Abstract
Developmental immunotoxicity (DIT) testing is centered around the concern that exposure to immunotoxicants early in development may result in enhanced susceptibility of, or unique or more persistent effects on, the immune system, in comparison to adult exposure. Developmental immunotoxicity has been the focus of numerous workshops and reviews for at least fifteen years. Most of these earlier activities have focused on both environmental chemicals and pharmaceuticals and have concluded that the best approach to DIT is to address the possible impacts of exposure during all of the critical windows of development. This article will emphasize the critical role played by exposure during the juvenile stage of development. This article will also highlight several key issues that distinguish DIT testing of pharmaceuticals. Representatives from the pharmaceutical, biotechnology, academic, and regulatory sectors (both FDA and EMA) were brought together during a two-day workshop in May 2010 to consider the current state of the science of DIT as it pertains to the testing of pharmaceuticals. It is important to emphasize at the onset that there are currently no regulatory guidelines for either drugs or nondrug chemicals specifically focused on assessment of DIT, although some general guidelines are included in both developmental and reproductive toxicity and general immunotoxicology guidance documents.
Introduction
This article is based on a presentation that was given during a symposium titled “The Immune System throughout Life” at the 40th Annual Meeting of the Society of Toxicological Pathology in Denver, CO. By way of background for an article focused on juvenile immunotoxicology, there are a number of characteristics of the developing human immune system, and of immunocompetence in early life, that should be emphasized at the onset (Burns-Naas et al. 2008; Holsapple et al. 2007):
It is known that common infectious diseases can occur more often and are usually more severe in the very young when compared with adolescents and adults.
Infants are easily immunosuppressed and are therefore potentially more susceptible to immune manipulations, including immune toxicities and immune-based therapies.
In spite of the above, it is known that infants can mount a vigorous immune response to tissue and organ allografts (e.g., “nonpathogenic” foreign antigens), and to vaccines.
Although it is known that the human immune system is functional at birth, it must be recognized that it undergoes considerable postnatal development.
It must be recognized that children are not small adults, and that development is a continuous process, and thus a “moving target”: neonates ≠ toddlers ≠ children ≠ adolescents ≠ adults.
Finally, it must be recognized that all phases of development must be considered when assessing toxicities and therapies for juveniles.
In light of all of the above, it is not surprising that a number of regulatory guidelines have emerged to address the safety of drugs intended for pediatric indications, including the United States Food and Drug Administration (FDA) Guidance for Industry Nonclinical Safety Evaluation of Pediatric Drug Products (FDA 2006; Tassinari et al. 2011), and the European Medicines Agency Guideline on the Need for Non-clinical Testing in Juvenile Animals on Human Pharmaceuticals for Paediatric Indications (Carleer and Karres 2011; EMA 2008). In Japan, the Ministry of Health, Labour, and Welfare (MHLW) started to prepare their Guideline for Nonclinical Safety Studies in Juvenile Animals in October 2010 (Shimomura 2011). In general, juvenile toxicity studies are conducted to identify potential latent signs of toxicity, to characterize the potential for chronic developmental toxicity that cannot be thoroughly assessed through standard developmental and reproductive toxicity (DART) protocols, and to ensure that children are not unnecessarily exposed to potentially adverse risk factors. The relationship of the various types of DART protocols and a typical juvenile animal study in the context of the developmental timeline of a rat are highlighted in Figure 1. Shown in blue are the three segments of the FDA / ICH DART protocols, which include the following: the Seg I test, which is effectively a test of fertility; the Seg II test, which is effectively an assessment of impacts on embryo-fetal development and of teratogenic potential; the Seg III test, which normally extends from late gestation to the early postnatal period, and which is an assessment of impacts on perinatal development; and a typical juvenile test protocol, which extends from pre-weaning (e.g., mid- to late postnatal) to pre-sexual maturation, and which is an assessment of impacts on postnatal development. Assessing the overall value of juvenile animal studies is beyond the scope of this article, but the ILSI Health and Environmental Sciences Institute (HESI) DART Technical Committee organized a workshop to address the impact of juvenile animal studies on pediatric drug development in May 2010 and have recently prepared a workshop summary (Leconte et al., in press).
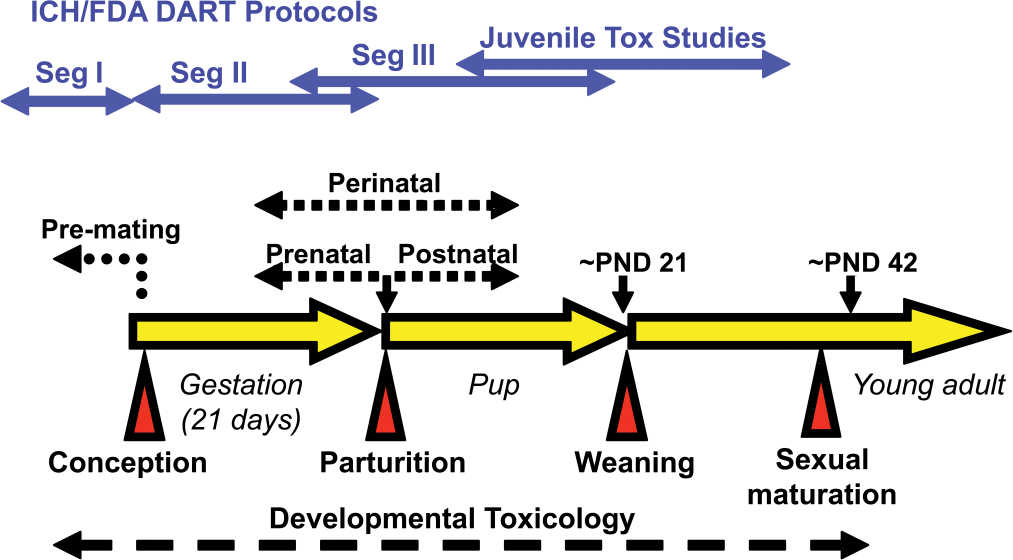
Developmental toxicology timeline in the rat.
In general, the objective of juvenile animal toxicity studies of pharmaceuticals is to obtain safety data, including information on the potential for adverse effects on postnatal growth and development (Cappon et al. 2009). Indeed, the FDA Guidance from their Center for Drug Evaluation and Research (CDER) addresses “the role and timing of animal studies in the safety evaluation of therapeutics intended for the treatment of pediatric patients” and emphasizes the following premise: “[i]t is thought that organ systems at highest risk for drug toxicity are those that undergo significant postnatal development” (FDA 2006). Importantly, the immune system was identified in the FDA Guidance as an important end point, which provides the basis for a discussion about juvenile immunotoxicity.
The basic tenet behind all of the regulatory guidelines discussed above is that children differ significantly from adults in their biological and/or physiological responses to chemical exposures. In the specific context of the immune system, this tenet could be stated as follows: the developing immune system may demonstrate greater susceptibility to chemical perturbation than the adult immune system. The primary “driver” for all of the attention on developmental immunotoxicity (DIT) testing is the following operational premise (Burns-Naas et al. 2008): exposure during the development of the immune system results in a qualitative or quantitative difference in the effect, or a greater persistence of the effect, and that a well-designed and well-executed “guideline” immunotoxicity study in young adult animals could miss this “window” of increased susceptibility. In that regard, there are some chemicals that exert greater effects on the developing immune system than the adult immune system (e.g., a quantitative difference) or that produce either a different profile of immunotoxic activity (e.g., a qualitative effect) or more persistent effects when exposure takes place during the development of the immune system, when compared with exposure in young adults (Luebke et al. 2006). However, no developmental immunotoxicants have been identified that have not also been immunomodulatory in the adult animal. Importantly, this observation may merely reflect the state-of-the-science; for example, all published DIT studies to date have been conducted on known adult immunotoxicants, and systematic studies comparing the effects across multiple developmental windows are still uncommon.
The State-of-the-Science of DIT
As noted above, there has been considerable attention on the state-of-the-science of DIT, including a number of workshops (Dietert et al. 2000; Holsapple 2002; Holsapple et al. 2005; Ladics et al. 2005; Luster et al. 2003) and reviews (Burns-Naas et al. 2008; Dietert and Piepenbrink 2006; Dietert and Holsapple 2007; Dietert and Burns-Naas 2008; Holsapple 2003; Holsapple et al. 2003; Holsapple et al. 2004; Holsapple et al. 2007; Luebke et al. 2006; Weinstock et al. 2010). A detailed summary of this material is beyond the scope of this article. Nonetheless, there were a number of conclusions that were consistently made during these workshops, or in these reviews, which include the following points:
It has been recommended that DIT studies should be added to already required DART toxicity studies, if possible.
It has been recommended that all “windows” be addressed at once, and then specific “windows” be “dissected” if an effect is seen.
It has been recommended that the DIT protocol could be extended to include juveniles and young adults, which is important to the context of this article; but, regardless of the specific protocol, it was considered important that exposure be continued until an assessment of immune effects was conducted.
Because of the extensive background on the use of rats in DART studies, it has been concluded that rodents—primarily rats—are the best choice for a DIT screen, but that this selection must consider the known differences in developmental timelines.
Finally, it has been concluded that immunotoxity is most appropriately assessed using functional tests, but it must be recognized that functional tests cannot be conducted in very young pups where possible effects could only be seen using histopathology.
Some additional perspective on a few of these points is provided below.
Figure 2 is a simple illustration of the fourth point, that is, the need to consider the known species differences in development (Holsapple et al. 2003). The developmental timelines of rats and humans are compared in Figure 2 using simply conception, birth, and weaning. The development of the immune systems in rats and humans is depicted using three developmental landmarks: the initial appearance of the first small numbers of B- and T-cells; the demarcation of splenic architecture; and the initial appearance of germinal centers. Using only a few landmarks, this comparison clearly indicates that the development of the rat and human immune systems is different. Although the basic anatomy of the rat immune system is largely established by postnatal day (PND) 0, the expansions of the various immune system components in response to environmental stimuli do not occur until after birth. In contrast, the appearance of these same components in the human immune system begins during mid-gestation. Ultimately, the postnatal appearance of some of the immune developmental landmarks in the rat would be comparable with an appearance in humans during the second trimester of pregnancy.
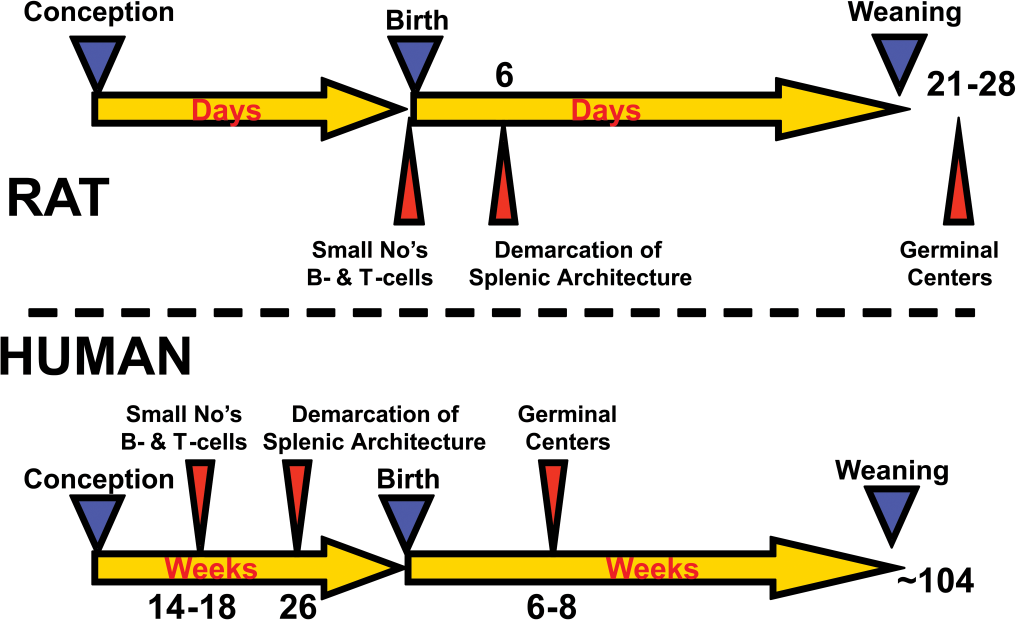
Comparison of the development of the rat and human immune systems.
Although previous workshops and reviews have emphasized the importance of assessing DIT using functional tests, as indicated in the fifth point, some additional context on the availability of functional tests, and on the use of histopathology, can be provided. Previous workshops and reviews on DIT have concluded that most of the functional tests have been optimized only for use in young adult rats (or mice), and that few of them have even been attempted at other developmental time points from early postnatal to weaning (Dietert and Holsapple 2007). Regarding histopathology, some elegant studies to better define the temporal development of the rat immune system have recently been done (Burns-Naas et al. 2008; Weinstock et al. 2010) and are ongoing. Current early data suggest that there is little potential for histopathologic detection of chemically mediated alterations in the developing immune system of rats prior to gestational day (GD) 15; but that major perturbations (e.g., failure to form) could be detected by GD 20. These studies also showed that although the immune system is intact by PND 22, the histologic features suggest relative inactivity (e.g., lack of stimulation by exogenous stimuli). The latter observation is consistent with the few studies that have been done to characterize the functional capabilities of rats pre-weaning (Kimura et al. 1985; Ladics et al. 2000). Although it has been concluded that, historically, most pathologists have far less experience with fetal and neonatal immune histology compared with adult immune histology, there is consensus that immunopathology end points are ultimately an important part of characterizing effects on the developing immune system.
As noted above in the second point, it has been recommended that all “windows” be addressed at once, and then specific “windows” be “dissected,” if an effect is seen. Such a “proposed” protocol is presented at the bottom of Figure 3B and is compared with a more traditional approach, the so-called “classical” DIT protocol, which is presented on the top of Figure 3A. The difference is immediately apparent, especially in the context of exposure. In the “classical” DIT protocol (Figure 3A), exposure is generally limited to an acute, or at best, a very short-term exposure of the pregnant dams, usually during the onset of organogenesis (e.g., GD 4). Because the immune tests have been optimized only in adults, as noted above, these tests have generally been conducted in rats at or about six to eight weeks of age. This protocol is an excellent measurement of any persistent effects associated with exposure during organogenesis, but it would miss any transient effects. Many of the earlier workshops and reviews on DIT had noted the importance of assessing both persistent and transient effects. The “classical” DIT protocol would also miss any effects associated with exposure during the prenatal, postnatal, and juvenile periods of development. In contrast, the “proposed” DIT protocol (Figure 3B) addresses the recommendations that exposure take place over all of the critical “windows” of susceptibility and that exposure be maintained until the immune tests are measured. There are some important caveats associated with the “proposed” DIT protocol. The first caveat is that it is assumed that exposure of the pregnant dams will result in sufficient transplacental transfer of the test material as to allow for possible effects on immune organogenesis and on the fetus. The second caveat is that it is assumed that exposure of the dams post-parturition will result in sufficient lactational exposure to the test material in the nursing pups. These caveats are certainly not trivial points to consider, especially because it has been argued that a negative DIT study, in the absence of confirmation that exposure has taken place, is considered incomplete, at best. Importantly, these caveats are not unique to a consideration of a DIT study design and are more generalizeable to a developmental toxicology consideration.
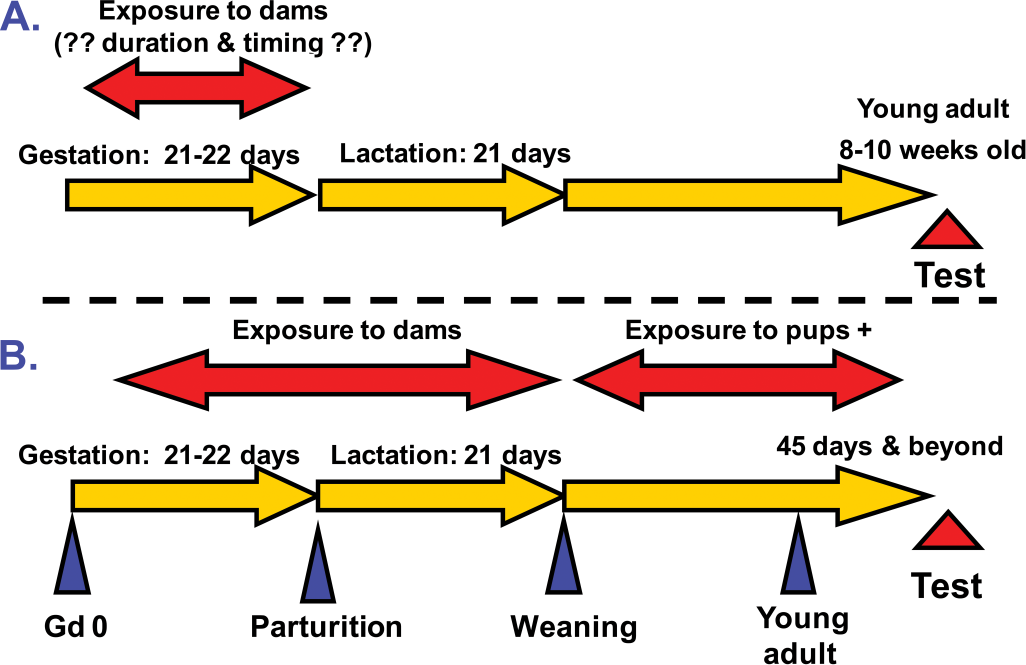
Comparison of DIT protocols.
Before leaving the discussion about the “proposed” DIT protocol, it is important to note that some considerable progress has been made in the context of implementing such a protocol. The HESI Agricultural Chemical Safety Assessment (ACSA) Technical Committee had previously proposed an extended one-generation protocol (Cooper et al. 2006). More recently, Vogel et al. (2010) described a modular one-generation reproduction study as a flexible testing system for regulatory safety assessment and highlighted the application of such an approach to the EU’s Registration, Evaluation and Authorization of Chemicals (REACH) program. Taken together, these results prompted the US EPA and the Dutch authorities to convene a working group to revise the OECD 415 protocol to embrace some of the principles articulated by Cooper et al. (2006) and Vogel et al. (2010). Indeed, in November 2010, the OECD released a draft protocol for a revised OECD 415, extended one-generation study, that included the following three cohorts: Cohort 1 assessed the potential impact on reproductive/developmental end points, and it could be extended to include an F2 generation on a case-by-case basis; Cohort 2 assessed the potential impact of chemical exposure on developmental neurotoxicity; and Cohort 3 assessed the potential impact of chemical exposure on developmental immunotoxicity. Although the revised OECD 415 protocol has been designed to assess the effects of chemical exposure in rats, more recently, evaluations of DIT for pharmaceuticals in nonhuman primates has been conducted as part of embryo-fetal development (EFD), pre-/post-natal development (PPND), or juvenile studies. With regard to critical immune system “windows,” the enhanced PPND (ePPND) study design in nonhuman primates (NHPs) appears optimal for DIT testing, and it has been shown to have similar benefit to the testing of monoclonal antibodies (Stewart 2009) and biopharmaceuticals (Martin et al. 2009).
One additional point to consider in this section on the state-of-the-science of DIT is the current status of regulatory approaches to DIT. As emphasized in the introduction, globally, regulatory agencies have not routinely required DIT during the drug development process. Rather, assessments are driven by a cause-for-concern approach (Holsapple et al. 2005) derived from a weight-of-evidence review of the totality of the available data. In the context of DIT, this cause-for-concern approach has generally focused on the following points:
The totality of the data derived from standard toxicity studies, which might indicate an effect on the immune system.
The pharmacologic properties associated with the drug in question, especially in the context of the potential to affect the immune system.
The intended patient population, including the potential to have an impact on the developing immune system (e.g., women of child-bearing potential, and infant [pediatric] and juvenile patient populations).
Drugs with structural similarity to compounds with known immunosuppressive effects.
A pattern of disposition of the drug that suggests that it is retained at high concentrations in components of the immune system.
Signs observed in clinical trials that might suggest an effect on the immune system.
It is important to note that this cause-for-concern approach is not unique for DIT; it applies to any organ system with continuing postnatal development. However, the possible impact of drug discovery on the developing immune system is increasing because novel therapeutics are beginning to emerge that have the immune system as a potential target organ, or that drive a redirected immune response as a pharmacologic end point.
Developmental Immunotoxicity Testing of Pharmaceuticals
As briefly mentioned above, most of the previous workshops and reviews on DIT have generally focused on both pharmaceuticals and environmental/industrial/agricultural chemicals. However, because novel therapeutics have already emerged and are continuing to be designed that target the immune system or the immune response, it is important to recognize that the reasons for performing DIT tests with pharmaceuticals are different from those for nondrug chemicals. The HESI Immunotoxicology Technical Committee (ITC) has taken steps to address the needs specific to DIT testing of pharmaceuticals.
There are clear risk/benefit differences for human exposure. Therapeutic applications are intended to treat a defined patient population, often for predefined periods of time, and there is a clear benefit to this intervention. In contrast, exposure of humans to nondrug chemicals used for industrial or agricultural purposes is typically unintentional, and often, there are no direct benefits to humans for this exposure. However, recognizing that there are clear risk/benefit differences does—and should—not imply that pharmaceuticals should be tested any less rigorously than industrial/agricultural chemicals. The HESI ITC recognized early in this activity that the ability to be flexible in determining both the need for DIT and the study design to assess DIT is a unique feature of pharmaceuticals. They also recognized that drug exposure to the developing system occurred via two general scenarios: first, inadvertent exposure in utero or via breast milk as a result of the mother taking the drug; and second, direct dosing of children to treat childhood diseases. Therefore, the goal in drug development is to assess the risk of developmental effects from these exposure scenarios, and not a general assessment of the hazard, as might be desired when considering the possibility for unintentional exposure to nondrug chemicals.
The HESI ITC formed a DIT Working Group that developed a two-stage approach. The first stage was to draft a manuscript titled, “Developmental Immunotoxicity (DIT) Testing of Pharmaceuticals: Current Practices, the State of the Science, Knowledge Gaps and Recommendations” (Collinge, M. et al., in preparation). This “state-of-the-science paper” addressed the following topics associated with DIT testing of pharmaceuticals:
triggers and causes for concern for DIT testing;
consideration of current guidance documents;
protocols and testing strategies for DIT;
specific issues relating to DIT testing of biopharmaceuticals; and
identification of major gaps in our knowledge and future directions.
This paper is anticipated to be submitted to a peer-reviewed journal by year end 2011.
The second stage by the HESI ITC DIT Working Group was to organize and host a workshop, which took place in Washington, DC in May 2010. The primary goals of the workshop, which brought together almost sixty scientists representing the pharmaceutical, biotechnology, academic, and regulatory (both FDA and EMA) sectors, were to discuss the key topics covered in the manuscript and to share the industry experience with and to gain regulatory perspective on DIT testing of pharmaceuticals. The overall intent of this activity was to capture the points of discussion, the areas of consensus, the areas of continued disagreement, and any other feedback, so that this information could be incorporated into a revised draft of the manuscript prior to publication.
The full content of the discussion at this workshop is beyond the scope of this paper but will be available in the revised and updated publication by Collinge, M. et al. (in preparation). In the final sections of this paper, some of the gaps in the state-of-the-science of DIT testing of pharmaceuticals identified during this workshop will be presented, which will be followed by some highlights of the key areas of consensus that were achieved at the HESI ITC 2010 workshop.
Gaps discussed by Collinge, M. et al. (in preparation) and identified at the ITC workshop included the recognition that there is an incomplete knowledge of comparative immune system development across species (e.g., functional as well as structural). In particular, it was noted that there are limited historical data on various immune parameters during development (e.g., lymphocyte subsets, cytokines, immunoglobulins). Another gap was the recognition that there is an incomplete knowledge of the placental transfer of biopharmaceuticals. In terms of the approaches to the DIT testing of pharmaceuticals, the following gaps were identified: the need for a robust, cell-mediated immunity assay; the need for further optimization and validation of current assays used in adults for use in a DIT test protocol; the need for in vitro (ex vivo) assays for evaluation of DIT (e.g., human peripheral blood mononuclear cells). The lack of translational biomarkers from nonclinical DIT assessments that can be applied to the clinical situation was also identified as a gap.
The key points of consensus from the workshop should be considered in light of some of the earlier perspectives presented in this paper. The following key points of consensus from the HESI ITC 2010 workshop were reminiscent of earlier conclusions and recommendations for DIT testing:
Assessments of DIT should be incorporated into existing developmental toxicity protocols (e.g., PPND studies) wherever possible.
Determination of recovery/reversibility (e.g., persistent vs transient effects) is expected for all DIT studies and should be included wherever possible.
The rat is the preferred species for DIT studies with pharmaceuticals.
It is recognized that although immunopathology and immunophenotyping have a role in DIT evaluations, immunotoxicity is most appropriately addressed with a functional evaluation, such as the T-dependent antibody response.
The following key points of consensus from the HESI ITC 2010 workshop reflect special considerations of DIT testing of pharmaceuticals:
Routine DIT testing for all pharmaceutical compounds is not advocated, and DIT testing with pharmaceuticals should only be conducted on a case-by-case basis, based on sound scientific rationale and establishing a cause for concern.
Sponsors should seek parallel advice from both the FDA and EMA prior to initiating DIT studies.
Only species that are pharmacologically responsive to the test material should be used for DIT studies with pharmaceuticals.
The potential to use the rat for DIT testing with biopharmaceuticals should be investigated and should not be automatically disregarded in favor of nonhuman primates, which should not always be the default species for DIT testing with all immunomodulatory biopharmaceuticals.
Use of one species for DIT testing of pharmaceuticals is considered adequate.
Transgenic models and homologs may be considered in the absence of a relevant species, as long as the pharmacologic/physiologic consequences in those test systems parallel those expected in humans.
The following key points of consensus from the HESI ITC workshop reflect the focus on juvenile immunotoxicology:
The timing of drug exposure relative to immune system development in nonclinical DIT studies should be relevant to that in the intended human pediatric/juvenile population.
The extended half-life of certain biopharmaceuticals should be considered in preclinical animal models of DIT since postnatal exposure in infants could occur following in utero exposure.
Finally, the paper by Collinge, M., et al. (in preparation) highlighted a few areas of research needs that were primarily intended to improve our understanding of the development of the human immune system, or the effects of immunotoxicants on the developing human immune system. First, they recommended that we work to attain a more complete understanding of when the various components of the human immune system reach functional maturity. Second, they recommended the creation of a database to monitor the incidence of autoimmunity, allergy/asthma, cancer, infection, or abnormal vaccine responses in children exposed to pharmaceuticals. Third, they recommended that further epidemiologic studies be conducted to evaluate the possible impacts of developmental exposures to known immunotoxicants. Finally, they recommended that an industry-wide survey be conducted on evidence of unique immunotoxicants in developing animals or in pediatric patients. The last recommendation extends beyond a focus on the human immune system and addresses a long-standing gap in the state-of-the-science of DIT, namely, the identification of test materials that are capable of DIT potential without being immunomodulatory in the adult.
Background on the HESI Immunotoxicology Technical Committee
HESI is the Health and Environmental Sciences Institute, a nonprofit, scientific organization based in Washington, DC, USA. The Health and Environmental Sciences Institute was created in 1989 as the global branch of the International Life Sciences Institute (ILSI). The mission of HESI is to bring together researchers from academia, government, and industry to design and execute scientific programs that improve the process of safety assessment for chemicals and pharmaceuticals.
The HESI Immunotoxicology Technical Committee (ITC) is one of the oldest components of the organization’s extensive scientific portfolio, as it was originally convened as a task force in 1990. The mission of the ITC includes the following:
To identify and address scientific issues related to the development and application of immunotoxicology to public health and human health risk assessment.
To promote the understanding and appropriate use of immunotoxicologic data to protect human health.
To contribute substantively to the scientific decision-making processes relative to the development of guidelines and regulations for immunotoxicologic testing at the local, national, and international levels.
Footnotes
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article. The authors received no financial support for the research and/or authorship of this article. Dr. Holsapple is now with Battelle, Health & Life Sciences Global Business, Columbus, OH.
