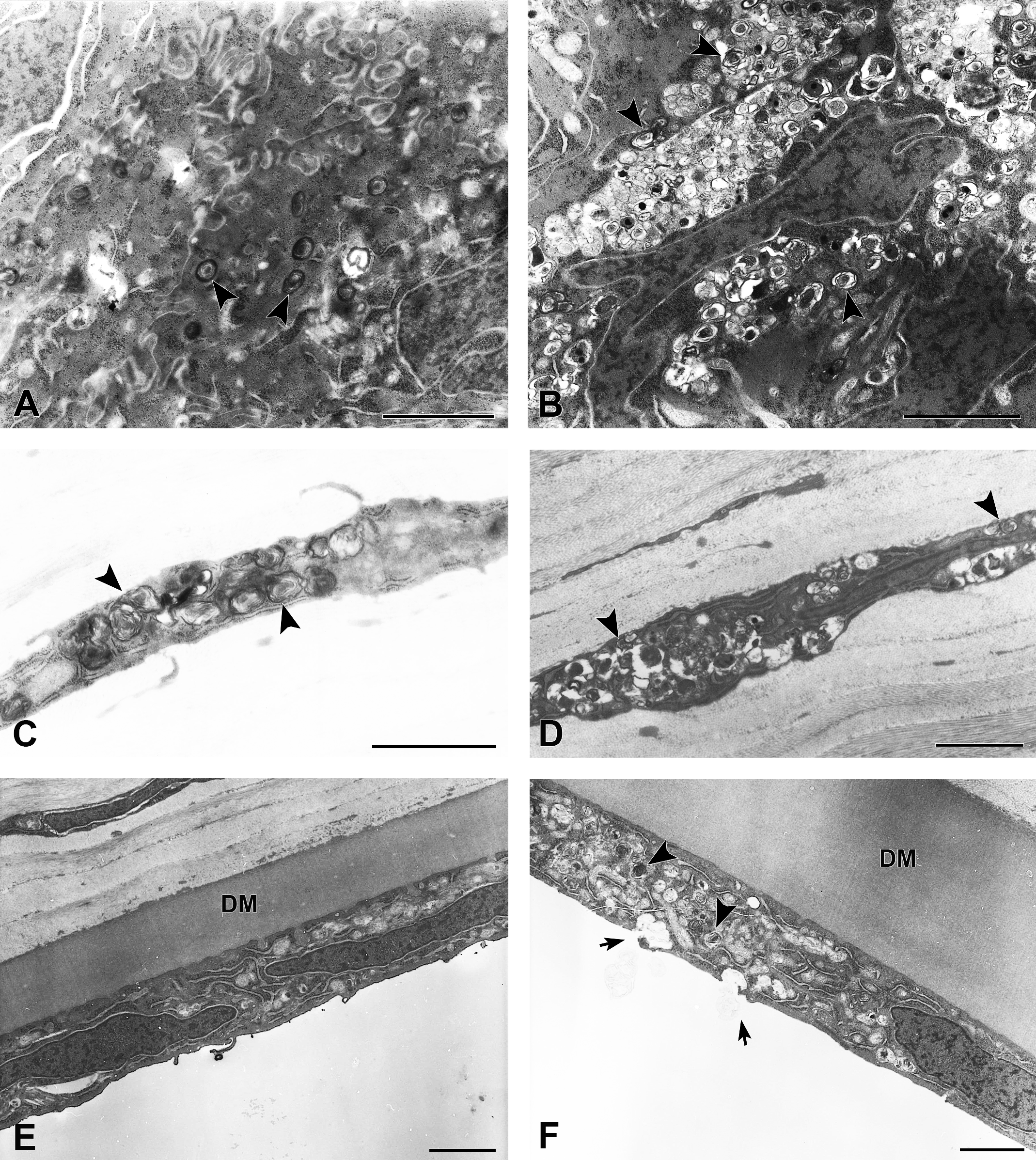
Ultrastructural changes in the corneal epithelium (A and B), stroma (C and D), and endothelium (E and F) in juvenile (A, C, and E) and young adult rabbits (B, D, and F) treated with ocular instillation of chloroquine for 14 days. Multilamellar bodies (arrowheads) are sparsely observed in the corneal epithelial cytoplasm of juvenile rabbit (A), whereas degradated multilamellar bodies were densely observed in lysosomes in young adult rabbit (B). Keratocytes are filled with multilamellar bodies in juvenile rabbit (C). Keratocytes of young adult rabbit exhibit less multilamellar bodies, and their cytoplasm is filled with other cellular materials (D). Multilamellar bodies appear in the corneal endothelium of young adult rabbit (arrowheads) and are excreted via exocytosis (arrows) (F). These figures are not observed in juvenile rabbit (E). DM = Descemet’s membrane; scale bar = 2 μm.
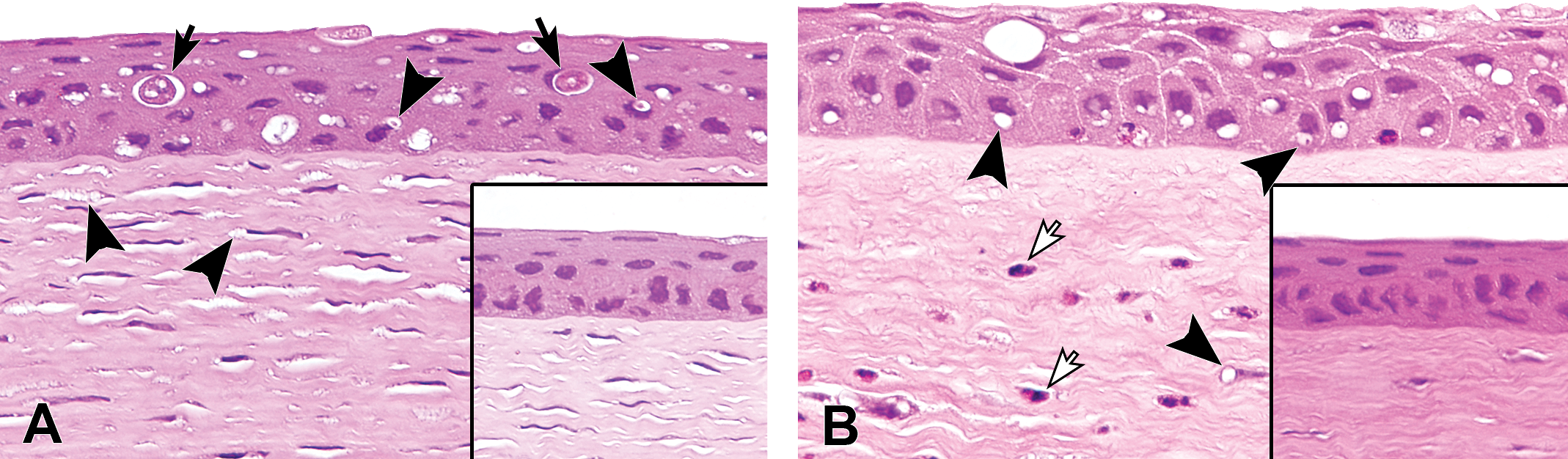
Representative hematoxylin and eosin–stained sections of corneas treated with ocular instillation of amiodarone for 14 days in juvenile (A) and young adult rabbits (B). Vacuolation is observed in the corneal epithelium and keratocytes (arrowheads) in both juvenile and young adult rabbits. In these images, single-cell necrosis of the corneal epithelium (black arrows) and inflammatory cell infiltration of the corneal stroma (white arrows) is also observed in juvenile and young adult rabbits, respectively. Each insert image presents nontreated cornea.
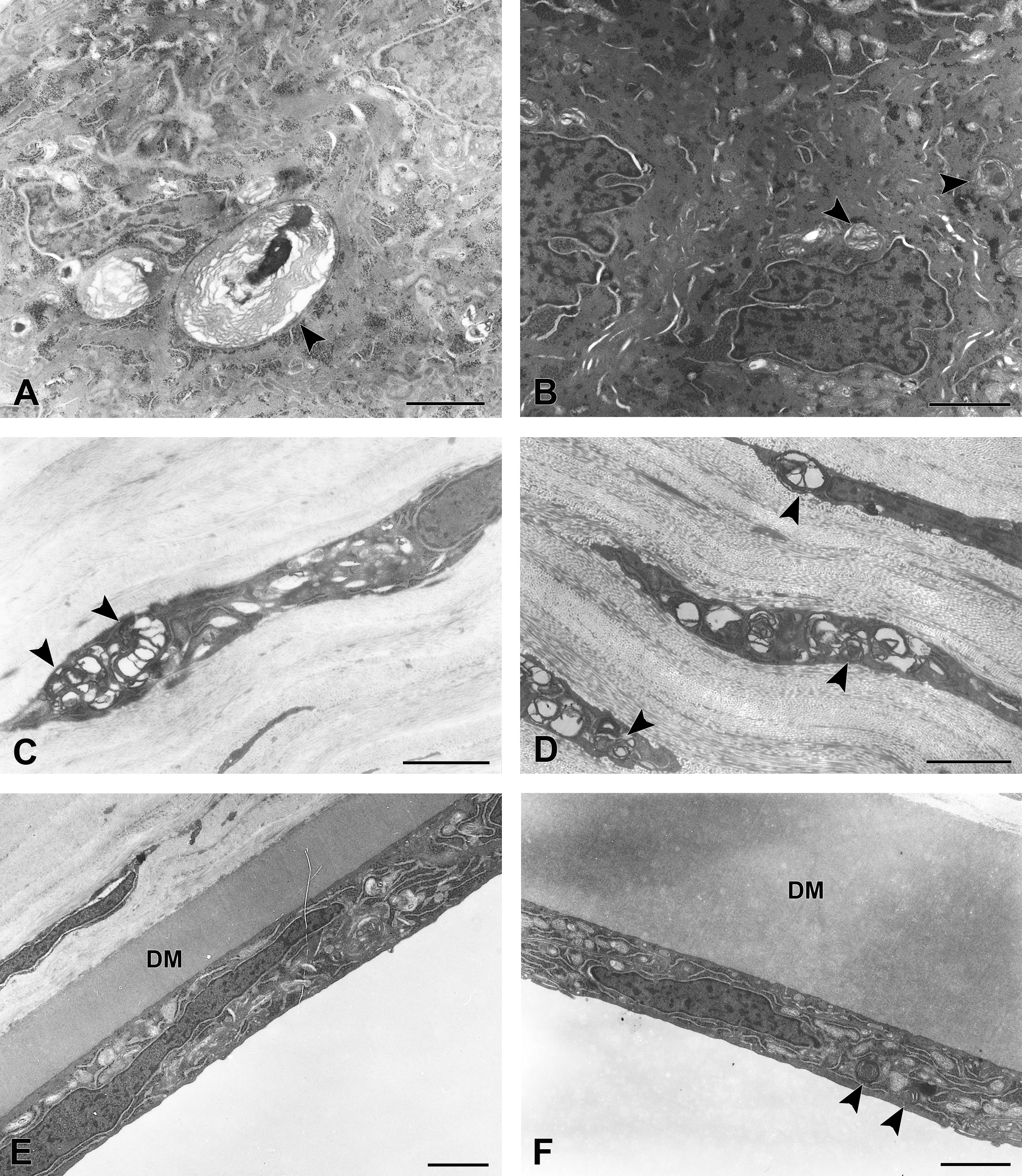
Ultrastructural changes in the corneal epithelium (A and B), stroma (C and D), and endothelium (E and F) in juvenile (A, C, and E) and young adult rabbits (B, D, and F) treated with ocular instillation of amiodarone for 14 days. Lysosomal multilamellar bodies (arrowheads) are observed in both juvenile and young adult rabbits (A–D and F), except for the corneal endothelium of juvenile rabbit (E). DM, Descemet’s membrane; scale bar = 2 μm.
Discussion
PLD occasionally appears in animal toxicity studies of pharmaceutical agents (Halliwell 1997; Nonoyama and Fukuda 2008), and the cornea is one of the common target organs affected by systemic administration of CADs (Greaves 2007). However, there was no evidence regarding whether PLD appears in juvenile rabbit eyes upon topical ocular application of CADs. This investigation is the first report demonstrating the effect in the juvenile rabbit cornea when CADs are topically applied to the eyes. Ultrastructural examination of the cornea in juvenile rabbits revealed cytoplasmic vacuoles composed of multilamellar inclusion bodies in lysosome-like vesicles, which is a characteristic of PLD (Halliwell 1997; Nonoyama and Fukuda 2008). This result indicates that topical ocular application of chloroquine or amiodarone induces PLD in juvenile rabbits, as previously noted in adult rabbits (Yamagiwa et al. 2017). Continuous dosing of chloroquine or amiodarone induces diffuse corneal cloudiness and inflammatory changes in adult rabbits (Yamagiwa et al. 2017). In this study, the incidence of diffuse corneal cloudiness was similar between juvenile and adult rabbits when chloroquine or amiodarone was applied to their eyes. Subsequent inflammatory changes in the cornea and surrounding tissues after repeat dosing of chloroquine or amiodarone were observed in both juvenile and young adult rabbits. This result suggests that the effects of drug-induced PLD on the cornea in juvenile rabbits are similar to those observed in adults.
Differences in the morphology of vacuolar changes in the corneal epithelium and the effect of topical ocular administration of chloroquine or amiodarone on the corneal endothelium between juvenile and young adult rabbits were noted in this study. Vacuoles were more prominent in the corneal epithelium in young adult rabbits compared with juveniles. In addition, lamellar bodies and their excretion via exocytosis were not observed in juveniles. Lysosome play an important role in cellular phospholipid metabolism (Samie and Xu 2014), and the mechanisms of the appearance of lamellar bodies might be due to the inhibition of phospholipid degradation or stimulation of phospholipid production (Nonoyama and Fukuda 2008). Lysosomal function and exocytosis in rabbits are potentially immature immediately after eye-opening, and lysosomal functional disorder is not observed. Although the level of cellular lysosomal functional maturity in juvenile rabbit eyes is unclear, the thickness of the corneal endothelium and the number of its Na+/K+-ATPase pump sites increase from birth to PND 30 (Stiemke, Edelhauser, and Geroski 1991). The growth of the rabbit corneal endothelium overlapped with the dosing period in this study. Thus, the age difference in the response might be due to prepuberty of the corneal epithelium and endothelium of juvenile rabbits. Despite these differences, minimal differences in ophthalmological changes were noted between age groups as described above.
In recent decades, the Food and Drug Administration (FDA) and the European Medical Agency (EMA) have revised regulatory guidance regarding pediatric drugs, and the guidance describes the nonclinical toxicology study using juvenile animals (FDA 2006; EMA 2008; Bailey and Marien 2011). According to the guidance, pharmaceutical companies have initiated juvenile animal toxicity studies to support clinical studies in children. Toxicological evaluations of intravitreally injected drugs have been conducted using juvenile rabbits (Zayit-Sondry et al. 2011; Fusco et al. 2012), but no study has been performed to evaluate the ocular toxicity of the topical application of drugs in juvenile animals. This study is the first toxicological examination performed by dropping the drugs into the eyes in juvenile rabbits. Repeat-dose topical ocular application of amiodarone resulted in ocular irritation changes, such as swelling of the eyelid, conjunctival congestion/edema, and corneal fluorescein–stained spots, in juvenile rabbits. This study revealed that ocular irritation caused by topical ocular administration of drugs could be detected in juvenile rabbits. As described above, rabbits are generally used for safety evaluation of ophthalmic drugs (Attar et al. 2013; Kurata et al. 2016). The size of the anterior segment of eyes in rabbits is relatively large, making ophthalmological examinations easier to conduct in rabbits than in rats and mice. These findings suggest that juvenile rabbits are a useful animal species for conducting nonclinical juvenile toxicity studies for pediatric ophthalmic drug development.
In summary, this study revealed that topical ocular application of chloroquine or amiodarone to juvenile rabbit eyes induces corneal PLD, which is similar to that observed in young adults. Although morphological differences in the corneal epithelium and endothelium were noted between the two age groups, the ophthalmological findings in the cornea were similar between juvenile and young adult rabbits.
Footnotes
Acknowledgments
The authors would like to thank the staff of Ina Research Inc. for all their help preparing the specimens for histological and ultrastructural examination. Their dedication and excellent efforts made these investigations possible.
Author Contributions
Authors contributed to conception or design (YY, SN, MK, HS); data acquisition, analysis, or interpretation (YY, YT, HK, HS); drafting the manuscript (YY); and critically revising the manuscript (YT, HK, SN, MK, HS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Senju Pharmaceutical Co., Ltd.
