Abstract
Rabbits are sometimes used for intranasal toxicology studies. We investigated the postnatal development of the nasal passage in juvenile Japanese white rabbits from just after birth to 6-week-old to provide information for conducting intranasal toxicological evaluation using juvenile animals. On postnatal day (PND) 1, the nasal passage consisted of the septum with mostly cartilaginous nasal wall and turbinates. The lining squamous, transitional, respiratory, and olfactory epithelia were already distributed similar to adults and were still underdeveloped. The nasal passage gradually expanded with age, as did the nasal wall, including the turbinates formed by endochondral ossification. The maxilloturbinate elongated, during which it branched complexly. The respiratory epithelium takes the form of columnar epithelium together with a reduction in goblet cells. In addition, the olfactory epithelium had clear cytoplasm in the ethmoturbinate, the olfactory nerve bundles thickened, and Bowman’s gland acini increased in size and number. Other tissues, including the vomeronasal organ, nasal-associated lymphoid tissue, and nasolacrimal duct, also developed histologically with age. This investigation characterized the postnatal histological development of the nasal passage in Japanese white rabbits, providing basic knowledge regarding the histological examination and rationale for appropriate study design of intranasal toxicology studies in juvenile rabbits.
Keywords
Introduction
The nasal passage is the entrance of air intake for respiration, and the nasal cavity is, therefore, a direct target for inhaled agents. Tissues in the nasal passage are also directly exposed to drugs during intranasal administration. The nasal cavity is also an exposure site for eye-drop drugs for drainage via the nasolacrimal duct after ocular instillation. Although the nasal passages are not common evaluation sites in routine oral or other systemically administered toxicity studies, it is examined microscopically when drugs or chemicals are inhaled or intranasally exposed to evaluate safety. Rabbits are sparingly used in safety assessments of nasally applied drugs or inhaled chemicals.1,2 Intranasal administration has recently become a method of drug delivery to the central nervous system, and several pharmacokinetic evaluations are conducted by administering drugs to rabbits intranasally.3,4 Rabbits are generally employed in nonclinical ocular instillation toxicity studies,5-7 and histological evaluations of the lacrimal drainage route, which is a direct drug exposure site and thus, should be evaluated histologically for the safety of ophthalmic drugs. From this situation, histopathological examination of the nasal passages in rabbits is suggested to be vital when assessing the safety of intranasal or ocular topical medicines.
The Food and Drug Administration (FDA) issued regulatory guidance regarding the safety of pediatric drug administration in 2000. The guidance discusses the necessity of conducting toxicological evaluations on these drugs using juvenile animals. The pharmaceutical industry should also consider the necessity of conducting juvenile studies on a case-by-case basis if drugs have the potential to induce postnatal developmental toxicity. 8 The guidance for conducting nonclinical toxicity studies using juvenile animals was issued by the FDA, 9 the European Medicines Agency, 10 and the Ministry of Health, Labour, and Welfare in Japan 11 in 2006, 2008, and 2010, respectively. Harmonized guidelines regarding nonclinical safety evaluations of pharmaceuticals intended for development in pediatric populations have been issued by the International Council for the Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) in 2021. 12 These guidelines state that juvenile toxicity studies are generally compulsory when conducting safety assessments on pediatric drugs unless a waiver was granted prior to a new drug application being submitted.13-15 When juvenile toxicity studies are designed, information regarding the timing of maturation in the species employed in the study, including organ or tissue development, must be used to provide a rationale for deciding the age of the animal being used. Study pathologists should have knowledge of both the normal histology of immature organs and tissues and their histological characteristics at each age. The postnatal development of the nasal passages of rats 16 and miniature pigs 17 has been investigated previously. The normal histology of the nasal passage in adult rabbits has also been reported 18 ; however, the characteristics of postnatal histological development of the nasal passage have not been fully described to the best of our knowledge and remain inadequate as references for histopathological examination. Histological studies regarding the postnatal development of nasal passages in rabbits would be valuable to elucidate the normal histological features at each age due to the current requirements, supporting nonclinical toxicity studies.
This study aimed to investigate the postnatal development of the nasal passages in juvenile rabbits and describe its histological characteristics to disseminate information on study conditions and the histological evaluation of juvenile toxicity studies.
Materials and Methods
Animals
Eight male and eight female neonatal and infantile Kbl:JW Japanese white rabbits were assessed in this study. Specific pathogen-free animals were obtained from Kitayama Labes Co., Ltd. (Nagano) with their dams or delivered by a purchased pregnant female rabbit at the animal facility of Senju Pharmaceutical Co., Ltd. Two 21-week-old female Kbs:JW Japanese white rabbits were also examined to evaluate the mature histology. Table 1 summarizes the postnatal age, number, and sex of the rabbits used in this investigation. The neonates and kits were housed in plastic cages (Tecniplast S.p.A, Lombardia) with their lactating dams, at 22°C ± 3°C and 55% ± 10% relative humidity with a 12/12-hour light and dark cycle in conventional animal rooms. A wooden box (Kitayama Labes Co., Ltd.) was placed in each cage to provide a suitable suckling environment. Cage-side observations were performed daily to monitor the animals’ health. The dams were fed a commercial diet (Lab R stock; Nosan Corporation, Tokyo) once daily, and all animals were supplied with tap water ad libitum. Animal care and treatment were provided in accordance with the standard procedures of the facility, which is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All experimental procedures were conducted in accordance with the institutional guidelines for animal experimentation at Senju Pharmaceutical Co., Ltd., and the protocol was reviewed by the Institutional Animal Care and Use Committee in accordance with the Institute of Laboratory Animal Research Guide for the Care and Use of Laboratory Animals.
Postnatal Age, Number, and Sex of Rabbits Examined.
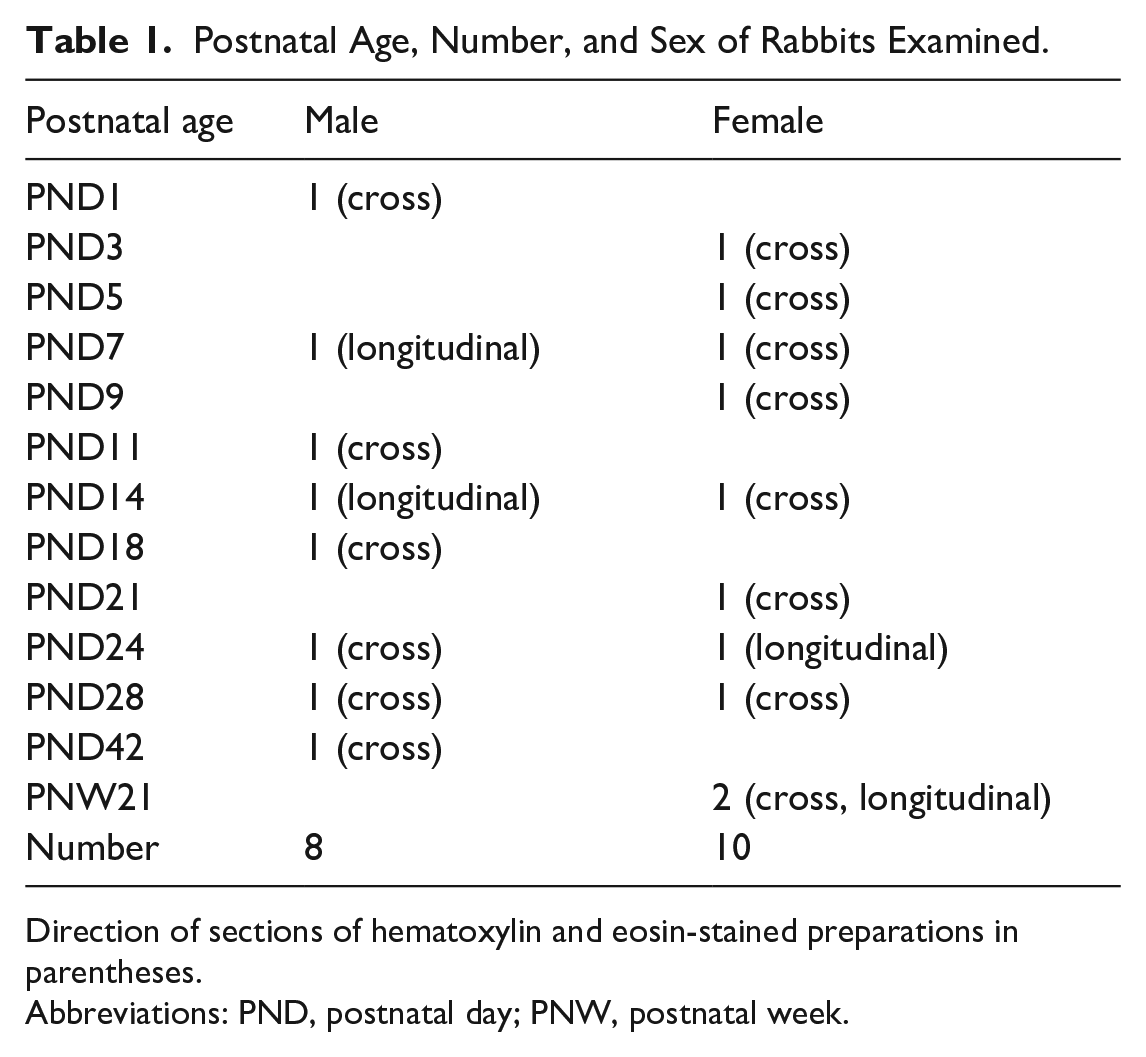
Direction of sections of hematoxylin and eosin-stained preparations in parentheses.
Abbreviations: PND, postnatal day; PNW, postnatal week.
Experimental Procedures
Clinically normal rabbit neonates and kits were randomly selected, and each rabbit was sacrificed on postnatal days (PND) 1, 3, 5, 7, 9, 11, 14, 18, 21, 24, 28, or 42, respectively. They were separated from their dams on the final day and euthanized by exsanguination through the abdominal aorta under inhalation anesthesia using sevoflurane (Mylan Inc., Pennsylvania). Adult female rabbits were sacrificed at postnatal week (PNW) 21 in the same manner. None of the animals presented with clinical respiratory abnormalities. The mandible was separated from all extraneous tissues after euthanasia, and the brain, eyes, skin, and muscle were removed. Next, 10% v/v neutral-buffered formalin (FUJIFILM Wako Pure Chemical Corporation, Osaka) was introduced into the nasal passage through the nasopharynx using a syringe with a plastic outer indwelling needle. The whole skull was then immersed in 10% v/v neutral-buffered formalin for fixation. After fixation, the skull was immersed in a 5% v/v formic acid/formalin decalcification solution for 6 to 26 days. Following decalcification, two levels of rostal and caudal cross sections of the nose (4 levels in total) as per methods described in previous literature regarding rabbit nasal cavities. 18 Levels I and II on the rostal side were sectioned immediately posterior to the incisors over the incisive papilla and at the first palatal ridge, respectively. On the caudal side, levels III and IV were located immediately anterior to the first upper premolar and the first upper molar or similar positions, respectively (Figure 1). Longitudinal sections on nare levels were also prepared at the ages of PNDs 7, 14, and 24, and PNW21. Each tissue was embedded in paraffin, and sections of approximately 3 µm were prepared and stained with hematoxylin and eosin (H&E). Several cross sections were not precisely obtained during the first sectioning despite these landmarks being set because the juvenile rabbits’ deciduous teeth were replaced by permanent teeth within 3 weeks postnatally. Therefore, several samples were additionally sectioned and stained in the same manner. The preparations were observed using a light microscope, and images were captured with the BX51 light microscope, equipped with a DP71 color charge-coupled device camera and the color imaging software cellSens Standard ver. 1.9 (Olympus Corporation, Tokyo). Whole slide images were captured with a Keyence All-in-one fluorescence microscope BZ-X700 using a 10× objective (Keyence Corporation, Osaka).
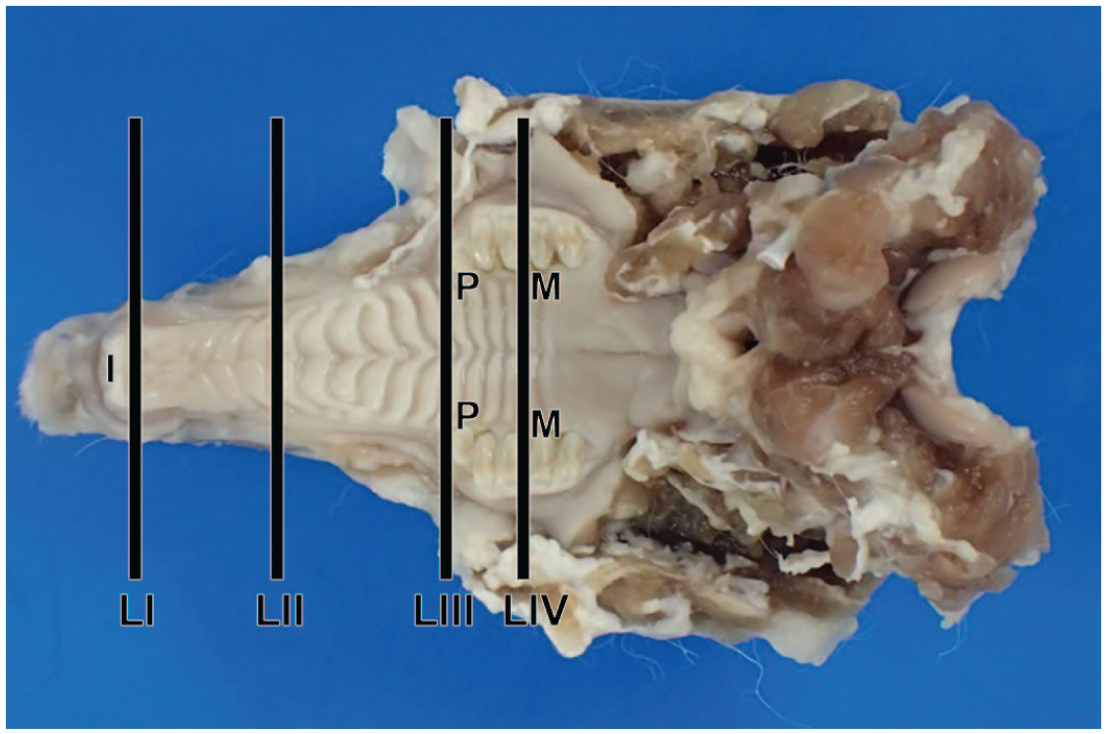
Ventrodorsal view of sampling positions of the rabbit nasal passage. I, incisor teeth; LI, level I; LII, level II; LIII, level III; LIV, level IV; M, first molar; P, first premolar.
Results
Overview
The H&E-stained longitudinal sections at PND7, PND24, and PNW21 are shown in Figure 2. After birth, the nasal cavity wall and turbinates mostly comprised cartilaginous tissue with immature nasal mucosa. The turbinates consisted of the nasoturbinate, maxilloturbinate, and ethmoturbinate, which were small and histologically immature. Although the epithelia in the nasal cavity were morphologically distinguished and lined in each region of the nasal wall, the transitional, respiratory, and olfactory epithelia were underdeveloped. The entire nose extended with age, while the nasal cavity increased in length and height. The maxilloturbinate and ethmoturbinates gradually enlarged, and complex branching occurred with age. The nasal cavity wall and turbinates were gradually replaced by osseous tissues.
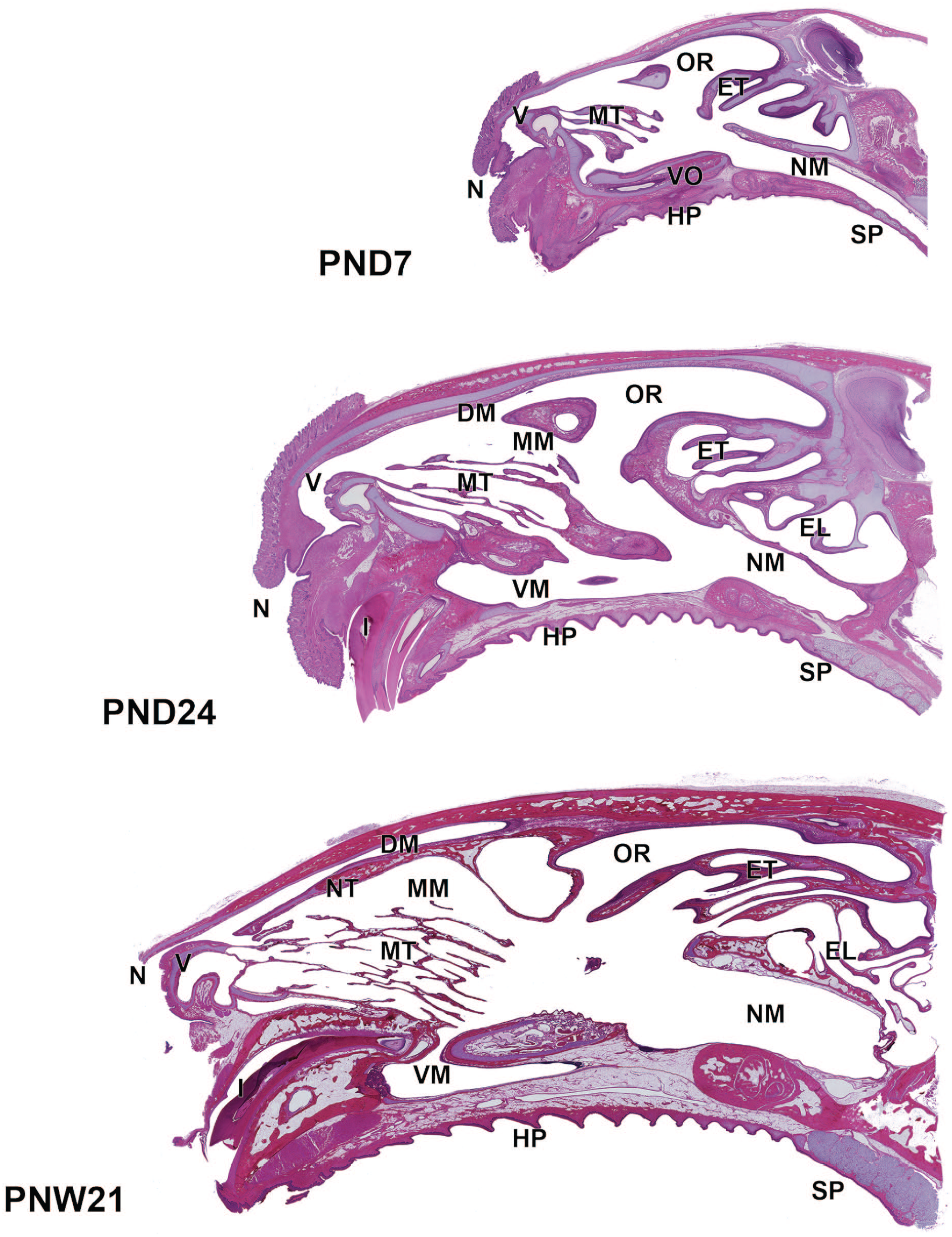
Longitudinal sections of the nose in Japanese white rabbits at postnatal days (PND) 7 and 24, and postnatal week (PNW) 21. The nose extends with age, and the nasal cavity increases in length and height. The maxilloturbinate and ethmoturbinates gradually enlarged with complex branching with age. The wall mostly consists of cartilaginous tissues at PND1, which are gradually replaced by bone tissue. Whole slide images captured with a Keyence All-in-one fluorescence microscope BZ-X700 using a 10× objective. Hematoxylin and eosin. DM, dorsal meatus; EL, ethmoidal labyrinth; ET, ethmoturbinates; HP, hard palate; I, incisor; MT, maxilloturbinate; MM, middle meatus; NM, nasopharyngeal meatus; N, nostril; NT, nasoturbinate; OR, olfactory recess; SP, soft palate; VM, ventral meatus; V, vestibule; VO, vomeronasal organ.
Rostal Side
The H&E-stained cross sections of the rostal side (levels I and II) at PND1, PND28, and PNW21 are shown in Figure 3. The nasal cavity at level I consisted of the nasal vestibule and was predominantly lined by stratified squamous epithelium. The epithelium in the nasal vestibule comprised four to six layers of squamous epithelial cells overlaid by keratinized epithelium, and some areas were lined with transitional epithelium. At PND1, the lamina propria of the transitional epithelial region was hypercellular, and contained vascular structure, termed the cavernous venous plexus and duct glands. The gland acini were not yet visible (Figure 4A). The connective tissue density had increased in the lamina propria at PND18 (Figure 4B), and gland acini gradually appeared in the lamina propria up to PND42 (Figure 4C). The lamina propria was histologically mature at PNW21, as evidenced by the completion of connective tissue development and increased gland acini (Figure 4D).
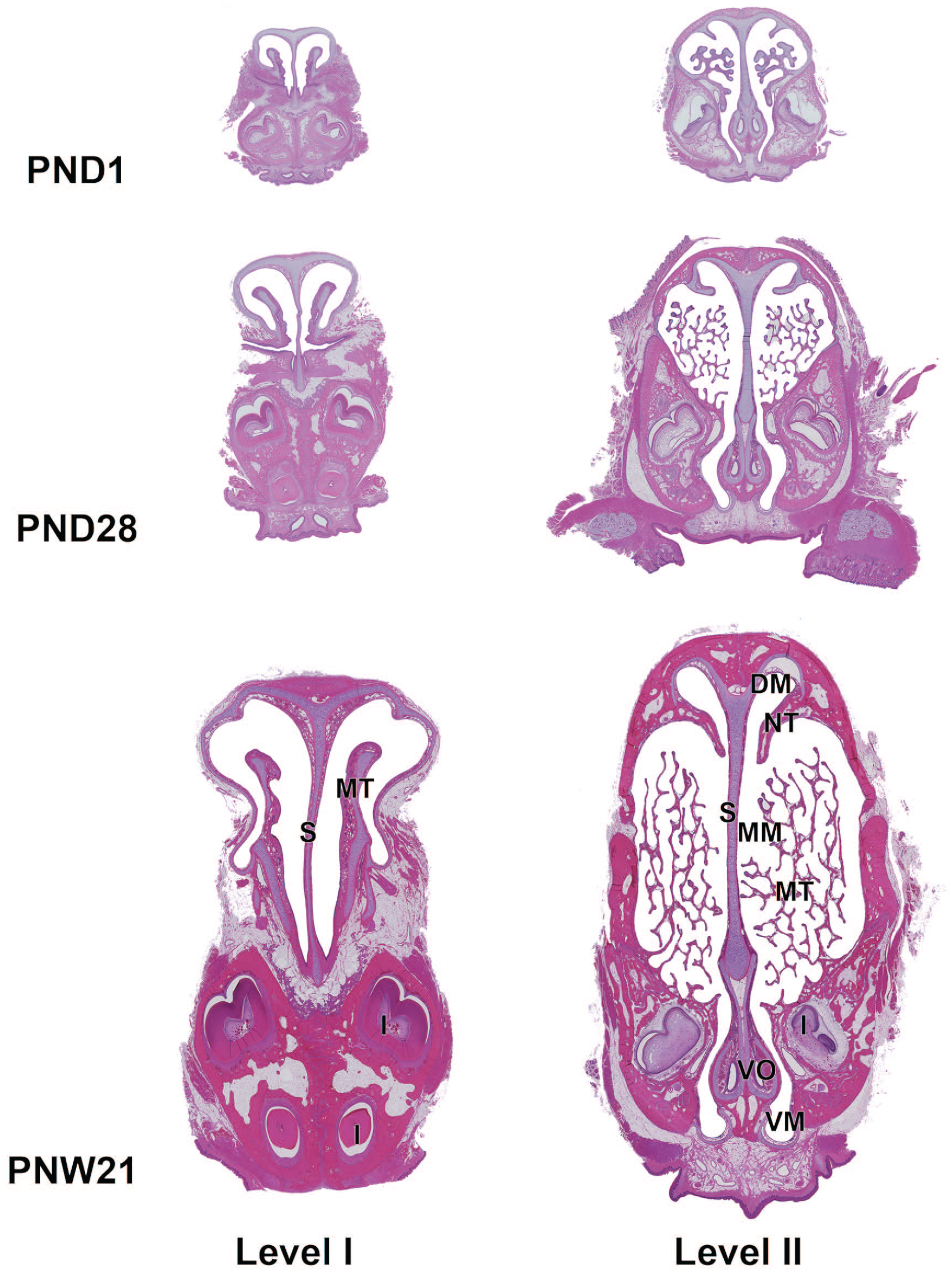
Cross sections of the rostal side of the nasal cavity in Japanese white rabbits at postnatal days (PND) 1 and 28, and postnatal week (PNW) 21. Level I, immediately posterior to the upper incisor; level II, through the first palatal ridge. The cross sections of the nasal cavity are small at PND1. The maxilloturbinate is short at level I and has several branches at level II. The nasal sinus expands especially in height with age, along with ossification of the dorsal and lateral walls of the nasal cavity. The maxilloturbinate and the nasoturbinate gradually enlarge. The maxilloturbinate enlarges with complex branching at level II. Whole slide images captured with a Keyence All-in-one fluorescence microscope BZ-X700 using a 10× objective. Hematoxylin and eosin. DM, dorsal meatus; I, incisor; MT, maxilloturbinate; MM, middle meatus; NT, nasoturbinate; S, nasal septum; VM, ventral meatus; VO, vomeronasal organ.
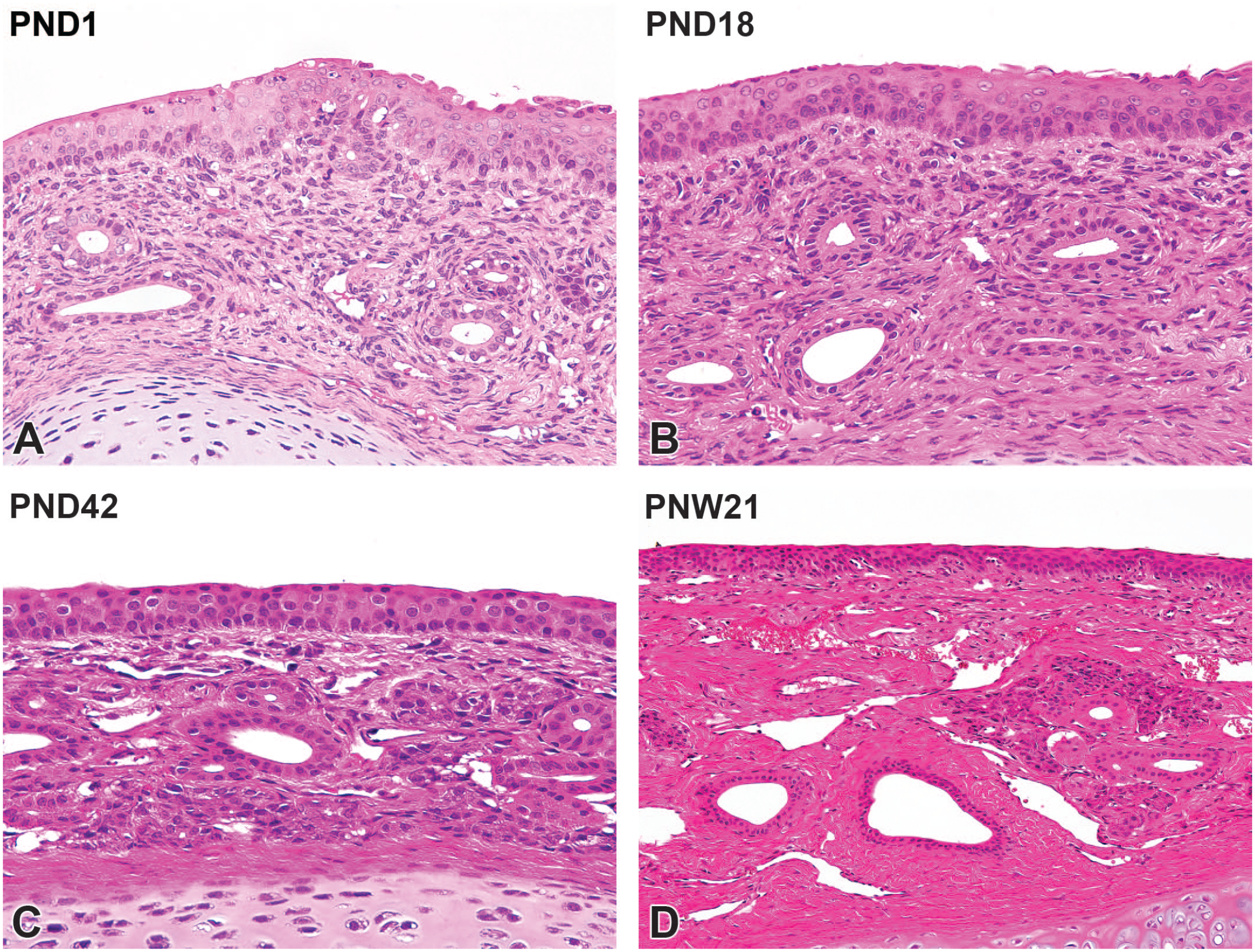
Postnatal development of the nasal vestibule mucosa on the maxillary turbinate in Japanese white rabbits. At postnatal day (PND) 1, the lamina propria of the transitional epithelial region is hypercellular, and contains vascular structure, termed the cavernous venous plexus and ducts of glands. The gland acini are not yet visible (A). At PND18, the connective tissue in the lamina propria increases in density (B). At PND42, serous gland acini and venous plexus appear in the lamina propria (C). The lamina propria is histologically mature at postnatal week (PNW) 21 by completion of connective tissue development and increases in gland acini (D). Original objective 40× (A-C) or 20× (D). Hematoxylin and eosin.
The maxilloturbinate protruded in the nasal cavity from level I and branched at level II and was lined predominantly by transitional epithelium with a thickness of up to six cells. At PND1, the maxilloturbinate was short, consisted of hyaline cartilaginous tissue, and had several branches at level II. The nasoturbinate, which divided the dorsal and middle meatus, appeared in the dorsal part of level II. At PND1, the nasoturbinate was composed of hyaline cartilaginous tissue lined by the transitional epithelium in the medial and dorsal surfaces or by the respiratory epithelium in the ventral surface at level II. The nasal sinus expanded with age, particularly in height, while ossification of the dorsal and lateral walls of the nasal cavity occurred simultaneously. The maxilloturbinate and nasoturbinate gradually enlarged. The maxilloturbinate at level II enlarged with complex branching with age (Figure 3). When higher magnification was applied at PND1, the maxilloturbinate consisted of cartilaginous tissue and turbinate bone formed by endochondral ossification. The tips were rounded and consisted of cartilaginous tissue (Figure 5A). The maxilloturbinate lengthened with age and branched complexly with the progression of endochondral ossification (Figure 5B). The maxilloturbinate became sharp at PND24, ossification of the turbinate bone was almost complete, and the cartilaginous tissue was located only in the tip (Figure 5C).
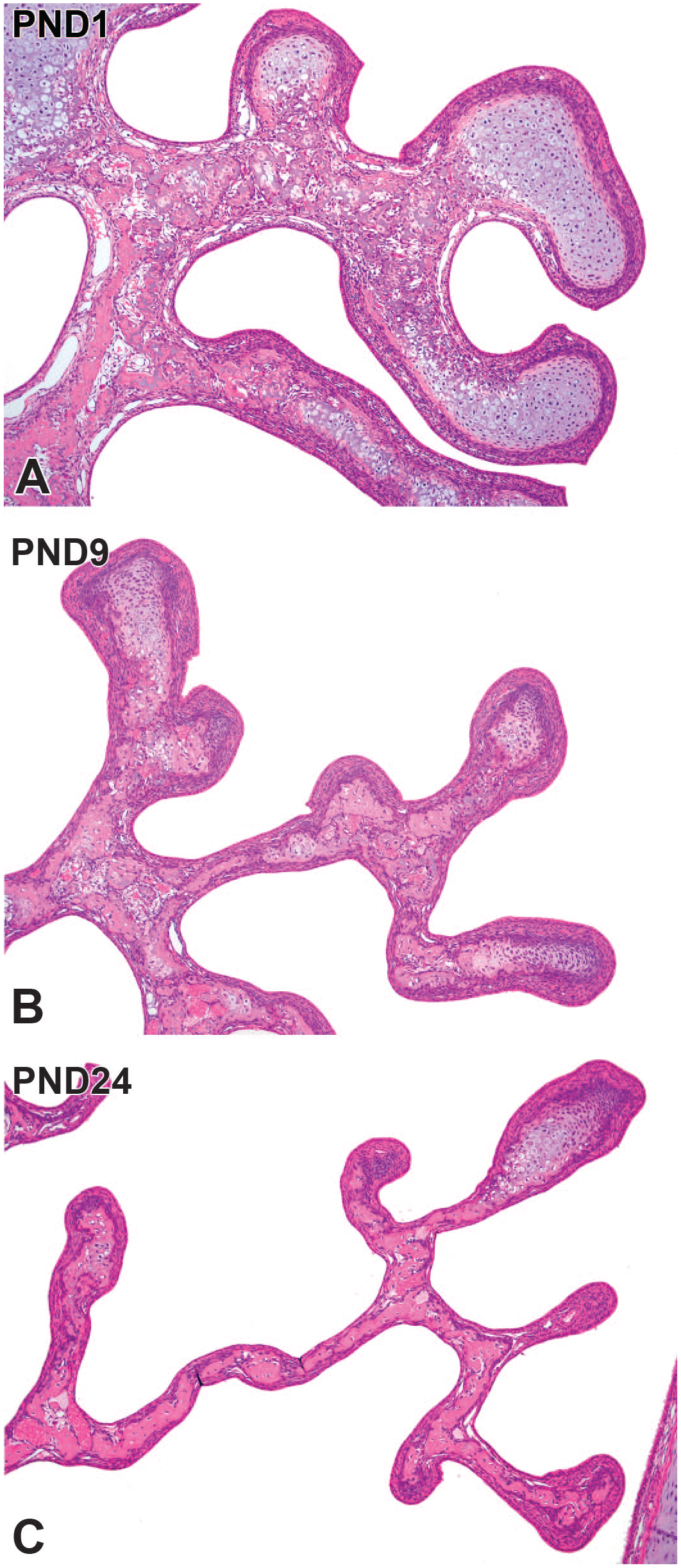
Postnatal development of the maxilloturbinate in Japanese white rabbits. At postnatal day (PND) 1, the maxilloturbinate consists of cartilaginous tissue, and the turbinate bone forms by endochondral ossification. The tips are rounded and consist of cartilaginous tissue (A). The maxilloturbinate elongates and branches complexly with age and progression of endochondral ossification (B). At PND 24, the maxilloturbinate becomes sharp. Ossification of turbinate bone is almost complete, and the cartilaginous tissue is only located in the tip (C). Original objective 10×. Hematoxylin and eosin.
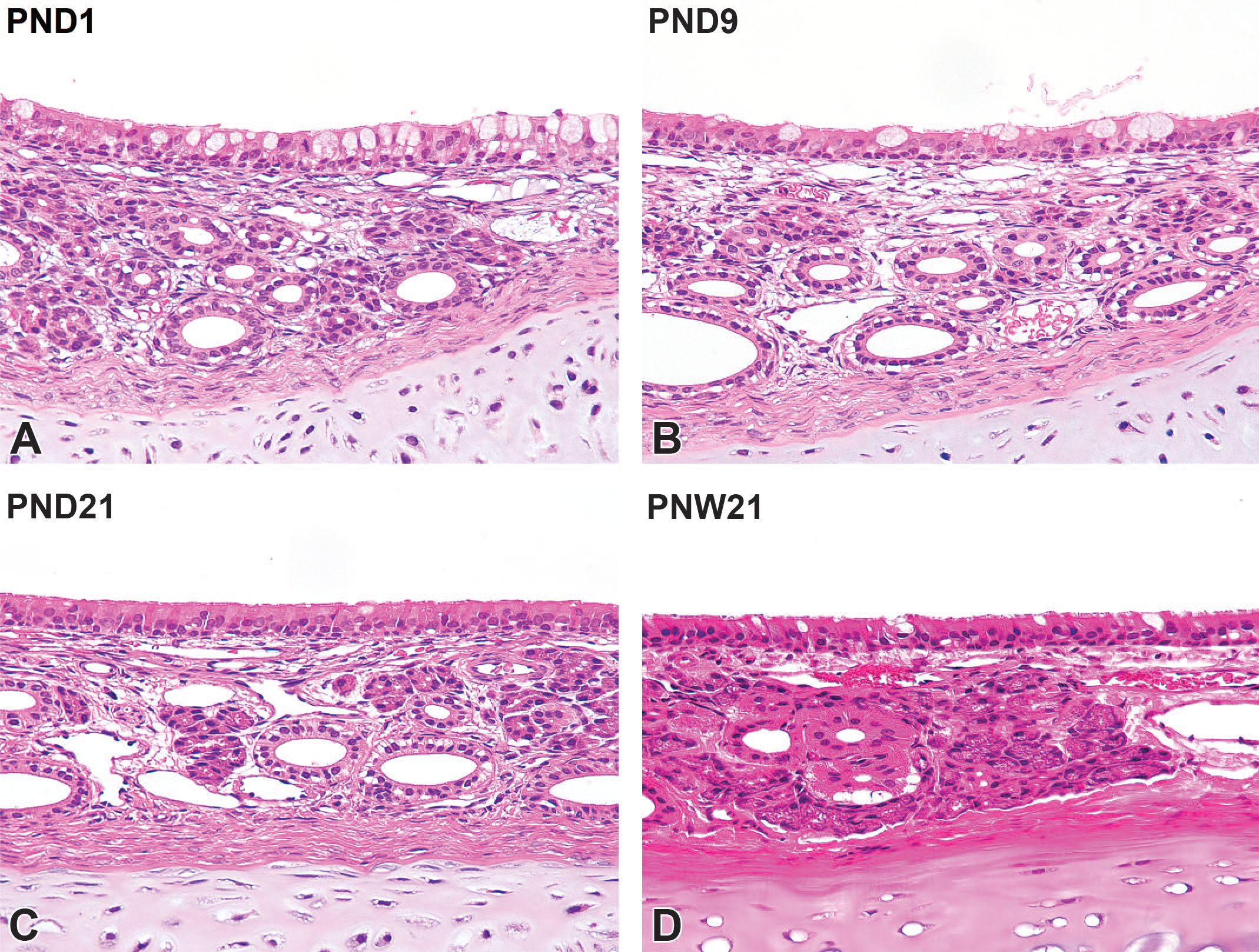
The nasal septum consisted of a hyaline cartilage plate lined with pseudostratified columnar ciliated respiratory epithelium in the surface mucosa at PND1. The respiratory mucosa consisted of pseudostratified cuboidal and columnar epithelium with abundant goblet cells. The lamina propria consisted of loosely arranged connective tissues and had the ducts of glands with vascular structure (Figure 6A). At PND9, the goblet cells in the respiratory epithelium decreased in density compared with PND1, whereas the connective tissue in the lamina propria increased in density. The increasing prominence of gland acini was observed in the lamina propria with age (Figure 6B). The goblet cell density was similar to that of adults at PND21. The gland acini were more prominent at this age (Figure 6C). At PNW21, further increases in gland acini were observed in the lamina propria, and development of the mucosa in the nasal septum region was completed (Figure 6D).
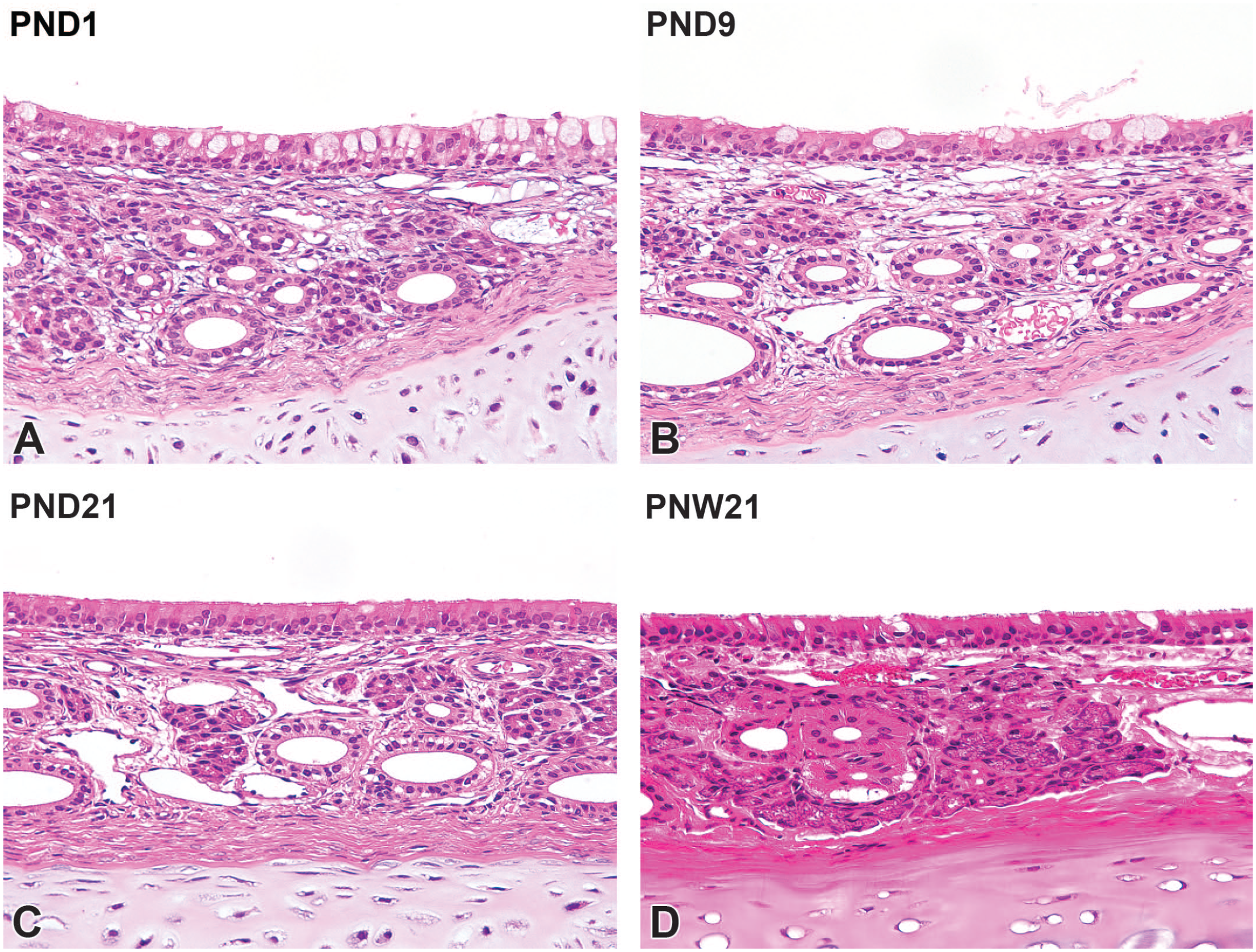
Postnatal development of the respiratory mucosa on the nasal septum in Japanese white rabbits. At postnatal day (PND) 1, the respiratory mucosa consists of pseudostratified cuboidal and columnar epithelium with abundant goblet cells. The lamina propria consists of loosely arranged connective tissues with gland ducts and vascular structure (A). At PND9, the goblet cells in the respiratory epithelium decrease in density compared with PND1, whereas the connective tissue in the lamina propria increases in density. Note increasing prominence of the gland acini in the lamina propria (B). At PND21, the goblet cell density is similar to that of adults. In the lamina propria, the gland acini are more prominent at this age (C). At PNW21, further increases in gland acini are observed in the lamina propria, and development of the mucosa is completed (D). Original objective 40×. Hematoxylin and eosin.
Figure 7 shows the H&E-stained cross sections of the vomeronasal organ at PND1 and PND24. The vomeronasal organ was already formed near the ventral aspect of the nasal septum at PND1 and encapsulated by the descending hook-shaped vomeronasal cartilage. The surface of the lumen was covered by the relatively thin vomeronasal epithelium, which comprised the olfactory and respiratory epithelia in the inner and outer lateral walls, respectively. The lamina propria of the vomeronasal organ had gland acini, vascular structure, and vomeronasal nerves; however, blood vessels were narrow, and nerve bundles were thin (Figure 7A). At PND24, the vomeronasal organ was morphologically mature, as shown by the increased vomeronasal olfactory epithelium thickness, expansion of blood vessels, and thickening of vomeronasal nerve bundles in the lamina propria (Figure 7B).
Cross sections of the vomeronasal organ (VO) in juvenile Japanese white rabbits. At postnatal day (PND) 1, descending hook-shaped vomeronasal cartilage already encapsulates the vomeronasal organ (A). The surface of the lumen is covered by the relatively thin vomeronasal epithelium, which consists of the olfactory and respiratory epithelia in the inner and outer lateral walls, respectively. The lamina propria of the VO has gland acini, vascular structure, and vomeronasal nerves, but blood vessels are narrow, and nerve bundles are thin (A). At PND24, the VO is morphologically mature by completion of increasing in thickness of the vomeronasal olfactory epithelium, expansion of blood vessels, and thickening of the vomeronasal nerve bundles in the lamina propria (B). Original objective 10×. Hematoxylin and eosin.
Caudal Side
The H&E-stained cross sections of levels III and IV at PND1, PND14, PND42, and PNW21 are shown in Figure 8. At PND1, the cross sections of the nasal cavity were small. The size of the nasopharyngeal duct and frontal and maxillary sinuses gradually increased with age as the ethmoturbinates increased in size. The ethmoturbinates elongated and formed with endochondral ossification of the turbinate bones. The turbinates at these levels were lined with respiratory and olfactory epithelia. The medial and dorsal aspects of the ethmoturbinates and most of the nasal septum were lined with olfactory epithelial cells. At PND1, the lamina propria contained hypernuclear olfactory nerve bundles and immature Bowman’s glands (Figure 9A). The olfactory epithelium had dark basophilic nuclei with dark eosinophilic cytoplasm. Mitotic figures were occasionally observed (Figure 9B). The olfactory nerve bundles became thicker with decreasing nuclear density as the rabbits aged (Figure 9C). Regarding the olfactory epithelium, the cytoplasm showed an irregularly pale color at PND11. Bowman’s gland acini increased in size and number with age (Figure 9D). At PND42, Bowman’s gland acini were more prominent, and further thickening of olfactory nerve bundles occurred (Figure 9E). The olfactory epithelium was histologically mature, as evidenced by the linear arrangement of olfactory and sustentacular cell nuclei (Figure 9F).
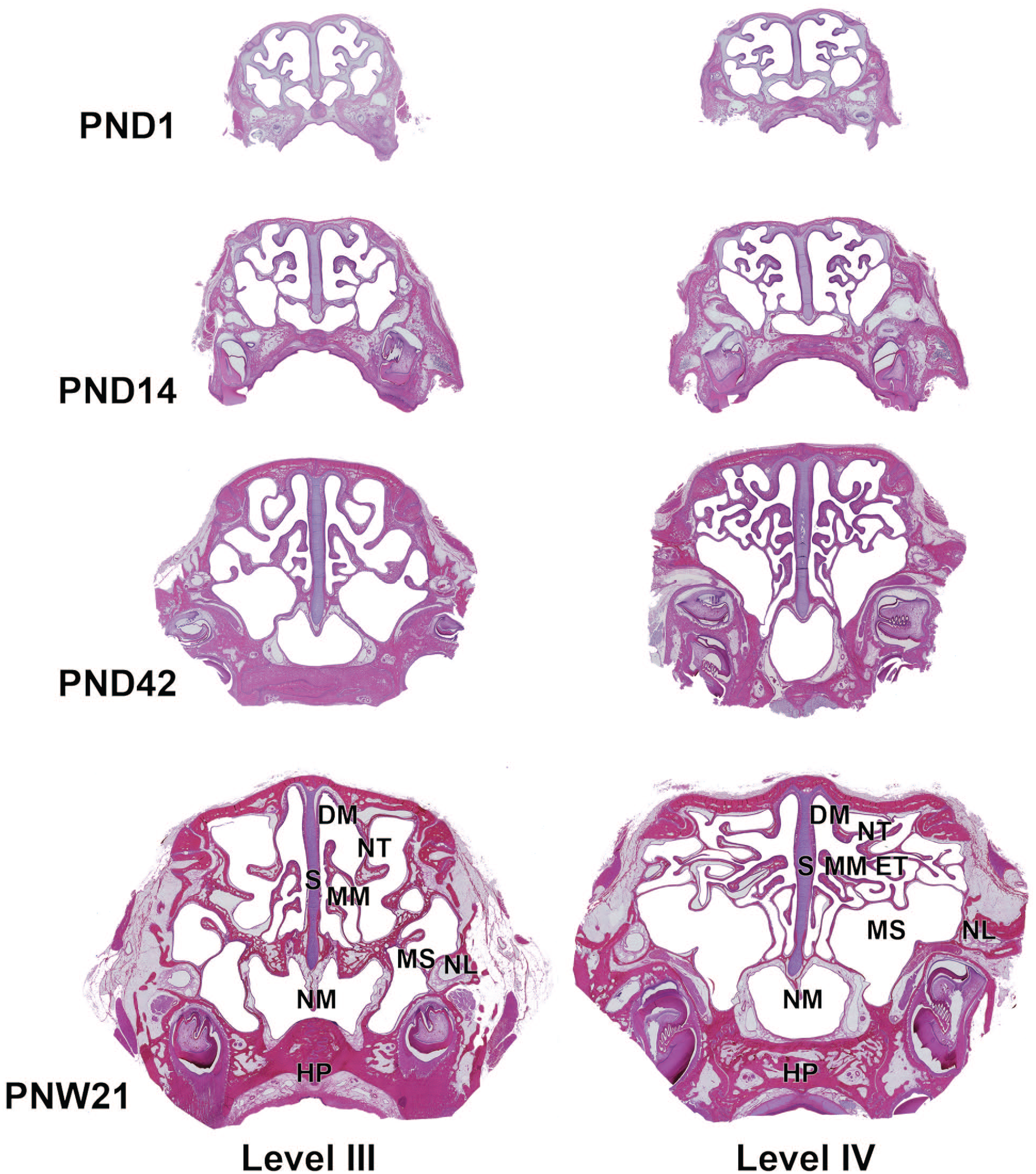
Cross sections of the caudal side of the nasal cavity in Japanese white rabbits at postnatal days (PND) 1 and 28, and postnatal week (PNW) 21. Level III, immediately anterior to the first premolar or resemble position; level IV, immediately anterior to the first upper molar or resemble position. At PND1, the size of the nasal cavity is relatively small. With age, the sizes of the nasopharyngeal duct and frontal and maxillary sinuses gradually increase with the increasing size of ethmoturbinates. The ethmoturbinates elongate and form with endochondral ossification of the turbinate bones. Whole slide images captured with a Keyence All-in-one fluorescence microscope BZ-X700 using a 10× objective. Hematoxylin and eosin. DM, dorsal meatus; ET, ethmoturbinates; HD, hard palate; MS, maxillary sinus; MM, middle meatus; NL, nasolacrimal duct; NM, nasopharyngeal meatus; NT, nasoturbinate; S, nasal septum.
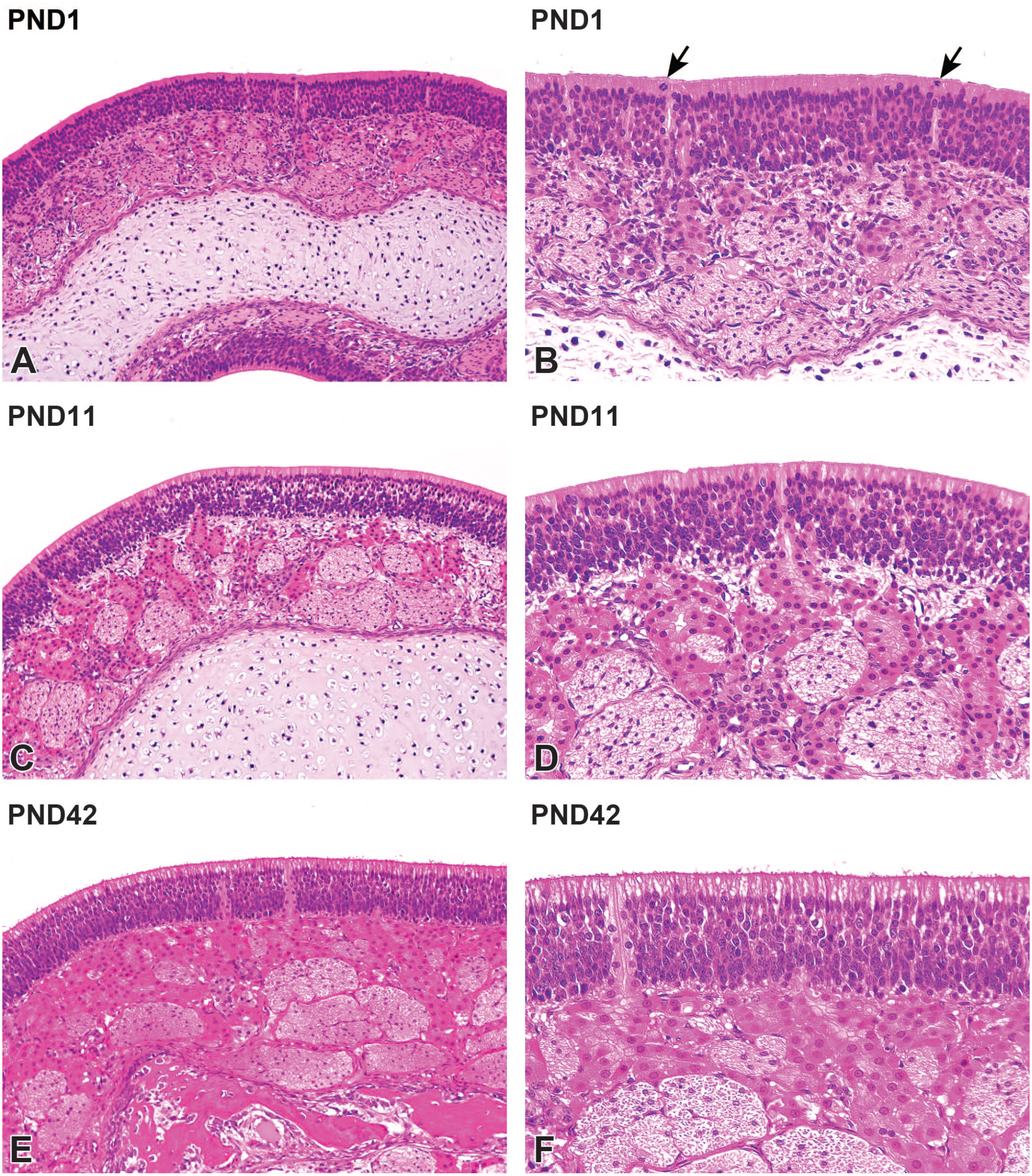
Postnatal development of the olfactory mucosa on the ethmoturbinate in Japanese white rabbits. At postnatal day (PND) 1, the lamina propria contains hypernuclear olfactory nerve bundles and immature Bowman’s glands (A). The olfactory epithelium has dark basophilic nuclei with dark eosinophilic cytoplasm. Note the appearances of mitotic figures (arrows) in the olfactory epithelium (B). The olfactory nerve bundles become thick with a reduction in nuclear density with age (C). In the superficial aspects of the olfactory epithelium, the cytoplasm shows irregularly pale color at PND11. Bowman’s gland acini increase in size and number with age (D). At PND42, Bowman’s gland acini are more prominent, and the olfactory nerve bundles thicken further (E). Note the linear arrangement of olfactory and sustentacular cell nuclei (F). Original objective 20× (A, C, E) or 40× (B, D, F). Hematoxylin and eosin.
The nasal-associated lymphoid tissue (NALT) was evident at the entrance of the nasopharyngeal duct at PND1 and on the caudal side of the dorsal meatus, mostly on the ventrolateral side of the lumen. At PND1, accumulation of lymphocytes was observed in the lamina propria of the respiratory mucosa at the entrance of the nasopharyngeal meatus (Figure 10A). A lymphatic nodule formed in the respiratory mucosa at PND7 (Figure 10B).
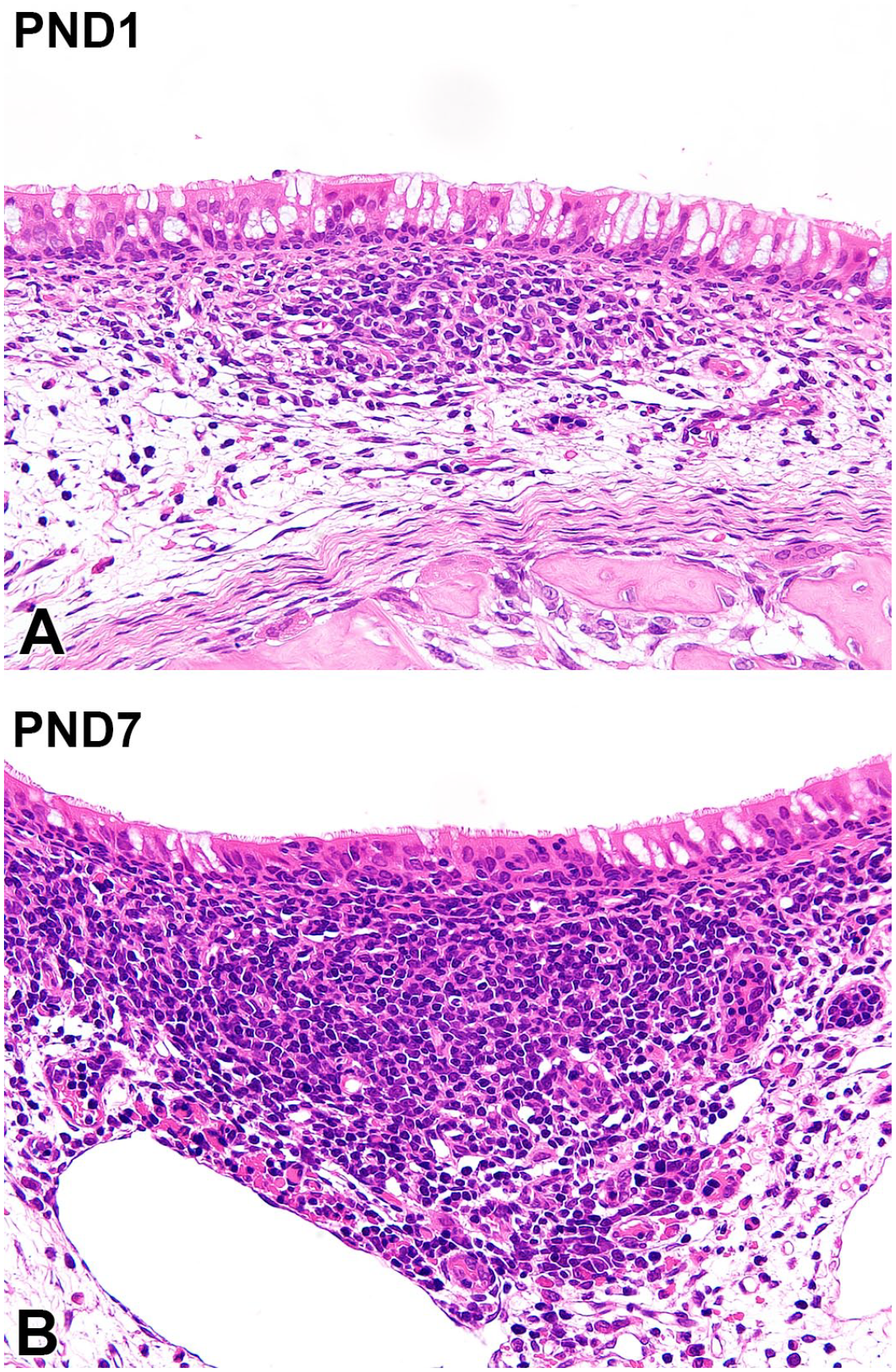
Histology of the nasal-associated lymphoid tissue in Japanese white rabbits. At postnatal day (PND) 1, accumulation of lymphocytes is observed in the lamina propria of the respiratory mucosa in the entrance of the nasopharyngeal meatus (A). The lymphatic nodule forms in the respiratory mucosa at PND7 (B). Original objective 40×. Hematoxylin and eosin.
The nasolacrimal duct was located in the lateral wall of the nasal cavity at levels III and IV (Figure 11A). At PND1, the nasolacrimal duct had lamina propria composed of sinusoidal capillaries with loosely arranged connective tissues around the ducts (Figure 11B). With age, the ductal diameter extended, and the development of the lamina propria progressed (Figure 11C). The expansion of sinusoidal capillaries in the lamina propria was complete on PND18 (Figure 11D).
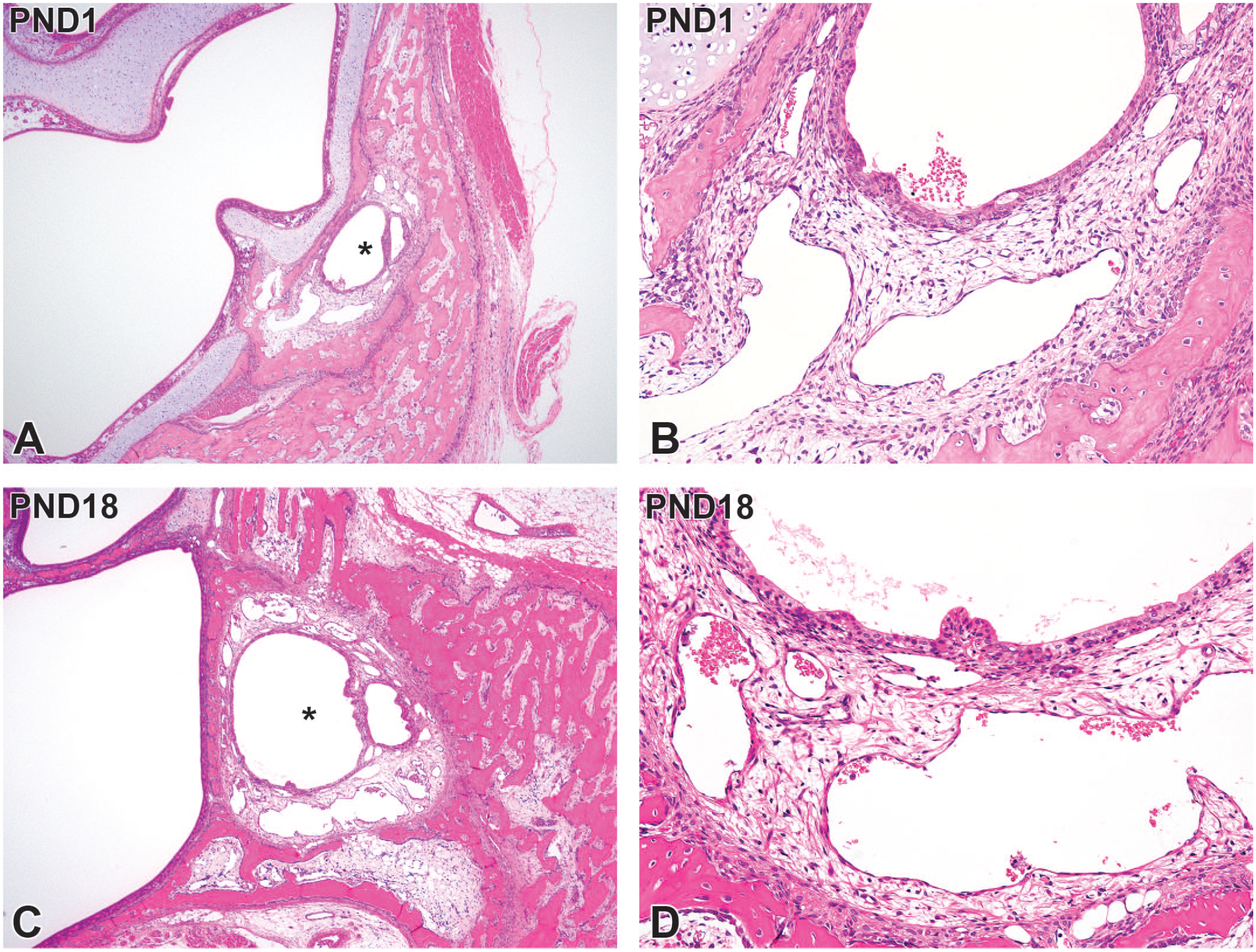
Postnatal development of the nasolacrimal duct on level IV sections taken from Japanese white rabbits. At postnatal day (PND) 1, the nasolacrimal duct (asterisks) forms in the lateral wall of the nasal cavity (A) and has lamina propria composed of sinusoidal capillaries with loosely arranged connective tissues around the ducts (B). At PND18, ductal diameter extends (C), together with the expansion of surrounding sinusoidal capillaries in the lamina propria (D). Original objective 4× (A, C) or 20× (B, D). Hematoxylin and eosin.
Discussion
In the present study, we investigated the postnatal histomorphological development of the nasal passage in Japanese white rabbits from just after birth to 6 weeks of age. The time-wise expansion of the nasal passage could be observed using preparations of longitudinal and cross sections of the nose. Four cross-section levels were selected for this histological investigation based on the methodology used during a previous study on adult albino New Zealand white rabbits. 18 Although several sections were necessary to be re-prepared due to imprecise sectioning caused by the replacement of deciduous teeth by permanent teeth in juvenile rabbits, the postnatal development of nasal cavity components such as the septum, turbinates, vomeronasal organ, NALT, and all types of lined epithelia could be histologically characterized by additional sectioning. The squamous, transitional, respiratory, and olfactory epithelia were distributed similar to those of adults and gradually developed with age. In addition, postnatal histological development of the nasolacrimal duct also occurred in the postnatal age. Therefore, preparing multiple sections of the nasal passage in juvenile toxicity studies in rabbits will contribute to the toxicological evaluation of pediatric drugs administered intranasally or instilled in the eyes.
As stated previously, rabbits are sometimes used in inhalation toxicity studies and pharmacokinetic evaluations for drugs administered intranasally.1-4 Several cases of pediatric medications by intranasal administration of drugs have been reported.19,20 In these cases, the drugs were used to manage pain during pediatric emergency care or for pre- or intraoperative sedation for pediatric patients. This dosing route is noninvasive and painless, and the drugs are effectively delivered to the central nervous system. The current regulatory ICH guidelines state that the necessity of juvenile toxicity studies should be considered before applying new drugs for potential use in children. 12 Therefore, the results of postnatal nasal histological development in this study will contribute to establishing the study design of juvenile toxicity studies using rabbits and their histopathological examination.
Postnatal developmental processes are essential for evaluating drug-induced developmental toxicity in juvenile toxicity studies. In the present study, the rabbits’ nasal passages gradually expanded with the ossification of the nasal walls. Turbinates, particularly the maxilloturbinates, were gradually replaced by bone tissue due to endochondral ossification, and the nose and turbinates increased in size. Endochondral ossification of the nasal wall and turbinates also occurs in rats 16 and miniature pigs. 17 These results suggest that endochondral ossification of the nose contributes to the expansion of the nasal passage.
Histological development of respiratory mucosa also occurs postnatally in rabbits. According to computed tomography and magnetic resonance imaging, the nasal airway gradually expands, and the airflow changes between the ages of 10 days and 5 years. 21 The nasal septum in human neonates is composed of cartilaginous tissue. 22 The cartilaginous septum develops from the posterior to anterior part by endochondral ossification with age. 23 In humans, turbinates consist of three parts, termed superior, middle, and inferior turbinates, and their development begins at week 5 of gestation. 24 Humans do not have maxilloturbinates, whereas rabbits have extremely complex branching. The maxilloturbinate in rabbits helps protect the lower respiratory tract by filtering airborne particles and gases via their branched structure.25,26 Other laboratory animals such as rats, dogs, and monkeys also have maxilloturbinates, although they exhibit far less branching.25,26 To establish the rationale of selecting the ages in employed animals and to extrapolate results to humans, anatomical differences between humans and laboratory animals should be considered when nonclinical juvenile toxicity studies are conducted. These structural differences between humans and animals imply that the targeted respiratory tract regions of drugs are not always matched. However, these are developed by endochondral ossification of the nasal wall in both humans and animals, including rabbits. These facts suggest that histological evaluation of endochondral ossification of the nasal wall and turbinates should focus on the safety of drugs administered intranasally to human children, and careful observation of the developmental toxicity of the nose is required when evaluating maxillary bone formation.
Very few animals were employed in the present study, and only 1 or 2 animals of each age were examined. Thus, histological examination was conducted without considering the sex of the rabbit, and sex differences could not be evaluated because cross-section samples of age-matched males and females could not be obtained, except at PND28. This is a limitation of the present study. Sexual maturation of rabbits occurs 4 to 8 months of age. 27 Indeed, the testes of male rabbits develop until 6 months of age, 28 suggesting that sexual development in male rabbits continues after 6 weeks of age. Nevertheless, the relationship between nasal development and sexual maturation has not yet been fully clarified in this study. Furthermore, nasal passage expansion was only noted by observing 2-dimensional histological sections, not by measuring nasal passage volumes. Applying 3-dimensional evaluation methods, such as computed tomography, will improve understanding of the postnatal morphological development of the nasal passage in rabbits.
The New Zealand white rabbit is one of the most common breeds employed as experimental rabbits 29 and is used in intranasal administration studies. 2 The Japanese white rabbit employed in this study is also used in intranasal administration studies. 30 New Zealand white rabbit strain is albino, as is the Japanese white rabbit strain; however, the histological differences in the nasal passages between these strains have not been examined. Further investigation regarding the strain differences in postnatal nasal histological development is needed to strengthen the rationale for using rabbits in juvenile toxicity studies.
In summary, this study describes the characteristics of the postnatal histological development of nasal passages in Japanese white rabbits. The results of this study provide basic knowledge for the histopathological examination of nasal passages in juvenile rabbits and the rationale for appropriate study design in juvenile toxicology studies.
Footnotes
Acknowledgements
The authors thank Mr Yoshihiro Takei and the staff of Ina Research Inc. for their help during the histological examination (preparation for the specimens for the examination). Their dedication and excellent efforts made these investigations possible. The authors also thank Dr Yu Haranosono from Senju Pharmaceutical Co., Ltd. for his valuable comments to aid the production of this manuscript. The authors thank Editage (![]() ) for English language editing.
) for English language editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
